Abstract
More than two million patients received haemodialysis (HD) in 2013. Many methods for improving dialysis therapy outcomes have been tested. Nevertheless, patients continue to experience high morbidity and mortality rates. We aimed to develop an animal model of HD treatment to study methods that would prevent the adverse effects of renal replacement therapy. The study was conducted using six male Merino sheep. The animals underwent a two-step bilateral nephrectomy, and a permanent dual-lumen catheter was inserted into the jugular vein. In each animal, 10 short, daily HD treatments were conducted. The dialysis prescription was adjusted individually to each animal. Measures of dialysis adequacy (spKt/V and urea reduction ratio [URR]) were calculated for each HD treatment. All animals remained in a good clinical state during the experiment. However, a sustained decrease in red blood cell count was detected. The average URR was 0.65 ± 0.01, whereas the calculated spKt/V was approximately 1.16 ± 0.03. Neither hyperphosphataemia nor a significant decline in serum albumin concentrations were detected during the study. A sustained increase in serum potassium concentrations was detected on consecutive days of the experiment. All sheep survived the treatment and were euthanized at the end of the experiment. In conclusion, we developed a reproducible sheep model of HD treatment. The gentle nature and specific anatomical features of sheep provided easy blood access and allowed us to perform HD without pharmacological intervention. However, some differences in sheep physiology relative to human physiology must be considered when interpreting the results of the study.
End-stage renal disease (ESRD) is a serious health problem worldwide. According to the Fresenius Medical Care Report, the global number of ESRD patients is growing; and in 2013, it reached 3,200,000, of which 2,250,000 were undergoing haemodialysis (HD) treatment. 1
Although efforts to improve dialysis therapy regimens are being made, patients continue to experience high morbidity and mortality rates and to suffer a reduced quality of life. On one hand, this issue may be due to the increasing average age of the population and the presence of comorbidities when dialysis therapy is initiated. On the other hand, it may also be the result of disorders that develop in connection with HD or that may arise from uraemia.2,3 Oxidative stress, elevated levels of inflammatory markers, volume overload, and anaemia are among the other conditions predisposing patients with ESRD to an increased risk of mortality.4–7 Moreover, HD is a treatment that involves the activation of immune cells and components of the coagulation system. This activation contributes to increased inflammation, induces oxidative stress, and promotes haemostasis disorders. In addition, it affects the survival time of red blood cells (RBCs) and contributes to the anaemia seen in chronic renal failure (CRF).8,9
For these reasons, more studies on the mechanisms of complications associated with HD treatment are needed. This need has prompted us to develop an animal model of ESRD with HD. Thus far, most of the studies on HD have been performed using dogs. However, ethical considerations and strong community feelings against the use of dogs as experimental animals highly limit scientific studies with this species. Therefore, more socially acceptable models are needed. Moreover, housing experimental dogs can be very costly. There are fewer ethical concerns regarding the use of sheep in research, and the costs to purchase and maintain them are relatively low. Their calm nature makes handling them easy and reduces the need for anaesthesia. Their size corresponds better to the size of a human, the anatomical features of their neck facilitate vascular access, and their coagulation system is more similar to the human system than that of dogs or pigs. These predominant factors contributed to our choice of sheep as our animal model.
Materials and methods
Study protocol
In the context of a larger project designed to determine the impact of near infrared radiation on blood in extracorporeal circulation, a sheep model of HD treatment has been developed. For this purpose, six out of 12 animals were randomly allocated to a control group by a researcher blinded to the study. The animals were subjected to a two-step bilateral nephrectomy under general anaesthesia with a one-week interval between procedures. Male animals were engaged in pairs to avoid isolation in the study. Each sheep underwent 10 short, daily HD dialysis sessions with a one-day pause between the sixth and seventh sessions. Animals were euthanized before a humane endpoint was reached. Since the purpose of this article was to describe a sheep model of HD, we did not include results from all the tests performed, such as markers of oxidative stress, inflammation, apoptosis, and biochemical and coagulation parameters. The study protocol was approved by a local II ethical review board in Wrocław (II Lokalna Komisja Etyczna we Wrocławiu, approval No. 10/2013). Each animal was provided with humane conditions and care according to the European Directive 2010/63/EU on the protection of animals used for scientific purposes.
