Abstract
Objective:
The aim of this study was to assess whether a general practitioner (GP), without cardiac point-of-care ultrasonography (POCUS) experience, could feasibly and accurately estimate left ventricular ejection fraction (LVEF), using AI guidance in both image acquisition and interpretation.
Materials and Methods:
A feasibility study was conducted with adult patients who had undergone recent standard echocardiography. A GP used a handheld ultrasound device (GE HealthCare Vscan SL), connected to an iPad Mini (with Vscan Air app and Caption AI), to obtain cardiac images and use AI to assess LVEF. Feasibility was defined as successful image acquisition from a modified POCUS set of views involving apical four-chamber, apical two-chamber, and parasternal long-axis views. Diagnostic agreement was assessed against reference LVEF from a standard echocardiography.
Results:
Among 25 patients (median age 74 years, 52% male), all three views were successfully acquired in 76% of cases using a maximum examination time of 10 minutes. Median AI-POCUS LVEF was 50% (IQR: 43–55) compared to 48% (IQR: 42–53) by reference (p = .14), with strong correlation (R = 0.95, 95% CI: 0.87–0.98). Bland–Altman analysis showed a mean difference of 1.0% (95% CI: –0.3 to 2.3), with acceptable agreement. Sensitivity and specificity for LVEF <50% were 90% (55.5–99.8) and 100% (66.4–100.0), respectively.
Conclusion:
AI-guided handheld cardiac ultrasound with a limited set of views for LVEF assessment may be feasible in a primary health care setting.
Echocardiography has evolved dramatically since its initial use to assess pericardial effusion, using a neurologist’s ultrasound transducer. 1 Today, it is a cornerstone of cardiovascular diagnostics and plays a central role in the evaluation and management of conditions such as heart failure, atrial fibrillation, and valvular disease. 2 However, the growing demand for echocardiographic assessments increasingly exceeds the capacity of hospital-based services and trained cardiac specialists.2,3 In the United Kingdom alone, more than 1.7 million outpatient echocardiograms are performed annually, placing considerable strain on the diagnostic infrastructure and contributing to longer waiting times. 3 Recent advances in handheld ultrasound devices and artificial intelligence (AI) offer an opportunity to address a potential “diagnostic bottleneck.” AI-enabled platforms can now guide (nonexpert) users through real-time image acquisition and generate automated measurements, at the point of care. 4 These developments may enable community-based clinicians, including general practitioners (GPs), to perform focused cardiac assessments, facilitating timelier triage and diagnosis in primary care settings.
This study was designed to evaluate the feasibility and diagnostic agreement of AI-guided handheld ultrasound device for a key cardiac parameter, left ventricular ejection fraction (LVEF), based on selected views from a bedside cardiac assessment, and performed by a GP without prior experience in point-of-care ultrasound (POCUS).
Materials and Methods
This prospective study involved a paired study design for diagnostic tests and followed the CONSORT extension guidelines for reporting nonrandomized pilot and feasibility studies. 5 It was conducted between November 2024 and May 2025 at a large primary care center in a diverse, underserved community in Amsterdam, Netherlands.
The recruitment of eligible participants was based on adults (≥18 years) who had received a standard transthoracic echocardiogram by a cardiologist (as part of routine care), within the previous month. Patients were included during aftercare follow-up consultations with their GP. A brief handheld cardiac ultrasound examination of up to 10 minutes was performed during these visits, either in the GP office or at home.
AI-POCUS: An Examination Protocol Based on a Modified Set of Views for LVEF Assessment
Sonography was performed using the Vscan Air SL (GE HealthCare, Waukesha, WI), a wireless handheld transducer (CE-marked, FDA-approved) paired with an iPad Mini 5 (Apple Inc. Cupertino, CA) running the Vscan app integrated with Caption AI AutoEF software (FDA 510[k] cleared). This software offers two types of AI support: real-time image acquisition guidance and automated LVEF estimation. Guidance goes as follows, as the user manipulates the transducer, the software provides real-time feedback to optimize transducer positioning and image quality, providing instructions such as rotate clockwise, or tilt, or shift laterally, which is based on live analysis of the video stream. This enables consistent acquisition of standard echocardiographic views even by users with limited prior sonography experience. The software allows the use of a rapid protocol that can be performed to estimate LVEF in a matter of minutes, using a three-view approach.
