Abstract
A preclinical study of 15
Carpal tunnel syndrome (CTS), also known as median mononeuropathy (MMN), is a musculoskeletal disorder characterized by the compression of the median nerve as it passes through the carpal tunnel and under the transverse carpal ligament. This is the most common nerve entrapment syndrome of the upper limbs, with approximately 849,000 new cases reported each year in the United States. 1 Nerve conduction studies are presently considered the gold standard in the diagnosis of CTS; however, imaging studies have shown a positive correlation between increasing cross-sectional nerve area measurements obtained using musculoskeletal sonography and a decrease in nerve conduction velocities.2–6
Sonography has also been demonstrated effective in evaluating other pathologies in the wrist such as masses or fluid collections, as well as the size and echotexture of the median nerve at distinct anatomical landmarks. 7 The National Guidelines Clearinghouse provides an evidence-based guideline for CTS diagnosed with sonography. This set of guidelines was the direct result of a meta-analysis published in 2011, which reported the diagnostic sensitivity of sonography to evaluate the median nerve at 77.6% and a specificity of 86.8%. 8 This indicates a role for high-resolution sonography, which can capture median nerve enlargement and increased hypoechogencity. 9 Recent studies have taken a different approach to improve the diagnostic accuracy of sonography of the median nerve. During the acute phase of CTS, the median nerve becomes enlarged and constricted by the transverse carpal ligament as it passes distally through the carpal tunnel inlet. Proximal to the inlet, a hyperemic response develops coupled with intraneural edema, causing an inflamed nerve. Swelling constricts the nerve as it passes through the inlet on its course to innervate the hand. By combining sonographic measures, including cross-sectional area, hypoechogenicity, and hypervascularity of the median nerve, the additive effect of these measures raised the probability of a correct CTS diagnosis to 87% to 99%.10,11
Preliminary studies have suggested that magnetic resonance imaging (MRI) of the carpal tunnel is more sensitive than sonography for detecting compression of the median nerve by surrounding anatomy or pathology. 12 The American College of Radiology (ACR) Appropriateness Criteria rank MRI as the imaging gold standard for CTS and is the recommended radiologic procedure with the clinical condition of chronic wrist pain with or without prior injury and normal or nondiagnostic radiographs. 13
The objective of the current study was to investigate the use of cross-sectional area measurements of the median nerve to determine whether longitudinal sonography measures could be used to detect physiologic change. By investigating the size of the median nerve measured using sonography and compared with MRI, a preclinical investigation might discern whether this imaging modality can detect physiologic change in the median nerve.
Methods and Materials
A study approved by the Institutional Animal Care and Use Committee (IACUC) board of review was completed using 15
MRI Examination
A modified musculoskeletal MRI protocol was completed on a BioSpec 94/30 MRI System equipped with a 9.4T horizontal bore magnet (Bruker BioSpin, Billerica, Massachusetts). The magnet operated at 400 MHz, running ParaVision 4.0 software platform adapted from the TopSpin 1.5 (Bruker BioSpin). Each subject was positioned in a warm plastic cylinder with a 3.5-mm quadrature coil placed anterior to the wrist. A 20-cm spatial gradient was used for signal acquisition. The subjects were also monitored throughout the scan with a Model 1025 small animal monitoring system (Small Animals Instruments, Stony Brook, New York). T1- and T2-weighted MRI scans were performed in the axial plane, obtaining 32 images of the median nerve as the scans progressed distally down the arm.
