Abstract
The effects of substrate composition on oxide layer properties following plasma electrolytic oxidation under similar conditions have been evaluated for α-cpTi, α/β-Ti6Al7Nb, β-Ti35Zr10Nb and β-Ti45Nb alloys. All oxidised surfaces revealed enhanced wettability, surface free energy and roughness relative to the non-oxidised surfaces. Nevertheless, the resultant oxides differed with respect to average pore size, pores density, layer chemistry and phase composition. The β-titanium alloys developed oxides with a larger average pore size and lower pore density relative to the α-cpTi and α/β-Ti6Al7Nb substrates. Anatase dominated the oxide layer formed on α-cpTi and β-Ti45Nb alloys, a mixture of anatase and rutile was present on the oxidised α/β-Ti6Al7Nb surface, whereas Ti2ZrO6 was the only phase detected on the oxidised surface of the β-Ti35Zr10Nb alloy.
Introduction
Titanium and its alloys are key biomaterials for load bearing medical devices like orthopaedic and dental implants. This is because titanium holds a unique combination of surface and bulk properties, which includes high TiO2 stability, Young's modulus closer to that of bone and high specific strength. 1 However, the new generation of bone implants will need to acquire multiple biofunctionalities. An example includes titanium implants that can simultaneously stimulate bone formation and prevent implant associated infections.
One approach towards biofunctionalisation is the incorporation of specific bioactives (e.g. growth factors, antibacterial agents, etc.) into titanium surfaces 2 followed by their controlled release at the site of implantation. Nevertheless, incorporation of bioactives imposes strict conditions in selecting the appropriate surface modification technique of titanium. Such techniques should satisfy a series of conditions, including (i) maintain the original functionality of the implant; (ii) ensure surface integrity during implantation and a stable interface with the host tissue thereafter; (iii) preserve the physical, chemical and biological properties of the bioactive during incorporation into titanium surface and subsequent implantation; and (iv) create enough reservoirs and ensure controlled release of bioactive over the desired duration. It is a real challenge for existing surface modification techniques to comply with all these requirements. A method that shows real potential is plasma electrolytic oxidation (PEO).3–5 The PEO process has the ability to convert the surface of titanium into a porous TiO2 layer through a set of electrochemical and plasma assisted reactions. As a consequence, a well adherent and stable oxide is created without impairing the chemical biocompatibility of the material. Furthermore, by adjusting the electrochemical parameters of the PEO process, the layer can be tailored with respect to thickness, chemistry and pore morphology, features that are important to control interfacial cellular events or generate reservoirs for bioactives at the implant surface. 5
As the anodic oxide is a conversion layer, its properties are affected by alloy composition and microstructure. Most of the research on PEO of titanium biomedical alloys is focused on standard (Food and Drug Administration approved) materials like commercially pure Ti (cpTi), Ti6Al4V or more recently Ti6Al7Nb alloys.4,5 Nevertheless, new alloys with relatively lower Young's modulus and better chemical biocompatibility are being developed.6,7 Understanding the responses of these alloys to the anodic oxidation processes and elucidating the properties of the resultant layers are prerequisites for the generation of surfaces with multiple biofunctionalities for bone applications.
The aim of this study was to assess the effects of substrate composition on relevant surface properties of PEO layers produced under similar conditions on α-cpTi, α/β-Ti6Al7Nb and the relatively new β-Ti45Nb and β-Ti35Zr10Nb alloys. The oxides have been obtained galvanostatically in a Ca/P based electrolyte, and the resultant layers have been investigated for surface chemistry, oxide phase composition, topography, wettability and surface free energy (SFE). The Ca/P based electrolyte has been selected to provide a bioactive surface for enhanced bone attachment. 5
Experimental
Titanium alloys and oxidation conditions
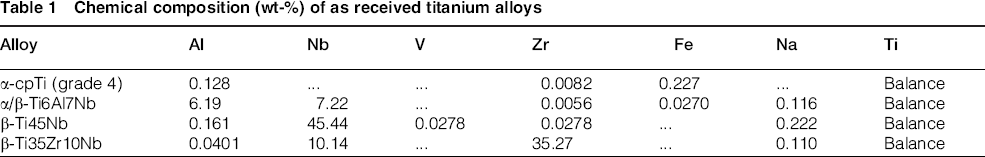
Four titanium alloys with different compositions and phase structures, that is, α-cpTi, α/β-Ti6Al7Nb, β-Ti35Zr10Nb and β-Ti45Nb, have been selected for this research. Their chemical composition, as determined by X-ray fluorescence, is shown in Table 1.
