Abstract
Early maternal separation has profound effects on nociception in rats. Cross-fostering is a standard husbandry procedure used by some commercial breeders. This study aimed to determine if cross-fostering altered nociception and the analgesic efficacy of buprenorphine and morphine. At seven and nine weeks of age, an elevated plus maze was used to assess anxiety and Hargreaves apparatus was used to measure thermal nociception at two intensities in cross-fostered and naturally-reared rats. At 10 weeks of age these rats were assigned to one of three treatment groups: saline, buprenorphine or morphine. The Hargreaves apparatus was used to evaluate the effect of analgesics on nociception. Differences were observed in nociception between the cross-fostered and naturally-reared rats at both intensities. At the lower intensity no significant differences were seen between the cross-fostered and naturally-reared rats post-administration of an analgesic. At the higher intensity significant differences were apparent. Morphine was less effective in inducing analgesia to thermal stimuli in cross-fostered rats compared with naturally-reared rats, whereas the opposite was found with buprenorphine which had a more pronounced analgesic effect in the cross-fostered rats. No significant differences in performance on an elevated plus maze were demonstrated between the cross-fostered and naturally-reared rats.
Cross-fostering of laboratory rats is a common procedure used by some commercial animal breeders, and involves the breeder taking all rat pups born on the same day, mixing them and reassigning them, at random, to a dam. This is usually done on the day of birth in order to produce litters of equal sizes. Maternal separation such as this is considered as an early life stressor 1 and has been shown to have lasting effects on a range of responses, including nociception and analgesic efficacy, which can extend into adulthood. 2 This process has also been shown to increase susceptibility to infections, 3 lead to neuroadaptive changes in the hippocampus, 4 alter maternal behaviour 5 and decrease the weight of the adrenal glands. 6 Consequently, this husbandry practice has the potential to affect the results of the studies that use such animals. Cross-fostering could also lead to increased variability in responses, thus requiring an increase in group sizes, which would be considered undesirable both for ethical and economic reasons.
It has been shown that maternal separation can alter both nociception and responses to analgesia. 7 Nociceptive response from the skin can be altered by both stress 8–11 and age. 12 In addition, rats that have undergone maternal separation are less likely to explore the open arms of the elevated plus maze 13 indicating a higher level of anxiety in comparison with rats that were not separated from the dam.
This study was undertaken to assess the effects of cross-fostering on baseline nociceptive thresholds and the response to two analgesic agents, morphine and buprenorphine. In addition, the response in an elevated plus maze was evaluated as a simple measure of anxiety.
Methods
Animals
Twenty-four male and 24 female Sprague Dawley rats (Charles River Laboratories, Margate, UK [specific pathogen-free according to the vendors health status]) weighing 138–265 g at the start of the study were used. Of these, 12 males and 12 females came from natural litters and 12 males and 12 females had been cross-fostered on the day of birth (a standard practice at the supplier for this colony of animals). They were housed in RC1 cages (Tecniplast UK, Ltd, London, UK) in same-sex groups of six according to their maternal experience. The animal room was maintained at 22 ± 2°C, 45% humidity and on a 12/12 h light/dark cycle with food (R&M No. 3, SDS Ltd, Essex, UK) and tap water provided ad libitum. Sawdust bedding was provided (Gold Chip, DB&M). There was a seven-day acclimatization period between the delivery of the animals and the first day of study. Each animal was weighed on a daily basis for the duration of the study. All procedures were carried out in accordance with the Animals (Scientific Procedures) Act 1986 and approved by the local ethical review process.
Elevated plus maze
At seven, nine and 10 weeks of age, the rats were tested using an elevated plus maze (two open arms (0.5 m × 0.1 m) and two closed arms (0.5 m × 0.1 m × 0.4 m), arranged perpendicularly and elevated 1.0 m from the floor). The rats were placed individually on the centre of the maze facing a closed arm and left for a duration of 180 s. During this time, the number of trips on to the open arm and the duration of each trip were recorded using a stop watch (Oregon Scientific C510-Y, Oregon, USA). The testing was carried out under the same light conditions as in the home room and a minimum of 48 h prior to nociceptive testing using the Hargreaves apparatus.
