Abstract
Summary
A one-year-old healthy sheep received an implant stenting the mural (‘posterior’) leaflet of the mitral valve. The experiment was authorized by the Cantonal Ethical Committee. The surgery was performed on the open, beating heart during cardiopulmonary bypass (CPB). Management of anaesthesia was based on isoflurane with mechanical intermittent positive pressure ventilation (IPPV) of the lungs, combined with intercostal nerve blocks and intravenous fentanyl and lidocaine. Marked cardiovascular depression occurred towards the end of CPB time and required high doses of dopamine, dobutamine, lidocaine and ephedrine to allow for weaning off the CPB pump. Moreover, severe pulmonary dysfunction developed when IPPV was re-initiated after CPB. Hypoxaemia persisted throughout the recovery from general anaesthesia. Multiple organ failure developed gradually during the three postoperative days, leading to euthanasia of the animal. As described in this case, marked lung injury associated with some degree of failure of other vital organs may occur in sheep after CPB. Intraoperative cardiorespiratory complications when weaning-off may indicate the development of ‘post-pump syndrome’.
Keywords
Mitral valve repair during cardiopulmonary bypass (CPB) is a routine procedure in human medicine for the treatment of degenerative mitral valve insufficiency. However, CPB contributes to peri- and postoperative morbidity and should be kept as short in duration as possible. To investigate a new time-saving concept of mitral valve reconstruction, a healthy sheep was anaesthetized, CPB installed and a mitral valve device implanted. The surgical procedure consisted of implanting a heterograft (MitroFast, Shelhigh Inc, Union, NJ, USA) to stent the mural leaflet of the mitral valve in combination with semicircular annuloplasty (Eckstein et al. 2005). A follow-up period of six months was envisaged as part of the original study design.
Materials and Methods
A one-year-old, healthy, female sheep weighing 54 kg, adequately vaccinated and dewormed, received 1500 IU of tetanus antitoxin (Tetanus-Serum Intervet, Veterinaria AG, Zürich, Switzerland) and was fasted 24 h prior to anaesthetic premedication (access to water was granted at all times). The results from blood cell count and serum biochemistry analyses were in the normal range.
Anaesthesia
After intramuscular premedication with 4 mg/kg ketamine (Narketan-10, Vetoquinol AG, Ittigen, Switzerland) and 1 mg/kg midazolam (Dormicum, Roche Pharma AG, Reinach, Switzerland), anaesthesia was induced and maintained at first with propofol (Disoprivan, AstraZeneca AG, Zug, Switzerland) intravenously (IV) and oxygen supplemented via a cuffed endotracheal tube (12 mm ID) during surgical preparations. Later in the operating theatre, general anaesthesia was maintained with isoflurane in oxygen and intermittent positive pressure ventilation (IPPV) was established. A tidal volume of 750 mL was administered seven times per minute. Ruminal regurgitations were collected by an orogastric tube to avoid aspiration. Intercostal nerve blocks using lidocaine 1% (Xylesin 2%, Amino AG, Neuenhof, Switzerland) and bupivacaine 0.5% (Bucain, Curasan AG, Kleinostheim, Germany) were performed in equal volumes to a total of 15 mL (intercostal spaces 3-7). Fentanyl (3-5 mg/kg/h; Fentanyl-Janssen, Janssen-Cilag AG, Neuss, Germany) and lidocaine (2 mg/kg followed by 1.8 mg/kg/h) were infused IV throughout the duration of surgery. Besides antibiotics, dexamethasone (Dexadreson, Veterinaria AG) and furosemide (Dimazon, Veterinaria AG) were both administered once IV at the beginning of the surgical procedure. Just after thoracotomy, aprotinin (Trasylol, Bayer Health Care AG, Leverkusen, Germany) was administered IV to limit complement activation during CPB (10,000 KIU/kg over 30 min followed by 10,000 KIU/kg/h). After the IV administration of unfractionated heparin (100 IU/kg; Liquemin, Roche Pharma AG), activated coagulation time (ACT; Hemochron ACT, Medtronic, Minneapolis, MN, USA) remained over 999 s for 210 min. No protamine was administered. Continuous monitoring of oxygen saturation (SpO2), heart rate and rhythm (electrocardiogram, lead II), respiratory rate, inspired and expired carbon dioxide and oxygen concentrations, airway pressures and volumes, caudal oesophageal temperature, urine output did not reveal any complication prior to the initiation of CPB. The systemic arterial blood pressure (measured at the auricular artery) and the pulmonary artery pressures were also continuously monitored and remained adequate (systemic systolic arterial pressure > 110 mmHg). The values of arterial blood gas analyses (PaO2, PaCO2, pH, HCO3-) as well as haemoglobin concentrations, electrolytes, lactate and glucose measurements (from systemic arterial blood) performed every 20 min were in the normal range.
