Abstract
Implantable microchips provide a secure, permanent and unique identification of individual animals. When performing fetal intervention studies in experimental animal models easy and secure identification of fetuses is desirable, as having test and control groups within the same uterus reduces the total number of animals used in a study. The aims of this study were: (1) to establish a protocol to identify porcine fetuses in utero by microchip implantation and (2) to assess postnatally whether clinical or pathological reactions to the implant occurred. Two Danish Landrace/Danish Large White crossbred sows at day 100 of gestation were used. The sows were sedated with azaperone and induced with propofol intravenously. Anaesthesia was maintained with isoflurane and oxygen. Antibiotics were administered intramuscularly (i.m.) at induction and analgesia was given pre-, intra- and postoperatively. A laparotomy was performed and the uterus exteriorized. The rump of the first fetus was recognized through the uterine wall and the thigh muscle of the fetus was fixed between the thumb and the forefinger. The microchip was then implanted into the fetus at an angle of 45° i.m. in the lateral hindleg using an insertion device with a 12G needle. The same procedure was done in every fetus. The uterus was returned to the abdomen and the abdominal wall closed. The sows gave birth to 24 liveborn piglets and one stillborn. None of the liveborn piglets were limping at the time of birth and no visible cutaneous or palpable reactions on the hindlegs were observed. Following euthanasia, the microchip was easily localized and no macroscopic reactions at the implantation site were seen. None of the piglets had more than one microchip implanted. Histology showed a chronic mild foreign body granulomatous inflammatory response with peripheral eosinophils surrounding the microchip. No inflammation was evident in the adjacent muscles. It is concluded that transuterine identification of piglets two weeks before delivery is feasible using a microchip implant as an effective, easy and reliable method for identification of individuals after birth.
When performing fetal intervention studies in experimental animal models, easy and secure identification of the fetuses is a must. This is especially called for when it is necessary to have test groups and controls within the same uterus. Conventional methods of identifying animals are not an option when working with fetuses, and other methods must be applied. Efforts to mark in utero injected fetuses for identification after birth have been conducted, but techniques used were not permanent or posed high risk to the fetus (Rubin et al. 2001). Implantable microchips were introduced in the late 1980s and have since then been routinely used in companion animals, production animals, laboratory animals, etc. for secure, permanent and unique identification of individual animals (Rao & Edmonson 1990, Ball et al. 1991, Murasugi et al. 2003, Le Calvez et al. 2006). The system is based on the radiofrequency identification technology and does not use batteries. Instead, it is energized by the electromagnetic field produced by the scanner. The inert microchip is easy to administer and the pre-programmed 10–15 digit code number is rapidly and error-free read with a scanner. The microchip is hermetically encapsulated within a smooth and strong biocompatible glass and is small enough to fit into a 12G hypodermic needle. The capsule is about the size of a grain of rice (12 mm long and 2 mm in diameter) and a 5 mm long part of the body is covered by a porous polypropylene polymer sheath as an antimigration measure (Rao & Edmondson 1990, Ball et al. 1991, Elcock et al. 2001, Le Calvez et al. 2006). This polypropylene cap causes development of a thin coherent membrane consisting of fibrocytes and collagen fibres thereby fixing the microchip (Rao & Edmondson 1990). Compared with other conventional means, such as ear notches, tags, toe-clips or tattoos which can be lost, may be painful or become difficult to see, the implantable microchip device is recommended as a fast and secure way of identifying animals (Le Calvez et al. 2006).
The aim of the present study was two-fold: (1) to establish a protocol to identify porcine fetuses in utero by microchip implantation and (2) to assess postnatally whether clinical or pathological reactions to the implant occurred.
