Abstract
Summary
Eighteen healthy skeletally mature (3 years old) ewes, with an average weight of 45 kg, of the Portuguese Churra da Terra Quente breed were used to evaluate the normal values of total and bone-specific isoform of alkaline phosphatase serum activities (ALP and BALP, respectively) and serum osteocalcin (OC) and their correlation with the serum minerals - calcium (Ca), phosphorus (P), magnesium (Mg) and ionized calcium (Ca2+). The sheep were maintained under controlled environmental conditions (constant diurnal photoperiod cycle and identical husbandry and feeding) for six weeks before the collection of the blood samples. The measurement of the total ALP and serum minerals was performed with automated biochemistry analysers using the BioMérieux® kits, the serum electrolyte Ca2+ Diametrics Medical, Inc® specific cassettes and the BALP and OC METRATM kits from QUIDEL® Corporation. The mean ± standard deviation values obtained were: total ALP 90.17 ± 85.72 U/L, BALP 15.0 ± 5.44 U/L, ratio BALP/ total ALP 29.28 ± 24.22, OC 13.02 ± 1.87 ng/mL, Ca 2.57 ± 0.37 mmol/L, P 2.13 ± 0.42 mmol/L, Mg 1.04 ± 0.13 mmol/L, Ca2+ 1.29 ± 0.04 mmol/L. Significant correlations were observed between the total ALP and Ca (r = 0.5939; P = 0.05) and OC and Ca (r = 0.5706; P = 0.05). Reference to the serum values of bone turnover parameters in sheep could be of great value in research and could provide complementary non-invasive information on the bone healing process, particularly with regard to obtaining an early prognosis of fracture healing.
The biochemical markers of bone metabolism reflect the bone turnover process and are usually divided into formation and resorption markers. Formation markers are proteins or enzymes secreted by osteoblasts during the bone formation process, such as serum total and bone-specific isoform of alkaline phosphatase (BALP), serum osteocalcin (OC) and serum procollagen type I C- and N-terminal propeptides. The latter two molecules are released during the type I collagen molecule formation. Bone resorption markers are those resulting from the breakdown of type I collagen during the bone resorption process, namely serum carboxyterminal telopeptide of type I collagen, urinary collagen type I cross-linked C- and N-telopeptide, urinary hydroxyproline, total and free urinary pyridinoline and deoxypyridinoline (Seibel et al. 2002).
The two most commonly used bone formation markers for clinical and research purposes in human beings in the evaluation of osteopenia, postmenopausal osteoporosis and metabolic bone diseases are the BALP and OC (Seibel et al. 2002). These markers are also used in several animal species in clinical and orthopaedic research, particularly in sheep (Newman et al. 1995, Turner et al. 1995, Chavassieux et al. 1997, Turner 2002, Taniguchi et al. 2003, Klein et al. 2004, Komnenou et al. 2005, Seebeck et al. 2005).
BALP is a tetrameric glycoprotein and a membrane-bound enzyme of osteoblasts (Price 1993) that may play a role in bone matrix mineralization (Price 1993, Whyte 1994). The major evidence of its involvement in bone extracellular matrix (ECM) mineralization is based on a human genetic metabolic bone disease, hypophosphatasia (Whyte 1994). There are numerous published hypotheses concerning the biological roles of BALP in bone ECM mineralization, i.e. as a catalyst in phosphate esters hydrolysis on the osteoblast cell surface in order to supply a high phosphate concentration for osteoid mineralization (Whyte 1994) and the breakdown of pyrophosphates, which are the inhibitors of the ionized calcium and inorganic phosphate deposition into osteoid (Whyte 1994, Robey 2002). The quantitative measurement of serum BALP activity has been used for clinical purposes as an indicator of osteoblastic activity, especially in the evaluation of postmenopausal osteoporosis and Paget's disease and also in the monitoring of the efficacy of antiresorptive therapies in osteoporosis (Delmas 1990, Seibel et al. 2002).
