Abstract
To achieve reliable experimental data, the side-effects of anesthetics should be eliminated. Since anesthetics exert a variety of effects on hemodynamic data and incidence of arrhythmias, the selection of anesthetic agents in a myocardial ischemic reperfusion injury model is very important. The present study was performed to compare hemodynamic variables, the incidence of ventricular arrhythmias, and infarct size during 30 min of ischemia and 120 min of reperfusion in rats using pentobarbital, ketamine–pentobarbital or ketamine–xylazine anaesthesia. A total of 30 rats were randomly divided into three groups. In group P, pentobarbital (60 mg/kg, intraperitoneally [IP]) was used solely; in group K–P, ketamine and pentobarbital (50 and 30 mg/kg, respectively, IP) were used in combination; and in group K–X, ketamine and xylazine (75 and 5 mg/kg, respectively, IP) were also used in combination. Hemodynamic data and occurrence of ventricular arrhythmias were recorded throughout the experiments. The ischemic area was measured by triphenyltetrazolium chloride staining. The combination of ketamine–xylazine caused bradycardia and hypotension. The greatest reduction in mean arterial blood pressure during ischemia was in the P group. The most stability in hemodynamic parameters during ischemia and reperfusion was in the K–P group. The infarct size was significantly less in the K–X group. Whereas none of the rats anesthetized with ketamine–xylazine fibrillated during ischemia, ventricular fibrillation occurred in 57% of the animals anesthetized with pentobarbital or ketamine–pentobarbital. Because it offers the most stable hemodynamic parameters, it is concluded that the ketamine–pentobarbital anesthesia combination is the best anesthesia in a rat ischemia reperfusion injury model.
Anesthetic agents have different actions on the myocardium. Pentobarbital sodium is the most widely used anaesthetic with the cardiodepressive effect of reducing heart rate (HR), stroke index and cardiac index in male rats and mice. 1 This drug is used in doses from 30 mg/kg to 100 mg/kg of body weight in many experiments. The depressive action of pentobarbital (P) has been shown to be dose-dependent. Therapeutic doses of P (30 mg/kg) do not exert this depressive effect on the SA nodal pacemaker activity and cardiac contractility. It is considered that an extremely large dose of P has an antiadrenergic effect on cardiac function. 2
Ketamine (K) has several pharmacological properties such as analgesic effects, and the cardiovascular-stimulating effects of increasing blood pressure, HR and cardiac output at the normal dose. 1 Owing to the short anesthetic duration and muscular rigidity of K, the use of K alone may be inappropriate during surgery of a long duration. Due to the opposing effects of these drugs on the cardiovascular system, a combination of K plus P (K–P) is more appropriate. In addition, by combining these two agents, their doses are reduced to levels at which the disadvantages of each are avoided. 3
Xylazine (X), an alfa2-adrenoceptor agonist, is the sedative analgesic most commonly used by veterinarians. Its adverse effects include bradycardia, cardiopulmonary depression and cardiac dysarhythmia. 4 It is an excellent agent for use in combination with K to provide muscle relaxation and analgesic effects.
The present study was performed to compare differences in the severity of myocardial ischemic reperfusion injury using different anesthetic agents under similar laboratory conditions, and therefore to demonstrate the best agents for use.
Animals, materials and methods
Male Wistar rats weighing 250 to 300 g were housed under standard conditions with free access to food and water. The rats were obtained from animal house of Shiraz University of Medical Sciences, Iran. The investigation was approved by the Animal Ethics Committee of Islamic Azad University of Medical Sciences.
Anesthesia
The three anesthetic protocols investigated in this study were P, K–P and K–X. Pentobarbital was purchased from Sigma (St. Louis, MO, USA). Ketamine and xylazine were purchased from Alfasan Company (Holland). Drugs were injected intraperitoneally. The depth of anesthesia was assessed by testing responses to pedal (foot pinch) stimulation.
