Abstract
Objective:
Arthroscopic dorsal capsuloligamentous repair (ADCLR) has emerged as an effective approach for dynamic scapholunate instability. This pilot study was designed to evaluate postoperative capsuloligamentous healing through sonographic assessment and to describe the early clinical outcomes following ADCLR.
Materials and Methods:
A retrospective analysis was conducted on 15 consecutive patients who underwent ADCLR, between September 2022 and February 2024. Dorsal capsule thickness was measured by sonography preoperatively, at 6 weeks, and at 3 months. Functional outcomes were assessed using the patient-rated wrist evaluation (PRWE) and the quick disabilities of the arm, shoulder, and hand (DASH) questionnaire.
Results:
The cohort included nine men (60%) and six women (40%) with a mean age of 33.4 ± 12.3 years (range: 16–56). Dorsal capsule thickness significantly increased from baseline (sagittal: 0.27 ± 0.05 cm; transverse: 0.22 ± 0.04 cm) to 6 weeks (sagittal: 0.60 ± 0.09 cm; transverse: 0.47 ± 0.09 cm), before partially regressing at 3 months (sagittal: 0.43 ± 0.10 cm; transverse: 0.39 ± 0.07 cm), suggesting a dynamic healing process. Mean PRWE improved by 27 ± 18.5 points at 3 months and 45.5 ± 9.9 points at 6 months, while Quick DASH scores improved by 12.7 ± 6.5 and 19.7 ± 5.9 points, respectively.
Conclusion:
Sonographic surveillance after ADCLR revealed transient dorsal capsular thickening, consistent with postoperative healing. These findings would suggest the relevance of sonographic monitoring and warrant future comparative studies, including failed cases, to clarify the correlation between thickening and successful ligamentous healing.
Ruptures of the scapholunate ligament are frequent and often disabling injuries for the patient. The advent of wrist arthroscopy has led to the development of surgical repair techniques that are less invasive, more reproducible, and reduce postoperative stiffness by allowing the ligamentous structures to heal. 1 Recent studies have led to a better understanding of the biomechanics of the scapholunate joint.2–7 It has become evident that its stability is ensured by the scapholunate complex, and not by the scapholunate interosseous ligament alone. 8 In cases of predynamic or early dynamic instability, arthroscopic capsuloligamentous repair of the dorsal capsuloligamentous scapholunate septum (DCSS) may therefore be suggested1,6,9 (See Figure 1).
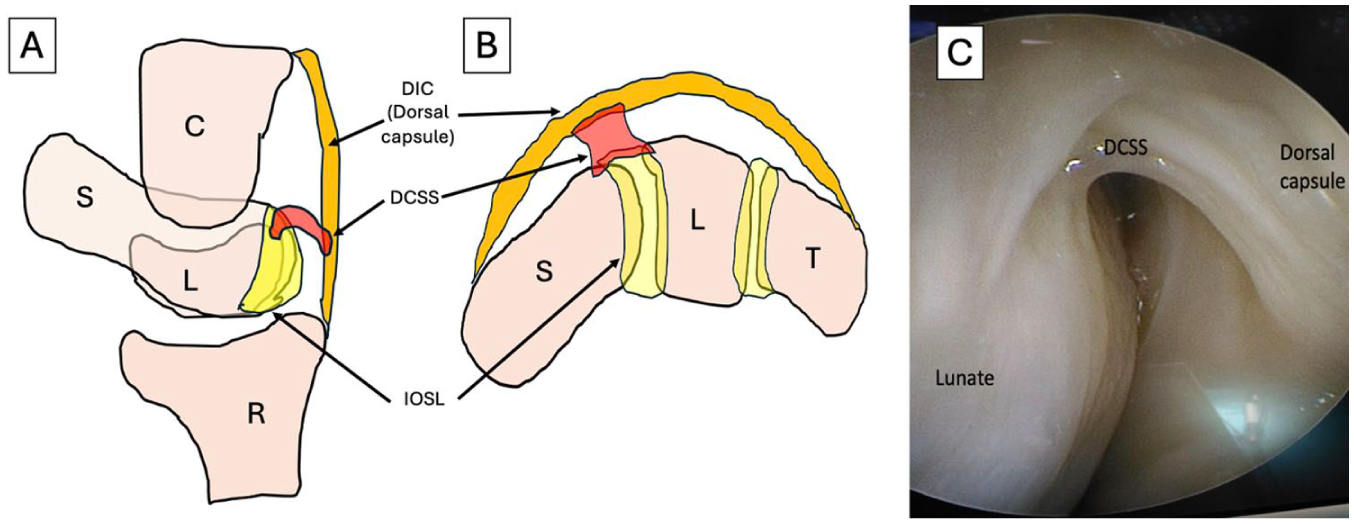
A schematic diagram is provided that illustrates the various capsuloligamentous elements of the scapholunate complex. (A) Anatomical sagittal section. (B) Axial anatomical section. (C) Arthroscopic visualization of the DCSS with radiocarpal views. C, Capitate; DCSS, dorsal capsuloligamentous scapholunate septum; DIC, dorsal intercarpal ligament (dorsal capsule); IOSL, InterOsseous ScaphoLunate ligament; L, lunate; S, scaphoid.
