Abstract
Summary
Animal models are necessary to evaluate new options for the treatment of fractures in osteoporotic bone. They permit both the biological response of a living system and the influence of the pathological processes to be taken into account. A sheep model for osteoporosis was established by combining oestrogen deficiency, calcium and vitamin D-deficient diet with steroid medication. Bone mineral density (BMD) was reduced by >30% after 12 weeks of combined treatment. Osteoporosis similar to the human situation with corresponding changes in the micro-architecture and mechanical properties of bone was observed. This publication focuses on the impressive results obtained with the model and contrasts them with considerations of animal welfare. Considerable side-effects associated with steroid medication became manifest. Animals in the treatment groups showed signs of infection of various degrees due to the immunosuppressive effect of the medication. The infections were mostly caused by Corynebacterium pseudotuberculosis. Antibody testing revealed a 100% prevalence of infection in this breed of sheep. A modification of the steroid treatment, i.e. less-frequent injections, reduced the incidence of side-effects. This sheep model shows a significant and reproducible reduction in cancellous BMD of >30%, including relevant changes in biomechanical properties and increased fracture risk. However, the severity of the side-effects cannot be overlooked. The model must be improved if it is to be used in the future. Options to reduce the side-effects are discussed.
Osteoporosis has become a major health problem worldwide (Consensus Development Conference 1993). Increased life expectancy and the high number of elderly people in western populations aggravate the situation. The demographic changes that are being predicted will increase the total number of fractures associated with osteoporosis, and hence the cost to society. Fractures commonly associated with osteoporosis are located at the proximal femur, lumbar vertebrae and distal radius. These injuries lead to prolonged hospitalization, an increased dependence on home care and higher mortality rates. Twenty percent of the osteoporotic patients die within one year of fracture because of long-term hospitalization (Cummings et al. 1989, Riggs & Melton 1995, Boyle et al. 2001). The treatment of fractures associated with osteoporosis is still not solved satisfactorily. The application of conventional implants for fracture fixation to osteoporotic bone is possible only to a limited extent and fixation failure occurs as a consequence of the weak bone structure (Barrios et al. 1993, Cornell 2003).
Major research efforts are being made around the world to improve the currently available treatment options and to develop new strategies for the surgical treatment of osteoporotic fractures. These include novel implants for osteosynthesis or arthroplasty as well as biological approaches, including biomaterials, cells and cytokines. Fracture treatment in osteoporotic bone can be regarded as a problem of stabilizing bones that have reduced bone mass. Mechanical testing using appropriate human specimens would then be sufficient to represent the osteoporotic situation and to allow investigation of new concepts for fixation and improved primary stability. However, this does not take into account the changes in the bone physiology related to osteoporosis, e.g. the changes in the remodelling cycle or the decreased sensitivity of osteoblasts to cyclic strains (Sterck et al. 1998). Although cell cultures and other in vitro assays may be appropriate to investigate the reaction of normal and osteoporotic cells to different kinds of stimuli, neither the effects of central control of bone turnover (Haberland et al. 2001) nor the feedback mechanisms involved in physical activity can be simulated. The local and systemic processes that occur in the presence of cytokines or growth hormones cannot be reproduced by these methods. Furthermore, the anatomical locations, physical dimensions and properties of the implants, their insertion techniques and methods of fixation have all been shown to be relevant to proper fracture healing.
A large animal model is needed to study fracture fixation in osteoporotic bone in vivo. Preclinical testing of new treatments or devices is required to prove efficiency and safety. The ‘Food and Drug Administration (FDA) Guidelines for Preclinical and Clinical Evaluation of Agents Used in the Treatment or Prevention of Postmenopausal Osteoporosis’ recommend that agents should be evaluated in two different animal species including ovariectomized rats and a second non-rodent large animal model with Haversian systems and remodelling patterns similar to the human situation (FDA 1994). Although the FDA guidelines refer primarily to pharmaceutical agents, preclinical evaluation is also required for new surgical treatment options, and therefore, there is a need for small as well as large animal models. The ideal animal for osteoporosis research should be large enough to permit serial sampling and experimental procedures to investigate fracture repair, have a mineral metabolism and bone structure similar to humans, be readily adaptable to the laboratory environment and be available at a reasonable cost.
The natural occurrence of osteoporosis is rare in animals, contrary to the situation in humans. Several strategies for the induction of osteoporosis in animals have been under investigation in recent years (Egermann et al. 2005). Sheep seem to be a promising model for the study of fracture treatment in osteoporosis (Hornby et al. 1995, Turner et al. 1995, Chavassieux et al. 1997, Turner 2002). They are docile, easy to handle and house, ovulate spontaneously and have hormone profiles similar to women (Newman et al. 1995). The size and basic anatomy of the sheep skeleton are generally comparable with the human situation (Wilke et al. 1997). The histological appearance and remodelling activity of sheep bone are similar to human bone (Newman et al. 1995). Ovariectomy alone induces a significant decrease in bone density in sheep, but the decrease is much lower in comparison with that in human osteoporosis (Turner et al. 1995). These observations led to the development of a combined induction protocol using known risk factors for osteoporosis (oestrogen deficiency, calcium- and phosphate-restricted diet, movement restriction and high-dose steroid treatment). Five consecutive studies were performed to evaluate sheep as a model for human osteoporosis using this combination approach for the induction. Three different induction regimens were first compared in a pilot study (Lill et al. 2000, 2002a) and the most efficient (combined) protocol was analysed in more detail in an induction study (Lill et al. 2002b). The osteoporotic sheep from this study were then used to investigate fracture healing in osteoporosis (Lill et al. 2003). As a next step, the long-term course and possible recovery of bone mass following the induction phase was determined (Goldhahn et al. 2005). Finally, efforts were made to improve the induction regimen in order to minimize the side-effects observed during earlier studies.
Corticosteroids are powerful drugs that can quickly reduce swelling and inflammation. Prednisone and similar steroid medications are widely used to control symptoms of asthma, inflammatory bowel disease, arthritis, autoimmune disorders or skin diseases. The potent effect of corticosteroids can result in serious side-effects (Dequeker 1999). The classic Cushing-like syndrome (oedema, puffy face, redistribution of body fat) is observed when excess corticosteroids are present. The common side-effects of long-term steroid therapy are osteoporosis, impaired wound healing and increased risk of infection (Walsh et al. 2002). One of the major side-effects of long-term steroid therapy, namely, osteoporosis was induced in the above-mentioned studies to reduce bone mass experimentally. The adverse effects of the long-term use of corticosteroids have been observed and are well documented (Dempster 1989, Lespessailles et al. 2000).
This publication summarizes our results and experiences with the steroid-based sheep model for osteoporosis and the impressive effect of steroid medication on bone mineral density (BMD), the mechanical properties and the histological appearance of bone. The goal is to give objective consideration to the advantages and limitations of this animal model with regard to animal welfare, side-effects, conditions of husbandry and options for improvement.
Materials and Methods
During the period 1998-2002 five studies were carried out at the AO Research Institute to establish a sheep model for osteoporosis. All studies were approved by the Animal Experimentation Commission of the Veterinary Office of the Canton of Grison, Switzerland, and strictly followed the guidelines of the Swiss Federal Veterinary Office for the use and care of laboratory animals.
Pilot study Three different induction regimens, i.e. ovariectomy + diet, ovariectomy + steroid medication and combined treatment of ovariectomy + diet + steroid medication were compared with an untreated control group (Lill et al. 2000, 2002a). Changes in BMD and micro-architecture were recorded over a period of six months for the two sheep in each group.