Sonography Examination
A musculoskeletal sonography protocol was refined and replicated to evaluate the size and shape of the median nerve quantitatively and qualitatively.15,16 Three sonographers used a GE Logiq
Analysis
Cross-sectional area (CSA) measurements were obtained from both imaging modalities in the axial plane, at the levels of the distal radius (proximal to the carpal tunnel) and the proximal pisiform (within the carpal tunnel). In previous studies, there has been some discrepancy in the manner with which sonographic CSA measurements were acquired. Several studies have used the outer edge of the median nerve epineurium to evaluate the CSA of the nerve,17,18 whereas others have advocated measuring inside the epineurium by tracking around the hypoechoic inner border.7,19–22 For this study, both measurements were obtained on the sonograms by tracing around the outer edge of the epineurium as well as inside the epineurium (Figure 1A,B). To address measurement error that could be attributed to the researcher, a direct trace of the CSA was completed five times at each anatomical location. The highest and lowest values were eliminated and the middle three data points were averaged.
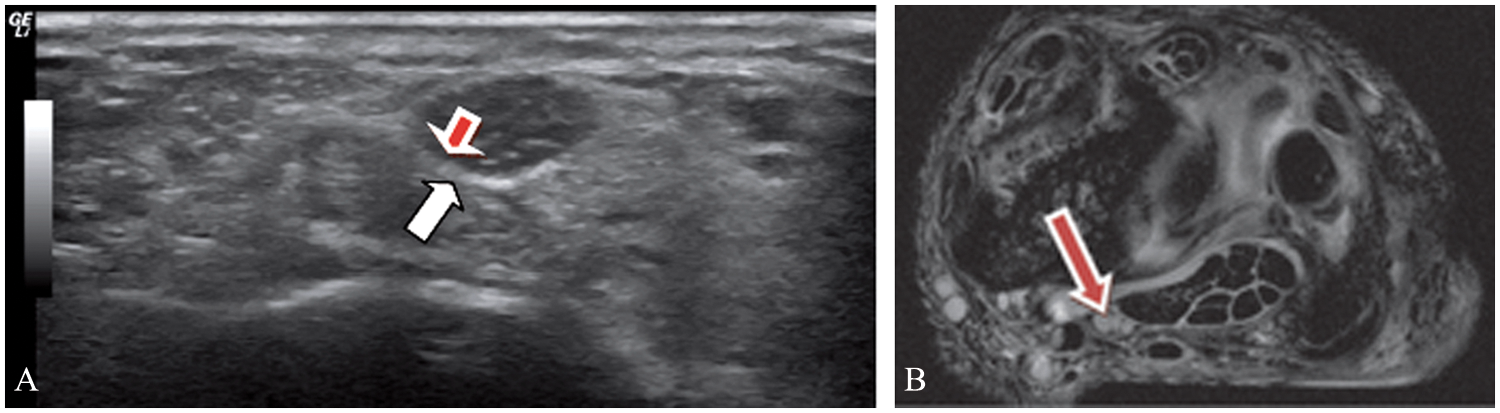
(A) Sonogram of the median nerve imaged in the axial plane. Note the echogenic outer edge (white arrow), known as the epineurium, which surrounds the nerve, and the hypoechoic inner edge (red arrow), which surrounds the perineurium and endoneurium. (B) Axial magnetic resonance image of the left working wrist of a subject that provides a high-resolution image of the outer border of the median nerve, at the level of the radius, proximal to the carpal tunnel inlet.
MRI Analysis
The appropriate T1-weighted MRI examination was selected and analyzed using the National Institutes of Health (NIH), Center for Information Technology medical imaging analysis computer software package, Medical Image Processing, Analysis, and Visualization (MIPAV; NIH, Bethesda, Maryland). A credentialed MRI technologist selected images of the median nerve in the axial plane at the anatomical landmarks of the distal radius (Figure 2) and the proximal pisiform (Figure 3). Proximal to the carpal tunnel inlet was identified in the axial plane as the slice at the most distal level of the radius, and within the carpal tunnel was identified by the most proximal portion of the pisiform. CSA measurements of the median nerve were then performed by using the direct trace function on the MIPAV software. The researcher carefully traced around the outer edges of the nerve epineurium at these two locations. Once the traces were completed, the computer software automatically calculated the CSA. The image number within the imaging sequence for both MRI and sonography was recorded on a customized worksheet to ensure reproducibility for further data analysis.