The follow-up with this technique can be lengthy, up to several months, and postoperative results can take a long time to correct.1,9 Unfortunately, there is no way of verifying whether postoperative healing is taking place correctly during follow-up, which may mean that the patient loses several months before a new surgical solution is proposed. In addition, persistent pain has led to a current clinical concern as to whether this is a diagnostic error (i.e., more complex scapholunate lesions, associated ligament injuries, etc.) or a ligament healing defect. In the absence of prognostic factors allowing the surgeons to confirm correct ligament healing, persistent pain at 6 months may cause the surgeon to hesitate about waiting longer. The availability of a prognostic tool would therefore help in deciding whether to perform a revision or simply wait for better healing, thereby saving the patient time. A diagnostic measurement tool, therefore, seems appropriate in order not to waste any more of the patient’s time and to guide the decision on whether to perform further surgery.
The only study to evaluate DCSS with sonography involved healthy subjects. 10 There are no radiologic imaging studies evaluating postoperative healing of the dorsal capsule. Thus, the aim of this pilot study was to evaluate capsuloligamentous healing evolution, after ADCLR using sonography. The hypothesis that was posed was that capsular thickness increases postoperatively via an inflammatory healing phenomenon. Secondly, an additional aim was to determine whether this healing differed between good and poor outcomes.
Materials and Methods
This prospective, pilot study included patients aged 15–65 years who had an indication for ADCLR and underwent surgery between September 1, 2022, and February 28, 2024. The indications included any predynamic or dynamic instability of the scapholunate complex (acute [<6 months] or chronic [>6 months]), with confirmation of the diagnosis based on arthroscopic visualization of the tear. The exclusion criteria were static scapholunate instability, SLAC osteoarthritis based on preoperative imaging or discovered arthroscopically, associated lunotriquetral instability, absence of inter-scapholunate ligament stump on arthroscopy, a surveillance of <6 months, and a lack of measurement of these data. The local ethics committee approved this pilot study protocol, prior to the investigation (IRB#2022-015724-88). After consenting, ADCLR was performed in each patient, under locoregional anesthesia and on an outpatient basis, following a previously described technique.1,11,12 Patients were operated on by a surgeon trained in arthroscopy. During surgery, the degree of instability of the scapho-lunate complex was quantified according to the European Wrist Arthroscopy Society (EWAS) classification. 13
Sonography and Functional Analysis
Sonography and clinical analyses were performed on each patient at the preoperative visit, as well as at follow-up visits every 6 weeks postoperatively for the first 6 months (e.g., 6 weeks, 3 months, 4.5 months, and 6 months). Sonography and clinical measurements were performed by the same surgeon, trained in musculoskeletal sonography and experienced in clinical practice (e.g., Level 4). 14 To avoid measurement bias, sonographic data were collected first, followed by functional results.
