Abstract
Hydatid disease, a zoonotic infection caused by Echinococcus granulosus, is endemic in pastoral regions and typically transmitted through exposure to infected canines (dogs), contaminated food, and water. A patient case is presented of a 29-year-old woman from urban Saudi Arabia who had chronic upper abdominal pain and lacked a classic exposure history. An Acuson ultrasound system (Siemens Healthineers, Erlangen, Germany), equipped with a convex transducer (3.5–7 MHz), revealed a pathognomonic thick-walled hepatic cyst that measured 88 × 78 mm, along with daughter cysts. This diagnostic finding was confirmed by a 64-slice computed tomogram (Siemens Healthineers), as well as demonstrating splenic extension that measured 120 × 90 × 70 mm and a calcified satellite lesion. This case highlighted the diagnostic challenges in non-endemic settings and underscores the critical role of multi-diagnostic imaging in detecting complications and guiding surgical planning. This is particularly important when adjacent organ involvement alters clinical management.
Hydatid disease (cystic echinococcosis) is a zoonotic infection caused by the larval stage of Echinococcus granulosus, a parasitic tapeworm endemic in pastoral regions such as North Africa, the Middle East, and South America.1,2 In 50% to 70% of reported cases, the liver is the main organ affected, followed by lungs with 30%. 3 The spleen or peritoneum rarely is affected during this disease. 4 Hepatic hydatids can even stay undiagnosed or asymptomatic for many years. In such cases, severe complexities could be developed including rupture, secondary infection, or mass effect leading to abdominal pain, anaphylaxis, or obstructive jaundice. 5
Currently, the diagnosis of this condition relies on diagnostic imaging. Sonography is the primary diagnostic imaging technique to detect this pathology, due to its high sensitivity (e.g., 90%–95%) for detecting pathognomonic features, such as thick walls, septations, and daughter cysts. 6 Computed tomography (CT) and magnetic resonance imaging (MRI) are reserved for assessing complications, extrahepatic extension, or preoperative planning. In the diagnostic process, differential considerations for large hepatic cystic lesions include simple cysts, pyogenic or amoebic abscesses, cystic metastases, biliary cystadenoma, and other parasitic infections. Sonography plays a pivotal role in narrowing these possibilities by enabling dynamic evaluation of cyst morphology, internal architecture, and organ relationships, while identifying characteristic features such as daughter cysts or hydatid sand that are well-documented hallmarks of hydatid disease.7,8
Despite characteristic diagnostic imaging signs, challenges in detection persist in non-endemic regions, due to low clinical suspicion, atypical presentations, or mimicry by cystic neoplasms. 9 Misdiagnosis of hydatid disease remains common in non-endemic areas, with up to 40% of cases initially mistaken for malignancies or abscesses. 9 This case study describes a rare occurrence that involved an infected female patient, from Morocco, with a large hepatic hydatid cyst that extended into the spleen. It underscores the critical role of multi-imaging techniques (i.e., sonography and CT) in identifying atypical anatomical involvements. Furthermore, it also highlights the significance of sonography in differentiating hydatid disease from other cystic lesions and emphasizes the importance of epidemiological context in diagnosis.
Case Report
A 29-year-old Moroccan woman presented with a 6-month history of dull, persistent upper abdominal pain. She reported no associated fever, vomiting, weight loss, jaundice, or changes in bowel habits. Her medical history was unremarkable, with no known chronic illnesses, recent travel to rural areas, or contact with livestock or dogs, which is a notable absence of classic risk factors for hydatid disease.
The initial abdominal sonogram was completed with an Acuson (Siemens Healthineers, Erlangen, Germany) and convex transducer, operating at 3.5–7 MHz. The examination revealed a large cystic lesion that measured 88 × 78 mm and had thick outer walls (>3 mm). The cystic lesion arose from the inferior aspect of the left hepatic lobe in the epigastric region. The lesion contained multiple smaller daughter cysts, a hallmark feature of hydatid disease (See Figure 1).
