Abstract
Infections with Helicobacter spp. are known to have serious effects on rodent health, especially in immunocompromised animals. In this study three approaches were used to eradicate Helicobacter spp. infection in mice with a deficiency in intestinal proteoglycan (mucin2), namely triple oral antibiotic therapy (amoxicillin, clarithromycin and metronidazole), cross-fostering of neonatal pups by surrogate mothers negative for Helicobacter spp., and in vitro fertilization (IVF) with embryo transfer into Helicobacter-free mothers. However, triple antibiotic therapy in mice with mucin2 deficiency was not effective and had negative effects on reproductive performance, and high susceptibility of mucin2-deficient mice to Helicobacter spp. during the first 12 h after birth rendered cross-fostering impossible. Only IVF with embryo transfer was effective in eradicating Helicobacter infection from transgenic mice with mucin2 deficiency.
Since 2014 quarterly testing for Helicobacter spp. has been included in the health monitoring program recommended by the Federation of European Laboratory Animal Science Associations (FELASA). 1 Three methods have been used to eradicate Helicobacter infection from a mouse colony, namely antimicrobial treatment, cross-fostering of neonatal pups to surrogate mothers, and embryo transfer. 2 In this report we summarized the results that were achieved using all the three mentioned approaches to eradicate Helicobacter spp. (H. billis, H. hepaticus and H. typhlonius) in a mucin2-deficient mouse colony.
Materials and methods
Animals
Helicobacter-infected immunocompromised C57BL/6-Muc2tm1Avel; Zbtb33tm1Bird double knockout mice (DKO), a potential mouse model of colon cancer, 3 and wild-type immunocompetent C57BL/6NCrl mice (WT) were transferred from the Nursery for Laboratory Animals (Pushchino, Russia) to the Institute of Cytology and Genetics, Siberian Branch of the Russian Academy of Sciences (SB RAS) (supported by grant RFMEFI61914X0005 and RFMEFI62114X0010). The mice were housed in the individually ventilated caging (IVC) systems (OptiMICE; Animal Care Systems, Centennial, CO, USA) with autoclaved dust-free birch bedding. Sterile water and diet (Ssniff, Soest, Germany) were provided ad libitum. Enviromental conditions were controlled at a temperature of 22 ± 2℃ and humidity at 35–60% with a photoperiod of 14 h light:10 h dark.
For the antibiotic therapy, 6–7-week-old mice were used (10 female and 10 male DKO, or 5 female and 4 male WT mice). Cross-foster rederivation of DKO mice involved five pairs of 8–12-week-old Helicobacter-positive DKO and 8–12-week-old Helicobacter-negative immunocompetent Hsd:ICR (CD-1) mice. Five 8–10-week-old WT Helicobacter-negative females and one DKO male were used for in vitro fertilization (IVF). Two 8–10-week-old female and one male Helicobacter-negative Hsd:ICR (CD-1) mice were used for the embryo transfer.
All experimental work with the animals was approved by the Committee on the Ethics of Animal Experiments of the Russian National Center of the Institute of Cytology and Genetics, SB RAS (No. 28 19.06.2015).
Antibiotic treatment
The dosage of the antibiotics added to drinking water was based on published antibacterial doses using an assumed daily consumption of 5 mL by an adult mouse. Mice were treated for two weeks with drinking water containing 0.6 mg/mL amoxicillin, 0.2 mg/mL metronidazole and 0.2 mg/mL clarithromycin. 2
Helicobacter-negative Hsd:ICR (CD-1) and Helicobacter-positive DKO mice were bred to produce dams and pups for fostering. Litters of Helicobacter-positive DKO dams were placed with Helicobacter-negative dams within 12–24 h after birth. Litters were weaned at the age of four weeks.