Chemical composition (wt-%) of as received titanium alloys
For the PEO process, titanium specimens were cut into disks and successively ground with 320, 800 and 1200 SiC grit paper. Thereafter, the disks were thoroughly cleaned in acetone, ethanol and deionised water for 5 min each step using an ultrasonic bath.
The oxide layers were produced in a double wall glass electrolytic cell (800 mL) using a bicomponent electrolyte, that is, 0.02M calcium glycerophosphate and 0.15M calcium acetate. A current density of 20 Adm− 2 has been applied via an AC power supply (50 Hz) for 300 s. The electrolyte was stirred with 500 rev min− 1, while its temperature was controlled at 22 ± 1°C using a thermostatic bath. Voltage–time (V–t) responses were recorded with a sampling rate of 1 s. Following PEO, the specimens were washed in running tap water for 5 min, ultrasonicated for 30 s in 70% ethanol and 5 min in deionised water and finally dried for 1 h at 110°C.
Characterisation of oxide layers
The surface morphology of PEO modified specimens was studied by scanning electron microscopy (SEM) using a JEOL JSM-6500F microscope. Energy dispersive X-ray spectrometry (EDS) analysis was applied on selected areas to determine the elemental composition of the layers. The porosity (i.e. surface porosity, pore density, average pore size, maximum and minimum pore size) was determined by SEM image analyses using Adobe Photoshop CS4 software. The measurements have been performed on 3000 × magnification SEM images using scanned areas of 30 × 40 μm2.
The phase composition of non-oxidised and oxidised titanium alloys was investigated by X-ray diffraction (XRD). The analyses were carried out on a Bruker-AXS type DX Advance Series 2 diffractometer using Co Kα radiation at 45 kV and 30 mA.
Surface roughness (Ra) was determined using a Surtronic 3+ (Taylor Hobson Precision, UK) profilometer. The evaluation length for each measurement was 4 mm. A number of 10 measurements for each reported roughness value were conducted.
The dynamic advancing contact angle measurements were carried out using deionised water and diiodomethane, based on which SFE was calculated according to Fowkes' theory. For the contact angle measurements performed with a DSA100 (Krüss, Germany) drop shape analyser, a volume of 10 μL liquid was placed automatically on the tested surface using a microlitre syringe. Upon contact with the surface, the increasing droplet was measured at intervals of 1 s for a duration of 33 s. For every specimen, four measurements were performed in two different wetting liquids. The reported values represent the average and standard deviations of contact angles in water and of the calculated SFE.
Results
Voltage–time response during PEO
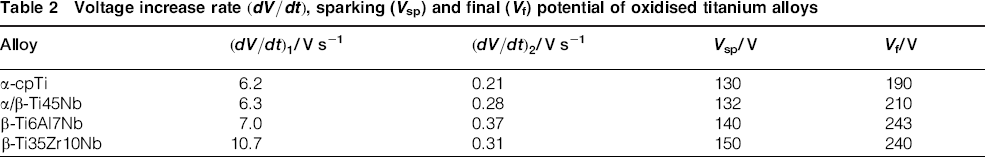
The evolution of voltage (V–t) during oxidation of α-cpTi and titanium alloys is shown in Fig. 1. The anodising voltage increased gradually for all titanium materials as a result of oxide layer growth. The rate of voltage increase, the sparking voltage (Vsp) and the final voltage (Vf) for each material are presented in Table 2. The anodising voltage increased with a high rate, that is, = 6.2–10.7 V s− 1, up to the onset of sparking (Vsp = 130–150 V) being followed by a much slower increase, that is, = 0.21–0.37 V s− 1, until the end of the process (Vf = 190–240 V). The highest final voltages have been recorded for the ternary titanium compositions. Oxygen gas evolution accompanied the entire anodic oxidation process.