Cardiopulmonary bypass
CPB was commenced using an extracorporeal membrane oxygenator by deviating blood flow from the caudal vena cava into the femoral artery. Blood temperature was maintained at 36°C (mild hypothermia). The heart maintained continuous but irregular beating activity (ventricular tachycardia) during the open-heart surgical procedure. No action was taken to restore the sinus rhythm until the end of the open-heart surgery. During the CPB, continuous positive airway pressure of 5 cmH2O was applied with oxygen in the lungs instead of IPPV to maintain the airway tissue oxygenation. The open-heart surgery lasted 36 min and the duration of the CPB was 52 min. Details of the mitral valve stenting technique are as published elsewhere (Eckstein et al. 2005).
After the heart surgery, three electrical discharges (20 Joules) applied directly onto the heart with a defibrillator followed by one minute of intrathoracic heart compressions and topical administration of lidocaine (0.5 mg/kg) were necessary to achieve cardioversion into sinus rhythm. In order to further support spontaneous heart beats in generating a minimum systolic arterial blood pressure of 75 mmHg, high doses of intravenous dobutamine infusion (3 mg/kg/min; Dobutrex, Asta Medica AG, Frankfurt, Germany) and repeated ephedrine boli (0.2 mg/kg; Ephedrin Streuli, Streuli Pharma AG, Uznach, Switzerland) were used in addition to a dopamine infusion (6-8 mg/kg/h; Dopamine, Fresenius-Kabi AG, Stans, Switzerland). Cardiovascular parameters were then judged acceptable with a peak central venous pressure of 8 mmHg and a left atrial pressure of 9 mmHg. Postoperative transoesophageal echocardiography revealed a competent mitral valve (transvalvular gradient below 3 mmHg) without visible inflow restriction. Dobutamine and dopamine infusions were continued postoperatively to maintain blood pressures until the sheep recovered.
Recovery
After complete weaning from the extracorporeal membrane oxygenation, IPPV of the lungs was restarted. For one minute, long inspiratory times of 15 s were set achieving an airway pressure of 40 cmH2O to allow for re-opening of the collapsed alveoli (alveolar recruitment). At this time, the arterial oxygen tension was low (7-10 kPa/50-80 mmHg, Figure 1) despite high inspired oxygen fraction (95-98%). In view of persisting hypoxaemia, four sustained lung inflations as described above were applied again, and high inspiratory and end-expiratory pressure was maintained (Figure 1). The thorax was then closed in a routine fashion and two chest evacuating tubes were placed and attached to a continuous vacuum generator. Peak airway pressure reached 40 cmH2O for 15 cmH2O of the end-expiratory pressure. After 60 min of these alveolar recruitment manoeuvres, arterial oxygen tensions progressively increased significantly (> 75 kPa/580 mmHg, Figure 1). The isoflurane was then decreased until spontaneous respiration returned. During three attempts to wean the animal off the ventilator, oxygen haemoglobin saturation decreased to <90% quickly after cessation of IPPV (Figure 1). Each time, assisted ventilation was transiently reinstated for a few minutes. During the fourth attempt, oxygen haemoglobin saturation was maintained within the physiological ranges. The animal was then extubated and the inspired air was supplemented with oxygen (6 L/min) via bilateral nasal insufflation and arterial oxygen tensions remained around 27 kPa (200 mmHg, Figure 1). After extubation, the sheep showed increased inspiratory efforts with a high respiratory rate and dramatic haemoglobin desaturation occurred whenever oxygen supplementation was interrupted.