Materials and methods
This study was performed on sows in which pig fetuses were treated with lipopolysaccharide at gestational day 100 as part of an atherosclerosis study. Fetal intervention was conducted in two pregnant, specific-pathogen free (SPF), Danish Landrace/Danish Large White crossbred sows (A and B) (Gundsoegaard, Gundsoemagle, Denmark) with an average weight of 250 kg. The sows were purchased from an SPF pig farm, health monitored according to the standards of SPF-Denmark (
The surgery area was prepared according to standard operating procedures for preparation of a surgical site (Division of Laboratory Animal Science and Welfare, Faculty of Life Sciences [LIFE], University of Copenhagen [KU]). A total of 100 mL local anaesthesia (lidocaine with noradrenaline 20 mg/mL, Pharmacy Services, LIFE, KU, Denmark) was given at the site of incision. A dorsoventral skin incision (25 cm) was made in the left lateral flank and the abdomen was opened by transecting the muscle layers. The uterine horns were palpated and the tip of one uterine horn was identified. A part of the uterus containing a maximum of two fetuses was exteriorized and covered with moistened sponges. The rump of a fetus was recognized through the uterine wall and the thigh muscle of the fetus fixed between the thumb and the forefinger. At the same time, another surgeon implanted the microchip (Pet ID microchip, E-vet, Denmark) in the fetus at an angle of 45° i.m. in the lateral hindleg (thigh muscle) using an insertion device with a 12G needle (Pet ID chip pistol, E-vet). An unused sterilized needle was used for each injection and the procedure repeated in every fetus by exteriorizing a new part of the uterus. Reimplantation was avoided by starting at the tip of the uterus horn on one side and ending on the other side. Monopolar electrocoagulation was used on the uterus at the site of injection if haemorrhage occurred.
The uterus was returned to the abdominal cavity, and the abdominal wall was closed with a simple continuous suture pattern in two layers (Dexon 0, absorbable). The skin was closed with a continuous interlocking vertical mattress suture (Polysorb 2, absorbable). The sows were allowed to recover and were closely observed throughout the rest of the gestation period. Under a thin bandage, a fentanyl patch (DurogesicTM, 100 μg/h, Janssen-Cilag A/S, Denmark) was attached proximal on the front leg for three days. In addition, the sows were treated with 1 mL progesterone (Progesterone 25 mg/mL, Pharmacy Services, LIFE, KU, Denmark) i.m. daily for a total of seven days to preserve the pregnancy.
Sow A delivered at term, whereas sow B on day 116 was treated with 0.175 mg cloprostenol i.m. (Estrumat®vet 0.025%, Schering-Ploug Animal Health) to induce delivery. Sow A delivered 12 piglets and sow B delivered 13 piglets. All but one piglet were liveborn. The piglets were assessed clinically (inspection and palpation) with respect to swelling, pain and evident reactions at the site of microchip implantation.
The presence and function of the microchip were checked in every piglet using an electronic animal identification system (AVID Mini TracKer II, E-vet).
At the age of five to eight days, the piglets were anaesthetized as a part of the other study. The piglets were sedated with 0.03 mL/kg/body weight of ‘swine mixture’ (125 mg tiletamine and 125 mg zolazepam (Zoletil®50, Chemvet, Denmark) + 125 mg xylazine (Narcoxyl®vet 20 mg/mL, Intervet, Denmark) + 125 mg ketamine (Ketaminol®vet 100 mg/mL, Intervet, Denmark) + 25 mg butorphanol (Torbugesic vet 10 mg/mL, Scanvet, Denmark) and then mask induced with 0.5–1.5% isoflurane and oxygen. A BD Neoflon (24G) (Becton Dickinson, Sweden) was placed in an ear vein and 2 mg/kg/body weight propofol was given to effect. Anaesthesia was maintained with 1.5–2.5% isoflurane and oxygen. At the end of the procedure, the anaesthetized piglets were euthanized by exsanguination.
Prior to tissue sampling, the hindlegs of the euthanized piglets were placed in plastic bags still covered by their skin for 48 h at 4°C. This was done in order to minimize artefacts from asynchronous muscle contractions during rigor mortis. As a result of putrefaction four piglets, which had been killed by the sow laying on them, were excluded from the study. In addition, the microchip could not be identified in one piglet. X-ray and transillumination confirmed that no microchip had been implanted therefore it was also excluded. Thus, a total of 20 piglets counting the stillborn (n = 20) were included in the histological evaluation.