A sonogram of the contralateral side was also performed during the preoperative consultation, to serve as a control. A V-scan Air (General Electric Healthcare, Chicago, IL, USA) with a 3–12 MHz linear transducer was used. The thickness of the dorsal capsule opposite the scapholunate space was measured in transverse and sagittal sections, with the wrist pronated and slightly flexed, as described by Gitto et al15,16 (See Figures 2 and 3).
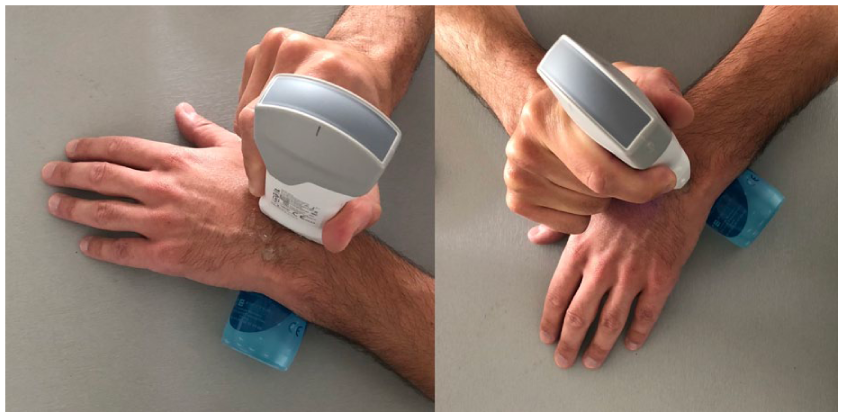
A sonographic examination of the dorsal capsule is demonstrated with transverse and sagittal views being obtained with a V-scan Air (GE Healthcare, Chicago, Ill.) and a 3–12 MHz linear transducer.
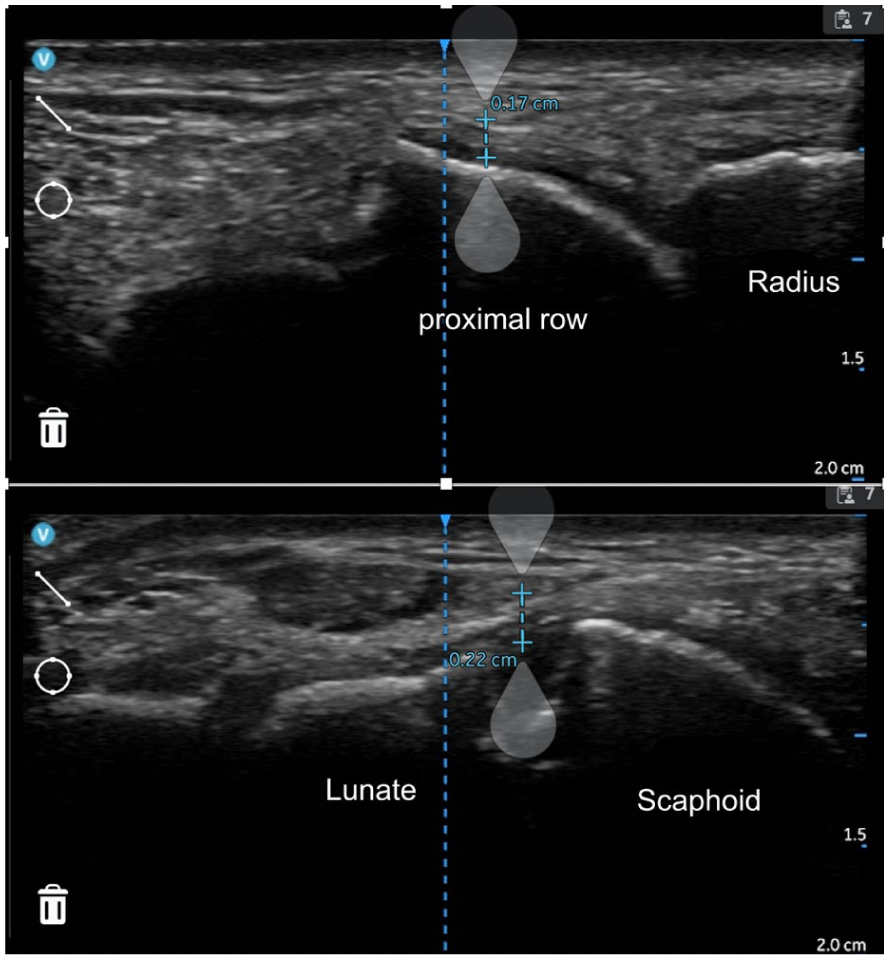
A sonographic measurement is provided of the thickness of the dorsal capsule, opposite the scapholunate joint line, in both the transverse and sagittal views.