Induction study Since the greatest bone loss was found in animals treated with ovariectomy + diet + steroid medication, this particular induction regimen was analysed in detail over seven months by means of a treatment and a control group of 16 animals each (Lill et al. 2002b).
Long-term study The combination treatment was administered to 10 osteoporotic and eight normal sheep that were then observed for 10 months in order to evaluate the long-term variations of bone mass, in particular, a possible rebound subsequent to the induction of osteoporosis (Goldhahn et al. 2005).
Fracture healing study Osteoporotic sheep (n = 7) from study 2 were also used to investigate fracture healing in osteoporotic bone and were compared with an appropriate control group (n = 7) (Lill et al. 2003).
Improvement study The purpose of this study was to modulate the existing induction regimen to minimize the steroid-induced adverse effects and retain the osteoporotic changes, such as reduction in BMD, microstructural deterioration and mechanical weakness of cancellous bone. Four different steroid application modalities were compared in 36 animals. The details and results of the improvement study have not been presented elsewhere and are therefore included here.
Side-effects due to steroids were observed in all studies. These five studies included a significant number of animals and therefore allowed conclusions to be drawn regarding the appropriateness of steroid medication.
Animals
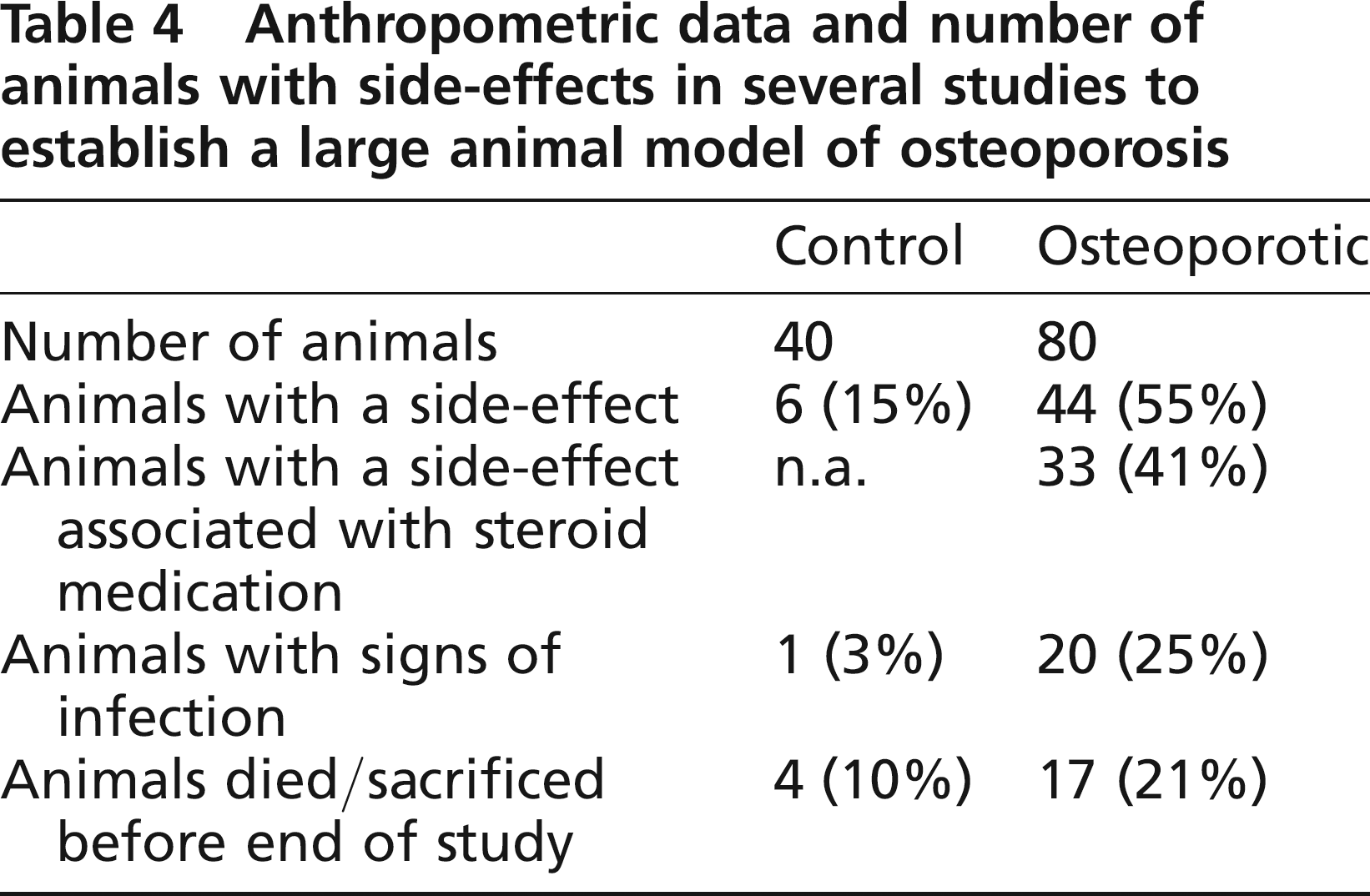
A total of 120 identically bred Swiss mountain sheep were investigated in these studies. Eighty sheep (age: 6.7 ± 0.8 years; weight: 63.8 ± 6.7 kg) received identical treatment to induce osteoporosis and 40 animals (age: 4.3 ± 1.8 years; weight: 59.1 ± 7.7 kg) served as healthy controls. Of the 80 experimental sheep, 36 animals (age: 6.2 ± 0.6 years; weight 65.4 ± 6.2 kg) were included in the improvement study. The sheep were housed indoors, in single boxes (1.8 m2) and under natural light conditions for the duration of the experiments (Davos: latitude: 46.48° N, longitude: 09.49° E). The clinical condition of the animals was recorded daily by the animal caretakers and all sheep were inspected by veterinarians at weekly intervals. Side-effects were meticulously reported and categorized as related to steroid medication (i.e. alopecia, dermal mycosis, infections, abscess, reduced food intake, weight loss >10%) or unrelated to steroid medication (i.e. aspiration during anaesthesia, haemorrhage during surgery, haematoma, implant failure).
Induction of osteoporosis
This animal model is based on three circumstances that are generally accepted to be major risk factors for osteoporosis in humans: menopause, calcium-restricted diet and drugs (corticosteroids). With the sheep in the supine position, bilateral ovariectomy was performed by means of a low median laparotomy under general anaesthesia with Isofluran™ (Halocarbon Laboratories, River Edge, NJ, USA) and intravenous Temgesic (buprenorphine HCl 0.3 mg/mL, Reckitt & Colman Pharmaceutics, Hull, UK). Care was taken to ligate the ovarian vessels twice. Postoperative pain medication was given as necessary.
The diet contained 1.5 g calcium and 100 IU vitamin D3 per day with a total food consumption of 1000 g per day per sheep (Eberle Nafag AG, Gossau, Switzerland). The animals in the control groups received standard feeding with normal calcium (5 g per day), normal vitamin D intake (1000 IU per day) and 1050 g of food per animal per day. The total amount of 1800-2000 mg methylprednisolone solution (Streuli & Co AG Pharmazeutika, Uznach, Switzerland) was given to the animals by intramuscular injection. Different muscles in the shoulder, neck and gluteal region were injected alternately. Steroid application and calcium-restricted diet was commenced two weeks after ovariectomy and was administered to the animals over a period of three months (induction phase).