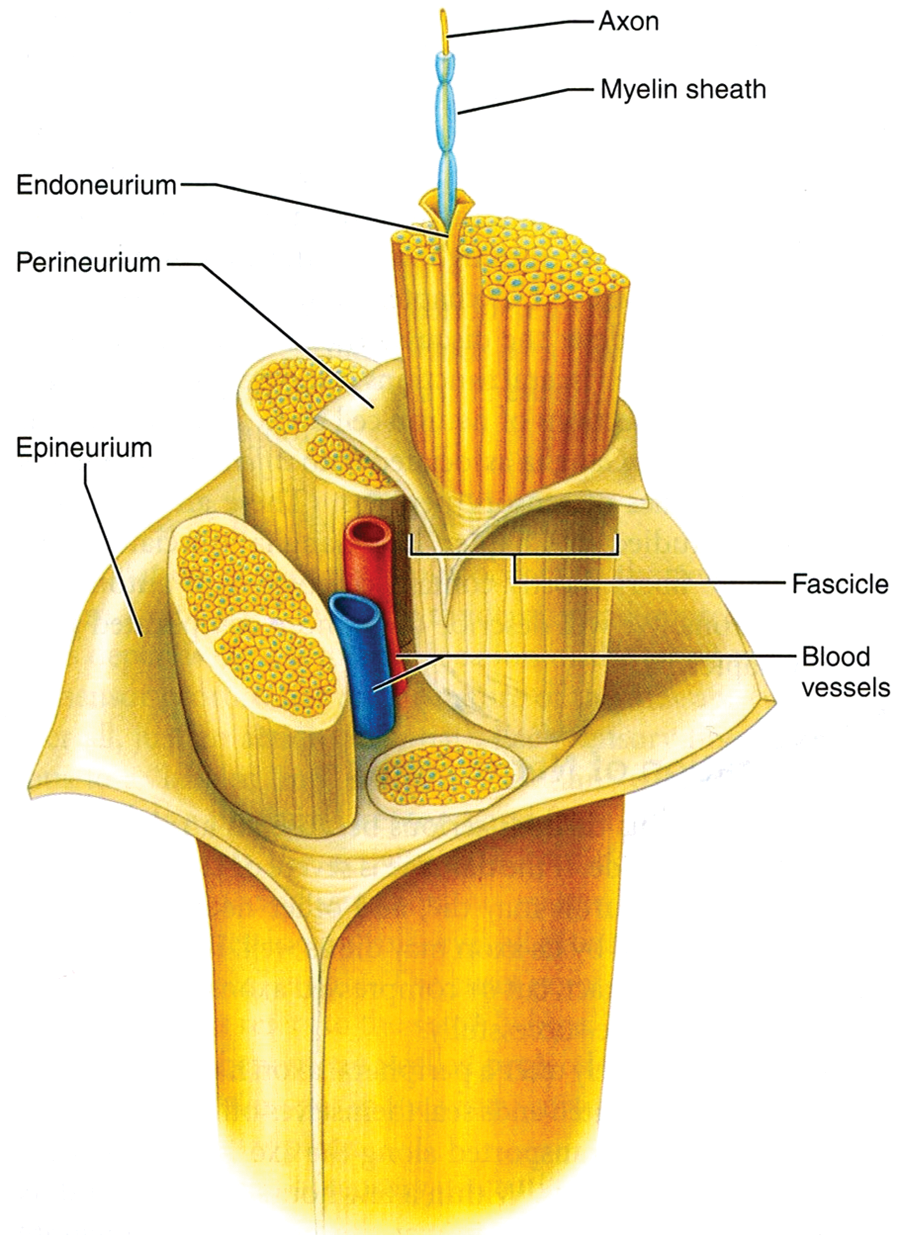
Anatomical structures that make up a typical peripheral nerve.