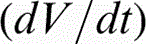
Variation of voltage amplitude versus oxidation time (V–t) of titanium biomaterials (Vs sparking voltage, Vf – final voltage,
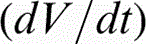 – voltage increase rate) (Colour version available online)
– voltage increase rate) (Colour version available online)
Voltage increase rate , sparking (Vsp) and final (Vf) potential of oxidised titanium alloys
Surface morphology and chemical composition of oxide layers
The morphology and elemental composition of oxide layers, as determined by SEM and SEM/EDS analyses respectively, are presented in Fig. 2. All the oxide layers revealed a porous topography (Fig. 2a–d). The larger pores with a round shape and smooth edges protruded slightly from the surface. Furthermore, surface microcracks were visible as indicated by the arrows in Fig. 2a–d.
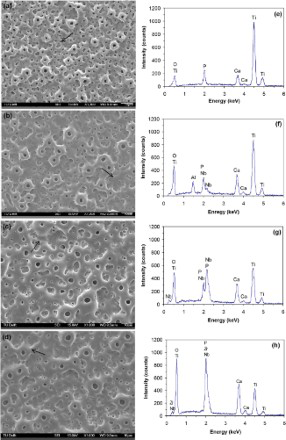
a–d SEM micrographs and e–h EDS spectra of oxidised titanium: arrows indicate microcracks: a,e cpTi; b,f Ti6Al7Nb; c,g Ti45Nb; d,h Ti35Zr10Nb (Colour version available online)
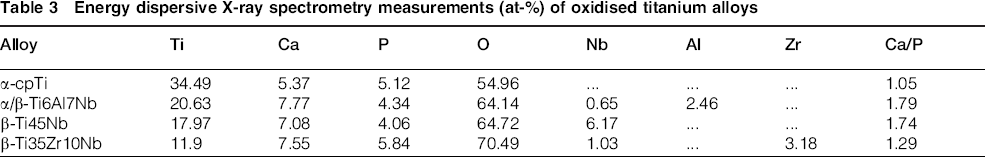
The EDS spectra (Fig. 2e–h) indicated the local elemental composition of each oxidised titanium material. Along with oxygen and constitutive elements of each material, incorporation of Ca and P from the calcium acetate/calcium glycerophosphate electrolyte was detected. Quantification of EDS peaks and the Ca/P atomic ratio are included in Table 3. The Ca/P ratio had values ranging from 1.05 to 1.79. Two oxidised titanium alloys, that is, α/β-Ti6Al7Nb and β-Ti45Nb, had a Ca/P ratio (1.79 and 1.74 respectively) closer to that of hydroxyapatite phase (1.67). This means that the type of titanium alloys may influence the incorporation of Ca and P in the layer.
Energy dispersive X-ray spectrometry measurements (at-%) of oxidised titanium alloys
After grinding to 1200 SiC grit paper, the substrates had Ra = 0.10 μm. The formation of pores and the presence of microcracks following the PEO process increased significantly the roughness of oxidised surfaces (Fig. 3). In addition, the roughness of oxidised titanium alloys was ∼2.2-fold higher than that of the oxidised α-cpTi surface.
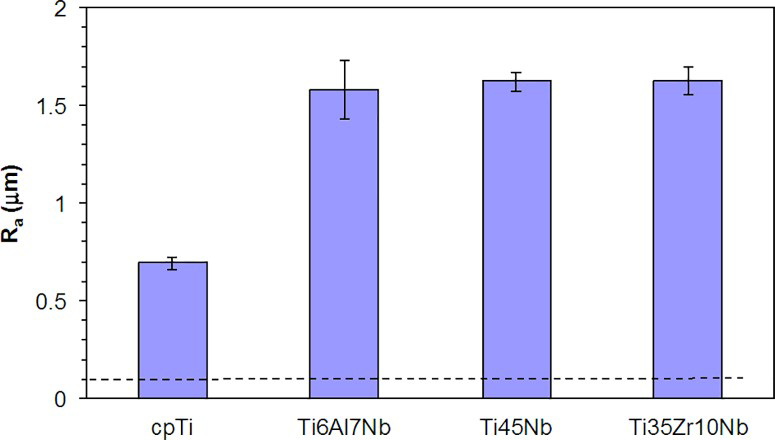
Surface roughness (Ra) of oxidised titanium alloys: broken line represents Ra of non-oxidised samples (Colour version available online)
Surface porosity (i.e. area covered by pores/total scanned area), pores density (pores/mm2) and average pore size are presented in Table 4. In addition, the pore size distribution of the oxide layers on the four different substrates is shown in Fig. 4. The β-titanium alloys revealed larger average pore size and lower pore density relative to the α and α/β-titanium substrates. Moreover, it has been observed that the α-cpTi and α/β-Ti6Al7Nb oxidised surfaces have most of the pores (∼70%) below1.0 μm with less than 10% of the pores larger than 2.0 μm, whereas the β-Ti35Zr10Nb and β-Ti45Nb surfaces have a significant fraction of pores (∼20–25%) above 2.0 μm.