The four different regimens of steroid application were compared in the improvement study. Group A received a daily injection of 20 mg methylprednisolone. In group B, 60 mg were administered every third day. Three portions of 500 mg and one portion of 300 mg were injected at three-week intervals into the animals in group C. Group D was injected with a daily dose of 70 mg methylprednisolone for one week, followed by a weekly reduction of 10 mg to a final dose of 20 mg after six weeks. Each animal received the total dose of 1800 mg methylprednisolone.
Analysis of BMD, bone structure and mechanical properties
BMD (g/cm3) was measured by peripheral quantitative computed tomography (pQCT) using a Densiscan 1000™ scanner (Scanco Medical AG, Bassersdorf, Switzerland). The mean BMD values from the distal radius and distal tibia were determined in vivo for longitudinal analysis. The BMD values in the spine were measured after necropsy. The distal radius and distal tibia were examined separately and the results were averaged. The pQCT measurements were done under general anaesthesia and the limbs fixed in the CT scanner with a custom-made fixation device to avoid motion artefacts due to movements of the sheep. A scout view was taken first to ensure identification of the identical region of interest adjacent to the joint throughout the whole experiment. The joint surface was defined as the starting point. Ten consecutive slices were obtained at 1 mm increments. A highly filtered low-energy X-ray source was used to enhance bone contrast. The effective energy of the X-ray beam was always set to 40 keV and 0.5 mA. The slice thickness was 1 mm and a matrix of 512 × 512 pixels was used resulting in a spatial resolution of 290 × 290 μm. The contour of the radius was outlined as a routine procedure on each cross-section. The 50 μ value was calculated using the inner 50% of the section to ensure that only cancellous bone was measured.
Cylindrical bone biopsies (diameter 10 mm) were taken in vivo from the iliac crest and postmortem from the iliac crest and the spine. The parameters of bone structure were obtained for these biopsies using the micro-CT 20 (Scanco Medical AG) at a spatial resolution of 28 μm. This 3D micro-CT system is based on a fan-beam and works in a multislice mode. Within the volumes of interest (4×4×4 mm), trabecular thickness (Tb.Th), trabecular number (Tb.N), trabecular separation (Tb. Sp), bone surface/bone volume (BS/BV) and bone volume/total volume (BV/TV) were determined.
To evaluate their mechanical properties the cylindrical bone specimens were placed in a materials testing machine (Instron 4302, series IX, Instron Ltd, High Wycombe, UK). The cylinders were compressed between two parallel plates in a craniocaudal direction at a rate of 1 mm/min. The structural stiffnesses (N/mm) were taken from the linear portions of the load-displacement curves.
Fracture model
A midshaft tibial osteotomy was selected as the standardized fracture model to study fracture healing in osteoporotic and non-osteoporotic sheep at six months after induction of osteoporosis. Steroid medication was stopped three months prior to osteotomy to avoid any effects on the healing process. The osteotomy was stabilized with a custom-made external fixator; the details of the model are described by Lill et al. (2003).
Results
Pilot study
The results of the three different strategies to induce osteoporosis, i.e. ovariectomy plus diet (O + D), ovariectomy plus steroid medication (O + S) and ovariectomy plus diet plus steroid (O + D + S), and those of the untreated control group are summarized in Table 1, which shows the changes in BMD during the six-month treatment period. There were no changes in the control group. The largest decrease in BMD was seen in group 3 (O + D + S) (Lill et al. 2000, 2002a).
Bone mineral density (BMD) (pilot study)
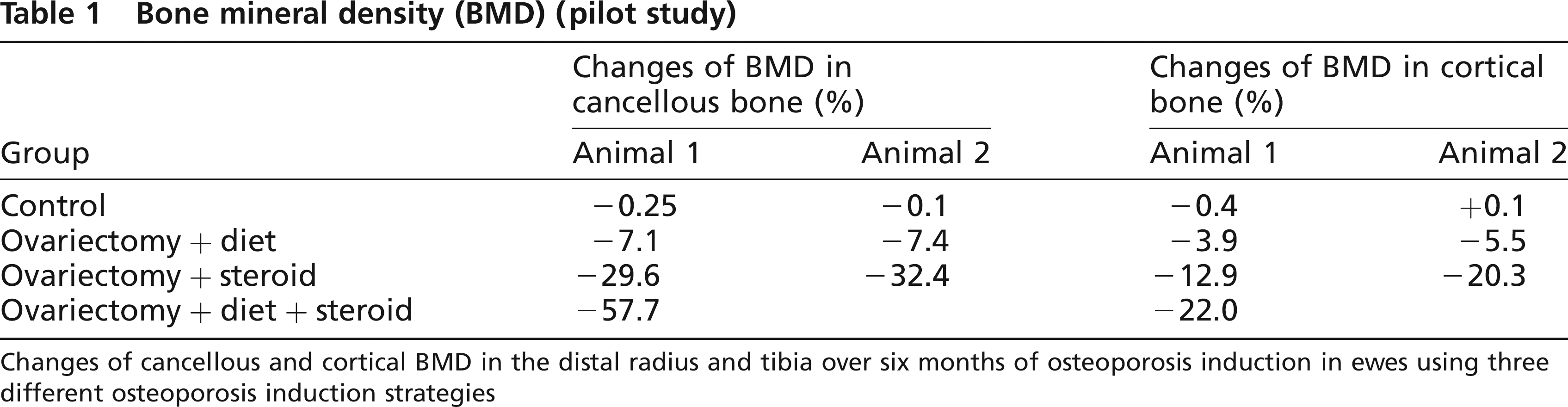
Changes of cancellous and cortical BMD in the distal radius and tibia over six months of osteoporosis induction in ewes using three different osteoporosis induction strategies
A distinct change in structural parameters was observed in the vertebral body biopsies (Tb.N -53%; Tb.Th -63%, Tb.Sp +150%). The changes were less distinct in the femoral head biopsies (Lill et al. 2000, 2002a). In compression testing of the vertebral body biopsies in group 3 (O + D + S), the stiffness values were found to be reduced by 40% and the failure loads were even reduced by 70% compared with the control group.
Induction study
The induction study on 32 adult female sheep was performed in order to establish a sheep model of osteoporosis using the steroid-based combination method of induction (Lill et al. 2002b).
After seven months of osteoporosis induction, the BMD of cancellous bone in the osteoporotic group (n = 16) was reduced by 33.2 ± 2.6% in the distal radius (Figure 1) and by 40.8 ± 3.3% in the distal tibia in comparison with the untreated animals (n = 16) (P < 0.001). A small, but non-significant decrease in BMD in cortical bone was found. The reduction in BMD was similar in the distal tibia (Table 2), but less prominent at other anatomical sites, such as the lumbar vertebral bodies (-12.7%, P < 0.01), femoral head or humerus (Lill et al. 2002b).