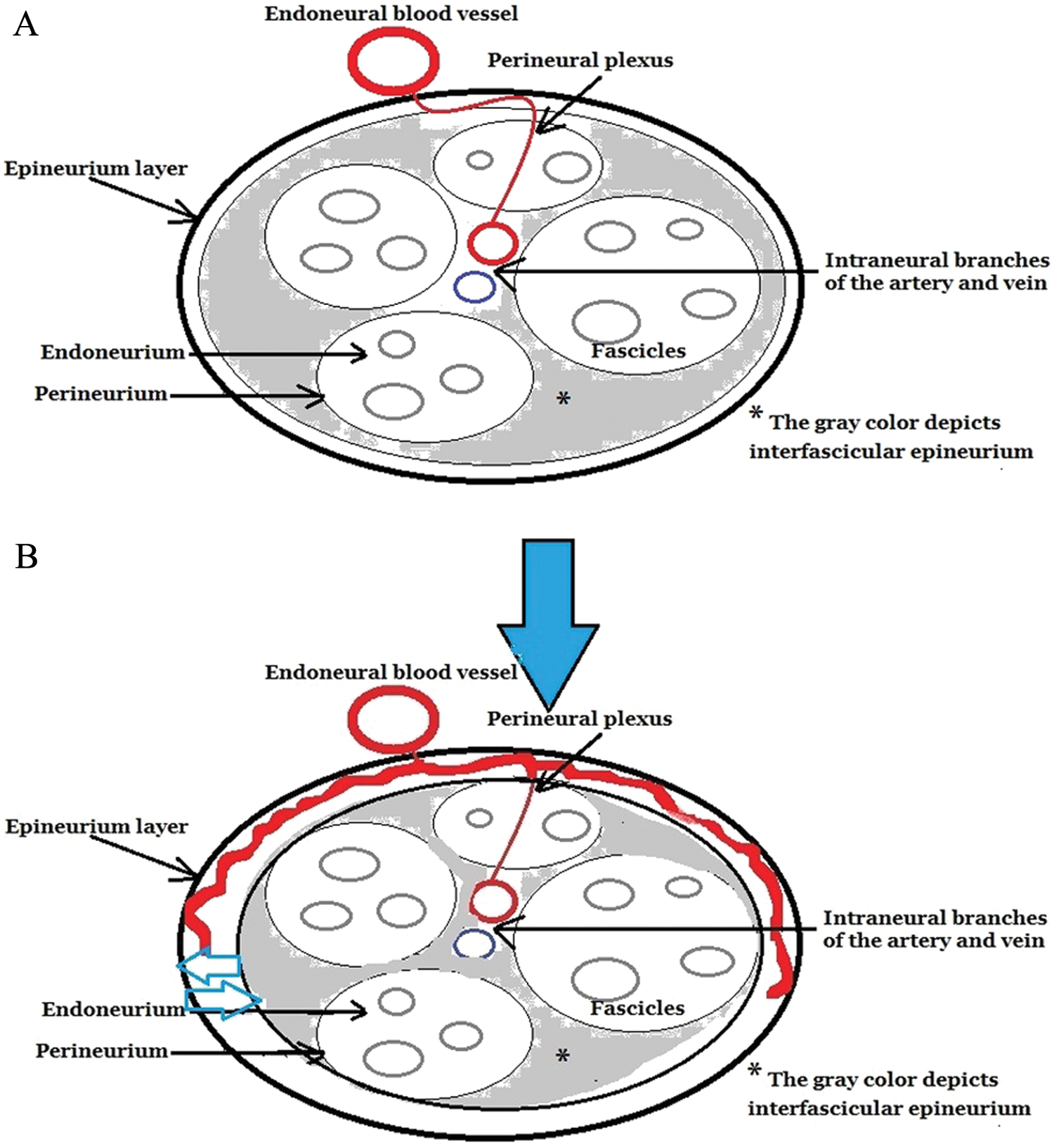
(A) Cross-sectional representative diagram of a normal peripheral nerve and the associated vascularity, without compression of the nerve. (B) A hypothesized cross-sectional representative diagram of an acute mononeuropathy of a peripheral nerve with associated hyperemia. The solid blue arrow depicts the compression on the nerve and the small open blue arrow demonstrates the swelling directed outward on the epineurium.