Experimental designs
A total of 30 rats were used in this study. They were randomly divided into three groups. In group P (60 mg/kg of P was used solely, IP); in group K–P (50 and 30 mg/kg of K and P, respectively, were used in combination, IP); and in group K–X (75 and 5 mg/kg of K and X, respectively, were also used in combination, IP).
Surgical procedure
The protocol used has been thoroughly described in detail in our previous publication. 5 Briefly, all animals were anesthetized and ventilated with room air enriched with oxygen at a rate of 70 per min. A standard limb lead II electrocardiogram was monitored and recorded throughout the experiment. Catheters were placed into the left carotid artery and tail vein for the monitoring of blood pressure and administration of Evans blue, respectively. After thoracotomy and the removal of the pericardium, a 6–0 silk suture was passed around the left anterior descending coronary artery. Following a stabilization period of 20 min, the left anterior descending artery was occluded for 30 min of ischemia and released for 120 min of reperfusion. Rectal temperature was continuously monitored and maintained at 37 ± 0.5℃.
Determination of infarct size and area at risk
At the end of reperfusion, the left anterior descending artery was re-occluded and 1 mL of 2% solution of Evans Blue dye (Sigma, St Louis, MO, USA) was injected into the tail vein to distinguish the non-perfused area, also known as area at risk (AAR), from the perfused area. The rats were then killed and their hearts were excised and frozen for one hour. The atria and right ventricles were removed and the left ventricles were cut into transverse slices of 2 mm thickness from the apex to the base. Tissue samples were then incubated with a 1% solution of 2,3,5 triphenyltetrazolium chloride (Sigma) for 20 min at 37℃, and subsequently fixed in 10% phosphate-buffered formalin for one hour. Viable myocardium was stained red by triphenyltetrazolium chloride, whereas necrotic myocardium appeared as pale yellow. In each slice, AAR and infarcted areas were determined by computerized planimetry using image analysis software (Image Tool, University of Texas, San Antonio, TX, USA). Infarct size (IS) was expressed as a percentage of the AAR ([IS/AAR]).
Assessment of ventricular arrhythmias
Ischemia-induced ventricular arrhythmias were determined in accordance with the Lambeth conventions: 6 ventricular ectopic beat (VEB) as a discrete and identifiable premature QRS complex (premature with respect to the P wave), ventricular tachycardia (VT) as a run of four or more VEBs at a rate faster than the resting sinus rate, and ventricular fibrillation (VF) as a signal for which individual QRS deflections could no longer be distinguished. Complex forms (bigeminy and salvos) were added to the VEB count and not analyzed separately.
Exclusion criteria
Experiments in which the following events occurred were excluded from the final data analysis: arrhythmias prior to coronary artery occlusion; and arterial blood pressure less than 60 mmHg, indicating a non-perfect surgical procedure.
Statistical analyses
The incidence of VF is expressed as a percentage, and statistical significance is assessed by using Fisher's exact test. After confirming the data for normal distribution, differences were tested for statistical significance using one-way analysis of variance (ANOVA) and significant differences by Tukey multiple comparison test. The data are expressed as mean ± SEM. Hemodynamic values within the same group were analyzed by repeated measures analysis of variance. A probability of P < 0.05 was considered to be significant.
Results
Hemodynamic parameters
Hemodynamics parameters in experimental groups.

Note: Data are presented as mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 compared with the K–X group. †P < 0.05, ††P < 0.01 compared with the P group. §P < 0.05, §§P < 0.01 compared with the pre-ischemic value. HR = heart rate; MBP = mean arterial blood pressure; P = pentobarbital; K–P = ketamine–pentobarbital; K–X = ketamine–xylazine.
Changes in mean arterial blood pressure (MBP) during different stages of ischemic (Isc.) reperfusion (Rep.) injury in the rat heart. Data are presented as mean ± SEM.
 P < 0.05, compared with pre-ischemic values. P = pentobarbital, K–P = ketamine–pentobarbital, K–X = ketamine–xylazine.