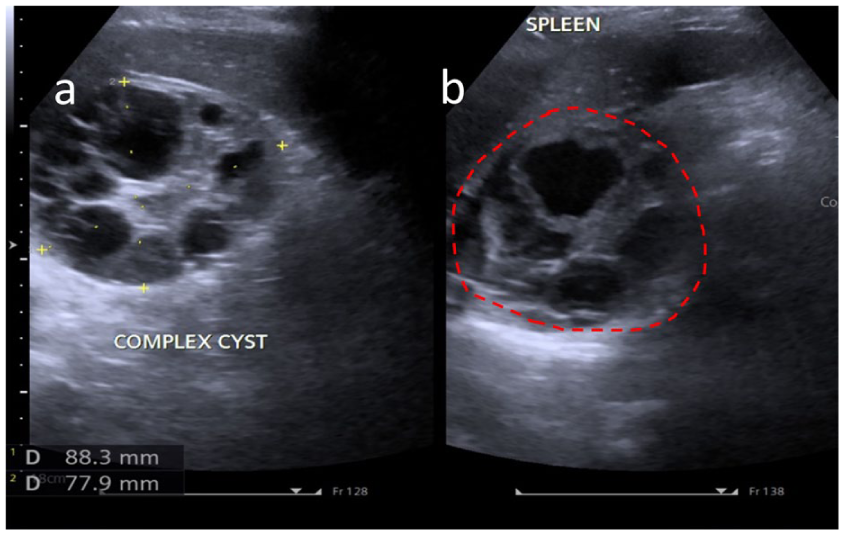
A large complex cyst in the epigastric region is shown with sonographic characteristics of hydatid cyst. These sonographic findings are suggestive of hepatic hydatid cysts; the larger one is extending to involve the superior pole of spleen.
To further characterize the lesion, a contrast-enhanced CT examination was completed on a 64-slice Sensation (Siemens Healthineers) unit and provided axial, coronal, and sagittal imaging planes of the liver. This confirmed a complex cyst measuring 120 × 90 × 70 mm in the left hepatic lobe with internal septations and daughter cysts, extending into the superior pole of the spleen (See Figures 2 and 3). A smaller calcified cyst, measuring 20 × 18 mm, was in segment IV of the right section of liver. This was suggestive of chronic disease shown in coronal and sagittal imaging planes (See Figure 4 and 5).
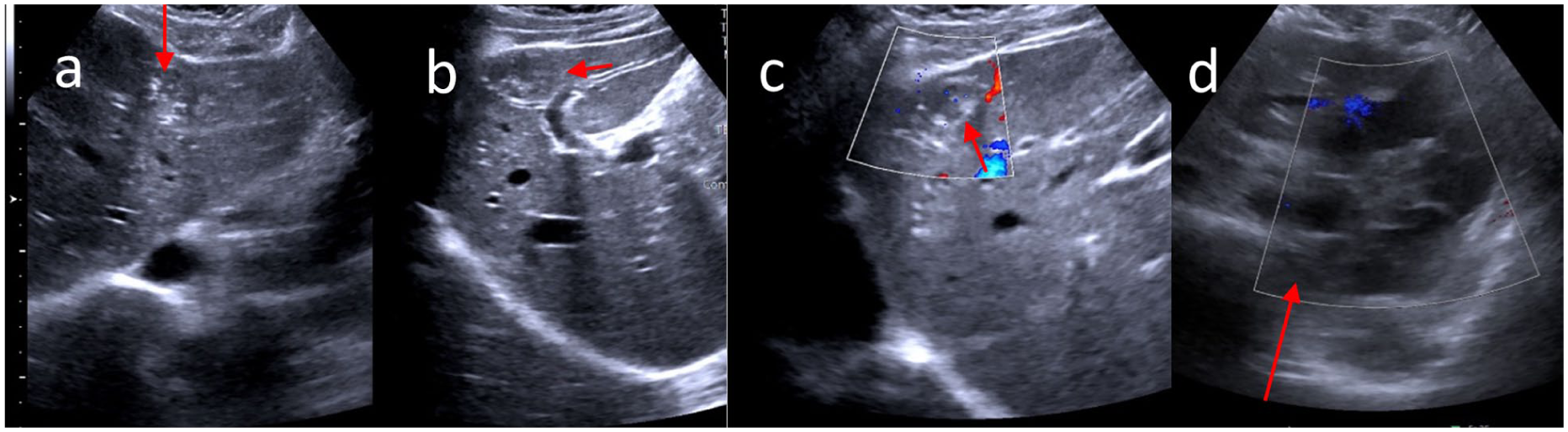
Sagittal sonographic imaging plane images were obtained from dome of diaphragm to pubic symphysis of this patient. These images represent key abnormalities such as hepatic hydatid cyst. Panels (a) and (b) show a hypodense area with calcific margins in the fourth segment of liver seems to be a hepatic mass. (c) Doppler demonstrated no vascular flow in the mass. (d) A large mass was seen in the epigastric region described as a large cyst with multiple internal daughter cysts.