Sonogram Analysis
The individual cases were analyzed by a credentialed sonographer on the same GE Logiq
Once both an outer CSA and an inner CSA were recorded, the inner CSA was subtracted from the outer CSA to obtain a residual area that represented the epineurial tissue.
Statistical Analysis
Descriptive statistics were generated using Excel 2010 (Microsoft Corp., Redmond, Washington) so that a mean CSA measurement could be objectively compared between both imaging modalities. This analysis also allowed for the detection of measurement changes involving the nerve that could be attributed to work exposure. Mean CSAs were organized and grouped by method of analysis (inner border, outer border, and residual), anatomical location (proximal to the carpal tunnel inlet and within the carpal tunnel), and the time point within the study the measurements were collected (prework vs postwork exposure).
Two-tailed
Results
Fifteen
Mean Direct Trace CSAs for the Subjects’ Left Hand Preintervention and Postintervention by Group
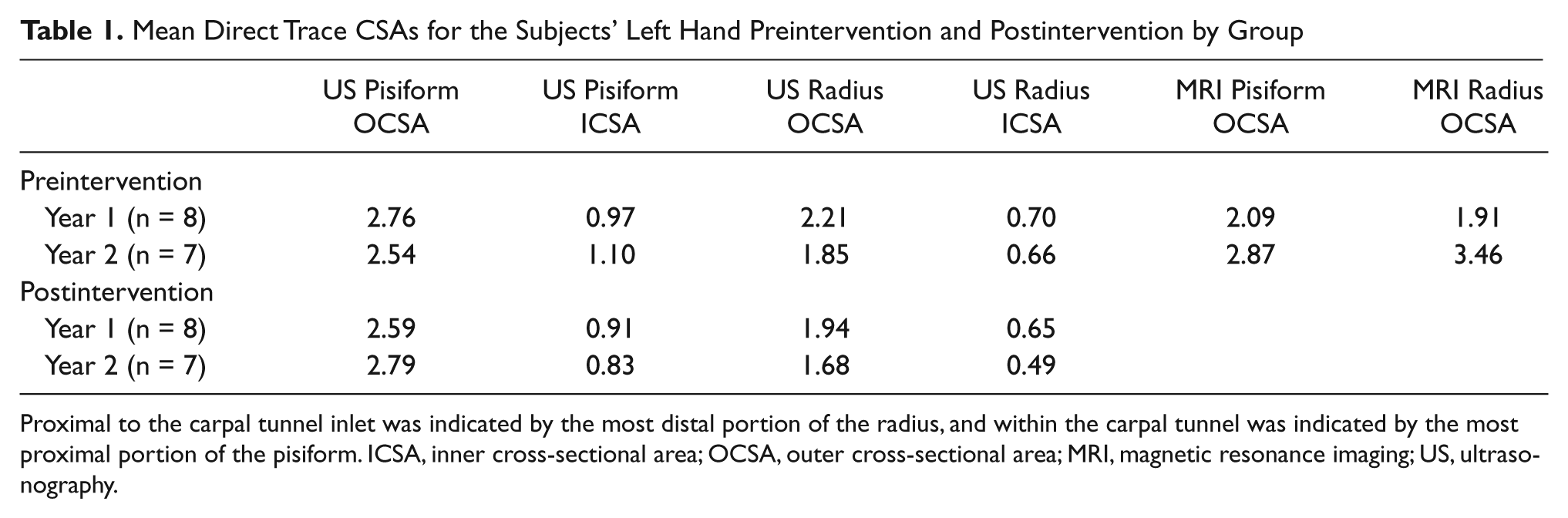
Proximal to the carpal tunnel inlet was indicated by the most distal portion of the radius, and within the carpal tunnel was indicated by the most proximal portion of the pisiform. ICSA, inner cross-sectional area; OCSA, outer cross-sectional area; MRI, magnetic resonance imaging; US, ultrasonography.