Bone mineral density (BMD) (induction study)
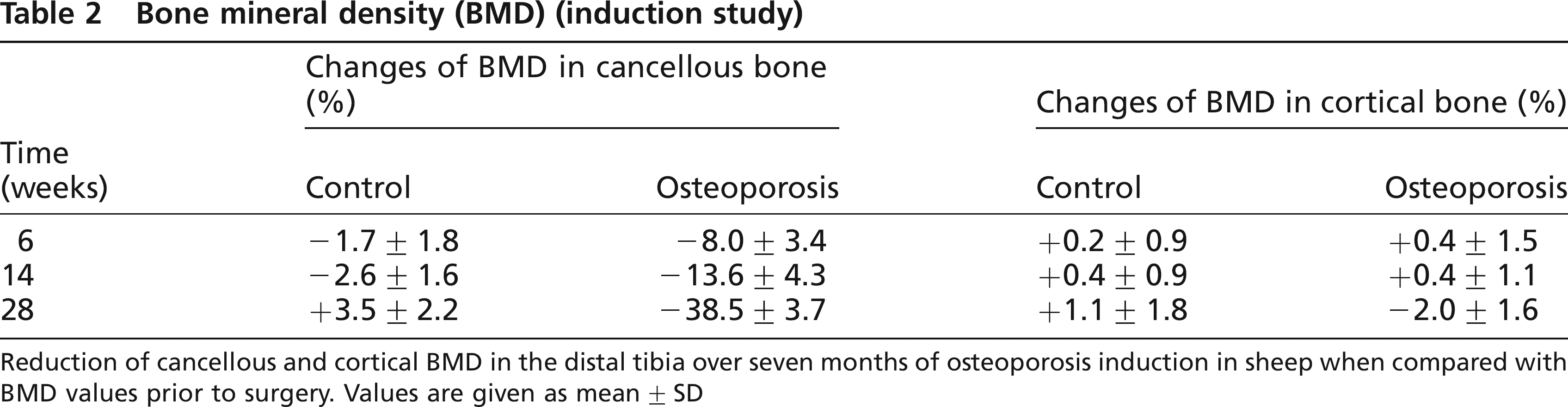
Reduction of cancellous and cortical BMD in the distal tibia over seven months of osteoporosis induction in sheep when compared with BMD values prior to surgery. Values are given as mean ± SD
Bone mineral density (BMD) of cancellous bone at the distal radius over seven months (mean ± SD). After seven months the difference between the untreated control (group 1) and the animals treated with ovariectomy, diet and steroid medication (group 2) was significant (P < 0.0001)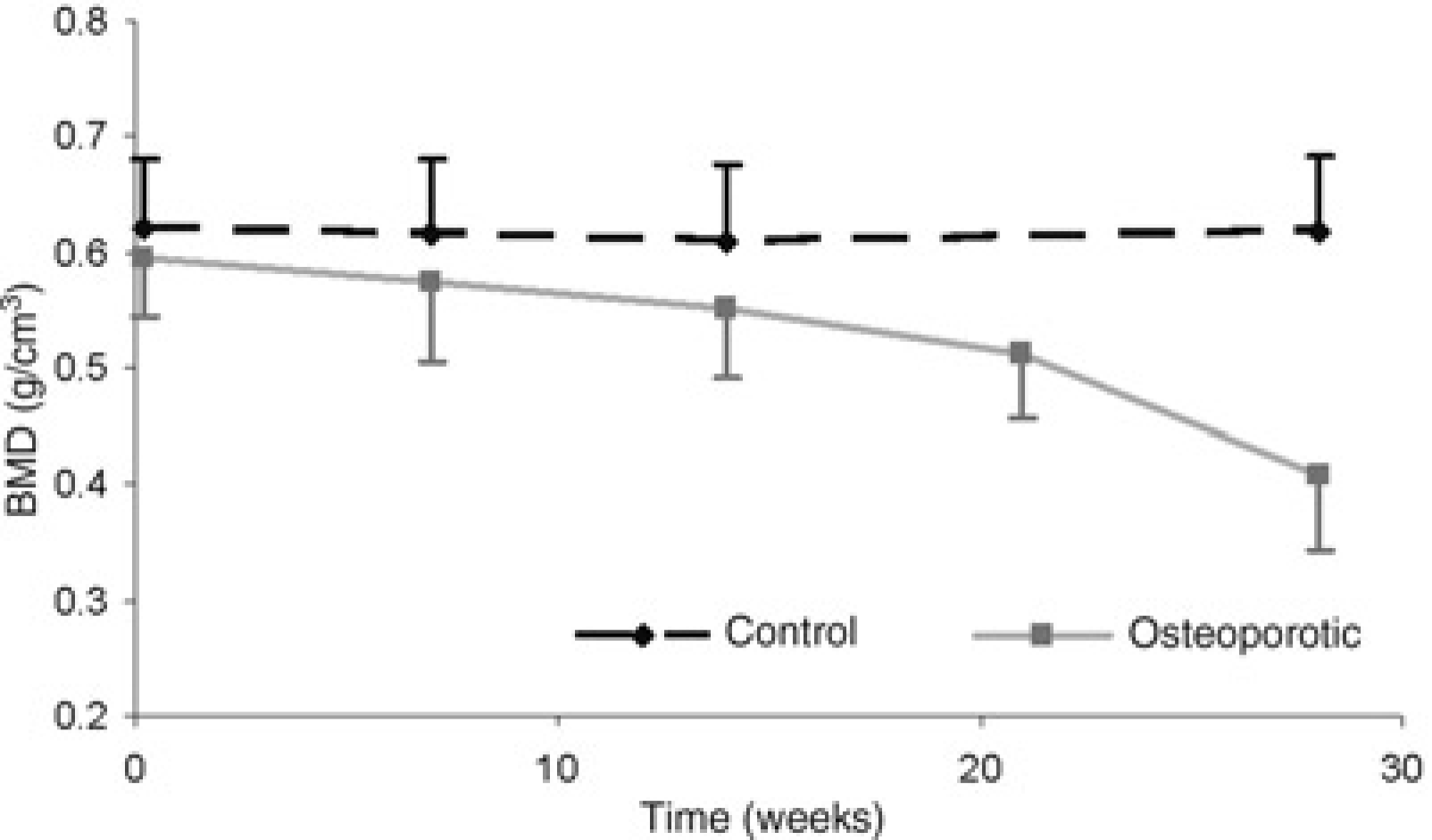
At seven months after osteoporosis induction, all structural parameters of the iliac crest had changed significantly: BV/TV (-42.2 ± 15.7%), Tb.N(-17.0 ± 13.1%), Tb.Th (-30.4 ± 15.1%) and Tb.Sp (+34.6 ± 25.7 %) (Table 3). 3D reconstruction of the micro-CT demonstrated the differences between the control group and the osteoporotic group (Figure 2).