Animals
The study comprised six adult male Merino sheep (16–18 months) with normal immunological status. The sheep were obtained from a breeding farm which was under the supervision of the Polish Union of Sheep Farmers (Agrominor, Świebodzice, Poland), an organization which ensures that the dergee of genetic polymorphism is adequate in sheep. The live weights (LWs) of the animals ranged from 50.5 to 75.1 kg (mean LW 63.2 ± 4.3 kg). The animals were examined by a veterinarian prior to the study to confirm that they were clinically healthy, and they were allowed to acclimatize and habituate to the conditions in the pens of the institutional vivarium for two weeks. Sheep were kept on a deep bedding in a room with mechanical ventilation and natural lighting at approximately 20℃. The animals were fed ad libitum with meadow hay and 0.2 kg/day of complete mixed ration (Golpasz SA, Golub-Dobrzyń, Poland); they also had unrestricted access to fresh water. The sheep also resided in these pens between surgeries. Twelve hours prior to surgery and between HD sessions, pairs of animals were kept in neighbouring, separate cages in visual contact of each other under the abovementioned environmental conditions. The floor of the cage was made of wide wooden slats.
Surgery
Initially, after 12 h of fasting, the left kidney was removed using a retroperitoneal approach. Each animal was sedated intramuscularly with xylazine hydrochloride (0.2 mg/kg LW, Vetaxyl®; Vet-Agro, Lublin, Poland). Subsequently, anaesthesia was induced intravenously with a propofol bolus (1 mg/kg LW, repeated when necessary; Scanofol®, ScanVet, Warszawa, Poland). Inhalation anaesthesia was initiated with 4 vol% isoflurane and was maintained with 1.5 vol% isoflurane. Analgesia was achieved by a constant-rate infusion of fentanyl (5–20 µg/kg LW/h; Fentanyl WZF®, Polfa Warszawa SA, Warszawa, Poland).
An approximately 12 cm long longitudinal incision was made in the skin between the costal arch and the knee fold, and 3–4 cm ventral from the transverse processes of the lumbar vertebrae. The kidney was exposed via blunt dissection of the appropriate muscles and the adipose capsule of the kidney. The renal artery, renal vein and ureter were ligated, and the kidney was excised. The surgical wound was closed in three layers. The animal was allowed to recover under supervision of a veterinarian. The sheep was closely observed for signs of pain, and the pain was controlled with meloxicam given subcutaneously (0.5 mg/kg LW; Metacam®, Boehringer Ingelheim, Ingelheim am Rhein, Germany). To prevent perioperative infections, a long-acting formulation of penicillin (8 mg/kg LW) and streptomycin (6560 IU/kg LW; Shotapen®, Virbac, Carros, France) was administered subcutaneously prior to surgery and 72 h after the operation.
After seven days, the right kidney was removed in a similar manner as described above. Before the main surgery, vascular access for HD was obtained. A tunnelled 14.5 F × 28 cm dual-lumen Hemo-Flow dialysis catheter (Medcomp, Harleysville, PA, USA) was placed into the right or left jugular vein using a modified Seldinger technique. The animals recovered from the surgery in cages under the supervision of a veterinarian.
Haemodialysis
Animal live weight, water intake and haemodialysis (HD) variables during the experiment.