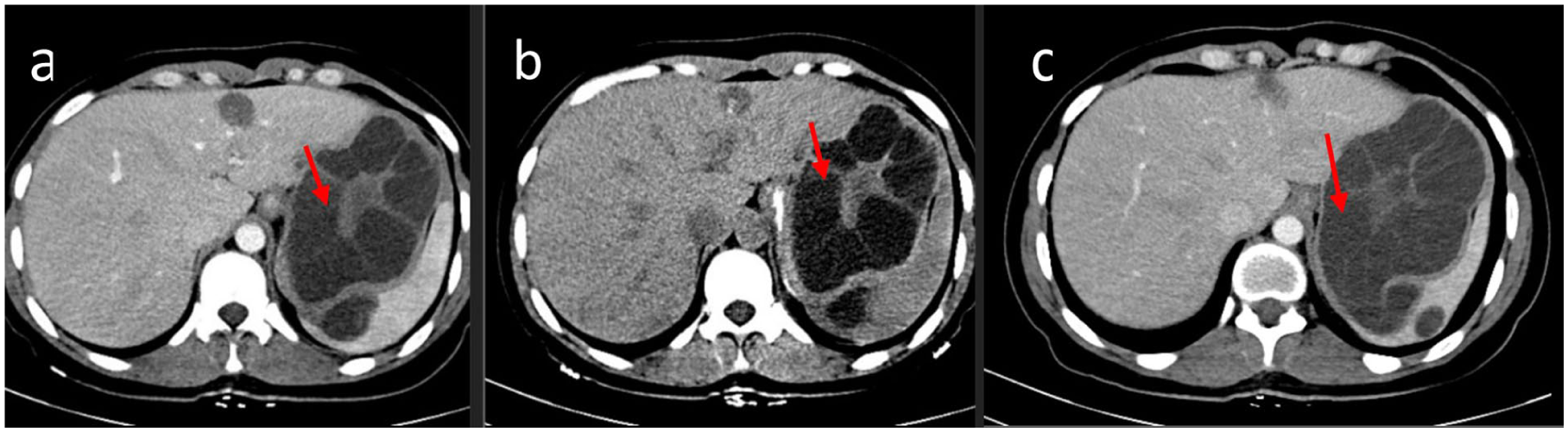
(a) A contrast computed tomographic (CT) scan of the upper abdomen suggested the presence of hepatic hydatid cysts. A calcified, small cystic lesion representing a hyperdense focus, located in the left lobe. Another large, thick-walled cystic lesion with multiple internal daughter cysts. (b) A non-contrast CT image of the upper abdomen of patient. (c) Lesion is seen on the lower segment of left lobe of liver and extending to involve superior pole of spleen. These diagnostic CT findings were suggestive of two hepatic hydatid cysts. No calcification was observed in the second large cystic lesion. The overall diagnostic imaging findings were suggestive of two hepatic hydatid cysts.