Arterial partial pressures of oxygen over time during the surgery. The difference between the arterial oxygen partial pressure (black line) and its ideal value (alveolar oxygen partial pressure, grey line) dramatically increased after cardiopulmonary bypass (CPB, black bar) as graded by the black fading between the curves. Time periods of classical ventilation (C, between 0 and 20 cmH2O), lung insufflation (I, at 5 cmH2O), lung recruitment manoeuvres (R, few long inspirations up to 40 cmH2O and repeated inspirations between 5-15 and 30-50 cmH2O) and spontaneous ventilation (S) are presented at the top of the figure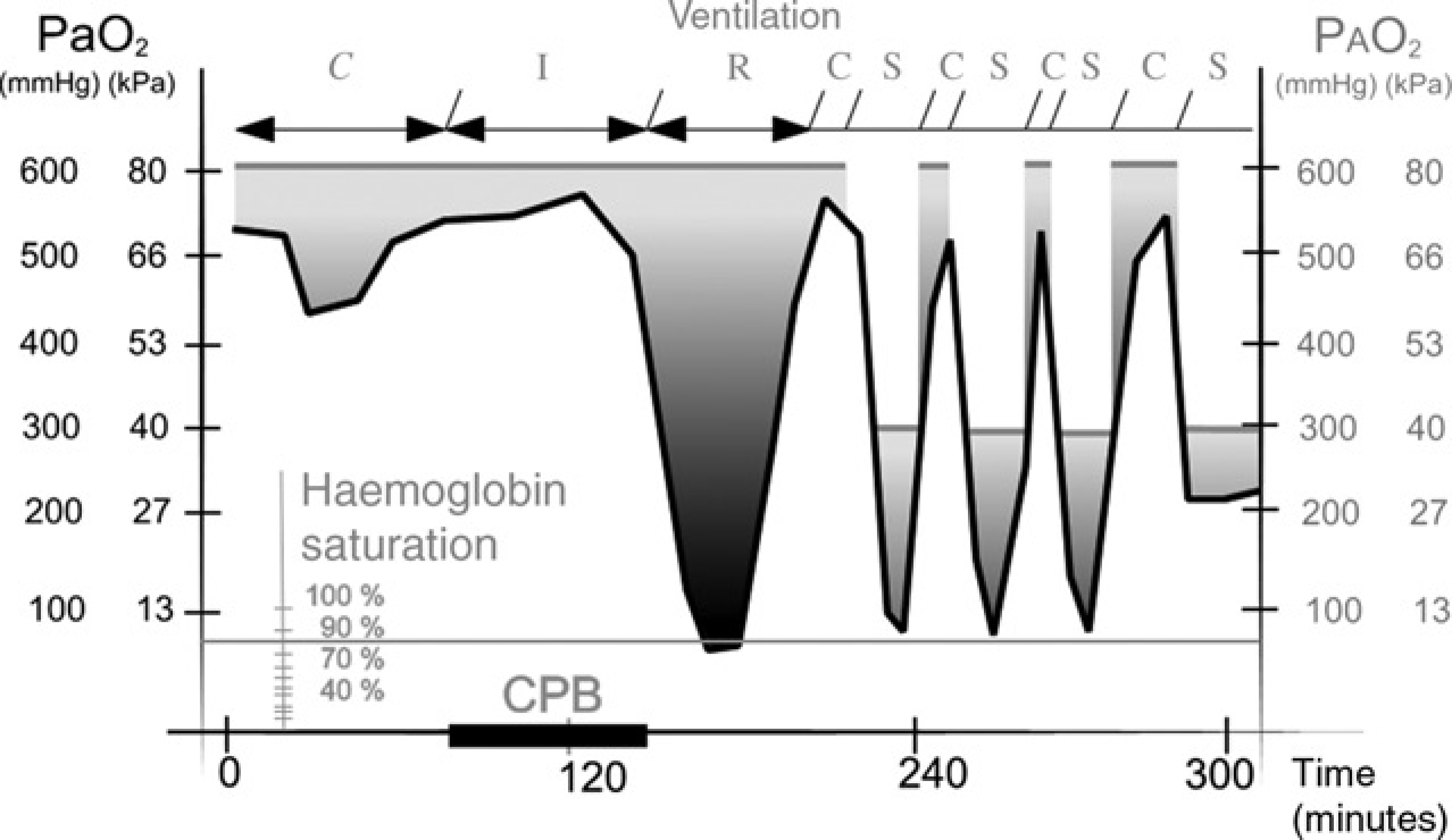
During the first two postoperative days, the sheep showed worsening signs of respiratory insufficiency with permanent requirement of oxygen supplementation, clinical and laboratory signs of renal insufficiency, haemorrhagic diarrhoea and marked signs of acute cardiac insufficiency. The sheep was euthanized eighty hours after surgery (100 mg/kg IV, Esconarkon, Streuli Pharma AG). Postmortem examination revealed multiple focal myocardial necrosis, moderate haemoglobinuric nephrosis, marked diffuse bronchopneumonia with marked intrathoracic sero-fibrinous effusion and several areas of pulmono-pericardial adherence.
Discussion
Consistent with the previous reports, cardiac arrhythmias occurred during the surgery and difficulties in restoring a normal sinus rhythm were encountered after CPB (Shofti et al. 2004). Propofol and isoflurane were used to induce and maintain general anaesthesia as this combination is reported to reduce heart sensitization to catecholamine-induced arrhythmias compared with barbiturates and halothane (Shofti et al. 2004). Additional administration of lidocaine IV was indicated not only as an analgesic adjunct but also to limit ventricular arrhythmias when directly manipulating the heart. As