OC, included in 10% of the non-collagenous proteins of the organic bone ECM, is synthesized by osteoblasts during the ECM mineralization period (Ducy & Karsenty 1996). It is possibly the only protein incorporated uniquely into bone ECM and dentin (Ducy & Karsenty 1996). OC is a vitamin K-dependent protein because its glutamic acid residues (gla residues) are γ-carboxylated by a specific γ-carboxylase that requires vitamin K as a cofactor (Suttie 1985). These modified gla residues give OC a high affinity for free Ca2+, Ca2+-containing proteins and hydroxyapatite crystals of ECM (Ducy & Karsenty 1996). Although the exact in vivo function of OC is still unknown, its affinity for bone mineral constituents implies a role in bone formation. Serum OC levels result from a fraction of the newly synthesized OC, which is released into the circulation, and can be used to determine the level of bone formation. In human beings, serum OC concentrations correlate with somatic growth and are higher in diseases characterized by a high bone turnover, such as acromegaly, primary or secondary hyperparathyroidism, hyperthyroidism, high-turnover osteoporosis or renal osteodystrophy (Delmas 1990, Seibel et al. 2002). In contrast, serum OC levels have a tendency to decrease in hypoparathyroidism, hypothyroidism, in patients subjected to corticosteroid therapy and in some patients with metastatic bone disease (Delmas 1990, Seibel et al. 2002).
The serum biochemical markers of bone turnover have been studied in order to develop experimental models of osteopenia (Newman et al. 1995), postmenopausal osteoporosis (Turner et al. 1995, Turner 2002) and glucocorticoid osteopenia in sheep (O'Connell et al. 1993, Chavassieux et al. 1997, Lill et al. 2002). Potentially, these markers may be used for the study of pathophysiological mechanisms and the management of osteoporosis, e.g. in order to test new bone-forming therapeutic agents. Sheep have also been used as an experimental model in orthopaedic studies, with assessment of bone healing usually acquired by means of imaging, histological and histomorphometric techniques and mechanical tests (An et al. 1999, An & Friedman 1999). Nevertheless, measurements of serum markers of bone turnover could possibly also aid in the evaluation of cellular function in fracture or bone defect healing research, supplying information about the organic response to the lesion and to the selected treatment (Taniguchi et al. 2003, Klein et al. 2004, Komnenou et al. 2005, Seebeck et al. 2005).
As there are few published reports on the relevance of markers of bone turnover in adult sheep under controlled environmental conditions, and on their potential application in clinical and orthopaedic research, the aim of this study was the measurement of total ALP and BALP serum activity and serum concentrations of OC in skeletally mature ewes under controlled environmental factors, and the study of their correlations with the serum minerals: calcium (Ca), phosphorus (P), magnesium (Mg) and ionized calcium (Ca2+).
Materials and Methods
Animals and environmental conditions
After experimental protocol approval by the National Ethics Committee for Laboratory Animals (Portugal), 18 healthy skeletally mature ewes (Portuguese Churra da Terra Quente breed) approximately three years old and with average weight of 45 kg, were acclimatized for six weeks for this study. The sheep were permanently housed indoors in group housing and were kept under a constant photoperiod cycle (light: from 07:00 to 19:00 h; dark: from 07:00 to 19:00 h), temperature (20 ± 2°C) and humidity (50 ± 10%) prior to the collection of blood samples. This husbandry protocol was used in view of published reports of seasonal variation in bone remodelling (Arens et al. 2006) and circadian rhythm variations of the biochemical markers of bone turnover, especially ovine OC (Newman et al. 1995, Liesegang et al. 2003). These data suggest an increase in the rate of bone formation during the evening and night (Gundberg et al. 1985, Liesegang et al. 2003). The animals were fed with hay and a daily dose of ruminant concentrate and water was provided ad libitum. When the animals were selected for the study, each one was subjected to a clinical examination, haemogram determination and general blood biochemical analysis. The animals were examined daily and their welfare monitored during the entire duration of the indoor housing period.
Collection of blood samples
Jugular venous blood samples were collected into serum tubes containing no anticoagulant (SARSTEDT, Monovette®, Serum Gel S; Nümbrecht, Germany), between 09:00 and 10:00 h, and before feeding, to avoid diurnal variations. Thirty minutes after collection, the blood was centrifuged at 3000 rpm for 10 min, the resulting serum harvested rapidly into 2 mL Eppendorf tubes and stored at -20°C for serum mineral analyses and at -80°C for bone turnover marker analyses.
The determination of ionized calcium was done in total venous blood collected into a special 1 mL syringe containing lyophilized lithium heparin normally used to obtain samples of arterial blood (RapidLyte™; Chiron Diagnostics Corporation, East Walpole, MA, USA). Immediately after collection, the needle was removed, the end of the syringe was covered with a luer cap, and the sample was thoroughly mixed for 20-30 s.