At the baseline stage, a
A
The designed intervention was an opportunity for the subjects to perform a repetitive pinching task that could affect the left wrist. Given the statistical differences in CSA from the baseline examination and the conclusion of the working phase at the level of the pisiform, a Pearson correlation was done to determine if changes in CSA measurements were associated with the work exposure and if it would be possible to gauge the changes that might occur in the CSA of the epineurial layer occurring at the level of the pisiform (Figure 1A).
After work exposure, a residual area, or area of the epineurium, at the postwork stage was calculated by subtracting the postwork inner CSA measurements from the postwork outer CSA measurements. This residual area was then correlated with both the postwork outer CSA and postwork inner CSA measurements. The results of a Pearson correlation demonstrated a statistically significant low-level strength of association (
Correlations Between CSAs of Sonographic Residual Area (Epineurial Layer) Outer and Inner CSA by Anatomical Location of the Pisiform, Postwork Exposure

Within the carpal tunnel is identified by the most proximal portion of the pisiform. CSA, cross-sectional area.
Further analysis was done to determine if the work exposure intervention presented a correlation between mean work exposure and the residual area (epineurial layer) of the median nerve at the level of the pisiform in the postwork stage. Again, a Pearson correlation was used and determined that a slightly positive correlation (
Discussion
A mononeuropathy is a disorder that involves a single peripheral nerve and is caused by an injury that can result in short- or long-term debilitation. Unlike the central nervous system, peripheral nerves are stronger and much more resilent.
23
This resilience to injury in a peripheral nerve has been attributed to a series of connective tissue sheaths that help to insulate and enclose the delicate sensory and motor fibers.
23
This preclinical study was designed to investigate the use of cross-sectional area measurements across imaging modalities in order to provide diagnostic information on physiologic change and the stage of injury to the median nerve in a controlled cohort of
This cohort of subjects began with prework measurements that were compared statistically using a
The choice to suspend further analysis of CSAs measured on the outer rim of the epineurium was the controversy and the need to standardize the measurement being explored in this experiment. The analysis of the CSAs measured inside of the epineurium with sonography is highly advocated; however, this could not be compared with MRI since this modality cannot accurately resolve the inner border of the epineurium. Therefore, by subtracting the sonographic outer-border CSA measurements, which included the epineurium, from the inner sonographic CSA measured along the inner border of the epineurium, a residual area representing epineurial layer thickness can be obtained. Outer sonographic measurements were used throughout this experiment due to the statistical similarity when compared with the gold-standard MRI measurements.
The next step was to try to understand the relationship of the inner-border sonographic CSA measurement with the outer CSA (outer-border epineurium) measurements. The connective tissue making up the epineurium, which serves to protect the nerve fibers, is known as epineurial connective tissue. Because sonography is able to accurately determine the CSA of the median nerve while excluding the epineurium,
7
it was possible to derive the interfascicular epineurial connective tissue area measurement, which surrounds the fascicles inside the nerve, by subtracting this from the sonographic measurements, which included the epineurium. This residual area or epineurium measurement would then allow for an analysis of the interaction between the outer and inner CSA measurements and the epineurial connective tissue area, all obtained through sonography. While examining the area of the nerve at the inlet to the carpal tunnel (distal point of the radius), a
Moving to the area of the nerve further distal within the carpal tunnel, identified by the most proximal portion of the pisiform, significant differences were seen comparing the pre- and postwork intervention. Using the calculated epineurial layer, defined as the difference between outer and inner median nerve CSA measurements, correlations could be made to allow for a better understanding of the relationship between the layers of the median nerve involved in the inflammatory process. A comparison of the outer CSA measurements, which included the epineurium, to the epineurial layer area itself demonstrated a strong positive correlation, suggesting that any increase in outer CSA could result in an increase of the epineurium layer area regardless of physiologic change. A second correlation between CSA measurements made along the inner border, which excludes the epineurial layer, and the same epineurial layer area as used in the previous correlation demonstrated a weak positive correlation, suggesting that as the epineurial layer area increased, the inner CSA measurements remained somewhat constant. These findings suggest that as inflammation of the median nerve occurs, it is the enlargement of the epineurial layer causing the overall increase in size, as opposed to the inner area of the nerve, which includes the fascicles and perineurium (Figure 3A,B). Continued study into alternative physiologic factors that could have influenced these results is important to explore, with the cautionary note that a strong statistical correlation does not necessarily represent causation. 24
Previous studies have reported the acute constriction of the median nerve at the site of entrapment resulting in swelling.