arrhythmias still occurred, lidocaine (0.5 mg/kg) was also applied topically onto the pericardium when attempts to restore sinus rhythm were performed. Furthermore, as severe cooling contributes to the occurrence of ventricular arrhythmias (Nesher et al. 2001), the sheep was maintained only under moderate hypothermia during the CPB (36°C). None of these interventions completely abolished the arrhythmias, and resulted in a delay in successful cardioversion.
Development of ventilation to perfusion mismatch during CPB is an expected complication of this procedure (Hachenberg et al. 1994, Verheij et al. 2005). The formation of lung atelectasis is thought to be of even more importance than the inflammatory insult in the development of post-pump pulmonary dysfunction of some patients (Magnusson et al. 1997, Weissman 1999). There is still no consensus on the strategies that aim at limiting the development of pulmonary dysfunction during the CPB in human patients. However, the use of continuous positive airway pressure by insufflation of oxygen in the lungs during the CPB seems to limit the post-CPB alveolar collapse (Loeckinger et al. 2000). This technique was used here and did not avoid the dramatic decrease of arterial oxygen tension after CPB.
Strategies to restore pulmonary function after the CPB by reducing atelectasis are better developed. High inspiratory pressures in combination with positive end-expiratory pressures following a few prolonged doubletidal volume inspirations (or ‘open-lung concept’) are described as an effective option to recruit atelectatic lung zones and keep them further opened (Tusman et al. 1999). Application of ‘open-lung concept’ ventilation has even been reported to reduce the pulmonary inflammatory response (Reis-Miranda et al. 2005). These manoeuvres may have contributed to the effective treatment of hypoxaemia in the hours following CPB.