Serum biochemical analysis
Serum activity of BALP was determined using an immunocapture method in a microtitre strip format with a monoclonal anti-BALP antibody adsorbed onto the strips that capture the BALP in the sample, with its enzyme activity detected by a paranitrophenylphosphate (p-NPP) substrate (METRA™ bone alkaline phosphatase enzyme immunoassay (BAP EIA) kit; QUIDEL® Corporation, San Diego, CA, USA). The measurement of OC was also determined using a commercial kit immunoassay with a competitive method that uses OC coated onto strips, a mouse anti-OC antibody, an antimouse IgG-ALP conjugate and a p-NPP substrate to quantify the serum intact OC (METRA™ Osteocalcin; QUIDEL® Corporation, Santa Clara, CA, USA). The assays were performed in duplicate, according to the manufacturer's instructions, and read on a microplate reader (Multiskan EX; MTX Lab Systems, Inc, Vienna, VA, USA), especially adapted for enzyme-linked immunosorbent assay (ELISA) and other colorimetric assays. The Metra™ immunoassays used have cross-reactivity with ovine species; the METRA™ BAP EIA test has been used for sheep (Turner et al. 1995, Chavassieux et al. 2001, Liesegang et al. 2003) and the cross-reactivity of ovine OC with human OC is reported as 83% (Mende et al. 1984).
Commercially available immunoassay kits were used for the measurement of total ALP serum activity (Enzyline PAL optimisé®, Ref. 63 609; BioMêrieux, Marcy l'Etoile, France) and for serum minerals, namely calcium (Ca-OCP®, Ref. 61 551; BioMêrieux), phosphorus (Phosphorus UV®, Ref. 61 571; BioMêrieux) and magnesium (Magnesium®, Ref. 61 411; BioMêrieux) using a colorimetric method, by means of molecular absorbance spectrophotometry, and read with an automated biochemistry analyser (LISABIO, Mascott Plus Model, Marcy l'Etoile, France). The determinations of Ca2+ were processed immediately after blood collection using an automated analyser (IRMA SL Series 2000 Model; Diametrics Medical, Inc®, St Paul, MN, USA), the operation principle of which is based on the ion-specific electrodes fixed in specific cartridges (IRMA SL, Blood Analysis System H3, Ref. ADM R 1262; Diametrics Medical, Inc®).