P < 0.05, compared with pre-ischemic values. P = pentobarbital, K–P = ketamine–pentobarbital, K–X = ketamine–xylazine.
Infarct size
Figure 2 shows AAR and IS following 30 min of regional ischemia and 120 min of reperfusion. There were no marked differences in AAR among the groups. IS of the K–X group was 23.7 ± 2.2 and this was significantly smaller than that of the other two groups, i.e. 47 ± 3.3 for the P group and 51 ± 4.1 for the K–P group (P < 0.001). There was no significant difference in IS between the P and K–P groups.
Infarct size and AAR following 30 min of regional ischemia and 120 min of reperfusion in different anesthetized rats. Data are presented as mean ± SEM. ***P < 0.001 versus the other two groups. LV = left ventricle, AAR = area at risk, P = pentobarbital, K–P = ketamine–pentobarbital, K–X = ketamine–xylazine.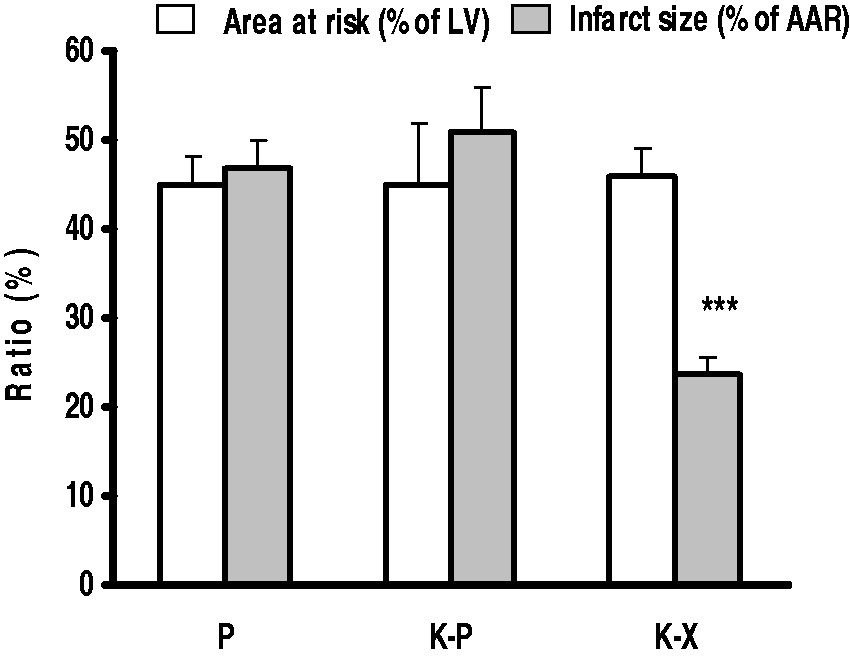
Ischemia-induced arrhythmias
Figure 3 represents the number of VEB (a), VT episode (b) and VT duration (c) during the 30 min ischemic period. The differences in the rate of arrhythmias were not statistically significant among the three groups. The arrhythmias in rats occurred after approximately 5–7 min of ischemia. Figure 3d shows that 57% of the rats subjected to ischemia reperfusion in the P and K–P groups developed VF; however, none of the rats in the K–X group fibrillated. Most fibrillation episodes stopped spontaneously without therapeutic intervention. Consistent with our previous work, there were no considerable reperfusion induced arrhythmias in the present study.
5
Mortality rates for the P, K–P, and K–X groups were 40%, 30%, and 0%, respectively, which did not reach statistical significance.