25
In the current study, measurements were repeated after work exposure to detect acute measurement changes within the nerve that may be attributed to inflammation. Inflammatory changes in the nerve have been described as eliciting a hypoechoic appearance with sonography and enlargement of the nerve due to endoneural edema.18,26 To determine the optimal diagnostic technique, it was necessary to repeat the statistical comparisons postwork exposure at both locations, proximal to the carpal tunnel inlet and further distal within the carpal tunnel. Repeating the
Because work exposure was the single intervention made between pre- and postmeasurements, the analysis of work exposure should be important to postulating what could be contributing to differences in the imaging measurements. Work exposure was the opportunity to complete the repetitive squeeze task, but it is not a direct measure of the actual force exerted by the subjects. This would be a more direct measure of the impact of the repetitive movements on the median nerve.
The work exposure proved to be influential in the difference between measurements taken before and after work at the anatomical level inside the carpal tunnel. Although the mean hours worked are a relatively “soft” measure of the intervention, this begins to describe the possible physiologic changes that may have been taking place within the median nerve. Increased CSA measurements traced outside the epineurium with concomitant residual CSA enlargement could be a contributor to impingement of the fascicules within the median nerve. Work exposure would be hypothesized to have more impact over a sustained time period, so the fact that this level of association was even demonstrated speaks to the influence of the repetitive pinch-pinch task.
These findings suggest a potential relationship between the physiologic response of the inflammation of the entire median nerve and that of just the epineurium, which is worth further investigation. The lack of entire nerve inflammation suggests that, as epineurial swelling compresses the interfascicular connective tissue area, this swelling in turn compresses the fascicles and affects electrical signaling. This ultimate compressive impact on the electrical signals is the underlying cause of the painful and symptomatic hands that are associated with CTS. 27 This would also explain the lack of sensitivity reported in a previous study in which CSA measurements were obtained excluding the epineurium. 28 Although the entire epineurial area of the nerve is accurately visualized through sonography, it is not the portion of the nerve where swelling is attributed. These data suggest that not only is the epineurium of the median nerve expanding and causing difficulty in moving through the carpal tunnel, but also the expansion internally within the nerve causes a decrease in electrical signals being transmitted distally to the hand and fingers.
An additional factor that can be attributed to intrafascicular compression is Renaut bodies. Renaut bodies are fibroblasts that are believed to be derived from endoneurial connective tissue and are often noted to be deposited around the median nerve as a response to repeated mechanical stress.29,30 In a cadaver study, Renaut bodies were found on all five females, and the deposition consistently surrounded the fascicles of the median nerve segments removed for microscopic investigation. 31 The presence of Renaut bodies is another potential cause for an increase in the epineurium and constriction of the perinerium from the connective tissue. Renaut bodies’ deposition has been linked with the development of MMN. 25
When examining the layers of a median nerve that has incurred a microinjury, the endonerium would react acutely under hyperemic conditions 26 and then progress to tissue repair once the trauma is relieved or, if the trauma is not relieved, become ischemic. An injury longitudinally can result in regeneration of nerve fibers depending on the distance that regrowth must take place. 32 Continued study is needed to address the reaction of the nerve tissue surrounding the individual fascicles, as physiologic changes may be varied.