Porosity characteristics of titanium materials following PEO process

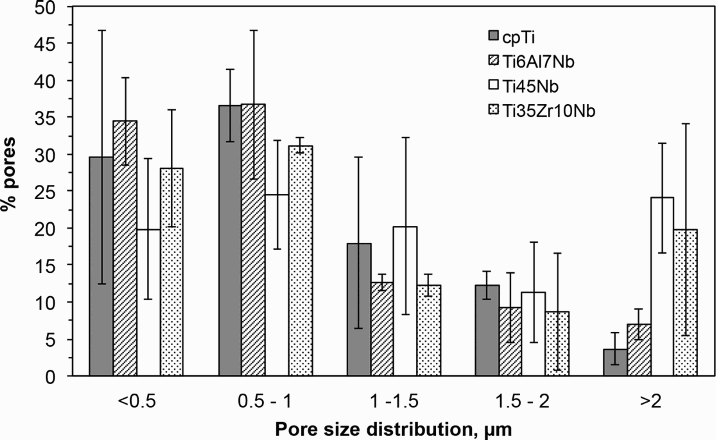
Surface pore size distribution of oxidised titanium surfaces
Oxide phase composition and wettability
Figure 5 shows the phase composition of non-oxidised and oxidised titanium materials as detected by XRD. Non-oxidised α-cpTi and α/β-Ti6Al7Nb revealed the XRD reflections of titanium. The β-Ti45Nb displayed the lines of Ti0.5Nb0.5 phase, whereas the β-Ti35Zr10Nb displayed those of the Ti0.7Zr0.3 phase. The XRD spectra of oxidised materials showed a few sharp peaks indicating the formation of well defined crystalline microstructures. The oxide phase composition of α-cpTi, α/β-Ti6Al7Nb and β-Ti45Nb alloys suggested a combination of anatase and rutile (Fig. 5a–c). The anatase peaks of α-cpTi and β-Ti45Nb oxidised alloys had higher intensities compared to rutile. This shows that the anatase phase dominates the oxide microstructure of these two materials. In contrast, the oxide of α/β-Ti6Al7Nb had a more balanced combination of anatase and rutile phases. The main crystalline oxide phase of β-Ti35Zr10Nb alloy was the orthorhombic Ti2ZrO6 phase (srilankite). Furthermore, a quite shallow XRD peak between 25 and 40° of the PEO modified β-Ti45Nb and β-Ti35Zr10Nb alloys suggested the presence of an amorphous oxide phase.
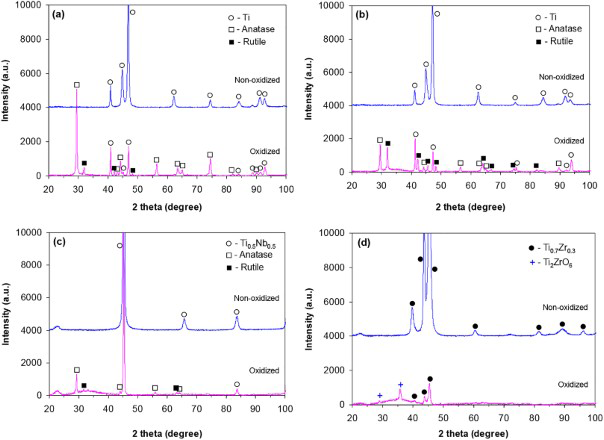
X-ray patterns of non-oxidised and PEO modified biomaterials: a cpTi; b Ti6Al7Nb; c Ti45Nb; d Ti35Zr10Nb (Colour version available online)
The contact angle and SFE findings for the non-oxidised and oxidised titanium materials are presented in Fig. 6. All oxidised specimens showed lower water contact angles (∼40°) as compared to bare titanium surfaces (∼70°) (Fig. 6a). Consequently, SFE of the oxidised surfaces increased relative to the bare titanium surfaces (Fig. 6b). No differences have been however observed between the four different oxidised surfaces.