Nociceptive testing (Hargreaves apparatus)
The foot withdrawal latency of the rats was measured using the Hargreaves apparatus (Ugo Basile, Italy, 2000). The cut-off time of the equipment was preset at 33.1 s. Each rat was placed into the testing enclosure (0.22 m × 0.17 m × 0.14 m) and allowed time to acclimatize before measurements were taken. Three measurements of foot withdrawal latency were made on each animal using alternating hind paws. Each trial was separated by a 2 min interval 14 to decrease the possibility of skin injury and alteration of sensitivity of cutaneous nociceptors. Following each trial, the equipment and enclosure were cleaned in order to remove all traces of urine to ensure that infrared (IR) transmission remained constant.
Foot withdrawal latency was assessed at two intensities (60 and 85) at both seven and nine weeks of age on both the naturally-reared and cross-fostered rats. These intensities were chosen following preliminary trials which demonstrated that at lower intensities the rats were able to tolerate the heat and so reach the cut-off time on each occasion. At the intensities used (60 and 85), data were collected without the latency being too close to the cut-off or too close to the start time of zero.
Analgesia
At 10 weeks of age, the animals were separated into three treatment groups: (i) Administration of 5 mg/kg morphine (CP Pharmaceuticals Ltd, Wrexham, UK; 30 mg/mL): (ii) 0.3 mg/kg buprenorphine (‘Vetergesic’, Reckitt-Coleman, Hull, UK; 0.3 mg/mL) or (iii) 1.0 mL/kg saline (B Braun Melsungen AG, Germany) was carried out subcutaneously in accordance with the schedule shown in Table 1. To ensure that effective analgesia was produced, known high doses of the two opioids were administered. 15,16 A cross-over experimental design was employed, with the rodents being separated into two groups each containing 12 cross-fostered (6 male and 6 female) rats and 12 naturally-raised rats (6 male and 6 female). The foot withdrawal latency of each treatment group was tested using the Hargreaves apparatus, as previously described, 1, 3 and 5 h after the administration of saline, morphine or buprenorphine. Following a minimum of 48 h washout period, the analgesic administered was switched between the two analgesic-treated groups (Table 1); group 1 and group 2 were switched so that 12 rats from group 1 received buprenorphine and 12 rats from group 2 received morphine.
Analgesia administration groups (48 rats)
CF: cross-fostered; N: naturally-reared; M: males; F: females
Following this study all rats were euthanized using a rising concentration of carbon dioxide (CO2) in accordance with schedule 1 of the Animals (Scientific Procedures) Act 1986.
Statistical analysis
Nquery Advisor Release 4.0 (Statistical Solutions Ltd, Cork, Ireland) was used in order to carry out power calculations to determine suitable group sizes for the study, using data from previous analysis. Data were analysed using SPSS software (version 12.0 for windows. SPSS Inc, Chicago, IL, USA). All data were tested for normality and homogeneity of variance, and were found to be normally distributed with homogenous variance. A one-way analysis of variance (ANOVA) was used to compare the anxiety and foot withdrawal latency data collected from the rats at ages seven and nine weeks. A repeated measures ANOVA was used in order to analyse the foot withdrawal latency at the three time points following the administration of the analgesics or saline. Area under the curve (AUC) was calculated (Excel macro; Microsoft Excel 2003, Reading, UK) for each treatment at each intensity for both the cross-fostered and naturally-reared rats and then compared using an independent t-test. An independent t-test was also used in order to determine differences between the weights of the cross-fostered and naturally-reared rats. Results were considered statistically significant if P < 0.05.
Results
Weight
There was no significant difference between the weight gain in the cross-fostered and naturally-reared rats throughout the duration of the study.
Elevated plus maze
At ages seven and nine weeks, no significant difference was found between the cross-fostered and naturally-reared rats in either the number of trips on to the open arms of the elevated plus maze or in the total duration of time spent on the open arms. A significant sex difference was found at both the seven- and nine-week time points, with female rats taking more trips on to the open arm (P < 0.0001) and spending more time on the open arm (P < 0.021) than male rats. At 10 weeks of age, the same pattern of results was observed. No significant difference was observed when comparing the cross-fostered and naturally-reared rats when considering the number of trips on to the open arms or the time spent on the open arms of the elevated plus maze. At 10 weeks of age a significant sex difference was still evident, with the female rats again taking more trips on the open arm (P < 0.049) and spending longer on the open arms (P < 0.031) than the male rats. Data are shown for the three ages in Table 2.