These views involved the apical four-chamber (AP4), apical two-chamber (AP2), and parasternal long-axis (PLAX) views. An algorithm provides a 0 to 100 scale for image quality for each view. Once acceptable views are acquired, the AI algorithm estimates the LVEF based on a deep learning approach trained on over 50 000 echocardiograms to visually estimate LVEF, mimicking expert human interpretation. No manual quality grading or external reference standard was used for these assessments, and no human raters were involved in image review. A detailed description of this device can be found in the referenced white paper. 6
Outcomes
Outcomes involved feasibility and diagnostic agreement. Feasibility was defined as the proportion of patients in whom adequate views (AP4, AP2, or PLAX) could be obtained, allowing the algorithm to produce an LVEF estimate. Diagnostic agreement involved comparing the AI-POCUS LVEF estimates to the cardiologist-reported LVEF from standard echocardiography (reference standard). The GP performing the handheld scan was blinded to the reference LVEF at the time of image acquisition and AI interpretation.
Data Handling and Ethics
This study represented early clinical adoption of a CE/FDA-approved medical device in routine primary care. The treating GP obtained verbal consent, documented in the electronic medical record, prior to scanning. All data were de-identified before analysis and transferred to a secure environment within the academic general practice network of Amsterdam UMC, under existing data-sharing agreements. Research analyses were subsequently conducted on these de-identified data. The use of routine primary care data for research purposes was granted a waiver of ethical approval and informed consent by the Medical Ethics Committee of the Amsterdam UMC (reference: W23_074, MEC-AMC, Amsterdam).
Statistics and Sample Size
For this paired study, no systematic difference was hypothesized in left ventricular ejection fraction (LVEF, %) between AI-POCUS and conventional echocardiography, with a mean difference of ≥5% considered clinically relevant. To detect this difference with 80% power at a two-sided α of 0.05, a sample of at least 18 paired subjects is required, based on an anticipated LVEF of 50% and a standard deviation of the paired differences of 7.5%. Agreement between methods will be assessed using the mean paired difference with 95% confidence intervals, paired t test, Pearson correlation, intraclass correlation coefficient (ICC), and Bland–Altman plots.
Results
Twenty-five patients participated with a median age of 74 years, and 52% of the participants were male (See Table 1 for additional demographics). Indications for conventional echocardiography included chest pain, dyspnea, palpitations, murmur evaluation, and heart failure or atrial fibrillation follow-up. The median interval between the standard echocardiogram and AI-POCUS was 24 days (IQR 18–29).
The Study Patients’ Demographics and Associated Characteristics (n = 25).
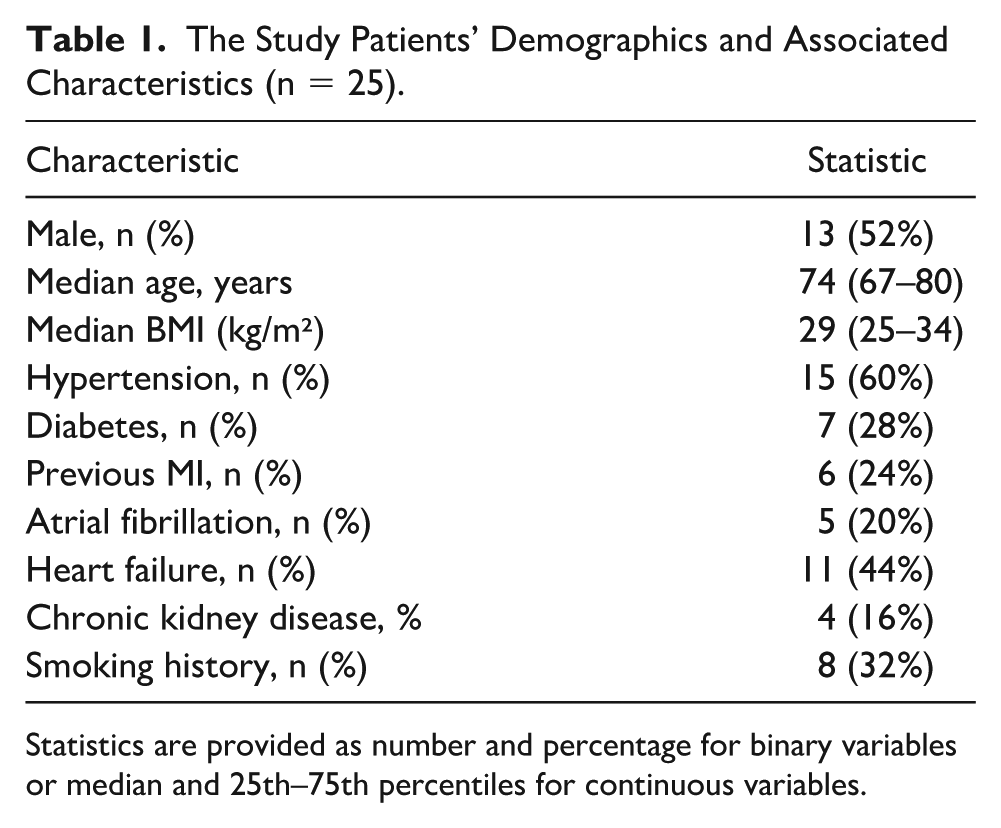
Statistics are provided as number and percentage for binary variables or median and 25th–75th percentiles for continuous variables.