Wound infections or delayed healing have been occasionally described as perioperative complication in sheep undergoing CPB (Kim et al. 1997). Interestingly, some authors refer to a morbidity rate following CPB in sheep related to lung dysfunction (Friedman et al. 1994, Kim et al. 1997, Stamler et al. 1998), which is significantly higher than previous reports in the literature. Complications like a ‘post-pump syndrome’ (PPS) are not described for sheep, while multiple organ failure after CPB is described in human patients (Hall et al. 1997). The origin of PPS is reported to be a strong inflammatory response to extracorporeal CPB (Friedman et al. 1994, Valenzuela-Flores et al. 2005). Complement activation may be a key trigger for the systemic inflammatory response syndrome (SIRS) (Chenoweth et al. 1981, Asimakopoulos 1999). This syndrome includes mostly lung injury (Asimakopoulos et al. 1999) and often renal failure with cardiovascular signs of shock, accompanied by multiple organ dysfunction syndrome (Valenzuela-Flores et al. 2005). Systemic corticosteroids, aprotinine, moderate hypothermia (instead of severe hypothermia) and avoidance of protamine were used in this case to reduce the systemic inflammatory response and complement activation (Carr & Silverman 1999, Mojcik & Levy 2001, Caputo et al. 2005, Rubens et al. 2005).
Appropriate systemic anticoagulation (ACT >500 s) is an absolute prerequisite prior to the initiation of CPB and was achieved here with a single bolus injection of 100 IU/kg heparin. After 210 min, ACT dropped slowly to its initial value without antagonization. Higher doses of heparin are common, but require protamine antagonization. However, the heparin-protamine complex is known to induce severe histamine release, complement activation and nitric oxide production with marked cardiovascular side-effects (Carr & Silverman 1999), including pulmonary vasoconstriction (Lowenstein & Zapol 1990). In this study, protamine was not administered and ACT returned to less than 90 s within 5 h of heparin administration. These measures were taken with the intent to reduce systemic inflammatory responses (Asimakopoulos & Gourlay 2003). Nonetheless, PPS developed in the case presented here and contributed to organ injuries.
The pulmonary function as assessed by alveolar to arterial oxygen tension differences was inadequate after CPB. Lung injury is the most common clinical expression of SIRS (Asimakopoulos et al. 1999). Some authors maintain that all patients undergoing CPB sustain some degree of pulmonary injury (Hachenberg et al. 1994, Weissman 1999). To outline the pathophysiology of this process in brief: complement activation leads to proteolytic enzyme and release of oxygen-free radicals in the pulmonary parenchyma, and various degrees of capillary leak, similar to what is reported in adult respiratory distress syndrome. The loss of hypoxic pulmonary vasoconstriction under inhalant anaesthesia and CPB worsens the amplitude of hypoxaemia. CPB-induced intestinal injury has also been reported to cause some degree of septicaemia and toxin release provoking further lung insults (Hall et al. 1997). The signs of haemorrhagic diarrhoea reported in this case report may have been due to this CPB-induced intestinal injury.
Post-CPB lung reperfusion injury can also happen when bronchial arterial blood flow is suppressed (Hall et al. 1997). During the period of low arterial pressure in the case presented here, some degree of reperfusion injury may have further worsened the insult to the lung parenchyma. The perioperative use of intravenous lidocaine to prevent reperfusion injury and subsequent multiple organ dysfunction syndrome (Cassutto & Gfeller 2003) was not sufficient to stop the development of the systemic disease that led to death of the sheep in the case reported here.
The authors believe that the intra- and postoperative complications described in this case report are comparable to a PPS in human patients, with very similar clinical features leading to death. The use of invasive monitoring and further description of complications are needed, since the actual morbidity and mortality of sheep after CPB is higher than what the literature may reflect. These additional data may lead to a better understanding of the underlying mechanisms associated with the development of PPS after mitral valve stenting in sheep.