A macroscopic evaluation of the tissue surrounding the implant was performed. For histological evaluation, an approximately 2 × 2 × 2 cm cube of muscular tissue containing the microchip in the centre was removed. The microchip had been implanted for a maximum of 23 days. The tissue samples were fixed for 48 h in 10% neutral buffered formalin. The microchip was removed by carefully teasing the tissue capsule at one end after fixation. The tissue without the device was then processed conventionally, embedded in paraffin and sectioned at 2–4 μm thickness. For comparison, control samples were taken from the contralateral femoral muscle. Sections were stained with haematoxylin and eosin (H&E) for an overall evaluation by light microscopy. In addition, selected sections were stained with special histochemical stains to evaluate fibrosis (Picrosirius Red for collagen [Pearse 1985]) and the Gordon and Sweets method for reticular fibres without gold reduction (Bradbury & Rae 1996). Lunas's method was used for evaluating erythrocytes and eosinophil granules (Luna 1968). A few sections were also stained to verify the presence of either DNA (Feulgen's nuclear reaction to DNA [Bancroft & Cook 1996]) or fibrin (Mallory's phosphotungstic acid-haematoxylin, PTAH [Luna 1968]).
During the study, the sows and their piglets were housed separately in pens on solid concrete floors with straw bedding and wood shavings. They were fed with a commercially pelleted dry feed (Sows feed No 3, NAG AmbA, Denmark) twice daily and water was given ad libitum. They were observed at least twice daily. The pigs used in this study were treated in accordance with the Animal Experimentation Act of Denmark, which is in accordance with the Council of Europe Convention ETS 123. The study was licensed by the National Animal Experimentation Board according to the law.