Feasibility
In 19 of 25 cases (76%), AI-POCUS produced images of sufficient quality to generate automated LVEF estimates based on three windows (e.g., AP4, AP2, PLAX) within 10 minutes scanning time. The median scanning time 4.5 minutes, ranging from 3 to 9 minutes. Figure 1 is an example of study image acquisition with both high- and low-quality images. Image acquisition failed primarily due to suboptimal apical transducer positioning for AP4 and AP2 views, especially in female patients with obesity or prominent breast tissue, and in one case breast implants. Five of six failed acquisitions occurred during the initial 15 examinations.
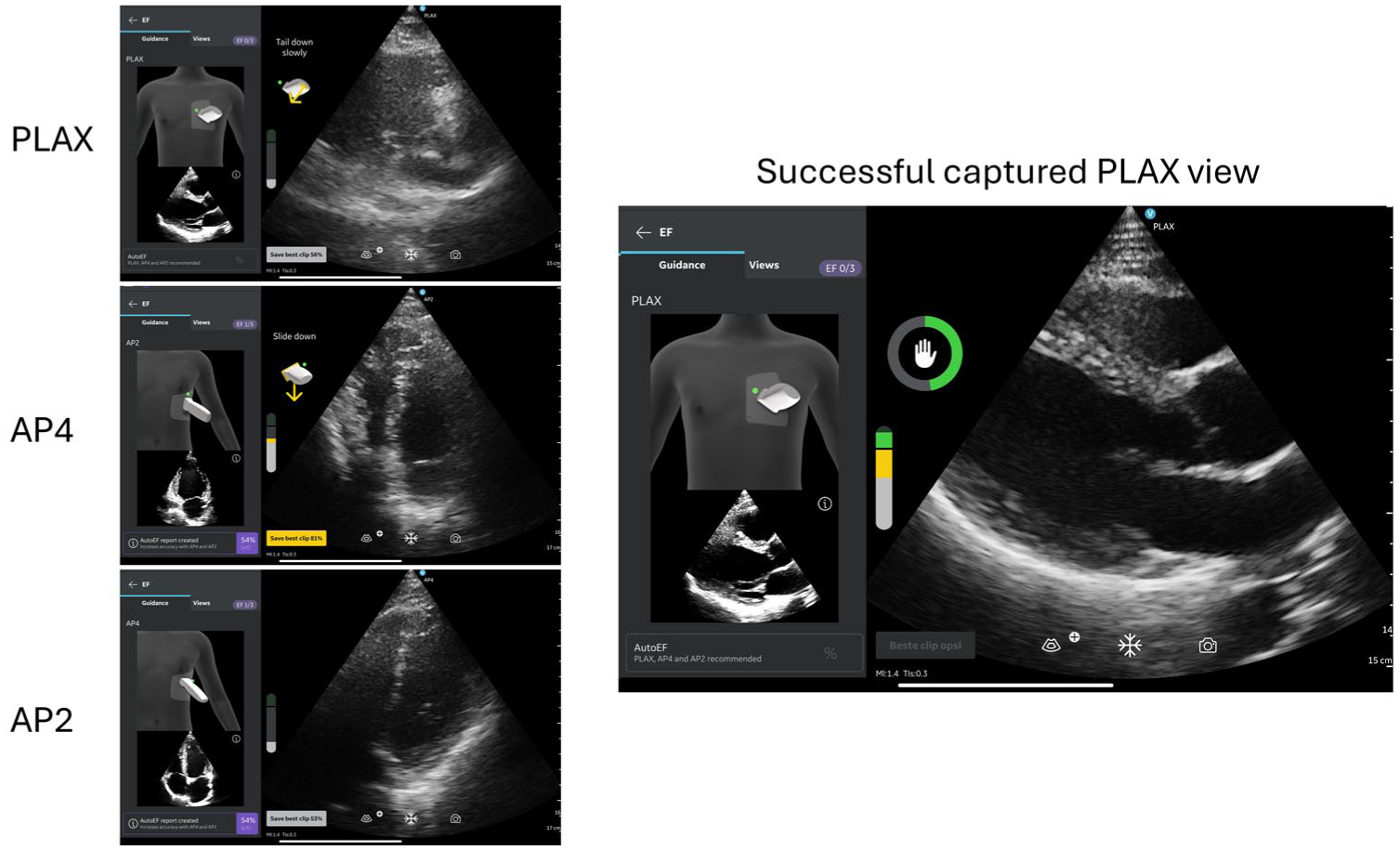
A sample echocardiographic image acquisition, which provided instructions when capturing the PLAX, AP4 and AP2 views (left panel) and successfully captured PLAX view (right panel).