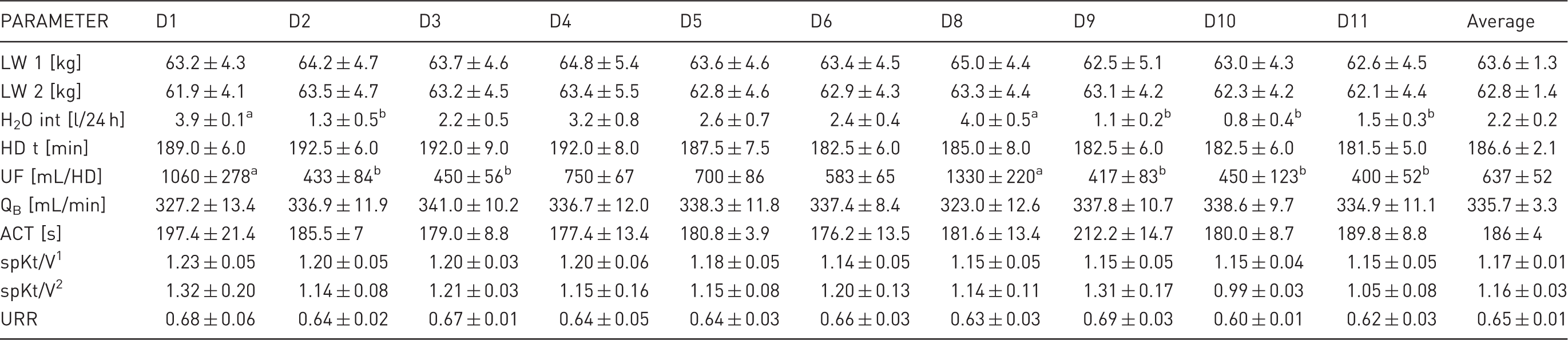
LW 1: live weight before haemodialysis; LW 2: live weight after haemodialysis; H2O int: daily water intake before haemodialysis; HD t: haemodialysis time; UF: ultrafiltration; QB: effective blood flow; ACT: activated clotting time; spKt/V1: single pool dialysis dose adequacy calculated from dialysis variables; spKt/V2: single pool dialysis dose adequacy calculated from Daugirdas equation; URR: urea reduction ratio; D1–D10: consecutive days of the experiment when HD was conducted. Values are presented as mean ± SEM for the six animals. Different superscripts in a row indicate significant differences among HD sessions.
Blood analysis
During each procedure, larger volumes of blood than were needed for the present study were drawn from the arterial and venous lines and were used as research material in other studies. Moreover, prior to and at the end of each HD session, venous blood samples were collected to monitor the health status of the animals. One millilitre of blood was drawn into sample tubes for blood morphology, biochemistry, electrolyte and gas analyses. Thirty minutes before the end of the procedure, one millilitre of blood was sampled to measure ACT.
Blood morphology was examined at the beginning and at the end of each HD session. Measurements were performed using an automated haematology analyser Scil Vet abc (Horiba, Kyoto, Japan). Creatinine (CREA), blood urea nitrogen (BUN), albumin (Alb) and inorganic phosphorus (PHOS) concentrations were measured in serum spectrophotometrically. Electrolyte analysis was performed using a Cornley AFT-500 electrolyte analyser (Meizhou Cornley Hi-Tech Co, Shenzhen, China). Venous blood gas analysis was conducted using an OPTI CCA-TS (OPTI Medical, IDEXX Laboratories, Inc., Roswell, GA, USA).
The urea reduction ratio (URR) was calculated from the serum BUN concentrations measured before and at the end of the HD session according to Equation 1 below. Dialysis adequacy, expressed as single pool Kt/V1 (spKt/V), was calculated as the function of the duration of the HD session (t), urea distribution volume (V) of the sheep and Fresenius HSPS 5 dialyser clearance (K) corresponding to the effective blood flow rate during HD. V was assumed to be 55% of the LW of the animal after HD. Effective blood flow velocity (QB) was calculated as the function of the total volume of blood transfused during HD read from the device (mL) and HD t (min). SpKt/V2 was calculated using the Daugirdas formula 10 (see Equation 2 below).
Equation 1:

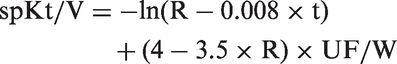
Statistical analysis
All data are expressed as means ± standard error of the mean (SEM). Differences among data collected over an 11-day period of the experiment were examined for significance using one-way analysis of variance (ANOVA), followed by the Tukey post hoc test. Data collected before and after HD sessions were compared using the Student's t-test. All statistical analyses were performed using OriginPro 8.5 data analysis software (Originlab Corporation, Northampton, MA, USA). Statistical significance was set at 5%.