Statistical analysis
All data were reported as mean ± standard deviation (SD) and the corresponding minimum and maximum values, respectively. The inter-individual coefficient of variation (CV) was determined by calculating the percentage of SD from the mean value for each of the biochemical parameters. The degree of correlation between the different biochemical parameters was assessed using a Fisher's test. All statistical analyses were performed with the statistical software program JMP (JMP Version 5, 1989-2003; SAS Institute, Inc.®, NC, USA). The r-value of correlations was considered significant at P < 0.05.
Results
Serum biochemical evaluation
The serum biochemical parameters values are presented in Table 1.
Values of serum biochemical markers of bone turnover, serum minerals and ionized calcium in adult sheep
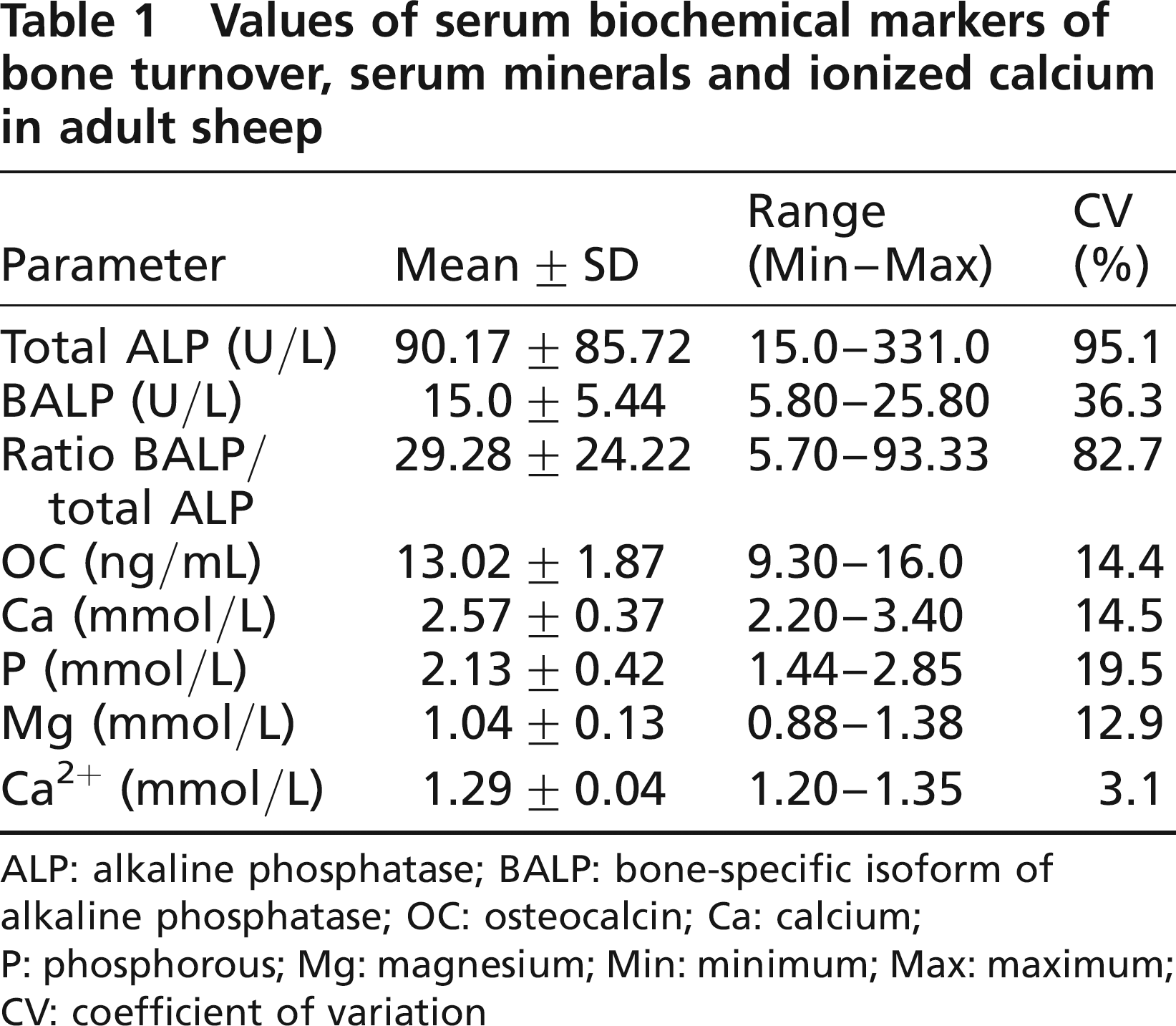
ALP: alkaline phosphatase; BALP: bone-specific isoform of alkaline phosphatase; OC: osteocalcin; Ca: calcium; P: phosphorous; Mg: magnesium; Min: minimum; Max: maximum; CV: coefficient of variation
There was an acceptable correlation, at the 95% probability level, between total ALP activity and Ca (r = 0.5939; P = 0.05) and between OC and Ca (r = 0.5706; P = 0.05) (Table 2).
Correlations between serum biochemical markers of bone turnover, serum minerals and ionized calcium in adult sheep
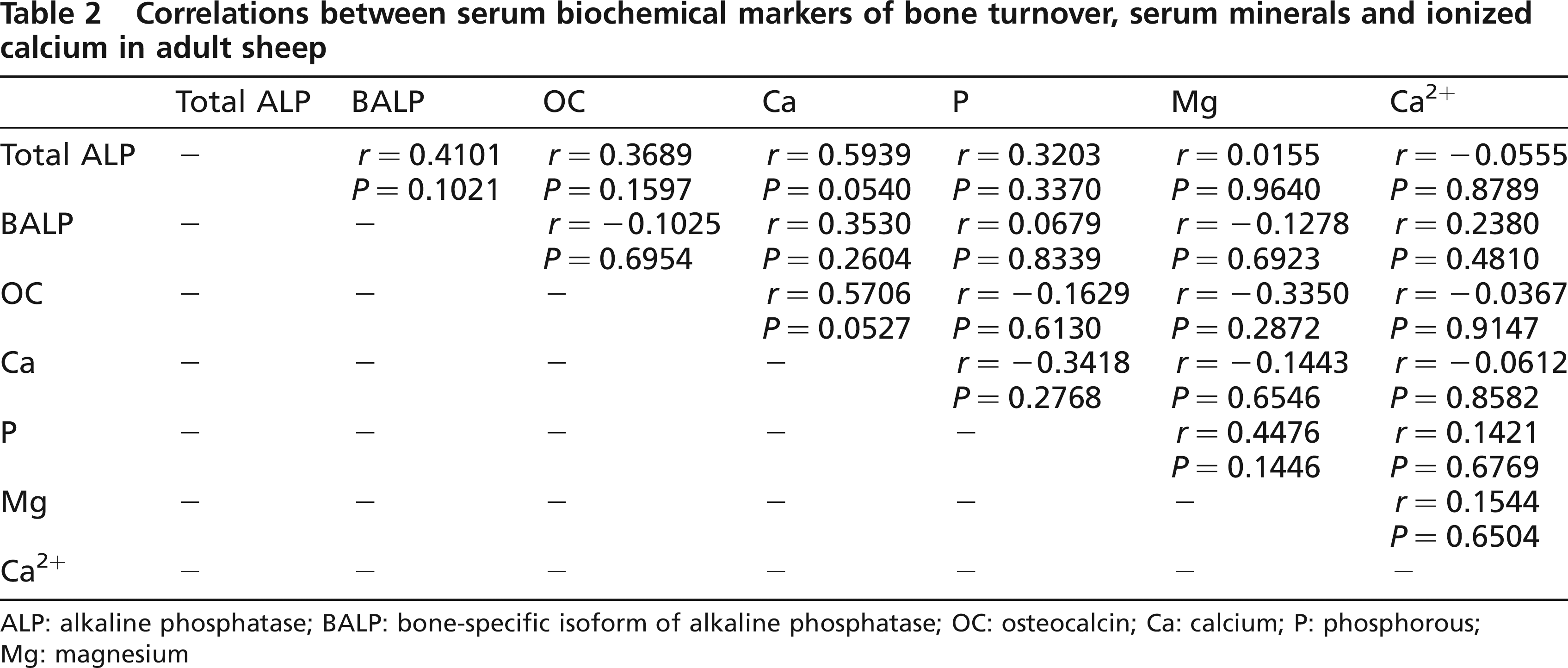
ALP: alkaline phosphatase; BALP: bone-specific isoform of alkaline phosphatase; OC: osteocalcin; Ca: calcium; P: phosphorous; Mg: magnesium
Discussion
The goal of this study is to determine the mean values of biochemical markers of bone formation and their correlation with the serum minerals in adult sheep, because there are very few reports in the scientific literature of bone turnover parameters (Chavassieux et al. 1997, Liesegang et al. 2003, Klein et al. 2004, Seebeck et al. 2005). The reference data for serum BALP and OC are not available for