Three-dimensional reconstructions from micro-CT measurements in a typical biopsy specimen from the iliac crest: control group (left) and osteoporotic group (right)
Micro-architecture of trabecular bone
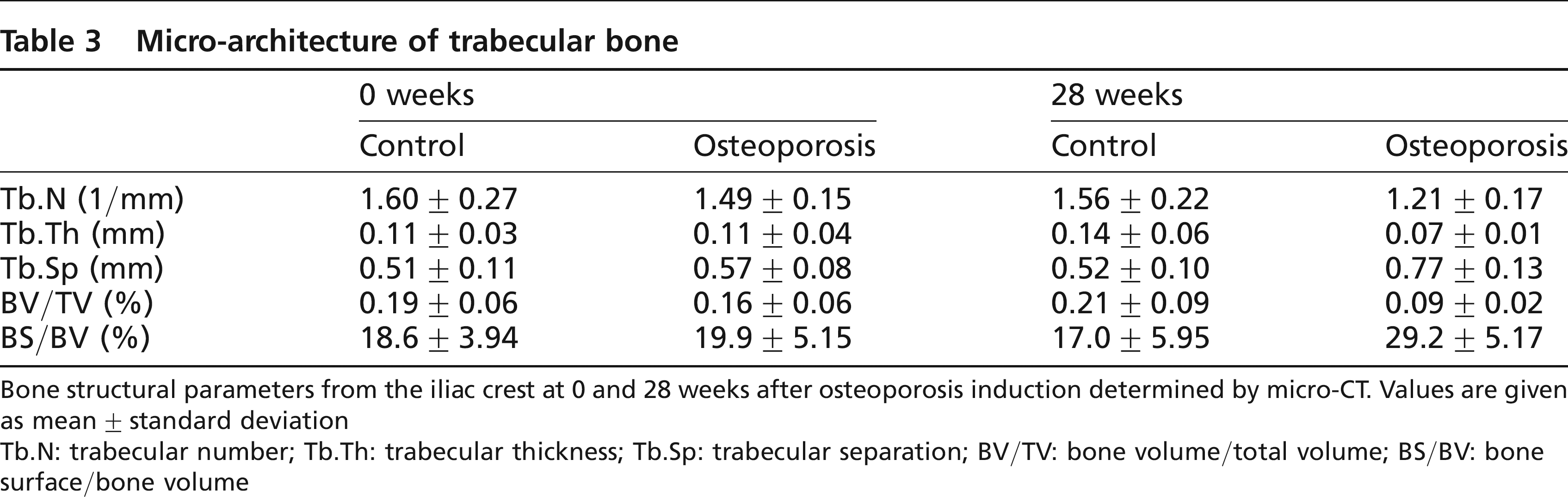
Bone structural parameters from the iliac crest at 0 and 28 weeks after osteoporosis induction determined by micro-CT. Values are given as mean ± standard deviation
Tb.N: trabecular number; Tb.Th: trabecular thickness; Tb.Sp: trabecular separation; BV/TV: bone volume/total volume; BS/BV: bone surface/bone volume
The biomechanical properties of cancellous bone are significantly different due to the induction of osteoporotic. In compression testing of vertebral body biopsies, the failure load (control: 3.2 ± 0.8 kN, osteoporosis: 1.9 ± 0.6 kN [P < 0.002]) and stiffness (control: 8.1 ± 1.0 kN/mm, osteoporosis: 5.9 ± 1.4 kN [P < 0.05]) were significantly lower in the osteoporotic group.
Long-term study
After 12 weeks, osteoporosis induction was discontinued in all animals, and the long-term course of BMD was monitored for another 28 weeks. Three osteoporotic and one healthy sheep were lost during osteoporosis induction or due to perioperative complications. The linear rate of bone loss during osteoporosis induction was 1.75% per week. Rebound of cancellous BMD started between 6 and 11 weeks after the end of steroid treatment with a linear coefficient of 0.22% per week (Figure 3) (Goldhahn et al. 2005).
The time line of the experiment, the progression of bone mineral density (BMD) at the distal radius in seven osteoporotic sheep during and after steroid application (a), and the levels of endogenously produced cortisone of seven sheep during and after osteoporosis induction and of seven healthy sheep (b)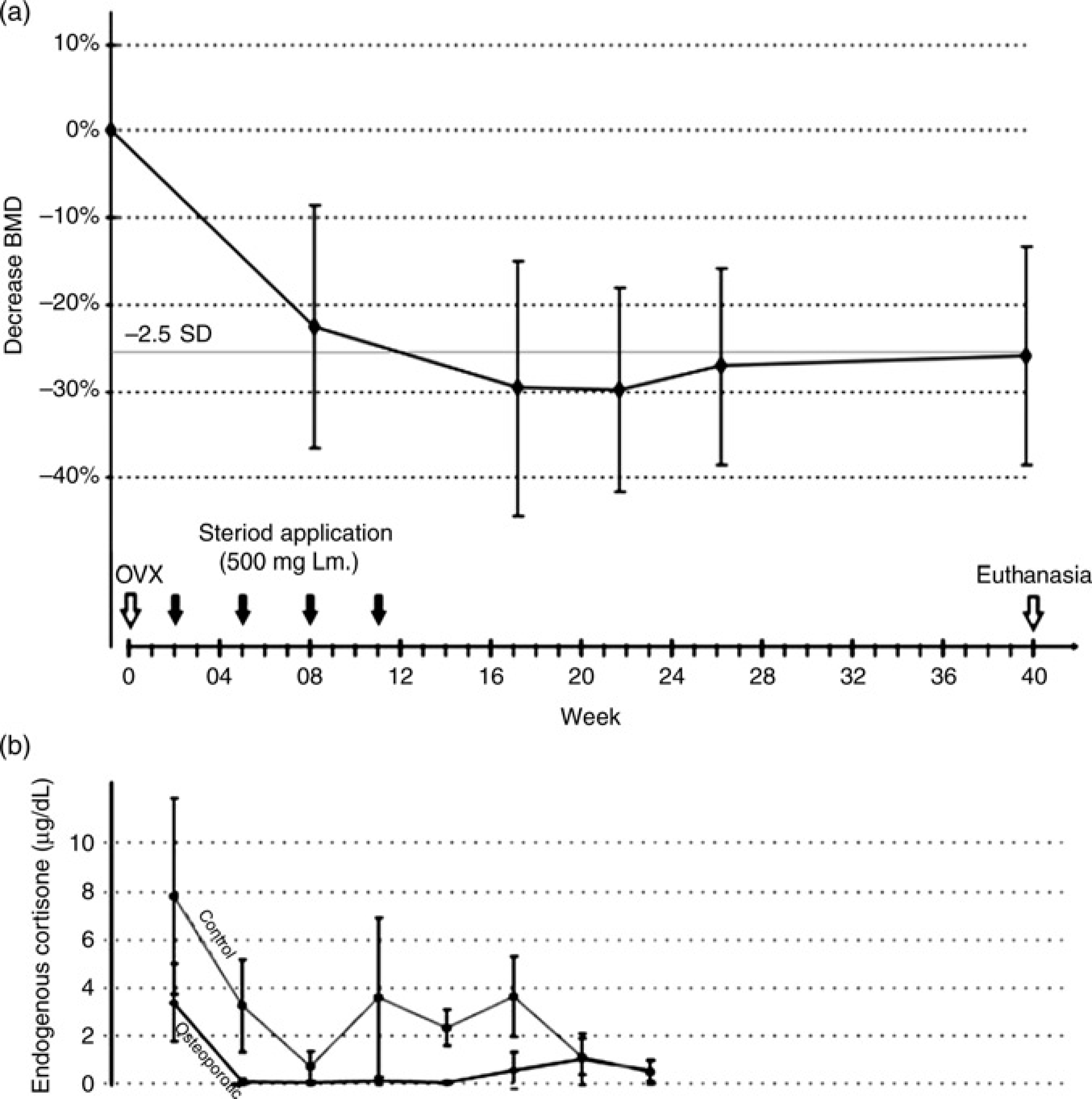
Fracture healing study
A standardized transverse midshaft tibial osteotomy stabilized with a special external fixator for eight weeks was used to investigate fracture healing in osteoporotic and normal sheep. In the control group, in vivo stiffness of the callus increased between weeks 3 and 4 after osteotomy. In the osteoporotic group, the increase in bending stiffness was delayed by about two weeks (Figure 4). Torsional testing of the tibia was performed after euthanasia and a significant difference of 33% in torsional stiffness was found between the osteotomized and the contralateral intact tibia in osteoporotic animals (P < 0.05).