Sonographic landmarks and cine clip locations were placed with the wrist pronated and slightly flexed. The transducer was centered over the scapholunate interval, identified by the hyperechoic cortical margins of the scaphoid and lunate, and the intervening scapholunate gap. On the transverse cine clip, thickness was measured at the midpoint of the scapholunate line, perpendicular to the capsule, avoiding anisotropy by fine tilt adjustments. On the sagittal cine clip, the transducer was aligned with the dorsal rim of the scaphoid, and thickness was measured orthogonally to the capsuloligamentous layer just dorsal to the joint line. Minimal compression was applied; settings (depth and focus) were standardized across time points to maximize reproducibility.
In addition, the patients completed the patient-rated wrist evaluation (PRWE) questionnaire and the quick disabilities of the arm, shoulder, and hand (DASH) functional tests.
Statistical Analysis
Statistical analyses were performed using SPSS Statistics 20.0 (IBM Corp., Armonk, NY, USA). Patient demographics, functional scores, postoperative mobilities, and sonographic measurements of the dorsal capsule thickness were presented as the mean ± standard deviation. Quantitative variables were tested for normal distribution using the Shapiro–Wilk test. Normally distributed quantitative variables (ultrasound measurements) were compared using Student’s t-test, and non-normally distributed variables (functional scores and mobility results) were compared using the Wilcoxon test. A p-value < .05 was considered to indicate a statistically significant difference.
Results
Fifteen patients were included continuously between September 1, 2022, and February 28, 2024. No patients were excluded or lost to follow-up. The population comprised nine men (60%) and six women (40%), with a mean age at surgery of 33.4 ± SD 12.3 years (range: 16–56 years). Intraoperatively, EWAS, III B lesions were found in 12 patients (80%), and EWAS IIIC lesions were found in three patients (20%). There were three minor complications (e.g., one extensor tenosynovitis and two cases of pain on palpation of the sutures). All the preoperative and postoperative clinical data and sonographic measurements had a normal distribution.
Sonographic Measurements
On sagittal sonographic views, the preoperative thickness of the dorsal capsule was 2.7 ± SD 0.5 mm (range: 1.7–3.4 mm), while the thickness for the contralateral wrist was 2.9 ± SD 0.5 mm (range: 2.1–3.9 mm). There were no significant differences between the preoperative and contralateral measurements.
During the follow-up phase, the authors recorded the following measurements for 15 patients: 6 ± SD 0.9 mm (range: 4.3–7.3 mm) at 6 weeks, 4.3 ± SD 10 mm (rang: 2.9–6.5 mm) at 3 months, 3.3 ± SD 0.7 mm (range: 2.2–4.7 mm) at 4.5 months, and 2.9 ± SD 0.3 mm (range: 2.3–3.4 mm) at 6 months (See Figure 4).