Diagnostic Agreement
Median AI-POCUS LVEF was 50% (IQR 43–55) versus 48% (IQR 42–53) by standard echo (p = .14). As shown in the lefthand panel of Figure 2, there was a strong correlation between AI-POCUS estimates and standard echocardiography (R = 0.95; 95% CI 0.87−0.98). As shown in the righthand panel of Figure 2, there was high agreement over the range of LVEF estimates. The mean difference in LVEF was 1.0%, with 95% confidence interval (CI) from −0.30 to 2.30, suggesting no significant systematic bias (p = .12). The limits of agreement ranged from −4.27% to 6.27% (95% CI: −6.52 to −2.01 for the lower limit, and 4.01 to 8.52 for the upper limit), indicating acceptable agreement.
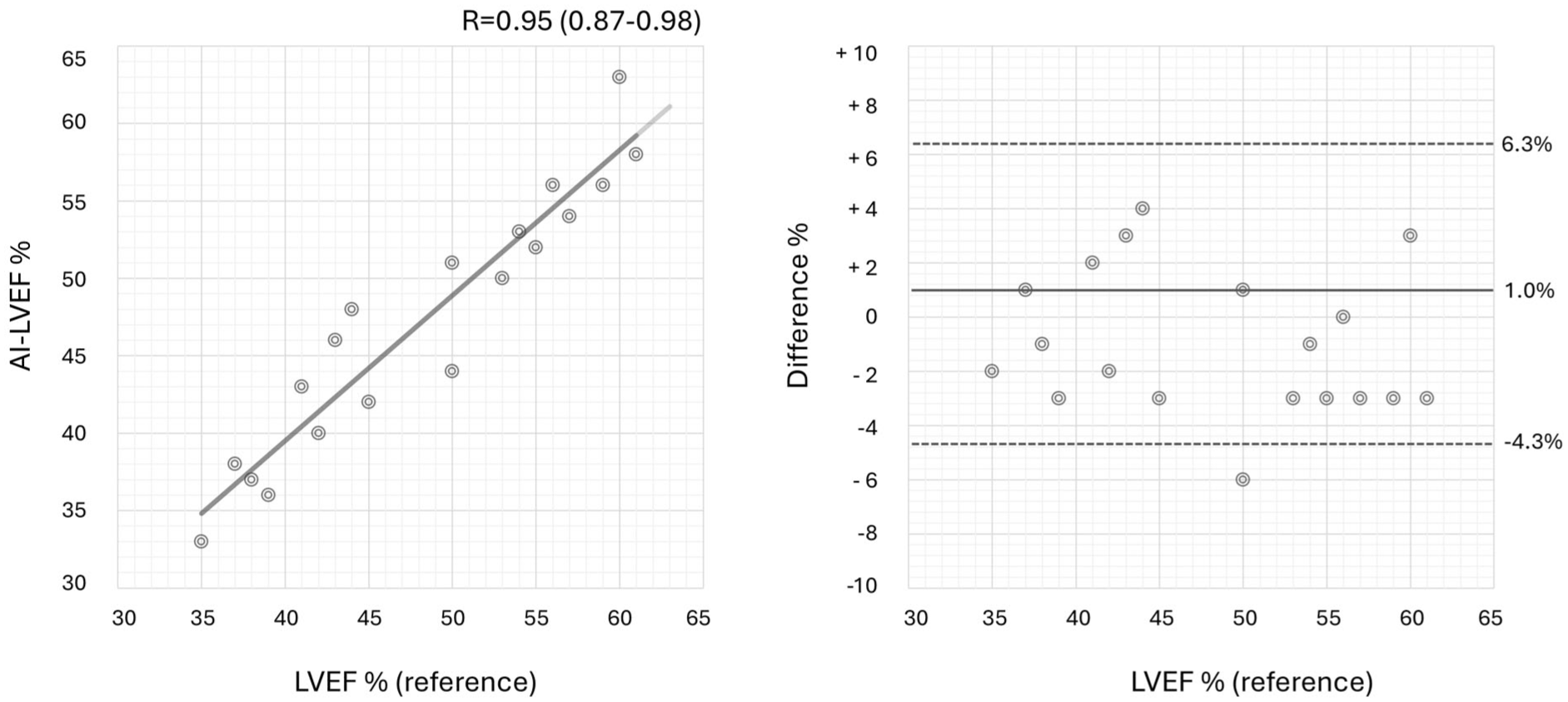