In vivo bending stiffness during eight weeks of bone healing in the control group and the osteoporotic group. The stiffness is given as a dimensionless factor. Before osteotomy (b.o.) the stiffness was set at 1 (a.o. = after osteotomy)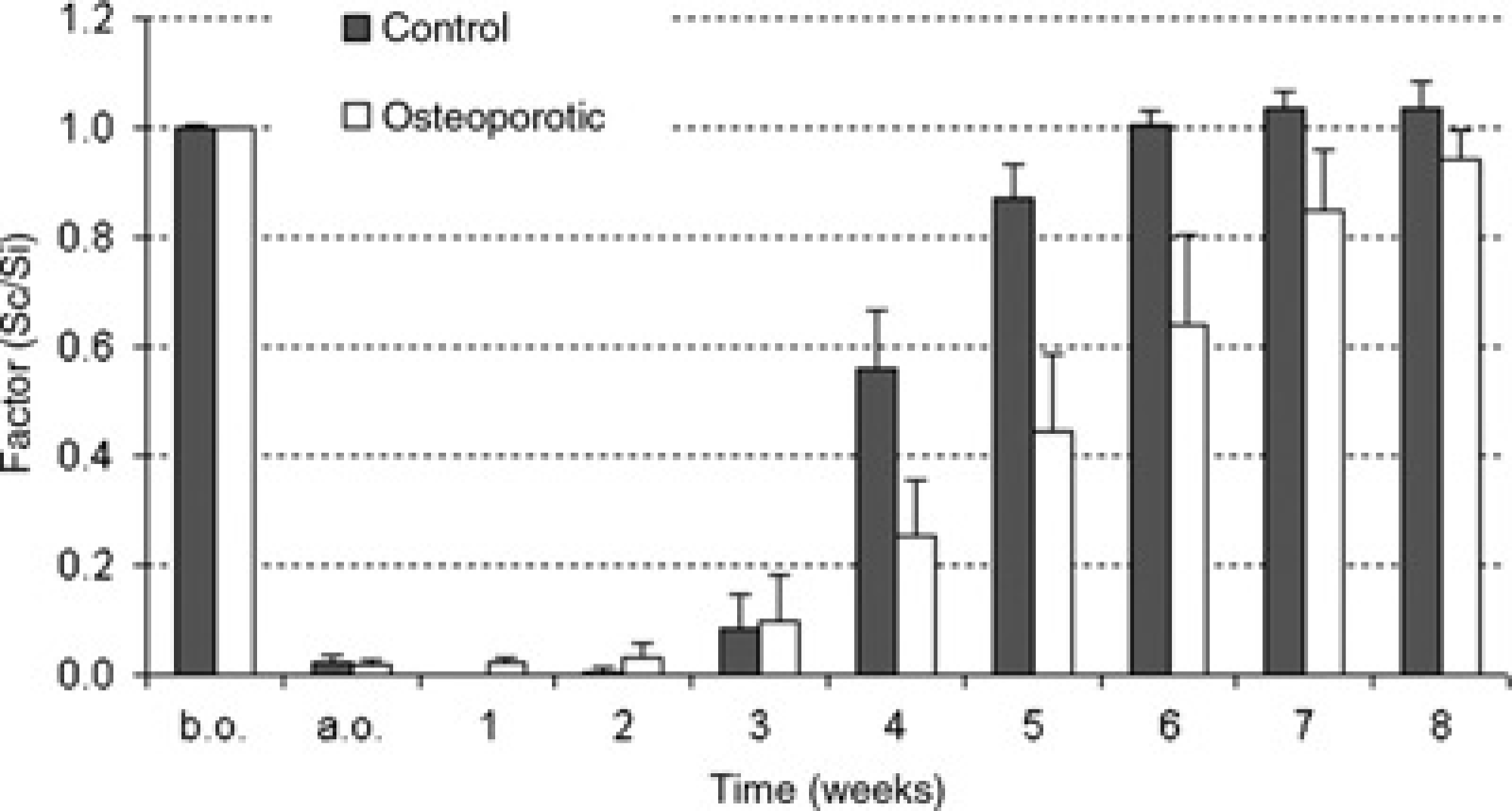
The callus density increased during healing both in the control and in the osteoporotic groups. The difference between the two groups was 16% (P = 0.015) at four weeks and 23% at eight weeks (P = 0.058). Less callus area was found in osteoporotic animals at four weeks (-29%, P = 0.023) and at eight weeks (-21 %, P = 0.052). This study demonstrated a delay of fracture healing in osteoporotic sheep tibiae with respect to callus formation, mineralization and mechanical properties (Lill et al. 2003).
Improvement study
The aim of this study was to improve the above-mentioned induction regimen with respect to bone metabolism and to reduce the adverse side-effects. All induction parameters were the same in all four groups, except the administration of steroid medication. After four months the BMD had declined by 16.7 ± 2.2% at the distal radius and by 22.5 ± 2.9% at the distal tibia. No difference in BMD or histomorphometrical parameters was detected for the four different induction regimens (Figure 5). Osteoporotic bone alterations were present in all four groups.
The decrease in cancellous bone mineral density (BMD) (mean ± SD) in the distal radius at monthly intervals. Values are given in percentages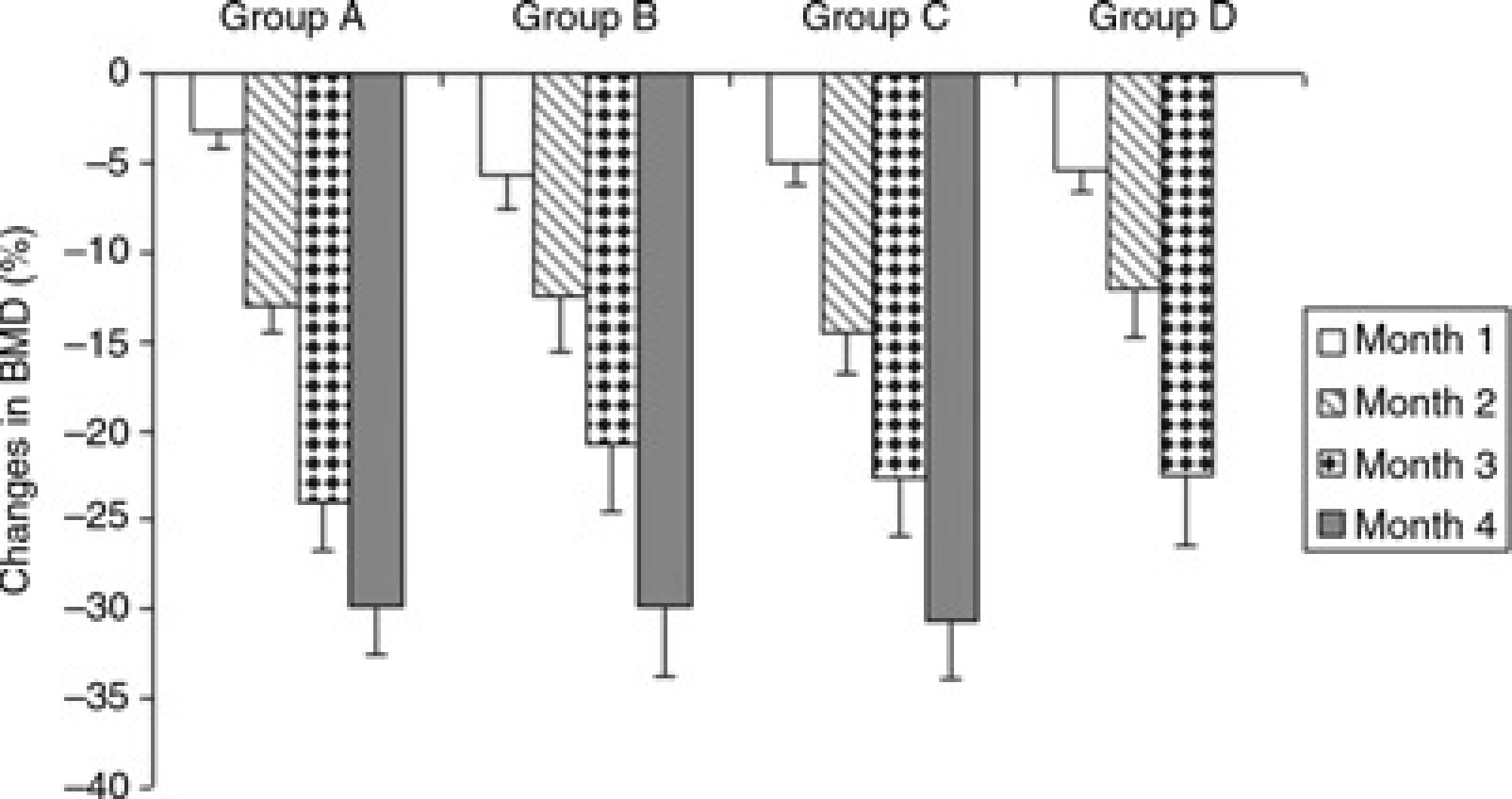
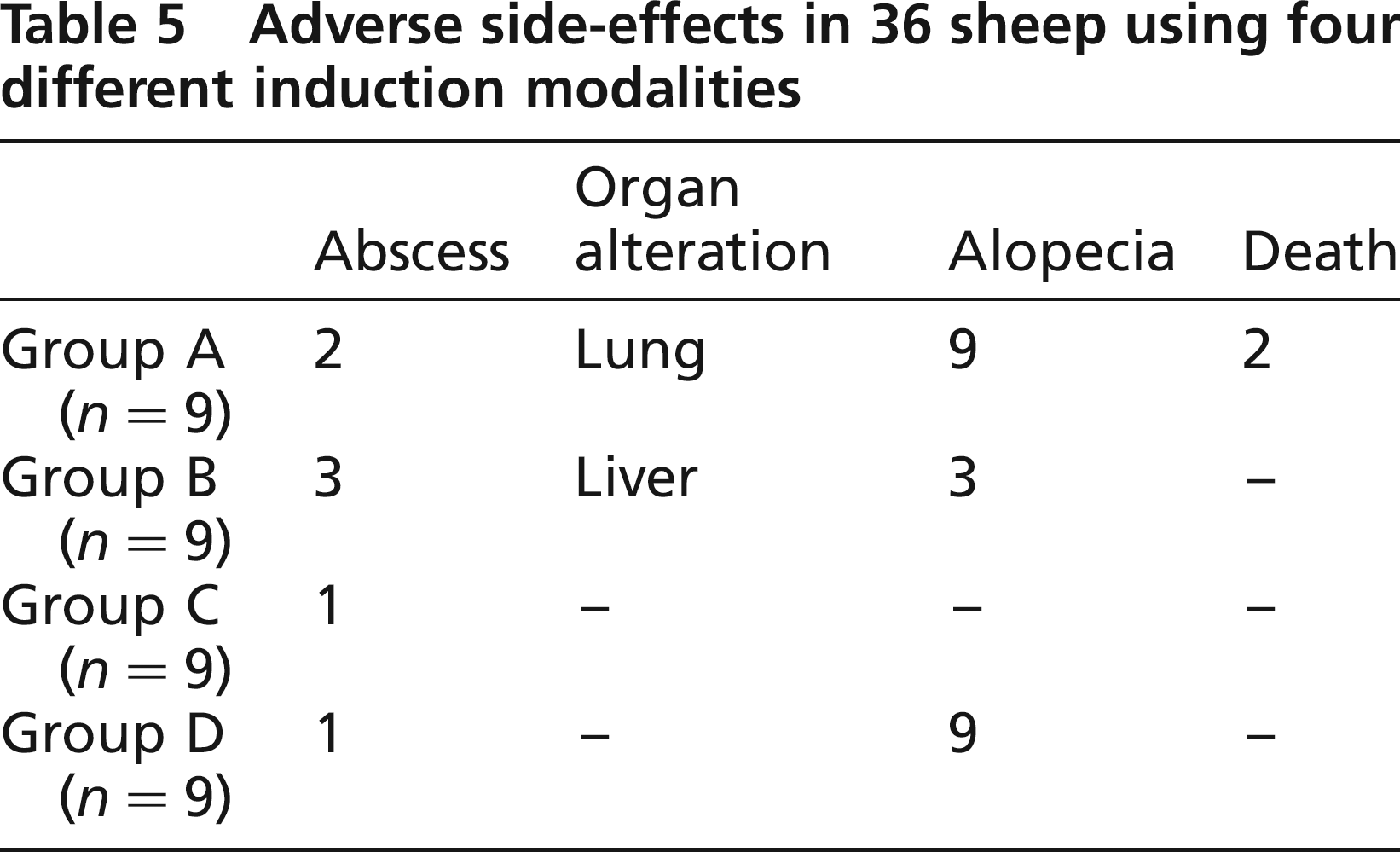
The side-effects observed in this study are listed in Tables 4 and 5. The prevalence of disseminated alopecia was 100% in groups A and D and 33% in group B. There was a total absence of alopecia and no effect on general health in group C. Organ alterations postmortem, e.g. lung and liver abscesses, were found in groups A and B, but not in other groups. There appeared to be substantial differences between the groups. Group C showed the lowest prevalence of side-effects.