Results
Clinical findings
Besides one stillborn, all piglets were healthy and developed normally. Unfortunately, some piglets died shortly after parturition due to trauma inflicted by the sow. One piglet had no detectable microchip, whereas the rest of the piglets each had one detectable microchip. None of the liveborn piglets were limping at time of birth. No visible cutaneous reactions on the hindlegs could be seen nor could any asymmetries or palpable difference between the left and right thighs be found.
Gross pathology
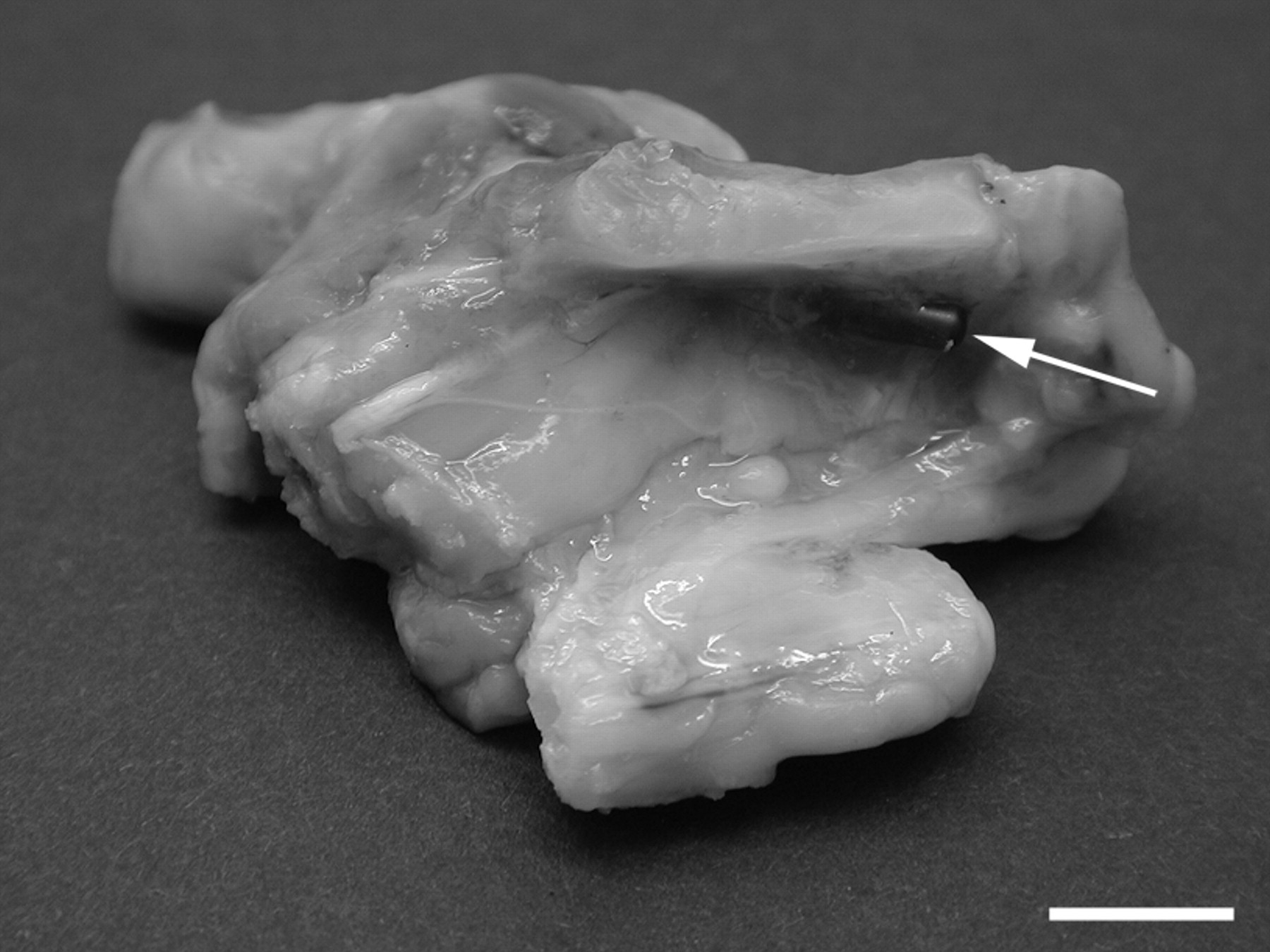
The microchip was easily found in the thigh muscles and no gross tissue reactions at the implantation site were observed. In three piglets, the microchip was situated close to and parallel with the femur or just underneath the ileum of the pelvis (Figure 1). In all but one piglet, the microchip was positioned in the connective tissue between muscle fibres (i.e. in the epi- or the perimysium). Only one microchip was found to be placed inside a muscle fibre.
Histopathology
Evident implantation sites were characterized by a focal granulomatous inflammation intermixed with moderate numbers of eosinophils (Figure 2). Foreign-body-type multinucleated giant cells (FBGCs) containing numerous haphazardly arranged nuclei throughout the cytoplasm were also present (Figure 3). The degree of collagen deposition surrounding the implant varied from minimal to mild and consisted mainly of mature collagen fibres, but thin reticulin fibres were also present. The reticulin fibres were mainly located as a thin rim close to the implantation site, whereas collagen fibres were situated more peripherally. No inflammatory cells were found in the adjacent muscles and no neoplastic changes were observed.