Results
Clinical status and survival rate
During the 11 days of the experiment, all animals remained in a good clinical state. None of the sheep died, and the experiment was completed before a humane endpoint was reached. Sheep were conscious, alert and had a good appetite. During the HD sessions, the animals did not show any signs of discomfort or pain and ate food most of the time. However, the animals decreased their water intake by approximately 50%, and a tendency toward weight loss was observed in all the subjects (Table 1). In one animal, transient diarrhoea and fever were observed. In another, an episode of weakness occurred during the sixth HD session, which was resolved after given a 150 mL bolus of 0.9% sodium chloride. Also in one other sheep, pruritus was seen during the first hour of the last four HD sessions.
The patency of the central venous catheters was maintained in all animals. However, in most animals, a reversed flow was used. No evident infection at the site of catheter insertion was seen in any of the subjects.
Blood morphology
Blood morphology in animals on consecutive days of the experiment and average blood morphology for the entire experiment.
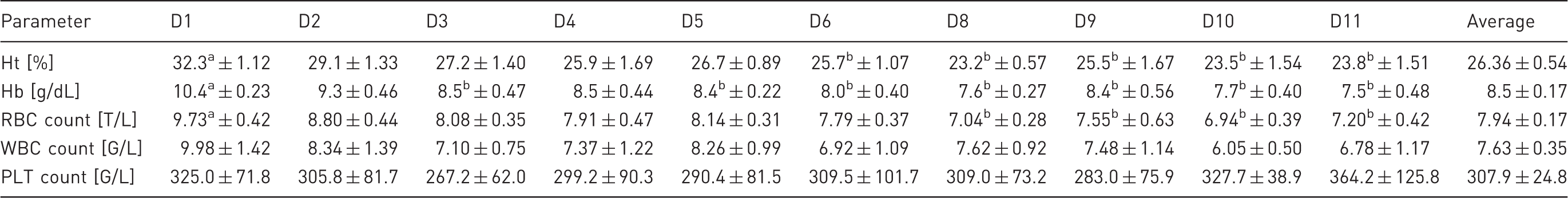
Ht: haematocrit; Hb: haemoglobin; RBC: red blood cell; WBC: white blood cell; PLT: thrombocyte; D1–D11: consecutive days of the experiment. Values are presented as mean ± SEM for the six animals. Different superscripts in a row indicate significant differences among haemodialysis sessions.
Biochemistry and haemodialysis adequacy
Biochemical blood serum parameters of haemodialysed sheep.
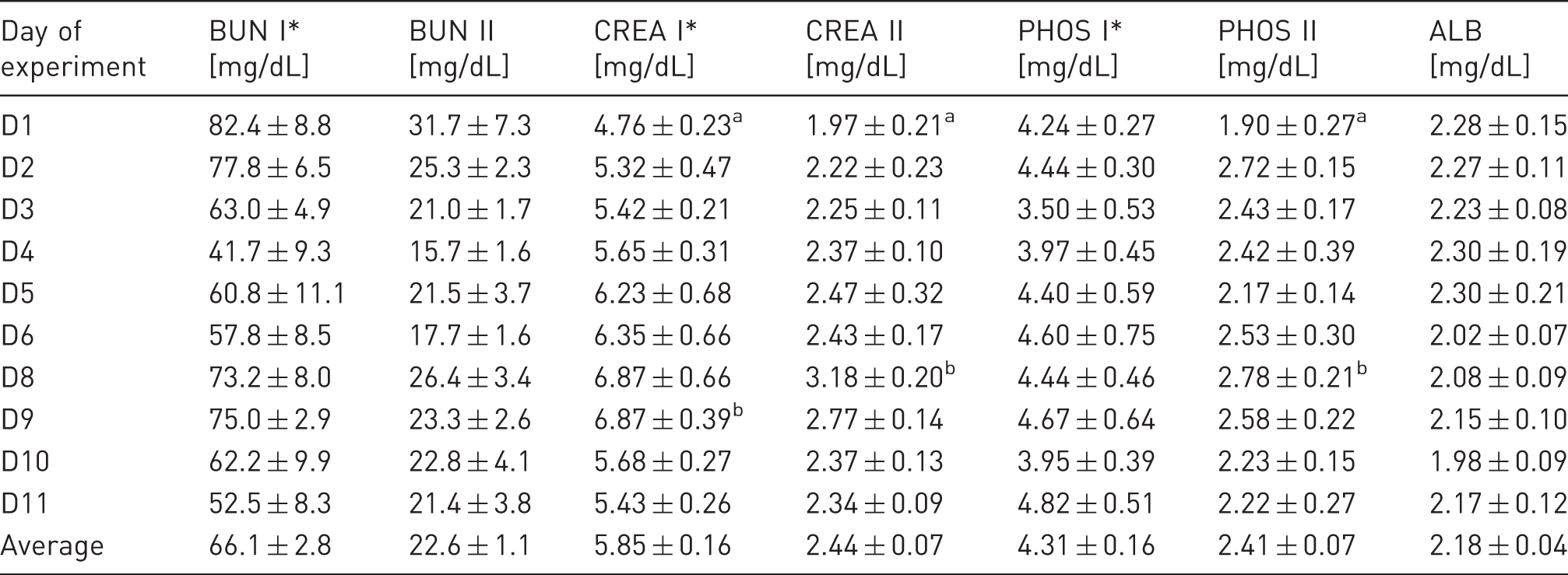