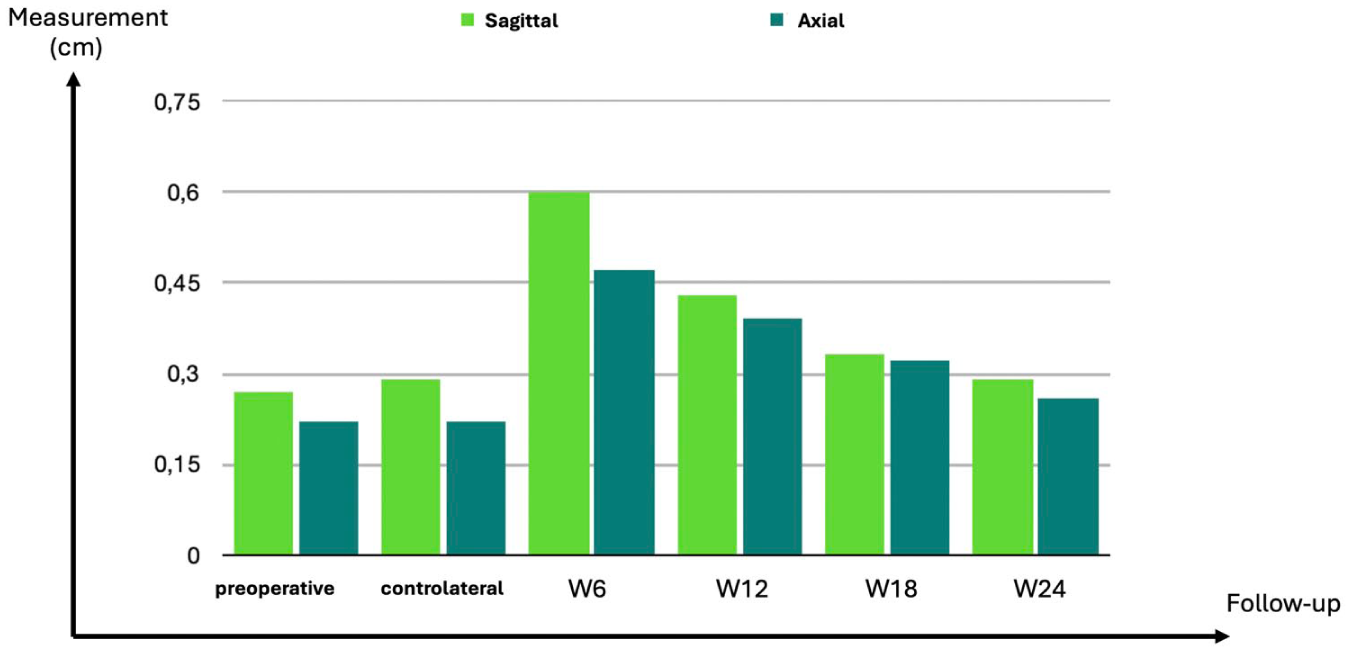
The mean thickness of dorsal capsule measured (in centimeters) with sonography in both the sagittal and transverse views. W, weeks.
On the transverse sonographic views, the preoperative thickness of the dorsal capsule was 2.2 ± SD 0.4 mm (range: 1.5–3 mm), and 2.2 ± SD 0.4 mm (range: 1.4–2.8 mm) for the contralateral side. During follow-up, the thickness was 4.7 ± SD 0.9 mm (range: 3.8–6.6 mm) at 6 weeks, 3.9 ± SD 0.7 mm (range: 2.8–5.4 mm) at 3 months, 3.2 ± SD 0.4 mm (range: 2.8–3.9 mm) at 4.5 months, and 2.6 ± SD 0.4 mm (range: 1.9–3.2 mm) at 6 months.
For both sagittal and transverse views, the mean thickness of the dorsal capsule was significantly increased (p < .05) from postoperative day 45 to 3 months compared with the preoperative measurement and with the measurement on the contralateral side. At 6 months, there was no significant difference between the preoperative and postoperative measurements for the sagittal and transverse sections.
Functional Results
Overall, the functional scores improved significantly (p < .05) at 3 and 6 months postoperatively (See Figure 5). For the PRWE, the score decreased from 53.9 ± SD 10 points (range: 37–72 points) preoperatively to 26.9 ± SD 16.3 points (range: 7.5–53 points) at 3 months and 8.4 ± SD 6.7 points (range: 2–27.5 points) at 6 months. For the Quick DASH, the score decreased from 32.7 ± SD 2.8 points (range: 27–40 points) preoperatively to 20 ± SD 5.9 points (range: 14–33 points) at 3 months and 12.9 ± SD 4.8 points (range: 6–22 points) at 6 months.