A graph of the study cohort’s data to determine the association between AI-LVEF compared to traditional LVEF values based on a Pearson correlation and Bland-Altman data plots.
One outlier showed discordant classification: AI-POCUS indicated moderately reduced LVEF (<50%), while standard echo indicated preserved function (See Table 2). In all other cases, classification categories (preserved, mildly reduced, reduced) matched. In terms of diagnostic test properties, this rendered a sensitivity and specificity of (mildly/reduced LVEF, <50%) 90% (55.5–99.8) and 100% (66.4–100.0), respectively.
Left Ventricular Ejection Fraction (LVEF) Classification Among Patients With an AI-LVEF Estimate Compared With Conventional Echocardiographic Estimate.
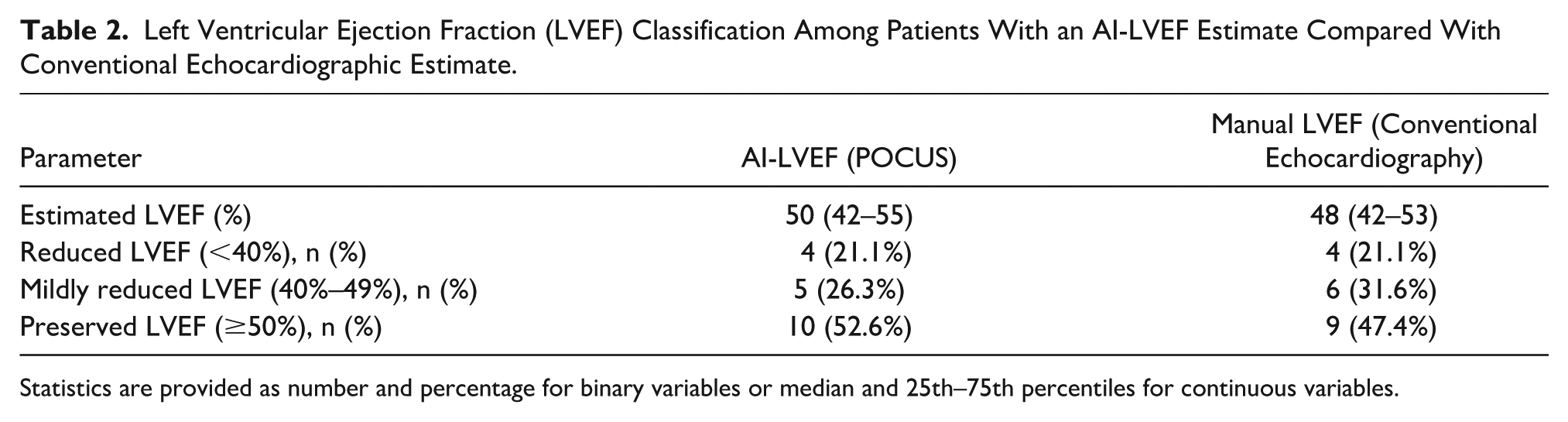
Statistics are provided as number and percentage for binary variables or median and 25th–75th percentiles for continuous variables.