Elevated plus maze data for cross-fostered (CF) and naturally-reared (N) rats aged seven, nine and 10 weeks
No significant differences were found between CF and N rats of any age
Hargreaves test – intensity 60
At seven weeks of age, there was no significant difference between the sexes shown when analysing the thermal intensity at 60. At this intensity and age there was a significant difference between the cross-fostered and naturally-reared rats (Figure 1). The withdrawal latency of the cross-fostered rats was significantly higher that that of the naturally-reared rats (P < 0.029). There was a significant interaction between the rat's history and its sex in the foot withdrawal latency at this thermal intensity.
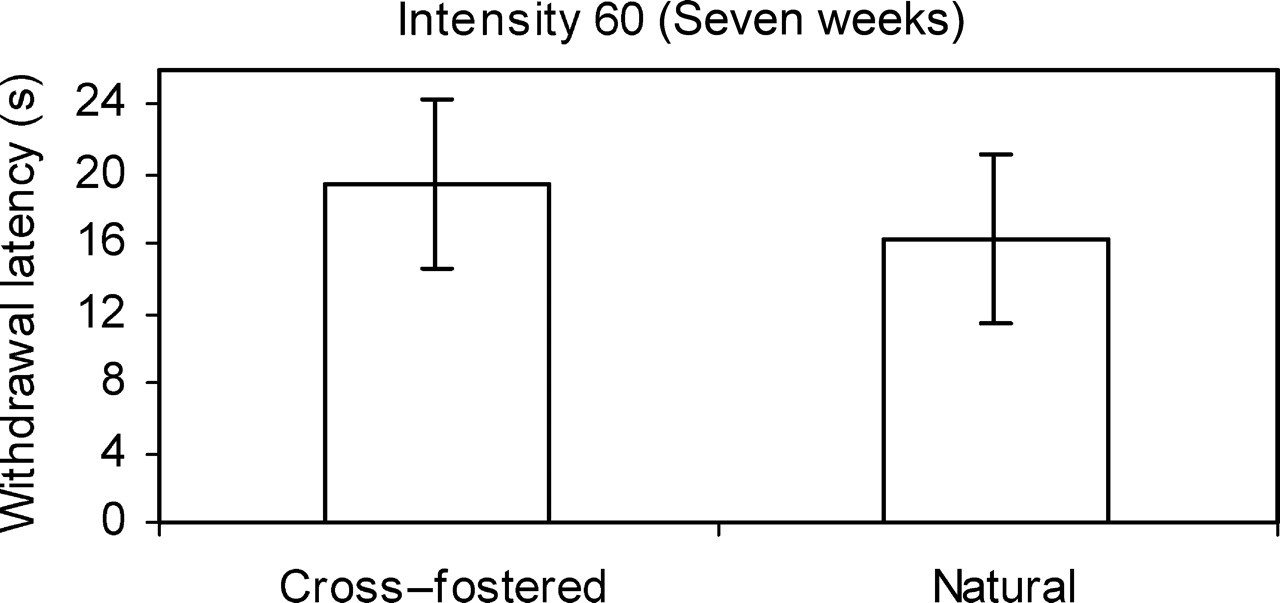
Mean foot withdrawal latency for cross-fostered and naturally-reared rats at an intensity of 60, aged seven weeks showing a significant difference between the groups
At nine weeks of age, there were no significant differences between the sexes or the cross-fostered and naturally-reared rats in their foot withdrawal latency.
Hargreaves test – intensity 85
At seven weeks of age, female rats exhibited significantly longer foot withdrawal latencies than males (P < 0.015) at a thermal intensity of 85. There was no significant effect of rat history. There was no significant interaction between the rats' history and their sex. At nine weeks of age there was no significant difference between the sexes, but cross-fostered rats had significantly lower withdrawal latencies than naturally-reared rats (P < 0.017).