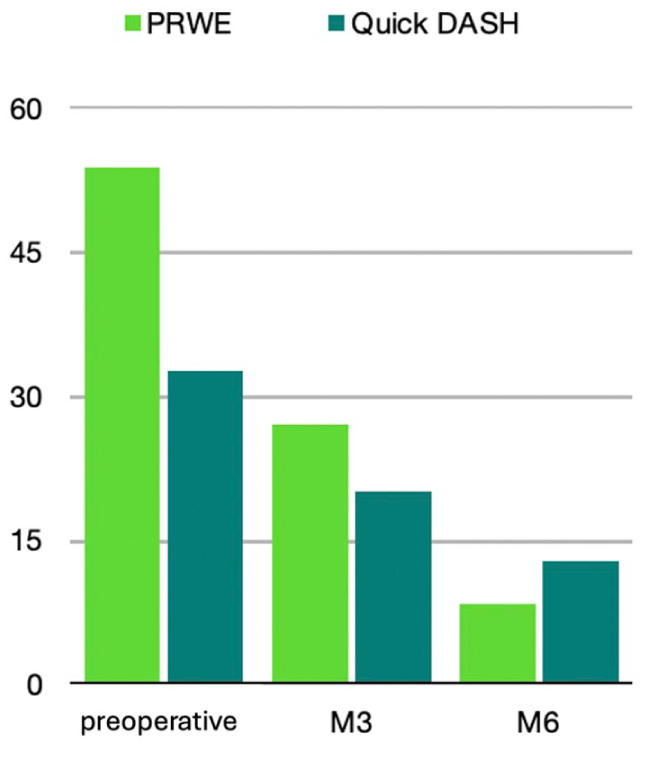
A bar graph is provided of the mean patient-rated wrist evaluation (PRWE) assessments and the quick disabilities of the arm, the shoulder, and hand (DASH) responses, preoperatively, at 3, and 6 months postoperatively. M, months.
The mean improvement in the PRWE score was 27 ± SD 18.5 points (range: −12.5 to 62 points) at 3 months and 45.5 ± SD 9.9 points (range: 28–64 points) at 6 months.
The mean improvement in the Quick DASH score was 12.7 ± SD 6.5 points (range: 0–24 points) at 3 months and 19.7 ± SD 5.9 points (range: 10–32 points) at 6 months.
Unfortunately, none of the patients were recognized as a clinical failure (i.e., all patients improved on their preoperative situation). A comparative analysis between successful and failed cases was initially planned; however, because no patient presented an unsatisfactory postoperative outcome, this analysis could not be performed.
Discussion
In this cohort of patients, ADCLR appeared to provide good results, even in advanced cases. 9 This approach offered a minimally invasive and conservative solution to the management of these injuries. It also offered the advantage of allowing natural ligament healing by the patient, rather than scapholunate biomechanics recreated by tendon reconstruction. 17 However, ADCLR requires surveillance for several months before success or failure can be confirmed. The main objective of this study was to investigate posterior capsule thickening, during ADCLR, for scapholunate complex rupture. The results showed that the thickness of the dorsal capsule, with a mean of 2.7 mm sagittally and 2.2 mm transversely, was significantly increased during the postoperative phase, peaking at 6 weeks after surgery (6.0 mm sagittally and 4.7 mm transversely), before returning to normal values at 6 months after surgery (2.9 mm sagittally and 2.6 mm transversely). These measurements support the existence of an inflammatory, hypertrophic reaction during the first postoperative weeks. The clinical results were also satisfactory, with a clear improvement in the mean PRWE score (from 53.9 points preoperatively to 8.4 points at 6 months postoperatively) and the mean Quick DASH score (from 32.7 points preoperatively to 12.9 points at 6 months postoperatively). However, the first aim of this study was not to compare the evolution of the capsule in the dorsal position based on a good or poor result, but simply to analyze the dorsal capsule using sonography. It was also deemed important to include the patients’ functional data to provide additional information to support the theoretical hypotheses. Thus, in this pilot study there is support for the idea that there is hypertrophic evolution of the dorsal capsule, after repair by ADCLR. Further comparative studies between good and poor outcomes are needed to support the idea that this hypertrophy occurs only in cases of satisfactory healing of the dorsal capsule. However, this deduction must consider that there was no ADCLR failure and therefore the current study did not have a comparative group with an unfavorable evolution. This is the major limitation of this work.