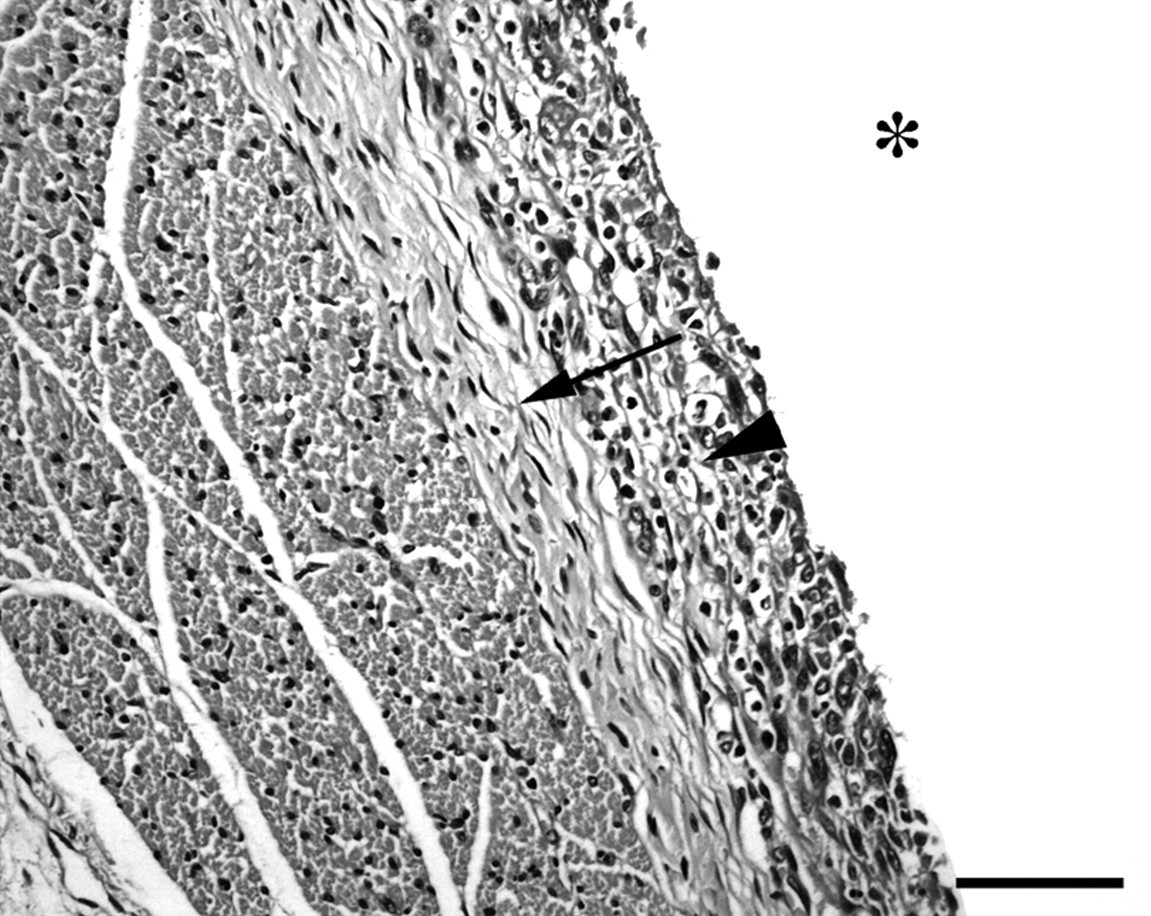
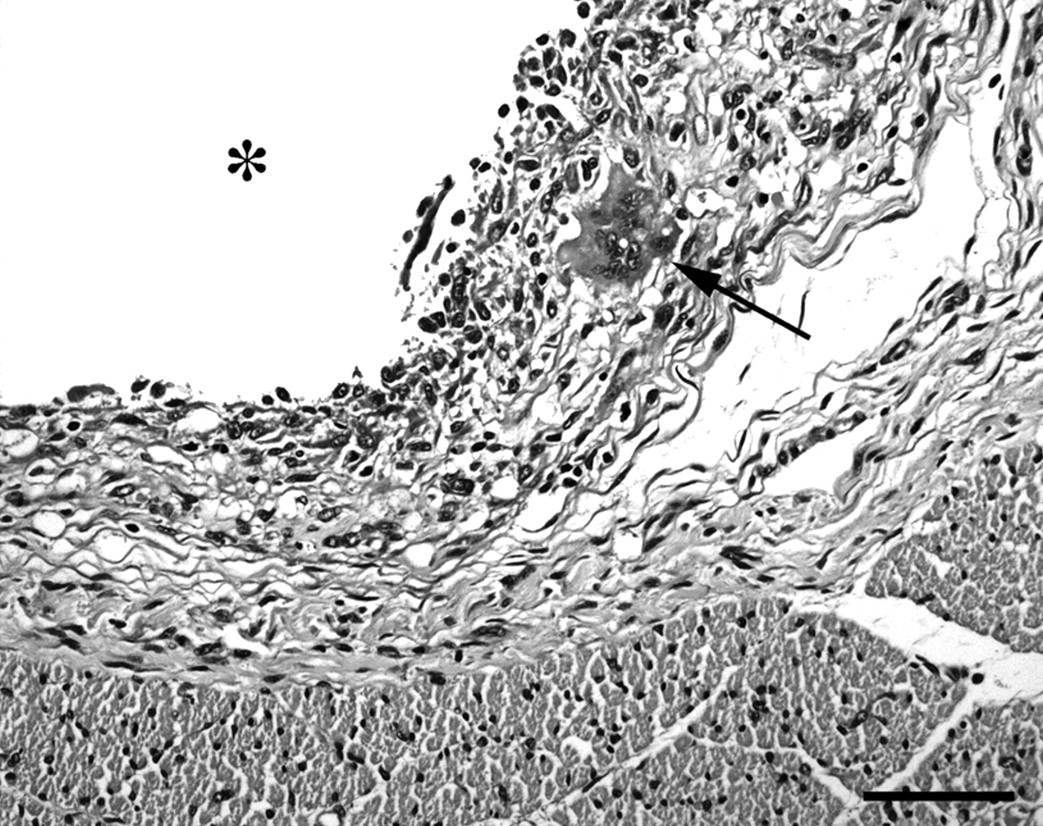
Luna-stained sections confirmed the presence of a mild to moderate number of eosinophils. These were mainly located in connective tissue around the implant. In two sections, a bit of homogeneous material was present on the inner side of the implantation site. One of these sections was positive for Feulgen's nuclear stain and the other for PTAH confirming that the material was DNA and fibrin, respectively.
Neither gross nor histological lesions were observed in the control muscle samples.
Discussion
Fetal intervention and surgery have been conducted in different animal models (Nelson et al. 1990, Swindle et al. 1996, Sangild et al. 1999, Dewan et al. 2000, Schmidt et al. 2004). In some of these studies, it is beneficial from a reduction point of view to have both test groups and controls within the same uterus in order to minimize the variability between sows. The transuterine identification method makes it possible to identify individual fetuses that have been treated through the uterus wall (e.g. injecting different test substances into the fetus). In this way, it is possible to reduce the total number of animals used by having more than one group within the same uterus. Thus, such identification, which to our knowledge has not been tried or described previously, fulfils the concept of the 3Rs.