BUN: blood urea nitrogen; CREA: creatinine; PHOS: inorganic phosphorus; ALB: albumin. I: values before haemodialysis (HD); II: values at the end of dialysis; D1–D11: consecutive days of the experiment. Values are presented as mean ± SEM for the six animals. Different superscripts in a column indicate significant differences among HD sessions.*Pre- and post HD serum CREA, BUN and PHOS levels are significantly different for each HD session.
Mean serum CREA concentrations before HD varied from 5.32 ± 0.47 to 6.87 ± 0.39 mg/dL, except on the first day of the experiment, when measured values were below 5 mg/dL. During HD, serum CREA concentrations were reduced by 53.7–59.6%, and at the end of the procedure, mean serum CREA concentrations changed from 1.97 ± 0.21 to 3.18 ± 0.20 mg/dL (Table 3).
Serum PHOS concentrations before HD were close to the lower normal limit and ranged between 3.50 ± 0.53 and 4.82 ± 0.51 mg/dL depending on the day of the experiment. No evident upward trend in pre-dialysis serum PHOS concentrations over the course of the experiment was detected. HD resulted in a decrease in serum PHOS concentrations below the reference range for sheep (4.0–7.3 mg/dL). Serum PHOS concentrations were reduced, on average, by 30.5–55.2% during the HD sessions (Table 3).
Mean pre-dialysis serum Alb concentrations varied between 1.98 ± 0.09 and 2.30 ± 0.21 g/dL depending on the day of the experiment (Table 3).
Electrolyte analysis
Blood serum electrolyte and venous blood gas parameters of haemodialysed sheep.
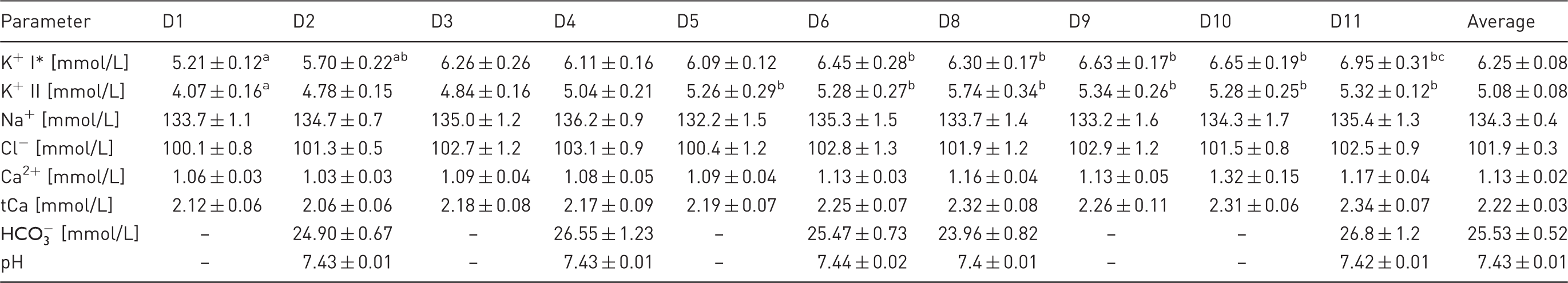
I: values at the beginning of haemodialysis (HD) session; II: values at the end of HD session; D1–D11: consecutive days of the experiment. Ca2+: serum ionized calcium concentration; tCa: total serum calcium concentration. Values are presented as mean ± SEM for the six animals. Different superscripts in a row indicate significant differences among HD sessions. *Pre- and post-HD serum K+ levels are significantly different for each HD session.