Age-related effects
When comparing rats of age seven and nine weeks, foot withdrawal latency was significantly affected by age (P < 0.024). At nine weeks of age the withdrawal latency, at both intensities, was significantly longer than at seven weeks of age. Foot withdrawal latency was also significantly affected by an interaction between age and thermal intensity (P < 0.037). There were significant differences between the sexes between weeks seven and nine of age (P < 0.008). At a thermal intensity of 60 both sexes had a longer foot withdrawal latency at nine weeks of age compared with seven weeks of age. Significant changes were found in cross-fostered and naturally-reared rats at these two ages (P < 0.002), showing at an intensity of 60, the rats at nine weeks had a longer withdrawal latency compared with at seven weeks of age.
Correlation of anxiety and baseline nociceptive measures
At seven weeks of age, both the cross-fostered and naturally-reared rats showed a strong positive correlation between the number of trips on the open arm of the elevated plus maze and the foot withdrawal latency at thermal intensity 60 (P < 0.001 for all comparisons) and at thermal intensity 85 (P < 0.001 for all comparisons). At nine weeks of age, both the cross-fostered and naturally-reared rats showed a strong positive correlation between the number of trips on the open arm of the elevated plus maze and the foot withdrawal latency at thermal intensity 60 (P < 0.001 and P < 0.002, respectively) and thermal intensity 85 (P < 0.001 for all comparisons and P < 0.002 for all comparisons, respectively). In addition, a strong positive correlation was seen, at both seven and nine weeks of age for the cross-fostered and naturally-reared rats, between the duration of time spent on the open arm of the elevated plus maze and the foot withdrawal latency at thermal intensity of 60 (P < 0.001 for all comparisons) and thermal intensity 85 (P < 0.001 for all comparisons).
Analgesia
At a thermal intensity of 60, the cross-fostered rats showed a significant difference between the AUCs of buprenorphine and morphine (P = 0.043) and between buprenorphine and saline (P ≤ 0.001). In each case, the AUC of buprenorphine was significantly greater. AUC of the morphine was significantly greater than the AUC of saline (P = 0.015). The naturally-reared rats at thermal intensity 60, showed saline to have a significantly lower area than both buprenorphine and morphine (P ≤ 0.001 and 0.008, respectively). However, there was no significant difference between the morphine and buprenorphine areas.
At a thermal intensity of 85, significant differences were observed between the AUCs. When comparing buprenorphine to morphine and buprenorphine to saline, a P value of <0.001 was obtained for the cross-fostered rats; in each case the buprenorphine had the largest AUC. There was no significant difference when comparing morphine with saline. At intensity 85, the naturally-reared rats showed no significant difference between the AUCs of buprenorphine and morphine, however, the difference between the buprenorphine and saline, and the morphine and saline still remained highly significant with a P value of <0.001 in both cases. In each case, the opiod had the larger AUC.
At a thermal intensity of 60, no significant difference was seen in the AUCs of buprenorphine between the cross-fostered and naturally-reared rats. This was also the case for both morphine and saline (Table 3).
Mean area under the curve (AUC) for the three treatments given to the cross-fostered (CF) and naturally-reared (N) rats
At an intensity of 85, a significant difference was seen between the AUCs of buprenorphine and morphine (Figures 2 and 3) for the cross-fostered and naturally-reared rats (P = 0.045 and 0.023, respectively). The cross-fostered and naturally-reared rats had however no significant differences between the AUCs of saline.