Limitations
This study is limited foremost by the level of evidence, which is correlative but not necessarily causative, and should be considered preclinical. In addition, a direct measure of the force during the repetitive squeeze task was not reported and would have made this a more robust analysis. Last, the positioning of the subjects for MRI in the warm plastic cylinder was difficult and could be the source of problems with consistent imaging of the wrist.
Conclusions
While comparing MRI CSA measurements and sonography CSA measurements, it was determined that without any manipulation of the nerve, these two measurements suggest no statistical difference and can be accurately compared. Because MRI is defaulted to as the optimal diagnostic tool for scanning the wrist, it was possible to continue with the experiment using sonography as an optimal imaging technique.
Because of the statistical difference inside the carpal tunnel following work exposure, the next step taken was to correlate both inner and outer CSA sonographic measurements with the epineurial layer area. Given the strong positive correlation between outer CSAs and the epineurial layer, combined with the weak positive correlation between inner CSAs and the epineurial layer area, it can be suggested that an increased size in the epineurium may be the cause of many CTS symptoms. This relationship is critical in demonstrating the pathophysiologic change of the nerve. Possibilities suggested for this physiologic result were intraneural edema, which could be adding to the inflammation of the median nerve and increasing the compression, 7 and Renaut bodies.25,29-30,32-34 Inflammation of the nerve was best displayed using a T2 MRI, which would be the next subsequent step in this research. Another important step would be to compare the blood flow in the perineural plexus using Doppler of the CSA areas to document any increased vascularity that could be linked to an increase in CSA. 33
Future Directions
Although this study was able to report statistically significant findings that somewhat contradict previous studies, further research is needed at the level of the intraneural vessels and surrounding tissue. The results of this pilot study should be considered preclinical evidence and as such cannot be generalized. Our team is committed to further investigation and replication of this study to advance the future of median nerve imaging and CTS diagnosis.
To move this line of research forward and determine whether musculoskeletal sonography can indeed be accurately compared with MRI measurements of proximal and distal segments of the wrist area, this study needs to be replicated with more participants and also translated to a human study. This current study involved only 15 animal subjects, yielding 30 scans at 60 locations to correlate, and more clinical data are needed if conclusions are to be drawn. Translating this work to humans is the next logical step in the progression of the research.
Another suggested direction that this research could undertake would be to compare these two imaging modalities using a direct overlay method. By manipulating images and matching anatomical landmarks, there would be less discrepancy in the measurements obtained because this would further facilitate a direct comparison. This would also remove the derived measurement of the connective tissue area and provide a more direct measurement of the area and shape during all time points throughout the study.
An intermediate step in the current study would be to obtain histological samples of the median nerve through dissection. This gold standard of measurement would provide an even higher level of comparison between the two imaging modalities. It would also allow for a search for the deposition of Renaut bodies. Fueling further research based on these preliminary data may affect the current ACR Appropriateness Criteria and influence an updated version that would reflect a more effective use of musculoskeletal sonography in evaluating the painful wrist for patients. This would advance the diagnosis capabilities of musculoskeletal sonography.
Musculoskeletal sonography also offers additional techniques for the detection of acute nerve trauma such as spectral Doppler. In an acute traumatic stage, the nerve will become hyperemic, therefore causing increased vascularity. 35 This physiological response can be detected and visually sampled at any portion of the nerve.36 The use of real-time scanning and the ability to see the live movement of the nerve and the way it interacts with surrounding tissues generates additional ways to monitor the physiologic process of nerve repair.
Footnotes
Acknowledgements
The research team would like to thank our veterinary technicians, Stephanie Moran and Rebecca Slattery, for their invaluable support and hard work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was made possible by a grant from the National Institute of Occupational Safety and Health R21 OH009907-02 and the ASRT Foundation.