As no abnormalities were noted on the clinical examination, the microchip implantation was considered to have exerted no obvious pain or discomfort in the neonatal piglets. Insertion of the microchip with the 12G injection needle caused a wound of approximately 1 mm in diameter and slight local haemorrhage was seen at the injection site on the uterus. However, no gross reaction was present either on the skin of the neonatal piglet or in the muscle surrounding the implant.
Despite clear advantages over other identification techniques some adverse reactions to microchips have been reported. These include migration, infection, neoplastic growth reaction, failure to work or loss from the body (Rao & Edmonson 1990, Burk & Eich 1995, Swift 2000, Elcock et al. 2001, Swift 2002, Le Calvez et al. 2006, Vascellari et al. 2006). To minimize the risk of adverse reactions in the fetus, it is important to follow the manufacturers' instructions and to fix the animal so the thigh muscle is accessible without harming the fetus. A new sharp sterile needle should be used for every fetus, both to minimize the degree of trauma and to reduce the risk of introducing bacteria. There is a high risk of bacterial infection (e.g. chorioamnionitis) and abortion when conducting fetal intervention studies (Rubin et al. 2001). Hence, the use of aseptic procedures cannot be over-emphasized because of the susceptibility of amniotic fluid and fetal tissues to infection. In our study, the two sows did not abort and no signs of infection were observed. As fetuses are predisposed to infection this type of intervention warrants prophylactic antibiotic administration even though a proper aseptic technique is sustained. Therefore, antibiotic therapy was given prior to surgery to reduce the risk of infection in the present study.