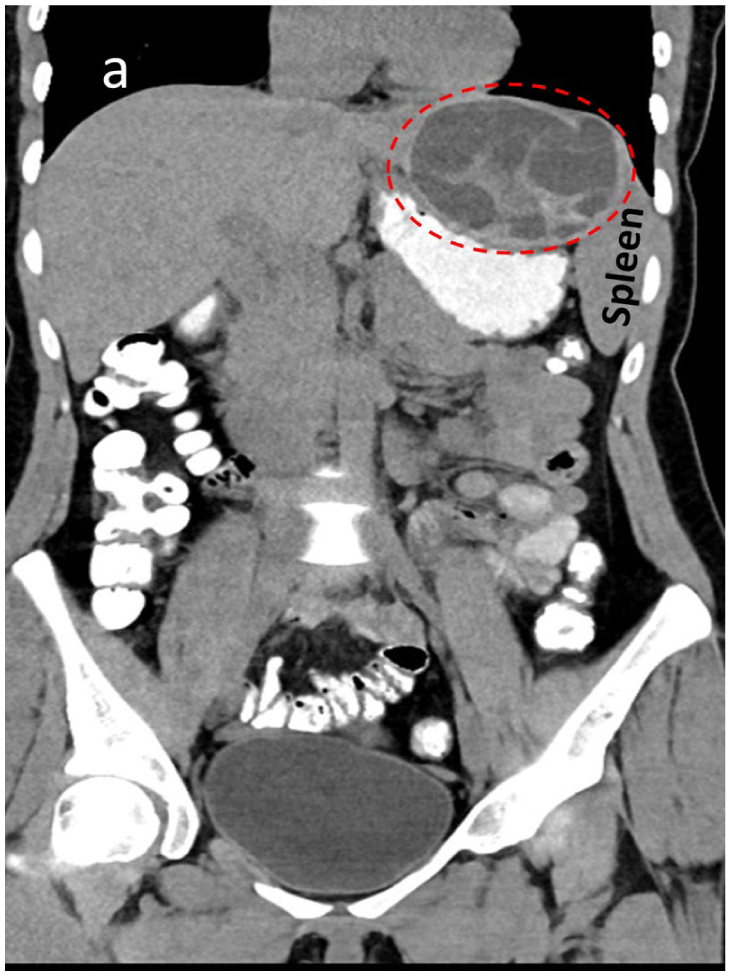
A hypodense large (120 × 90 × 70 mm), thick-walled cystic lesion is noted on this coronal computed tomographic image. The lesion was in the left hepatic lobe and contained multiple daughter cysts, extending from the left lobe of the liver to the upper part of spleen. In addition, it had a smaller calcified cyst in segment IV of right liver.
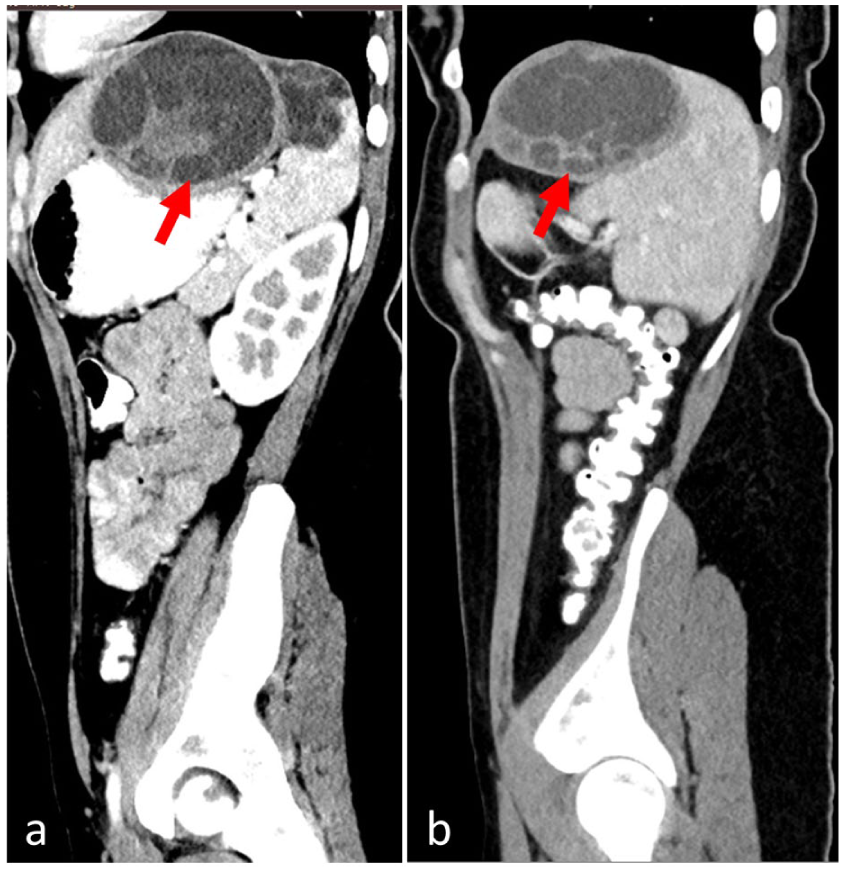
A computed tomographic (CT) sagittal plane image demonstrated multiple cystic lesions with anterior-to-posterior depth, suggestive of hydatid cyst involvement.