Anthropometric data and number of animals with side-effects in several studies to establish a large animal model of osteoporosis
Adverse side-effects in 36 sheep using four different induction modalities
Clinical condition and adverse side-effects
The clinical condition of the sheep was meticulously documented during all studies. Adverse side-effects appeared in both groups, although the frequency was significantly higher for the treated animals (P < 0.001) (Table 4). Multilayer complications were observed in the control animals, i.e. failure of the fixation system during fracture healing, minor skin lacerations during shearing, haematoma or refusal to take food after anaesthesia. Since the osteoporotic sheep were being treated with steroid medication, the side-effects were categorized according to their association with the pharmaceutical treatment. In the osteoporotic group, 83% of side-effects, mainly infection, abscess and alopecia, were related to the steroid medication. In this group, the majority of side-effects (62%) occurred during the first 12 weeks of osteoporosis induction. The incidence of side-effects was related to the age of the animal (>7.0 years: 61% of animals with side-effects; <7.0 years: 38% of animals with side-effects), but did not correlate to body weight.
The majority of side-effects in osteoporotic sheep were classified as infections including organ alterations in the form of abscess. In 82% of these infections, Mycobacteria pseudotuberculosis was found to be the causative agent. In order to identify ways of decreasing the infection rate, serum antibodies to M. pseudotuberculosis from 20 randomly chosen healthy sheep were investigated. These sheep were not involved in the studies, but had been bred identically. Interestingly, all animals (100%) had variable titres of antibody to this bacteria indicating prior contact to this agent during their lifetimes.