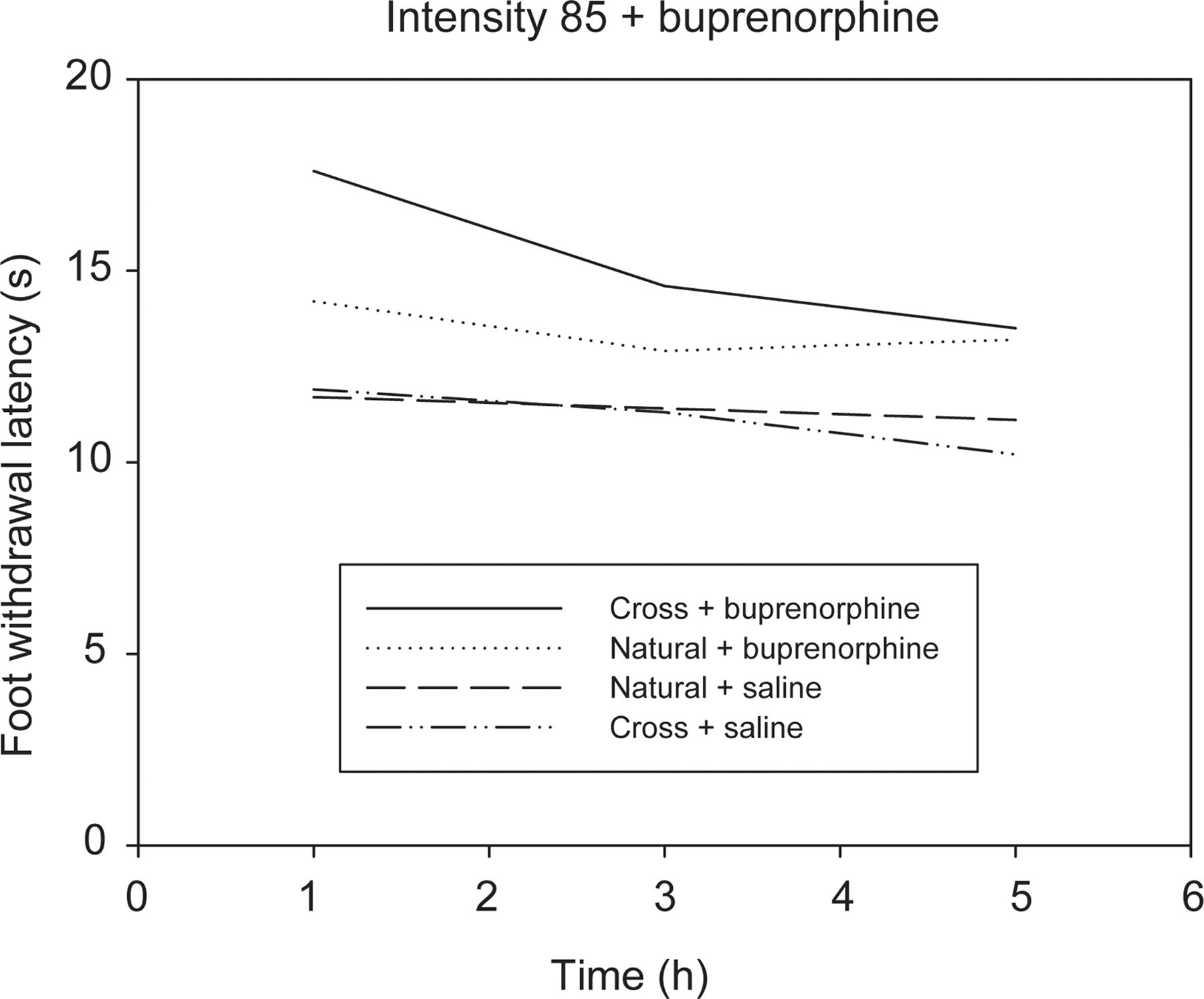
Effect of buprenorphine over time on the withdrawal latency of cross-fostered (Cross) and naturally-reared (Natural) rats aged 10 weeks, intensity 85, showing significant differences between the area under the curve between Cross and Natural rats following buprenorphine administration
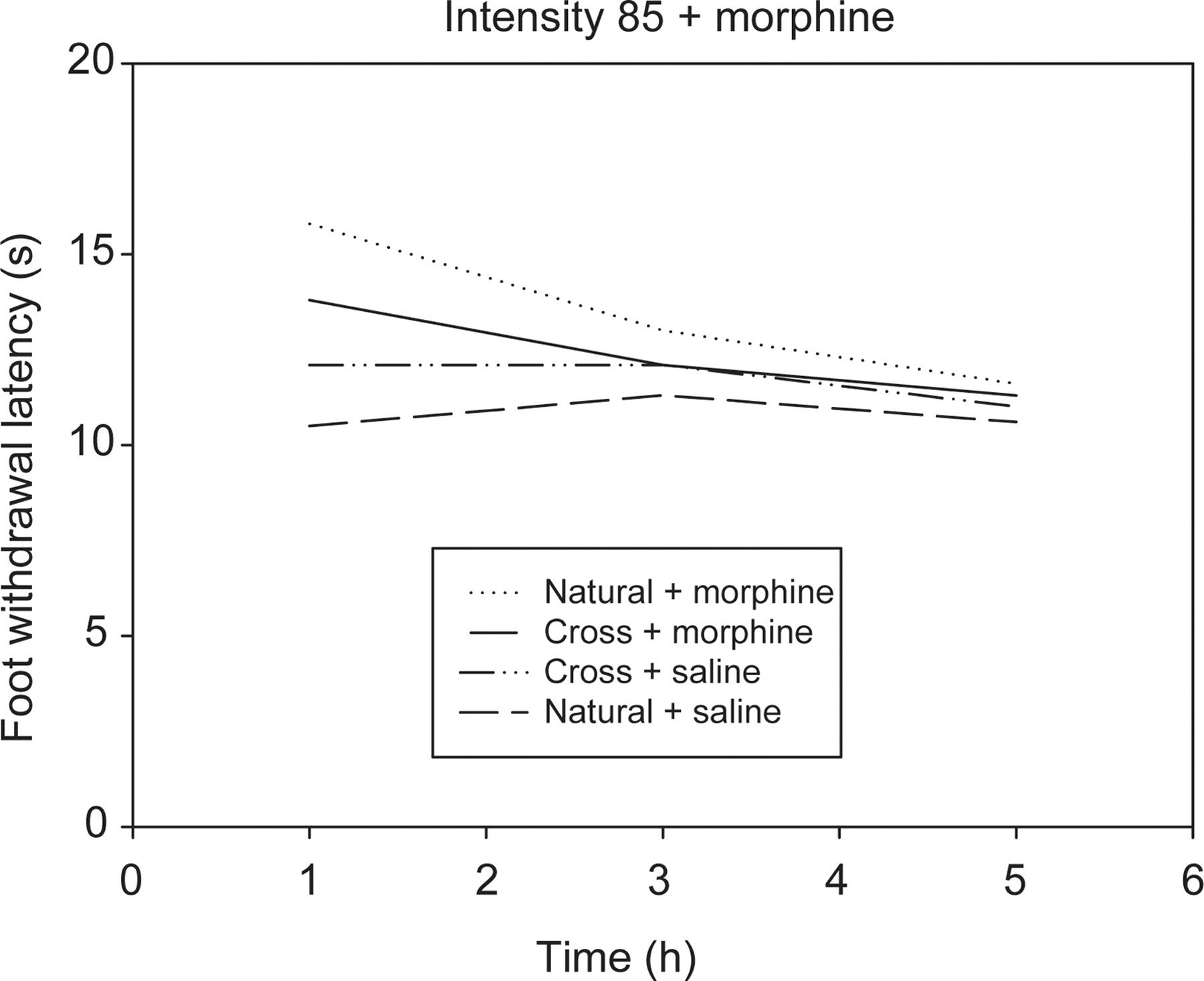
Effect of morphine over time on the withdrawal latency of cross-fostered (Cross) and naturally-reared (Natural) rats aged 10 weeks, intensity 85, showing significant differences between the area under the curve between Cross and Natural rats following morphine administration
Correlation of anxiety and nociceptive measures
At each time point following analgesic administration there was no correlation seen between the Hargreaves (intensity 60) data and the number of trips on the open arms of the elevated plus maze for the cross-fostered rats. Under the same conditions at one hour post-administration, the naturally-reared rats showed a significant negative correlation between the two factors (P = 0.048). At 3 h and 5 h post-administration, there was no correlation seen.
At a thermal intensity of 85, there was no significant correlations for either the cross-fostered or naturally-reared rats at any of the time points, between the withdrawal latency and the number of trips on the open arms.
There was no significant correlation between the time spent on the open arms of the elevated plus maze and the Hargreaves data at a thermal intensity of 60 or 85 for either the cross-fostered or naturally-reared rats.
Discussion
The common practice of cross-fostering involves the breeder taking all rat pups born on the same day, mixing them and reassigning them to a dam in order to produce litters of equal sizes. Consequently, all rat pups purchased from these commercial breeders will have at least been briefly maternally separated, many will have been cross-fostered, but some would have been reassigned by chance to their natural mother. The chance of reassignment to their natural mother will decrease as the size of the breeding colony increases. The aim of this study is to determine if this practice of cross-fostering influences nociceptive thresholds, the responses to nociceptive testing and the effects of opioid administration. Although significant effects of these early life stressors were demonstrated, the magnitude of the changes were small.