Total serum calcium (tCa) concentrations were low compared with the reference range for sheep (2.3–2.9 mmol/L). The mean tCa concentration for the whole study was 2.22 ± 0.03 mmol/L. From the eighth day, tCa concentrations were slightly higher than during the first days after nephrectomy, however this change was not significant. Similar changes in ionized serum calcium concentrations were noted (Table 4).
Blood gas analysis
Venous blood gas analysis revealed stable plasma bicarbonate concentrations and blood pH in animals before HD sessions throughout the study. Both parameters were within normal ranges established for sheep (plasma
Discussion
We developed a reproducible model of HD in uremic sheep. To the best of our knowledge, this is the first detailed report on HD modalities and outcomes in nephrectomized sheep. Thus far, many animal species, such as pigs, dogs, goats, sheep, rabbits and rats, have been used in various experiments investigating HD. However, only dogs and goats have been used as experimental models of repeated HD after nephrectomy.13–15 In our study, 14.5 F wide tunnelled dual-lumen catheters were easily inserted into the animals, and no adverse reactions were observed after drawing blood into standard bloodlines and dialysers. Neither pharmacological sedation nor anaesthesia was necessary for handling the animals during HD sessions. Moreover, this model allowed us to collect a sufficient number of blood samples over the course of the experiment. With smaller animal models of HD, such as dog models, more complex studies would require the use of larger numbers of animals due to the smaller volume of circulating blood, which is a strongly limiting factor in experiments of extracorporeal circulation. Although small laboratory animals are suitable for studying renal disease and pharmacological treatments, their use in HD research is limited. 16 Other authors 17 were able to perform HD in rats with CRF using a specially designed dialyser. However, only a single HD treatment was performed on each animal. It is not known whether this model would be adequate in the study of long-term HD. Moreover, in rats, due to their low volume of blood, researchers were unable to examine a large number of blood parameters. 17 The impact of HD on leukocytes has also been tested using rabbits and purposely modified dialysers. However, only non-uremic animals were included in the study and, similar to studies with rats, single HD sessions were performed. Moreover, general anaesthesia was maintained during the experiment. 18 The most common large model animals used in studies are swine, which are successfully used in cardiovascular research. However, reports on HD in uremic swine are rather sparse. 19 In studies using a pig model for extracorporeal circulation research, the animals were deeply sedated or anaesthetized, which could possibly hinder long-term experiments. Moreover, obtaining vascular access and maintaining it can be rather difficult due to the anatomy of the neck of this species.
During the entire period of our experiment, the animals remained in good health. Although a slight weight loss was reported, there was no decrease in appetite, and the implementation of assisted nutrition was not necessary. In experiments on bilaterally nephrectomized sheep, inappetence has been shown to appear as soon as 24 h after an operation. 20 In a nephrectomized sheep model of continuous ambulatory peritoneal dialysis, the animals ceased eating 4–6 days after nephrectomy. 21 In a study 22 in which nephrectomized haemodialysed sheep were used to evaluate the rennin–angiotensin system for nine days, no data on the condition or appetite of the animals were provided. However, the number of animals tested decreased from 10 on day 0 to five on day 9, which might suggest a high mortality rate. In a goat model of HD, continuous weight loss and rumen function disorders were manifested in the animals. 13 Anephric dogs on HD were fed parenterally to provide adequate nutrient delivery. 14
We applied a daily HD regimen in our study because, in a pilot experiment, we have observed a strong decline in appetite in sheep haemodialysed every two days. In most studies involving bilaterally nephrectomized animals, this method of HD was also used.13,14 As mentioned above, a previous study did not provide detailed data on the survival rate of nephrectomized sheep that were haemodialysed every two days for nine days. 22 In a sheep model of CRF achieved by unilateral nephrectomy combined with an infarction in the other kidney, HD was implemented every 2–3 days when the serum CREA concentration rose above 8 mg/dL. This procedure allowed the animals which maintained residual renal function to survive for the duration of the experiment. 23 However, daily HD seemed to be the best option in anephric animals.