One piglet had no detectable microchip. This microchip had probably not been implanted correctly and had perhaps been injected into the lumen of the uterus instead of the thigh muscle of the fetus. This technical error is a risk when implanting the microchip transuterine. Consequently, it is important to fix the thigh of the piglet close to the uterine wall to avoid this error. In our study, some microchips were found located along the femur and just beneath the pelvis. Whether this was a result of migration or due to implanting too close to these bony structures is unknown. However, in both circumstances the microchip was found to be encased with connective tissue and no reactions were seen on the bones.
One of the potential disadvantages of the microchip implantation is the possibility that foreign-body-induced tumours may develop in long-term studies. Such tumours have been described for instance in laboratory rats and mice (Rao & Edmondson 1990, Elcock et al. 2001, Le Calvez et al. 2006). Previous studies indicate that mice, rats and to some extent dogs are more susceptible to foreign body tumourigenesis than guineapigs, chickens and hamsters (Rao & Edmondson 1990). This negative long-term possible outcome was not included in the scope of this study. However, such a possibility should be considered when choosing this type of identification system especially for long-term studies.
Implanted foreign biomaterial, such as a microchip, triggers acute and chronic inflammatory responses (Anderson 1988, Rao & Edmondson 1990, Ball et al. 1991, Tang et al. 1998, Murasugi et al. 2003). Even though the microchip had only been implanted for a maximum of 23 days including the fetal period, a chronic mild foreign body reaction enclosing the microchip was evident. This inflammatory response toward the microchip was not unexpected, as pig fetuses are known to have a relative mature and functional immune system already at midgestation, and this type of response is characteristic for foreign bodies (Anderson 1988, Tlaskalova-Hogenova et al. 1994, Trebichavsky et al. 1996, Rothkötter et al. 2002).
Macrophages and FBGCs were present at the surface of the implant while eosinophils mainly were found in the subjacent connective tissue. These inflammatory cells are often present in foreign body reactions and are a part of the normal inflammatory response following biomaterial implant (Ball et al. 1991, Tang et al. 1998, McNally & Anderson 2003, Murasugi et al. 2003, Gonlugur & Gonlugur 2006). Foreign body granulomatous inflammatory responses are maintained in a dynamic state. These are manifested by continuous changes in the cell composition throughout the phases of evolution and involution of the inflammatory response (Boros 1978, Anderson 1988). We only evaluated the response at one time point and found that the state of the granulomatous response was very similar in all piglets (i.e. mature mild granulomatous inflammation with a clear fibrous encapsulation). Further studies are needed to evaluate the response at different time points in long-term experiments.
In conclusion, transuterine identification of piglets two weeks before delivery is feasible using a microchip implant. The identification of the individual animal is effective, easy and reliable. However, proper fixations of the thighs of the piglets are of importance for a successful implantation of the microchip. The implanted microchip in this study produced no adverse clinical or gross pathological signs. As in other animals a mild granulomatous inflammatory response was evident around the microchip.
Footnotes
Acknowledgements
We thank Petru Liuba and Erkki Pesonen for allowing us to include their pigs in this study. Furthermore, Laboratory of Pathology and Veterinary Reproduction and Obstetrics, Faculty of Life Sciences for technical and laboratory assistance, and Jens Flescher and Pia Adolfsen for their excellent care of the animals, as well as assistance with surgery and postoperative treatment.