Mature Helicobacter-negative WT female mice were superovulated. Fourteen hours later the mice were sacrificed by cervical dislocation, and their oviducts were removed. Cumulus–oocyte complexes (COCs) obtained from the ampulla of the fallopian tubes were collected and placed in an HTF medium (Cosmo Bio, Tokyo, Japan) covered with mineral oil (Sigma Aldrich, St Louis, MO, USA) and incubated at 37℃ under 5% CO2 in air. Spermatozoa from the epididymal caudae of one Helicobacter-positive DKO male mouse were suspended in an HTF medium covered with sterile mineral oil and incubated at 37℃ under 5% CO2 in ambient air for one hour. Insemination was carried out by adding the sperm suspension to the COCs. For embryo transfer, two-cell embryos were transferred to Helicobacter-negative Hsd:ICR (CD-1) pseudopregnant females, and the recipients were allowed to give birth naturally. 4
Reproductive efficiency
The number of days to birth was calculated from the date a male breeder was introduced into the cage of a female breeder until the offspring was born. The number of pups that survived until weaning was recorded 21 days after birth.
Health status
According to the recommendations by FELASA, since 2014, pathogens to be tested every three months were investigated in sentinel mice quarterly, whereas pathogens to be tested annually were investigated once a year at QM Diagnostics, Nijmegen, The Netherlands. 1 Colonies of mice were shown to be negative for all tested pathogens, except for Helicobacter spp.
Helicobacter spp. assay
Fecal DNA from individual mice was tested for Helicobacter spp. Fecal DNA was extracted using the QIAamp Fast DNA Stool Mini Kit (Qiagen, Hilden, Germany). Helicobacter spp. were detected using real-time polymerase chain reaction (PCR) kits for the detection of Helicobacter spp., H. bilis, H. hepaticus and H. typhlonius, according to the manufacturer’s instructions (Belki-Biotechnologies, Novosibirsk, Russia). Briefly, 50 ng of sample DNA was added to 15 µL of a corresponding Master-mix containing specific primers and SYBR Green I for real-time detection. The sample was considered positive if the Ct value was within the detection limit of the kit and if the melting temperature was identical to the one of the positive control. 5
Statistical analysis
Helicobacter-positive and negative data are presented as totals. The reproductive efficiency data are presented as mean ± standard error. The statistical comparisons between different treatments were performed using Mann–Whitney tests.
Results and discussion
Helicobacter spp. in mice after different approaches to eradication.
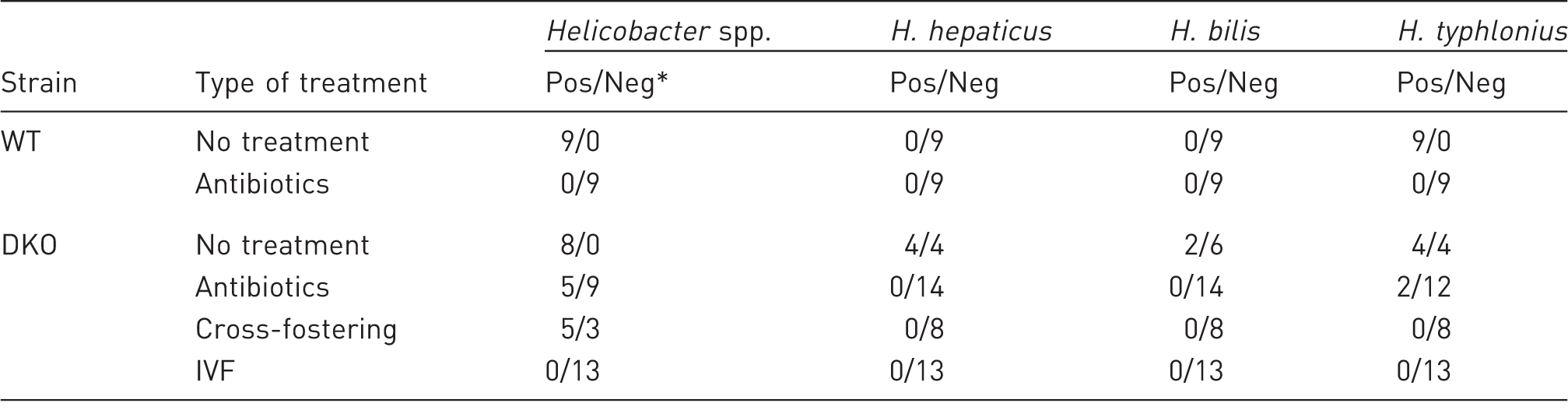
Number of positive results versus number of negative results of Helicobacter spp. assay. IVF: in vitro fertilization, WT: wild-type mice, DKO: double knockout mice.