The HD parameters (mean blood flow velocity 335.7 ± 3.3 mL/min, mean HD duration 186.2 ± 2.1 min) allowed us to achieve an estimated Kt/V of 1.16 ± 0.03 and an average URR of 0.65 ± 0.01, and to minimize uraemia, as well as uraemia-associated symptoms. In anephric goats and healthy sheep, blood flow was maintained at approximately 200 mL/min.13,24 In nephrectomized sheep dialysed every second day, a single HD session lasted 4 h, with a blood flow rate fixed at 300 mL/min. The average serum BUN concentration in those animals varied from 105.7 ± 15.6 to 144.7 ± 19.2 mg/dL, and was significantly higher than in our study. 22
We used a standard anticoagulant comprising a heparin bolus and a continuous heparin infusion. In our preliminary studies, we determined that the appropriate dose to prevent blood clotting during HD was a 5000 IU heparin sodium bolus and a 1250 IU/h heparin infusion, which allowed us to keep the ACT at 186 ± 4 s. The larger doses of heparin required for sheep reflect the greater tendency of their blood to clot compared with that of human blood; however, pigs and dogs are even more hypercoagulable and are not considered ideal for the study of the blood-surface interactions 25 that are present during extracorporeal circulation. In healthy haemodialysed sheep, similar doses of heparin were used. 24 However, in other studies on uremic sheep, heparin was infused at a rate of 5000 IU/h after receiving a 10,000 IU heparin bolus. 22 Information about ACT was not provided in any of these studies.
In our study, we detected a sustained decline in RBC counts and Ht and Hb values during the 11 days of the experiment. We believed that this was the result of a depletion of erythropoietin as well as the collection of large volumes of blood. It was reported 23 that anaemia developed within one month after experimentally-induced CRF in sheep, and the severity depended upon whether the sheep required dialysis. In anephric goats, anaemia was also a substantial problem affecting the condition of the animals. 13
During the entire experiment, we did not observe any significant decrease in serum Alb concentrations over time. It has been shown that a decrease in the serum Alb concentration is an indicator of malnutrition and an inflammatory state in humans with CRF. 26 Therefore, a relatively stable serum Alb concentration in sheep during our study might be an indicator of the good clinical state of the animals.
In the electrolyte analysis, we noticed low serum PHOS concentrations throughout the experiment. This result was consistent with observations that showed that hyperphosphataemia did not develop in bilaterally nephrectomized ruminants. 20 This phenomenon was most likely related to the secretion of large amounts of phosphates with saliva into the rumen. In addition, we noticed a continuous gradual increase in serum potassium concentration. The reason for this significant increase was probably due to the high content of potassium in the feed. During the experiment, no alterations in measured venous blood gas parameters were seen, presumably due to the intensive treatment and the lower tendency towards acidosis in herbivores.
In conclusion, bilaterally nephrectomized sheep provide a good model for studying the adverse effects of HD treatment. Their docile behaviour and specific anatomical features allowed us to conduct HD procedures using commercially disposable equipment without the risk of severe hypotension, and allowed us to obtain sufficient blood volumes for further evaluation. However, due to the unique physiology of the alimentary tract and the nature of their nutrition, some differences must be considered when comparing sheep with humans. Furthermore, assisted nutrition and oral medications could be difficult to administer. Experiments of longer duration are necessary to determine if this model is suitable for assessing the long-term effects of HD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication is part of the ‘Wrovasc – Integrated Cardiovascular Centre’ project, co-financed by the European Regional Development Fund, within the Innovative Economy Operational Program 2007–2013, realized in the Provincial Specialised Hospital, Research and Development Centre in Wroclaw as part of the ‘European Funds – for the development of innovative economy’. This work was also partially supported by statutory funds of Wroclaw University of Science and Technology, and by Wrocław Center of Biotechnology, Program Leading National Research Center (KNOW) for the years 2014–2018. The authors received no other financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