When anxiety levels in the cross-fostered and naturally-reared rats were tested, no significant differences were found. However as with previous studies, female rats were less anxious than the male rats; visiting the open arms more frequently and spending significantly longer on the open arms of the elevated plus maze. 13,17 The differences were inconsistent in the foot withdrawal latencies between the cross-fostered and naturally-reared rats. At seven weeks of age, the withdrawal latencies were longer in the cross-fostered rats than in the naturally-reared rats at thermal intensity 60 but not at thermal intensity 85. At nine weeks of age, the withdrawal latencies were longer in the cross-fostered rats than in the naturally-reared rats at both thermal intensities, however this difference was only statistically significant at thermal intensity 85. Although these differences may have been statistically significant, they may be of little biological relevance due to the small differences in the actual responses observed. This corresponds to other studies investigating early maternal separation in rats. 18 Stressful stimuli such as maternal separation at an early age has been suggested to cause stress-induced analgesia. 10 Despite the observed differences, the data were not sufficiently robust to conclusively state that cross-fostering rats decreases sensitivity to thermal stimuli. Therefore, it seems that cross-fostering is not sufficiently stressful to cause major alterations in the rats' responses to thermal nociception in this case.
Buprenorphine is a potent and long-acting analgesic, 19 which is a partial agonist of μ, κ and δ opioid receptors. 20–22 Morphine is an opioid analgesic which is known to act on μ receptors. 23
A fast onset of action was seen following the administration of both buprenorphine 15 and morphine, with the peak effect being seen at the one-hour time point for both drugs (Figures 2 and 3).
At a thermal intensity of 60, no significant differences were shown in the AUC between the cross-fostered and naturally-reared rats for any of the three treatments tested. At intensity 85 this was also true for saline, however significant differences were shown for both buprenorphine and morphine. This difference showed that when administered the same dose of morphine, the cross-fostered rats showed a decreased effect in its action. A previous study 10 showed a decrease in effect of morphine on nociception in rats that were stressed following a period of restraint in comparison with non-stressed rats. It is thought that this change in morphine sensitivity may be due to modulation of opiate receptors following repeated stimulation from endogenous opioids which are released when the animal is stressed. 10 This could be the case for rats that have been maternally separated at an early age. Kalinichev et al. 24 reported that morphine is less potent at inducing antinociception in rats that have been maternally separated in comparison with control rats. This could be the result of alterations to endogenous opioid systems 24 causing long-lasting changes in opioid responsiveness. 25 This alteration could be due to a decrease in the number of opioid receptors which would naturally lead to a decrease in the analgesic efficacy of morphine. 7
At a thermal intensity of 85, the effect of buprenorphine was significantly greater in the cross-fostered rats than in the naturally-reared rats. A previous study has indicated that in a situation of chronic pain, adaptive changes are made to the opioid system resulting in upregulation of various receptor types including δ receptors. 22 Based on the results collected, it may be the case that stress, such as when maternally separated, may also induce this upregulation of opioid receptors leading to an increase in sensitivity to buprenorphine. As this was only demonstrated at one of the two intensities tested, further study is needed in this area.
The morphine and buprenorphine findings may appear contradictory as both analgesics act upon the same opioid receptors. 26,27 This is therefore an area which requires further investigation. However, the fact remains that following the cross-fostering process, alterations are found in the sensitivity to these analgesics.
In conclusion, the process of cross-fostering leads to changes in thermal nociception and sensitivity to buprenorphine and morphine. It is demonstrated that at specific ages and intensities, rats that had been cross-fostered had an increased nociceptive threshold. However, the magnitude of these changes is relatively small and so should have limited effects on analgesic studies. Cross-fostering may have other effects on nociception and analgesia in rats and this warrants further investigation. It may also be important to study other strains of rat as the animals' genotype may influence its response to early life stressors.
Previous study in rats has shown a range of alterations following early maternal separation. These include facilitation of primary infection by Nippostrongylus brasiliensis,
3
a decrease in the weight of the adrenal glands,
6
neuroadaptive changes in the hippocampus such as a decrease in the expression of N-methyl-
Footnotes
ACKNOWLEDGEMENTS
The authors thank the British Pharmacological Society for financial support, Charles River UK for supply of the animals and the staff of the Comparative Biology Centre, for technical support. Amy Dickinson continues to be funded by the BBSRC and Pfizer plc.