The current study results align with the literature, which provides evidence of good results for acute and chronic lesions of the scapholunate complex classified as EWAS II and III.1,4,9,13,18–20 Given the correlation between the current study’s positive results and the postoperative evolution of the DCSS thickness, it can be suggested that the significant increase in DCSS thickness between days 45 and 90 favors the healing of this ligamentous structure. These preliminary findings may support the reliability of sonography as a noninvasive surveillance tool to document the expected transient thickening of the dorsal capsuloligamentous complex, during early healing after ADCLR. Thus, the absence of an increase in the thickness of the dorsal capsule during this follow-up period could encourage the surgeon to propose a sooner new therapeutic solution to the patient, and to avoid prolonging the follow-up with several weeks of physical therapy.
To the authors’ knowledge, dorsal capsular thickness has not been investigated during the postoperative period, although Debril et al 10 examined the standard anatomy of the healthy DCSS with sonography. The current study provides a novel contribution, namely further evidence in favor of ADCLR for dynamic or predynamic scapholunate complex ruptures. Gitto et al 15 described and standardized the measurement technique employed in this study.
Various publications have highlighted the value of sonography for surgeons to aid in diagnosis and ultrasound-guided surgery.21–23 The latter presents sonography as a diagnostic tool for postoperative follow-up and could provide evidence of the presence or absence of postoperative scarring during follow-up. In the future, the widespread use of musculoskeletal sonography could enable orthopedic surgeons to anticipate or diagnose surgical failure earlier. This approach could allow the proposal of an alternative or a revision surgery and thus enable patients to return more quickly to their previous activities.
Limitations
This pilot study has major limitations due to the research design and threats to internal and external validity. The clinical results of this cohort are unique to them and cannot be generalized. Additionally, this cohort only consisted of 15 patients, so the analysis was completed with low statistical power. Prospective studies with larger sample sizes are needed to confirm these findings. Another key point is that this pilot study did not have a comparison patient group, which could have provided a therapeutic failure. Thus, it is difficult to conclude that an increased DCSS thickness is clearly in favor of healing and correlates with good functional results. The pilot study did collect only functional scores rather than amplitudes, strength, or pain. These type of data have been extensively studied in the literature9,20; however, the inclusion of additional data may not have aligned with the main aim of the pilot, which was to assess sonographic evaluation of healing after ADCLR. However, it would be interesting to collect data from a larger cohort and to retrospectively evaluate healing after arthroscopic repair in patients considered to have therapeutic failure.
While it appears that sonography holds great diagnostic promise, particularly in terms of facilitating patient follow-up by the surgeon, it remains highly operator dependent. An inter-observer evaluation would have been interesting to check the reproducibility of the diagnostic measurements. The fact that only one surgeon performed the measurements would be considered a measurement bias.
These results should be interpreted with caution. The absence of failed cases in the present cohort stems from the limited size and pilot nature of the study rather than from a selection bias. Accordingly, the study was not intended to determine predictive thresholds or to compare successful versus failed repairs; instead, it aimed to describe the temporal pattern of postoperative capsular thickening that can inform the design of adequately powered comparative studies.
Finally, this is the first study to assess postoperative healing of the DCSS. This work was conceived and conducted as a pilot study, intended to generate preliminary observations and to assess the feasibility and reproducibility of sonographic surveillance, after ADCLR.
Conclusion
The results of this pilot study demonstrated a thickening of the dorsal capsule in the first 3 months after ADCLR, associated with good functional results. The use of sonography could enable the surgeon to check whether dorsal capsuloligamentous healing is taking place during surveillance and thus guide the patient for appropriate follow-up. However, a comparative study with a sonographic evaluation with poor results is necessary to support the pilot study hypothesis.
These preliminary findings may support the reliability of sonography as a noninvasive diagnostic surveillance tool to document the expected transient thickening of the dorsal capsuloligamentous complex, during early healing after ADCLR.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was obtained from Comite De Protection Des Personnes Sud Méditerranée I (IRB number 2022-015724-88).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