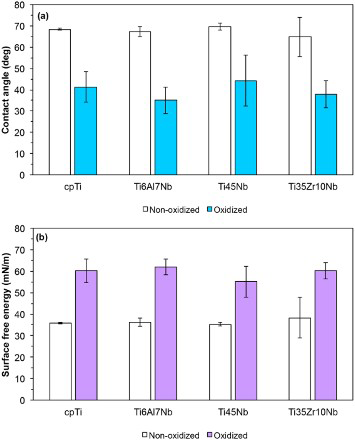
a contact angle and b surface free energy of non-oxidised and oxidised titanium materials (Colour version available online)
Discussion
As displayed in Fig. 2, the oxide morphology of β-titanium alloys (i.e. Ti45Nb and Ti35Zr10Nb) resembles that of α-cpTi and α/β-Ti6Al7Nb materials. The layers present typical PEO oxide features such as porosity, microcracks and roughness.3,8,9 Initially, a dense, barrier-like oxide film 10 is formed upon reaching Vs values (Fig. 1). This layer forms mostly at the metal/oxide interface by inwards migration of O2 − and outwards movement of Ti4+ under high electrical fields (>106 V cm− 2 [Ref. 11]) and is associated with high rates of voltage increase. Above the sparking voltage, the layer grows in the presence of microdischarges and intense gas evolution, leading to the formation of a porous structure and enhanced levels of Ca and P incorporated from the electrolyte through plasma assisted processes. 12 The formation of Ca and P rich material in the presence of sparks contributes to enhanced outward growth of the oxide and thicker oxide layers.
Since the layer is formed by conversion of the substrate into the corresponding oxides and the oxidation conditions were similar for all titanium materials, the differences observed in the surface characteristics of the resultant oxides may be attributed to the type and concentration of alloying elements present in each titanium alloy.
The findings of the present study indicate that surface roughness, wettability and SFE increased after PEO treatment relative to the non-oxidised surfaces regardless of the type of substrate. This is expected due to the formation of a porous structure in the presence of sparks and the change in surface chemistry. When comparing the four different surfaces after the PEO process, it is noticed that the β-titanium alloys led to a lower pore density, larger average pore size and enhanced surface roughness relative to the α-cpTi. In addition, the ternary β-alloy also revealed different oxide chemistries. The α/β-alloy showed the same trend as the β-alloys except for the average pore size that was similar to that on the α substrate. These findings suggest that alloying of titanium to form binary and ternary compositions influences the growth of anodic oxide layers. Much rougher surfaces with less but relatively larger pores and changed oxide chemistry have been obtained in the present study. This could be associated with the α/β and β phases present in the structure of these alloys having a different anodising behavior relative to the α-titanium phase. The extent of changes in oxide characteristics and the possible local variations along the surface are expected to depend on the type of phase, number of phases and their surface distribution, as well as on the type and concentration of the constitutive elements.
The presence of Nb in the β-Ti45Nb composition altered, as mentioned above, all the investigated oxide characteristics when compared to the α-cpTi substrate, except the crystalline phase composition of the resultant oxide. Nb was not identified by XRD to form crystalline phases on the oxidised β-Ti45Nb alloy. It indicates that Ti is preferentially oxidised during PEO of this alloy. Nevertheless, the addition of Zr in the β-Ti35Zr10Nb alloy changed the composition of the crystalline phases by the formation of the Ti2ZrO6 mixed oxide. This suggests a higher reactivity of Zr relative to Nb during PEO that may be related to its lower free Gibbs energy of oxide formation. 13 Further, a higher pore density was observed on this surface relative to the oxidised β-Ti45Nb surface. The β-Ti45Nb surface seems the most reluctant to pore formation among all the materials studied, as evidenced by the lowest pore density and surface porosity measured (Table 4).
When comparing the ternary α/β-Ti6Al7Nb with the ternary β-Ti35Zr10Nb composition, it seems that replacement of Al with Zr led not only to a change in structure from α/β to β phase but had effects on oxide characteristics following PEO, including the porosity parameters and oxide chemistry.