Differential considerations included liver abscess, simple hepatic cyst, biliary cystadenoma, and cystic metastases. These were excluded based on the diagnostic imaging features and clinical presentation. The lesion’s thick walls, internal septations, and daughter cysts distinguished it from a simple cyst. Absence of fever or elevated inflammatory markers helped rule out pyogenic abscess. No biliary dilation, peritoneal seeding, or signs of rupture were present.
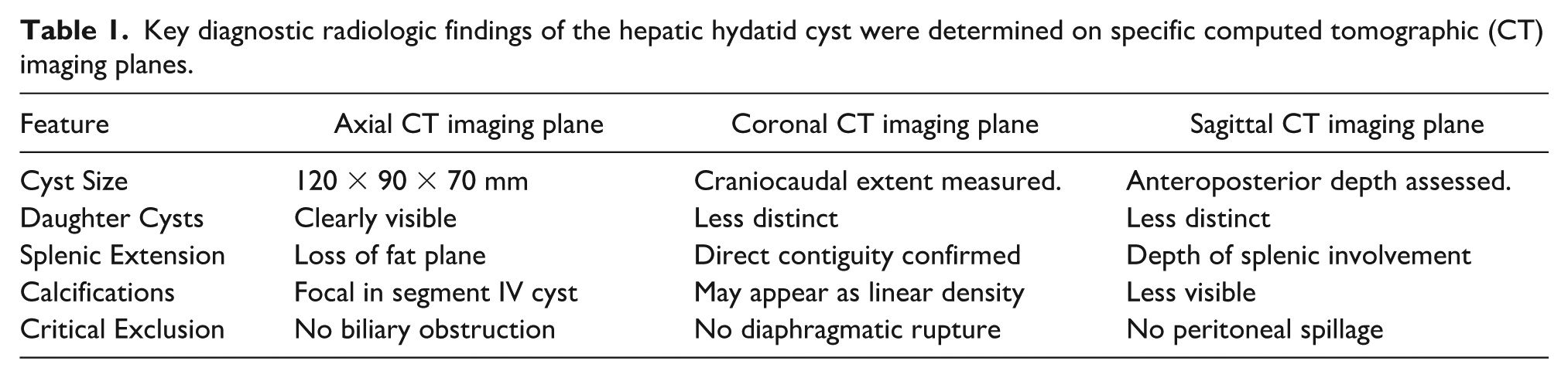
Multiplanar CT imaging confirmed the hepatic hydatid cyst with splenic extension, demonstrating daughter cysts most prominently on axial views. Calcifications in segment IV of the liver were better appreciated on sagittal and coronal imaging planes (See Figures 2 and 4). Table 1 summarizes the key diagnostic radiologic findings. The rare hepatic splenic involvement significantly impacts surgical planning due to increased risk of intraoperative complications compared to isolated hepatic cysts.
Key diagnostic radiologic findings of the hepatic hydatid cyst were determined on specific computed tomographic (CT) imaging planes.
Surgical Management
An exploratory laparotomy was performed on this patient. A large hydatid cyst measuring 12 × 9 cm was identified in the left hepatic lobe, along with a smaller 3 × 2 cm cyst in segment IV of the right lobe. No rupture into the biliary tree or peritoneal cavity was observed, and other abdominal viscera appeared normal. A midline incision was made, and the peritoneal cavity was entered under sterile conditions. Packs soaked in hypertonic saline were placed around the liver to prevent spillage. The cysts were aspirated and irrigated with hypertonic saline, and the germinative membranes with daughter cysts were evacuated completely. Each cavity was inspected for biliary communication, after which pericystectomy was performed. The residual cavity was managed with omentoplasty. Thorough peritoneal toileting with warm saline was performed, hemostasis was secured, and a subhepatic drain was placed. The abdomen was then surgically closed in layers.
Histopathological Findings
The surgical specimens consisted of fragments of cyst wall and membranes from both hepatic hydatid cysts and were submitted for examination. On evaluation, multiple whitish, laminated membranous fragments were observed, the largest measuring 4 × 3 cm, along with several small translucent cystic fragments that exhibited collapsed walls and granular contents consistent with “hydatid sand.” No malignant or solid tumor components were identified. Microscopic analysis revealed acellular laminated eosinophilic cyst walls characteristic of hydatid disease, with an inner germinal layer containing brood capsules and multiple viable protoscolices. The surrounding tissue demonstrated a fibrocollagenous reaction accompanied by lymphocytic infiltration and multinucleated giant cell reaction. Importantly, no evidence of dysplasia or malignancy was detected.