sheep. The interest in bone metabolism serology comes from its possible application in the evaluation of osteopenia and postmenopausal osteoporosis, in metabolic bone diseases and in the course of bone fracture healing. Bone biochemical markers allow a dynamic assessment of bone healing that can facilitate early decision-making in the treatment of bone fractures.
In this study, the total ALP was found to be within the reference range (69-387 U/L) published by Kaneko et al. (1997) and Meyer and Harvey (1998).
As far as BALP is concerned, the published information available for serum levels of this parameter in healthy adult sheep is very scarce. In a paper published by Chavassieux et al. (1997), BALP values of 2-4 ng/mL are quoted in mature ewes (8-10 years), whereas Liesegang et al. (2003) have quoted a reference value of 35-40 U/L in growing female sheep (6 months). Recent studies by Klein et al. (2004) and by Seebeck et al. (2005) in skeletally mature female sheep (2 years), in which the bone healing process was assessed, have indicated a range reference of 2.4-33.2 ng/mL in the first study and 6.4-42.9 ng/mL (17-112 U/L) in Seebeck's study. In our study on three-year-old healthy adult sheep, under a controlled diurnal photoperiod cycle, the mean (± SD) value for BALP activity was 15.0 ± 5.44 U/L.
The ratio between BALP/total ALP (12.1-59.0) was also within the range quoted in the study by Seebeck et al. (2005). The large range of inter-individual variations observed for total ALP, BALP and the ratio BALP/total ALP in this study, even under strictly controlled conditions of husbandry, feeding and light/dark cycle exposition, was compatible with previous studies and similar to the literature of other species (Klein et al. 2004, Seebeck et al. 2005). Nevertheless, BALP measurement is advantageous when compared with other bone formation markers, because its serum activity has a half-life of approximately 40 h (Crofton 1982) and also because the diurnal variation of BALP activity is very small when compared with OC (Nielsen et al. 1990).