Anatase and rutile are common phases present in the oxide structure of PEO layers of titanium.8,9 Crystallisation of TiO2 layers may start with the formation of anatase nuclei within the amorphous structure of the barrier layer at low voltages. Habazaki et al. 10 found anatase crystals of < 1 nm at the metal/oxide interface during anodising of sputtered titanium in ammonium pentaborate electrolyte. This may be caused by compressive stresses arising from electrostriction given by high electrical fields within the film. 11 The anatase phase develops further with layer growth and undergoes allotropic transformation (600–1100°C) 14 to rutile probably due to local annealing effects in the layer.9,11The results in Fig. 5 confirm the presence of anatase and rutile phases in the oxidised structure of three titanium materials: α-cpTi, α/β-Ti6Al7Nb and β-Ti45Nb. However, the presence of ∼35 wt-%Zr led to the formation of orthorhombic Ti2ZrO6 (srilankite) instead of anatase and rutile phases. Srilankite has been previously reported by Feng et al. 15 in the anodic oxide layers of Ti28Zr10Nb produced below sparking but subsequently annealed in air at 600°C for 3 h.
As reported by Das et al., 16 the free (Gibbs) energy of adhesion of water on the anatase phase (ΔG = − 705.6 mJ m− 2) is lower than that on the anatase/rutile surface (ΔG = − 151.4 mJ m− 2). This may allow faster nucleation of apatite on anatase than on mixtures of anatase and rutile phases. Uchida et al. 17 revealed a more pronounced apatite deposition on anatase gels than on rutile gels. The authors found a better crystallographic matching for the epitaxial growth between apatite (0001) plane and anatase (110) plane than that between the apatite (0001) plane and the rutile (101) plane. Furthermore, Sollazzo et al. 18 showed that anatase coated titanium dental implants inserted in rabbit tibia proved enhanced osseointegration relative to titanium control surfaces. On the other hand, there are studies indicating that rutile and/or the anatase/rutile combination produced by anodic oxidation of α-cpTi also favour apatite formation 19 or implant osseointegration. 20 While the presence of these TiO2 phases is considered beneficial for bone implants, the effects of the newly detected Ti2ZrO6 phase on cell behavior and bone tissue response are yet to be established.
It is known 21 that at voltages exceeding Vsp, microdischarge channels are developed in the anodic oxide layers. Within these channels, high temperatures (103–104 K) and pressures (102–103 MPa)21,22 are generated, which reinforce incorporation of electrolyte species into the oxide layer. 5 The presence of Ca and P in all oxide layers, as revealed by EDS analysis (Fig. 2e–h), in addition to the anatase/rutile phases, is expected to induce apatite formation, as suggested by Han et al. 23
These findings suggest that the PEO technology can be harnessed for these novel Ti alloys in order to produce (bio)functional surfaces for various applications. Nevertheless, since the resultant oxides differed with respect to elemental and phase composition, as well as porosity parameters, which are highly relevant properties for bone implants, dedicated in vitro and in vivo studies are needed to establish the optimal layer characteristics for such an application.
Conclusion
Following oxidation under similar conditions, the four different titanium substrates investigated in this study (i.e. α-cpTi, α/β-Ti6Al7Nb, β-Ti35Zr10Nb and β-Ti45Nb) revealed the characteristic PEO morphology with micropores and surface microcracks, leading to enhanced roughness. The incorporation of Ca and P species from the electrolyte has been observed in all oxidised titanium surfaces. Following oxidation, the surfaces of the alloys were two times rougher than those of α-cpTi. Furthermore, the PEO treated β-Ti35Zr10Nb and β-Ti45Nb alloys exhibited a broader pore size distribution and a lower pore density relative to the other oxidised compositions. The presence of Zr favoured the formation of orthorhombic Ti2ZrO6 phase in the oxide structure of β-Ti35Zr10Nb, whereas Nb did not result in a new crystalline phase but revealed the lowest pore density on the oxidised β-Ti45Nb surface.
The findings of this study indicate that emerging titanium compositions lead to PEO layers with different characteristics relative to the already known alloys, with possible consequences on their biocompatibility. Therefore, the resultant oxides must be evaluated and optimised for the intended biomedical application prior to clinical use.

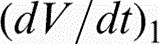 / V s− 1
/ V s− 1
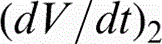 / V s− 1
/ V s− 1