Discussion
The present patient case underscores the diagnostic approach of hydatid disease in non-endemic urban settings, where atypical presentations may delay detection. It also highlights the pivotal role of diagnostic multi-imaging, such as sonography and CT, in guiding clinical management. It is also particularly useful when anatomical complications, such as splenic extensions, necessitate precise preoperative assessment. The epidemiological context of hydatid disease is particularly noteworthy. Cystic echinococcosis (CE) is a notifiable disease monitored by the Saudi Ministry of Health through surgical records from public hospitals in Morocco. Between 1980 to 1992 and 2003 to 2008, the national registry documented 23, 512 surgical CE cases, showing a rise in mean annual incidence from 3.6 to 5.2 per 100,000 population during this period. 10 A meta-analysis of 28 cross-sectional and 14 case-control studies revealed key CE transmission pathways that direct contact with dogs accounted for 26.1% of cases (confidence interval [CI] = 13.8%–39.6%), while contaminated water sources contributed to approximately 29.4% (CI = 12.1%–51.7%). 11 Foodborne transmission was responsible for 23.4% of infections (CI = 2.1%–47.3%). The remaining 21.1% of cases likely involved environmental exposures, although specific studies on this pathway were lacking. These findings highlight the multifactorial nature of CE transmission in endemic regions.
Sonographically, hepatic hydatid cysts can present with a wide spectrum of appearances depending on the stage of the disease, the presence of complications, and the World Health Organization–Informal Working Group on Echinococcosis (WHO-IWGE) classification. 12 In early stages, the cyst may appear as a well-defined, anechoic lesion with a thin wall (e.g., cystic echinococcosis 1 or CE1), while more advanced stages often show internal septations and multiple daughter cysts giving the classic “honeycomb” or “rosette” pattern (e.g., cystic echinococcosis 2 or CE2). Detached laminated membranes floating within the cyst fluid may produce the “water lily sign” (e.g., cystic echinococcosis 3a or CE3a), whereas partially degenerated cysts with mixed echogenicity can mimic complex masses (e.g., cystic echinococcosis 3b or CE3b). In chronic or inactive stages, the cyst may appear solidified or calcified, producing echogenic foci with posterior acoustic shadowing (e.g., cystic echinococcosis 4 or CE4).13,14 Additional diagnostic findings may include hydatid sand with shifting echoes upon patient repositioning, and in complicated cases, irregular walls, heterogeneous internal contents, or adjacent organ involvement may be visualized.15,16
Hydatid disease progresses slowly, often over several years, as the larval stage of E. granulosus develops into fluid-filled cysts within the host. After ingestion of parasite eggs, the oncospheres penetrate the intestinal wall, enter the portal circulation, and most commonly lodge in the liver, which serves as the first filter. The disease typically begins as a single cyst, but multiple cysts or multiorgan involvement can occur, especially in untreated or recurrent cases. Over time, cyst growth exerts a mass effect on surrounding tissues, potentially causing pain, obstruction, or functional impairment. The liver is affected in 70% of cases, typically in the right lobe due to portal blood flow patterns. 17 If the larvae bypass the hepatic filter, they may reach the lungs, which act as the second filter. From there, systemic dissemination can occur via hematogenous spread, leading to involvement of other organs such as the spleen, kidneys, peritoneum, brain, bones, heart, and muscles, although these are relatively rare sites.4,18 Complications can arise from rupture into adjacent structures such as the biliary tree, pleural space, or peritoneal cavity, leading to secondary infection, dissemination of protoscolices, or severe allergic reactions, including anaphylaxis. 19 Chronic disease may also result in calcified, inactive cysts, which can persist as residual lesions even after treatment.