VEB (a), VT episode (b), VT duration (c) as mean ± SEM, and the incidence of VF (d) as percentage during 30 min of regional ischemia in anesthetized rats. *P < 0.05 versus the other two groups. VEB = ventricular ectopic beat, VT = ventricular tachycardia, VF = ventricular fibrillation, P = pentobarbital, K–P = ketamine–pentobarbital, K–X = ketamine–xylazine.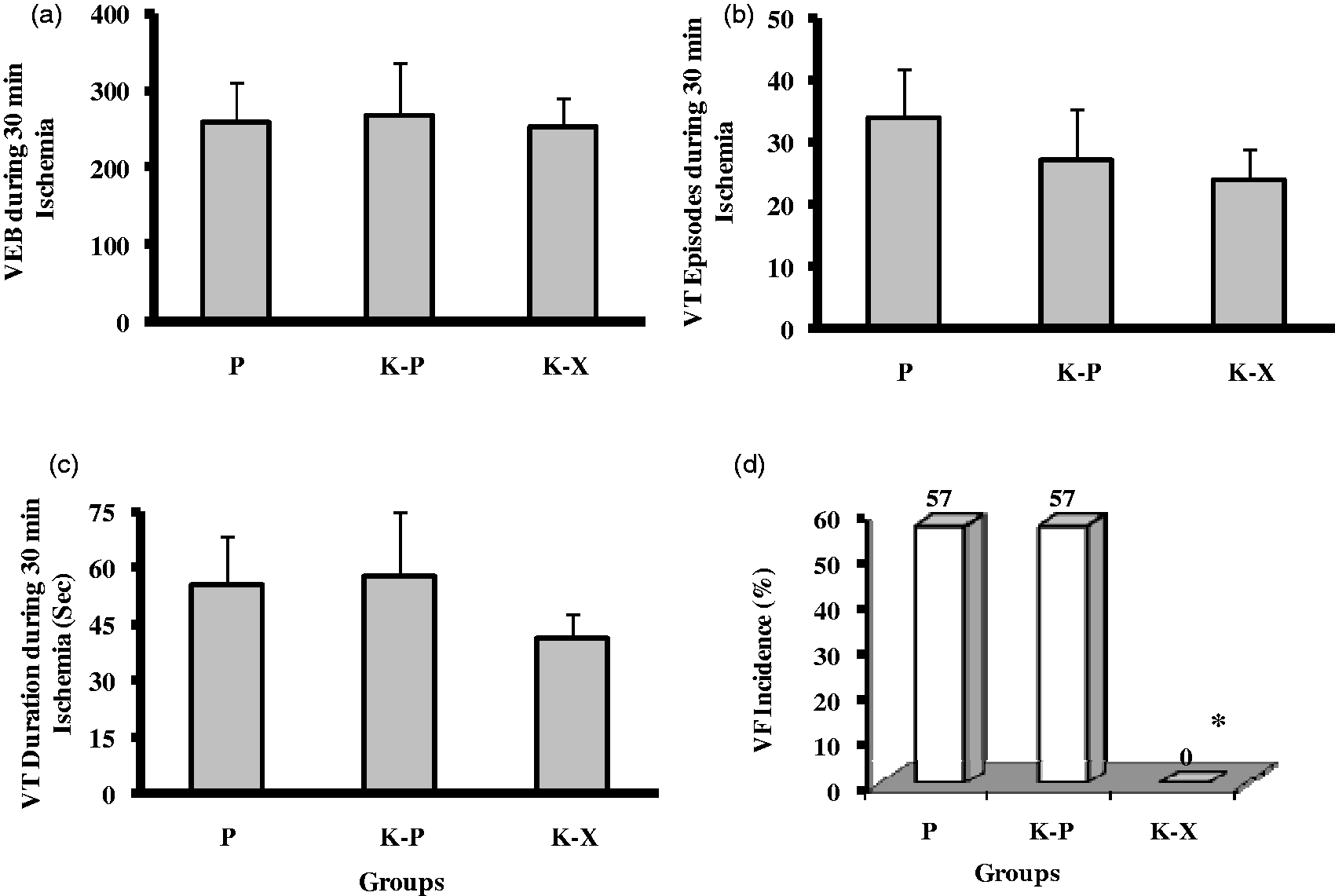
Discussion
The major findings of this study may be summarized as follows: (1) the K–P combination caused more stable hemodynamic parameters during surgery than P alone or the K–X combination. (2) The greatest reduction in blood pressure during ischemia was seen in the P group. (3) The K–X combination induced bradycardia and hypotention at baseline. (4) No VF was observed in the K–X group. (5) IS was significantly less in the K–X group.