Serum levels of intact OC in adult sheep are reported in some osteoporosis studies (Pastoureau et al. 1988, 1991). These indicate a variation that is inversely correlated with age, and consider a mean value of 12.5 ± 2.6 ng/mL as normal in adult sheep (3 years). The study of Seebeck et al. (2005) indicated a range of 2.8-15.7 ng/mL also in adult sheep (2 years). In our study, mean value for intact OC was 13.02 ± 1.87 ng/mL. In serum OC measurement, it is necessary to take into account its short half-life of approximately 20 min in the circulation, as it is rapidly eliminated by the kidneys (Delmas et al. 1983, Delmas 1990). In addition, one must bear in mind the circadian rhythm of this molecule (Gundberg et al. 1985) and its high level of instability, as it undergoes proteolysis in the circulation and during the sample processing and storage (Seibel et al. 2002). The intact molecule of OC represents one-third of the serum immunoreactivity of this protein, whereas the other two-thirds are made up of the large N-terminal mid-molecule fragment and several other smaller fragments (Seibel et al. 2002).
In this study, the observed BALP value in sheep was within the range of that of humans, as indicated by the Metra™ BAP immunoassay (25-44-year-old women: 11.6-29.6 U/L; >44-year-old women: 14.2-42.7 U/L; >25-year-old men: 15.0-41.3 U/L), whereas the level of intact OC was slightly higher when compared with human data as indicated by the Metra™ OC immunoassay (men: 3.7-10.0 ng/mL; women: 3.4-9.1 ng/mL) (Quidel Corporation, San Diego, CA, USA).
The values of serum minerals in this study were within the normal general reference values published for sheep by Kaneko et al. (1997) - Ca: 2.88-3.20 mmol/L, P: 1.62-2.36 mmol/L, Mg: 0.90-1.31 mmol/L. Only the value of serum Ca was slightly above the reference range published for sheep (Kaneko et al. 1997), but identical to the value in the study of Seebeck et al. (2005). The ionized serum Ca2+, which represents about 50% of the serum and extracellular Ca and its biologically active form, was also within the reference range (1.25-1.60 mmol/L) published by Rosol and Capen (1997).
A significant correlation between total ALP and Ca was also verified, which may be due to the fundamental role played by Ca in the enzymatic reactions of this enzyme. The correlation between OC and Ca, which was also significant, supports the existence of a possible relationship between these two parameters, and at the same time, the involvement of OC in the mineralization of skeletal tissues. The absence of a correlation between BALP and OC, also reported in a previous study (Liesegang et al. 2003), could be due to the fact that these two markers of bone metabolism are synthesized by different osteoblasts at different stages of the osteoblast differentiation phase (Schlemmer et al. 1992, Aubin & Liu 1996). The literature states that BALP usually increases during the osteoblast proliferation and ECM development and maturation stages and decreases when mineralization is advanced in time and that OC is synthesized during mineralization (Stein & Lian 1993, Stein et al. 1996, Aubin 1998).
The determination of reference values of bone turnover parameters in adult sheep could also have special interest in experimental studies of osteopenia and postmenopausal osteoporosis performed in sheep as an experimental model. These markers also have potential interest in bone healing research, as they may provide complementary non-invasive information on the bone remodelling process during fracture healing, in addition to imaging, mechanical, histological and histomorphometric analyses. In this field, the studies of Klein et al. (2004) and Seebeck et al. (2005) in a normal and delayed ovine fracture healing model have suggested that the bone turnover parameters are relatively less suitable for monitoring the course of fracture healing than previous clinical studies in humans (Ohishi et al. 1998, Emami et al. 1999, Kurdy 2000, Taniguchi et al. 2003, Akesson et al. 2005, Nakagawa et al. 2006). Nevertheless, these authors have also confirmed the suitability of these markers for estimating the extent of the osteogenic response at the fracture site, and indicating healing disturbances, and their value in the acquisition of non-invasive data on the healing process in experimental orthopaedic studies. As a consequence, further studies are currently being performed to assess the usefulness of the biochemical markers of bone metabolism in the differentiation between an experimental normal fracture healing model and an experimental critical size bone defect model similar to a clinical situation of a non-union process.