In the present case study, the patient presented with chronic dull upper abdominal pain, which highlights the diagnostic and management challenges of hepatic hydatid disease, particularly in populations from endemic regions. Diagnostic imaging played a pivotal role in determining the differential diagnosis. Despite the absence of classic risk factors such as direct animal contact, her epidemiological background of Morocco being a highly endemic zone for E. granulosus raised strong suspicion for cystic echinococcosis. Sonography revealed a large, thick-walled cystic lesion, in the left hepatic lobe, containing multiple daughter cysts, as well as smaller calcified cyst in segment IV of right liver. These pathognomonic “spoke-wheel” or “waterlily” like appearance cysts have been reported to have 98% specificity for hydatid disease. 4 The CT examination further confirmed hepatic involvement with extension into the spleen, a rare but serious complication seen in less than 2% of abdominal hydatid cases. 20 This anatomical involvement significantly alters surgical planning, as studies have reported higher complication rates in cases with adjacent organ involvement compared to isolated hepatic cysts.21,22
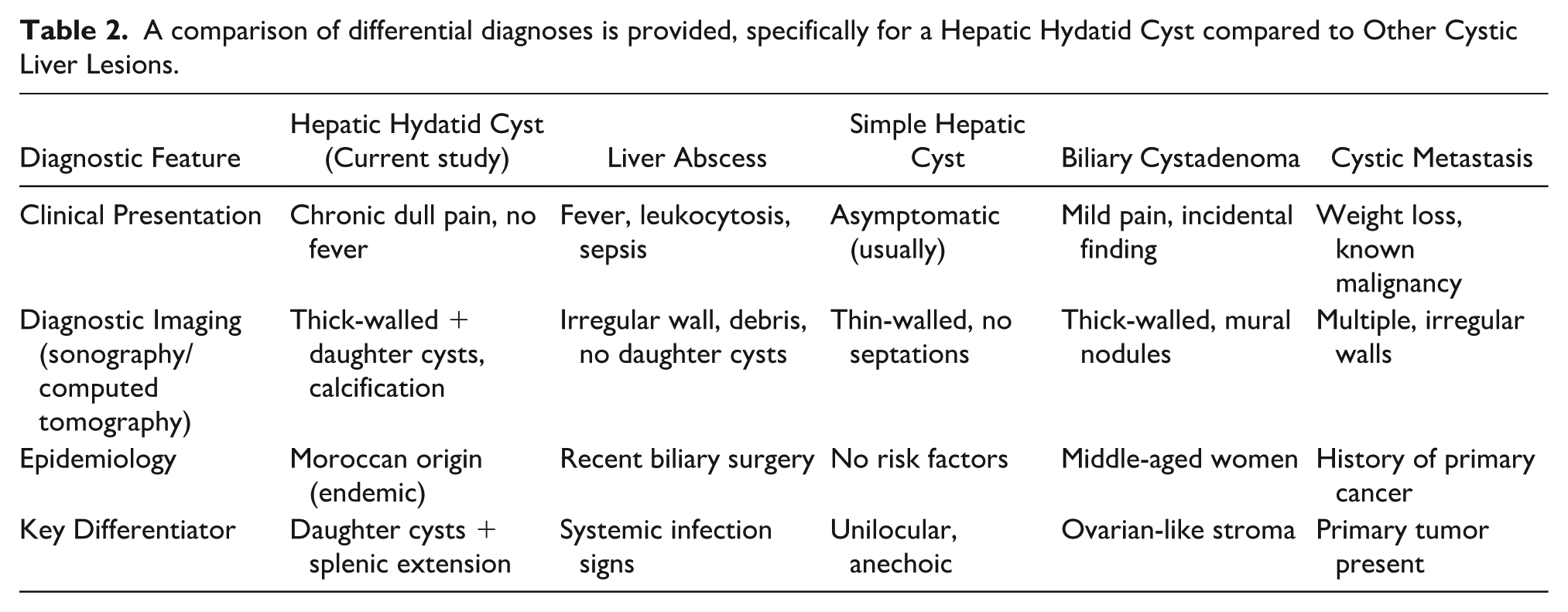
The differential diagnosis included liver abscess, simple hepatic cysts, biliary cystadenoma, and cystic metastases, but these were effectively excluded based on imaging characteristics and clinical presentation. Simple hepatic cysts, for instance, are typically thin-walled and anechoic, lacking septations or daughter vesicles, while abscesses usually present with fever and heterogeneous internal debris. Pyogenic liver abscesses typically present with systemic symptoms such as fever (e.g., 78%–90% of cases) and elevated inflammatory markers (e.g., C-reactive protein [CRP], erythrocyte sedimentation rate [ESR]). 23 Imaging characteristics further differentiated pyogenic liver from hydatid disease. While hydatid cysts demonstrate well-defined walls with daughter cysts, liver abscesses typically show irregular margins, internal debris, and heterogeneous enhancement on contrast imaging. Biliary cystadenomas are rare, accounting for less than 5% of cystic liver lesions. 24 While they may demonstrate thick walls like hydatid cysts, a key distinguishing feature is the presence of mural nodules, which can be noted in 60% to 70% of cases. Other diagnostic features are the internal vascularity, interrogated by Doppler, and the absence of daughter cysts or calcifications unless malignant transformation has occurred. In the present case, the imaging findings did not effectively support biliary cystadenoma, as there were no enhancing mural nodules, and the presence of both daughter cysts and calcifications strongly favored hydatid disease. Table 2 is provided and systematically compares the characteristic features of the current study patient’s hepatic hydatid cyst with other common cystic liver lesions. This comparison highlights the clinical, radiologic, and epidemiological findings that confirm the diagnosis of cystic echinococcosis while excluding alternative pathologies.