The selection of anesthetic agents in a myocardial ischemia reperfusion injury model is very important, since anesthetics exert a variety of effects on hemodynamic data and the incidence of arrhythmias. No single drug is known to be an ideal anesthetic agent for cardiovascular research. The use of a combination of drugs is appropriate, such that the advantages of small amounts of drugs are gained without the disadvantages of large doses of any one drug. 7 These results showed that MBP in the K–P group was significantly higher than in the P group and remained more stable during the ischemic and reperfusion periods. This finding is in agreement with other reports.8,9 The results of open-heart surgery and bypass surgery in minipigs have shown that K–P anesthesia is associated with more stable cardiovascular conditions than is fentanyl–pentobarbital anesthesia. 3
It has been well documented that an intravenous administration of P causes hypotension and tachycardia.2,10 It has also been demonstrated that concentrations of P (≤30 mg) do not exert depressive effect on the heart. An extremely large dose of P has an antiadrenergic effect on cardiac function. 2 Therefore a dose of 30 mg/kg for P was chosen for combination with K.
Ketamine is known to be a cardiovascular-stimulating anesthetic. That is why Worek et al. considered it to be suitable for studies on endotoxin-induced cardiovascular and lung failure in the pig. It was demonstrated that the survival rate was five times as high in the septic K group as in the septic P group. 8
Goldman et al. showed that by combining K with P, the concentrations of each could be reduced to non-toxic levels and pigs could survive with this anesthesia for 96 h without any additional cardiovascular-supporting medication and without any obvious signs of disease. 9
Hypotension and bradycardia were observed in rats using the K–X combination. HR differences between the groups under P and K–X anesthesia were preserved during the ischemia and reperfusion periods. These facts can be explained by the activation of central and presynaptic sympathetic neuronal alpha-2 adrenoceptors. X potentially produces marked cardiopulmonary depression. Respiratory and HR decrease after the administration of X. 11
Our findings of heart arrhythmia and bradycardia with the K–X combination were in agreement with other reports.11,12 No differences in VEB, VT episode and duration were noted among the P, K–P and K–X groups; but in the K–X group, no VF was observed in any of the animals and IS was significantly less. A positive correlation has been shown between pre-ischemic HR with occurrence of VF during ischemia 7 and mortality. 13 That is, tachycardia potentiates the risk of ischemia-induced VF. HR reduction is an approach to consider for the treatment of stable angina and the prevention of sudden death due to VF induced by ischemia which is often associated with tachycardia. In addition, HR reduction has been associated with a reduction of IS. 14 It is therefore hypothesized that the reduction of IS, lack of VF during ischemia, and the lower mortality rate in the K–X group are relevant to bradycadia caused by X.
The IS and the incidence of arrhythmias in the K–P and P groups were the same. Results from our present study reveal that the addition of K to P can reduce the required dose of P, and hemodynamic parameters are more stable during ischemia reperfusion injury. So, K–P anesthesia preserves cardiovascular function better than P anesthesia.
To achieve reliable experimental data, the side-effects of anesthetics should be eliminated. Clearly, the depressed myocardial function caused by P and X should be reason enough to exclude this anesthetic from use in such studies. Among tested agents, the K–P combination had the least hemodynamic effects on the in vivo rat heart. Considering all our results together, it is suggested that under the conditions of this study, a K–P combination appears to be the most reliable anesthetic agent when the purpose is to evaluate interventions for cardiac protection.
Footnotes
Acknowledgment
The authors thank Mrs Zahra Jahanbakhsh for her kind help.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research work in this paper was supported by a grant from the Islamic Azad University, Arsanjan Branch.