Discussion
This article provides an overview of our results and experiences in establishing a large animal model for osteoporosis. We present the changes in BMD, bone microstructure and mechanical properties recorded during the induction of osteoporosis in sheep. In addition, we focus on the clinical condition of the animals and discuss ways to reduce the number and severity of side-effects.
A large animal model is very much needed to test new treatment options for osteoporosis and associated fractures with a view to meeting future medical care needs. Since osteoporosis does not occur naturally in animals, bone loss needs to be induced. Loss of oestrogen is the main cause of the rapid bone loss that women experience during the first years after menopause. Osteoporosis is defined as a decrease in BMD by >2.5 standard deviation (SD) below the mean. In female sheep, ovariectomy alone causes a reduction in BMD of 5-12% (Hornby et al. 1995, Turner et al. 1995, Geusens et al. 1996, Lill et al. 2000), which represents a reduction of 0.8 SD and is not comparable with the human situation. We established a sheep model of osteoporosis based on the recommendations of the FDA by combining several treatments to induce osteoporosis: oestrogen depletion by ovariectomy, calcium and vitamin D-restricted diet, reduction in physical activity and steroid medication. All treatment components are known to be high-risk factors for osteoporosis in humans. BMD, one of the strongest predictors of subsequent fracture in humans, shows significant changes of -33% at the distal radius or -13% at the lumbar spine. This bone loss is higher than that due to ovariectomy alone (Turner et al. 1995) and similar to other reports on steroid-induced osteoporosis (Chavassieux et al. 1997, Schorlemmer et al. 2003). The cancellous bone mass represented by BV/TV was decreased (-58%) and the absolute and relative changes are similar to those seen in humans (Kimmel et al. 1990, Dalle et al. 2001).
A linear bone loss was found to be 1.75% per week during the 12-week period of osteoporosis induction (Goldhahn et al. 2005). Following cessation of steroid treatment, a slow rebound of cancellous BMD was observed. Although oestrogen deficiency persisted the rebound still occurred indicating that steroid treatment and diet influence bone loss the most. It is highly likely that the initial level will never again be attained, possibly because of the residual effects of ovariectomy. An osteoporosis-like situation persists in the affected sheep for more than four months after cessation of steroid treatment.
Fracture healing was investigated three months after the cessation of steroid treatment while the bones were still osteoporotic. The results confirmed recent data from several studies in humans and animals, which showed a delay of bone healing in osteoporosis (Barrios et al. 1993, Walsh et al. 1997, Kubo et al. 1999, Meyer Jr et al. 2001, Namkung-Matthai et al. 2001). This sheep model allows the testing of new strategies to stimulate fracture healing in osteoporosis like the implementation of growth factors (Egermann et al. 2006).
The successful induction of osteoporosis using this method was demonstrated at our institute (Lill et al. 2000, 2002a,b) and reproduced by another group using the same protocol (Schorlemmer et al. 2003). In order to study fracture fixation in osteoporosis an animal model that includes a significant decrease in BMD and changes in biomechanical properties is needed, as these factors are associated with increased fracture risk. To our knowledge, this is the only large animal model with a reliable reduction in BMD of >30% including significant changes in biomechanical properties indicating increased fracture risk. This model meets the WHO criteria of osteoporosis since the BMD of all osteoporotic sheep is below 2.5 SD of the mean value of the control group. Our model simulates a mixed postmenopausal and glucocorticoid-induced osteoporosis that is not comparable with the more prevalent postmenopausal osteoporosis. Histological analyses must take into account this mixed type of osteoporosis. However, fracture risk and implant holding strength are not related to the type of osteoporosis, but clearly depend on trabecular structure and mineral density. With the focus on these parameters, this model highly resembles the human situation. Recent attempts to establish a large animal model for osteoporosis using the inhibition of bone formation by central application of leptin (Pogoda et al. 2006) or inbred selection of sheep with low bone mass (Campbell et al. 2003) have resulted in minimal bone loss only and have not achieved an osteoporotic situation.
The clinical health condition of the animals was meticulously recorded during all studies and changes like alopecia or abscess were observed. The side-effects seem to be related to the immunosuppressive potential of the steroid medication. The drop-out rate and numerous adverse effects are ethically questionable. This publication is the first one to our knowledge to present details on animal welfare although similar protocols to induce osteoporosis have been used repeatedly (Chavassieux et al. 1993, 1997, Ikeda et al. 2003, Schorlemmer et al. 2003). The degree of discomfort for the experimental animals has to be assessed and carefully considered. Investigations using an animal model are only acceptable provided that the advance in knowledge is recognized as important, applicable and directly beneficial to animals or humans, that there is minimal disability to the animal and that no in vitro alternatives are feasible (Murray 2002).
We therefore conducted an additional study to modulate the existing induction regimen, to minimize the drug-induced adverse side-effects and to retain the osteoporosis changes such as reduced BMD, microstructural deterioration and mechanical weakness of cancellous bone. The group with only a few high-dose steroid injections showed fewer side-effects than the other groups. A high number of injections seemed to be the main reason for many side-effects, especially abscess at the injection sites.
In Switzerland, >80% of all sheep are infected with C. pseudotuberculosis (Williamson 2001), although clinical symptoms rarely become manifest. Symptoms can occur, for example, due to reduced general condition or immunosuppression. These bacteria were detected in almost all abscesses reported in our studies. The steroid-induced immunosuppression reinforced persistent infection with pseudotuberculosis causing detrimental effects on animal welfare. Screening tests to detect antibodies to C. pseudotuberculosis to find animals with no prior contact to this agent might reduce the infection rate. The modified administration schedule (reduced number of injections, increased single dose) might prevent the occurrence of adverse side-effects without reducing the amount of bone loss. A discontinuous application schedule seems to allow regeneration of the immune defences. The turnover of granulocytes only takes 7-10 days and mature neutrophils have a short lifetime of four days, whereas the physiological bone turnover cycle takes 60-70 days. As shown in this study, the number of side-effects could be clearly diminished by administering the steroid medication at longer intervals.
In conclusion, this study presents the extensive experience at our institute of sheep models for osteoporosis. Although the sheep has been used as a model for bone loss before, most report osteopenia with changes in markers of bone turnover. This is the only large animal model that yields a reliable and reproducible reduction in BMD of >30% including significant changes in biomechanical properties leading to increased fracture risk. It is not necessary for an animal model to mimic the human situation identically. It is only necessary that it answer the specific question or questions being studied. For a model to be scientifically valid, it is necessary to show that the knowledge gained from the model can be extrapolated to the condition in the original species, mainly humans. With regard to models for osteoporosis and associated fractures, an example of this extrapolation is a biomechanical competence as the endpoint in preclinical studies that should be comparable with the fracture rates measured as the endpoint for bone quality in clinical studies. The advantages of severe bone loss and reduction in biomechanical competence are associated with disadvantages in the general condition of health. These side-effects need to be considered carefully and are, therefore, reported in this paper. However, modulation in steroid application and breeding of sheep in conditions free of C. pseudotuberculosis are able to reduce these side-effects. If the side-effects could be further reduced, the model would be a viable option for the study of orthopaedic fracture treatment in osteoporotic patients.
Footnotes
Acknowledgements
This study was fully supported by the AO Research Institute which is partly funded by royalties from licences granted to Synthes GmbH and Mathys Medical Ltd. The authors receive nothing of value and wish to thank Roland Wieling and Urban Lanker and his team for assistance with surgery and animal care.