A comparison of differential diagnoses is provided, specifically for a Hepatic Hydatid Cyst compared to Other Cystic Liver Lesions.
Biopsy or needle aspiration of hydatid cysts is generally not recommended due to the significant risk of intra-cystic fluid leakage, which can lead to spillage of viable protoscolices, into surrounding tissues.5,25 This not only increases the risk of secondary echinococcosis through local dissemination but can also trigger a severe anaphylactic reaction, which may be life-threatening. In addition, puncturing the cyst disrupts its protective laminated membrane, potentially complicating subsequent surgical excision and increasing the likelihood of recurrence. For these reasons, diagnosis is primarily based on imaging findings supported by serology.
Treatment for hepatic hydatid disease usually involves surgical removal, either by total pericystectomy or partial hepatectomy. 26 A less invasive option is the procedure of puncture, aspiration, injection, reaspiration (PAIR), which is combined with anti-parasitic medicine, such as albendazole. 27 However, the disease can reoccur, especially if the cyst fluid spills during surgery or if the removal is incomplete. 28 Using albendazole before and after surgery greatly lowers this risk.29,30 If the disease is not treated or is misdiagnosed, serious problems can occur. These include rupture into the bile ducts, abdomen, or chest, secondary infection, or even a dangerous allergic reaction called anaphylaxis. Detecting the disease early with diagnostic imaging and treating it promptly is the best way to prevent complications and ensure long-term recovery.
In the present case study, surgical management involved pericystectomy with omentoplasty following aspiration and irrigation with hypertonic saline, performed with meticulous care to avoid spillage. Intraoperatively, no biliary communication or peritoneal rupture was identified, indicating an uncomplicated surgical course. Histopathology confirmed hydatid disease, which demonstrated the classical laminated cyst wall, germinal membrane with brood capsules, and viable protoscolices. Importantly, the absence of intraoperative or postoperative complications emphasizes the role of preventive measures such as saline-soaked packing and peritoneal toileting in achieving a favorable outcome. It has been reported that biliary leakage is a notable complication, affecting about one in ten treated children, making it the most frequent surgical complication observed. 31 In contrast, the current case demonstrated that adherence to meticulous operative precautions can reduce such risks, underscoring the importance of standardized surgical protocols. Therefore, the current case not only exemplifies classic diagnostic imaging and pathology concordance but also illustrates how careful surgical technique directly impacts prognosis, aligning with global evidence on best practices in hydatid disease management.
Conclusion
This unique case exemplified both the classic and evolving aspects of hepatic hydatid disease, highlighting the critical value of high-resolution diagnostic imaging (e.g., sonography and CT) in detecting atypical features such as splenic extension, which significantly altered management. It emphasizes the importance of maintaining a high index of suspicion for hydatid disease in patients from endemic regions, even without a classic exposure history. Early recognition through diagnostic imaging facilitates timely diagnosis, enhances clinician awareness for targeted screening in high-risk demographics, and helps improve diagnostic accuracy while preventing complications in non-endemic settings.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study because all patient information was de-identified.
Informed Consent
Written informed consent was obtained from the subjects before the release of the report for review.