Reproductive efficiency of mice after Helicobacter spp. eradication.
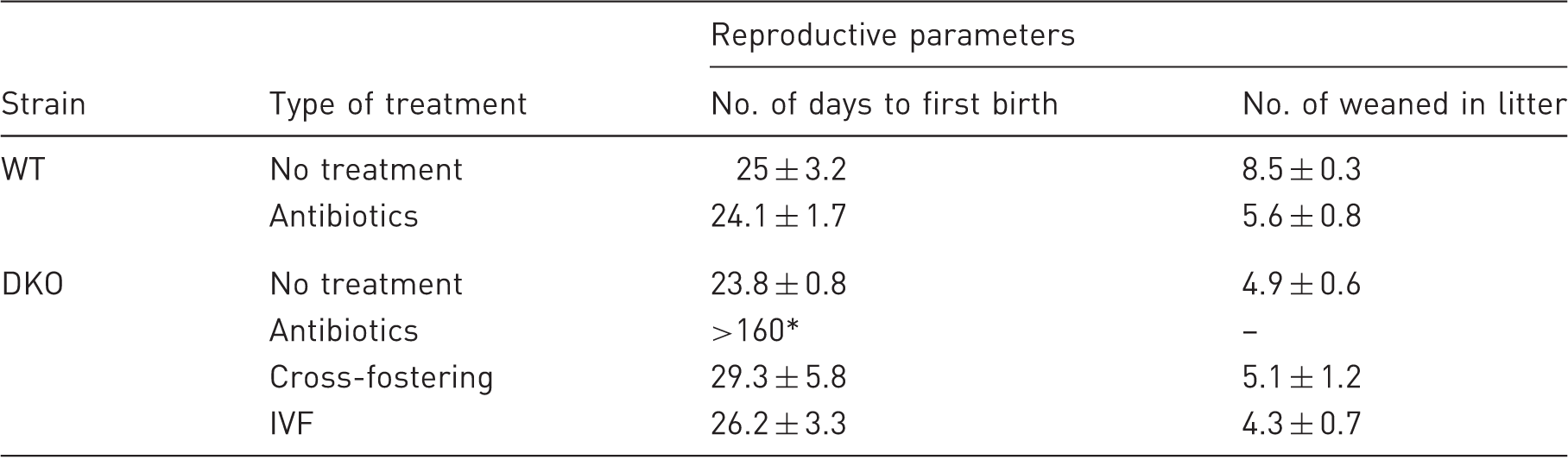
P < 0.001 as compared with other groups, Mann–Whitney test. IVF: in vitro fertilization, WT: wild-type mice, DKO: double knockout mice.
As previously published, IVF is more effective for the elimination of bacterial and viral infections in mice, but this is labour-intensive and costly. 7 Indeed, IVF successfully eliminated 100% of Helicobacter spp. in DKO mice (Table 1) and in two succeeding generations.
The primary reason for eliminating Helicobacter spp. from mouse colonies is the negative impact on reproductive performance of immunocompromised mice. 8 After antibacterial treatment of immunocompetent C57BL/6NCrl mice, the reproductive performance was the same as in the untreated mice (Table 2). Nonetheless, antibiotic therapy affected reproduction of mucin2-deficient mice: Helicobacter-negative DKO mice had no litters over the course of six months. We concluded that antibiotics affected reproductive function of mucin2 knockout mice only (Table 2).
In conclusion, some of the attempts to eradicate Helicobacter spp. in mucin2-deficient mice are in no way effective for eliminating this infection, and can be detrimental to their reproductive performance. IVF is a more effective method of eliminating Helicobacter infection from immunocompromised mice.
Footnotes
Acknowledgement
We thank Pr. E B Prokhortchouk for providing the knockout mice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Russian Foundation for Basic Research grant No. 15-04-07653 and by project No. 0324-2015-0004 of the Federal Research Center, Institute of Cytology and Genetics, SB RAS.