Discussion
This study demonstrates that an AI-guided handheld echocardiography can be feasibly performed by GPs and provides early evidence that it is possible to produce accurate LVEF assessments, compared to standard examination. Real-time AI support enabled successful image acquisition in most cases, with a strong correlation to reference measurements. Moreover, a learning curve was observed, which was consistent with training recommendations by the American Society of Echocardiography (i.e., advising at least 30 scans for competency in focused cardiac sonography). 7
Comparison With Previous Work
The findings of this study are consistent with earlier feasibility studies indicating that nonspecialists can acquire diagnostically useful cardiac ultrasound images with AI assistance.8–10 For example, a study using Caption AI’s AutoEF algorithm in 99 patients demonstrated strong agreement with expert-reviewed LVEF values (r = 0.95), with high sensitivity (90%) and specificity (92%) for identifying LVEF ≤ 35%, closely matching clinician-acquired estimates (r = 0.94). 8 Similarly, in an intensive care setting, another study using the Exo Iris handheld device and ExoAI software reported successful LVEF estimation in 96% of patients, with high inter-rater reliability (ICC 0.88–0.94), despite novices requiring more time than experts. Notably, the PLAX view was faster and easier to acquire than the AP4 view, taking novices about 1.5 minutes versus 3 minutes, respectively. 9
Consistent with the current study, others have also reported that novice users often struggle to obtain high-quality apical views.6 In such cases, AI-LVEF estimation using a single PLAX view may offer a pragmatic alternative. For instance, a study of 166 clinical examinations using Caption AI’s AutoEF showed that machine learning algorithms could accurately estimate LVEF using either PLAX and/or apical views in 85%–90% of cases, further supporting the feasibility of AI-guided POCUS. 10 Nonetheless, caution is warranted. Data from ExoAI indicate that while AI-derived LVEF estimates from the PLAX view alone had high specificity (90%) for detecting reduced LVEF (≤40%), sensitivity was substantially lower (56%–70%), suggesting higher reliability for ruling in rather than ruling out systolic dysfunction. 9
The added clinical value of AI-enhanced POCUS compared to standard care has also been explored. In a randomized trial by He et al, 11 AI-guided LVEF assessments were found to be non-inferior to those performed by sonographers, with comparable diagnostic accuracy, greater time efficiency, and indistinguishable from human estimates according to the reviewing cardiologists. Even without AI support, POCUS demonstrates clinical utility in outpatient settings. A systematic review reported that POCUS detected left ventricular dysfunction in 92% of cases and outperformed cardiac auscultation in identifying valvular disease. 12 Another review comparing POCUS-assisted versus standard clinical assessments found improved sensitivity without a loss of specificity for detecting left ventricular dysfunction as well as aortic and mitral valve disease. 13
Implications for Future Research
AI-guided POCUS holds considerable potential to improve early cardiac diagnosis and optimize care pathways in primary care, particularly in underserved or resource-limited settings where access to formal echocardiography is limited. By enabling accurate, real-time assessment of cardiac function at the bedside or consultation room, AI-POCUS may facilitate more timely referrals, thereby reduce diagnostic and therapeutic delays. An interesting concept herein would be low-threshold expert consultation by integration of sharing images (and/or real-time guidance) via telemedicine platforms.
While the findings of this study, as well as prior research are promising, further validation through large, multicenter trials are needed to ascertain the reliability as well appropriate use of AI-POCUS in primary care settings. Future studies should examine interoperator variability, diagnostic accuracy across diverse primary care settings, and the generalizability of findings across various devices and AI platforms. 4 Blinded expert evaluations and assessments of downstream clinical impact will be crucial to determine the real-world value of AI-POCUS. Studies comparing the cost-effectiveness and workflow integration of AI-POCUS versus conventional referral-based echocardiography could inform reimbursement strategies and implementation frameworks for primary care settings. Additionally, ongoing development of AI algorithms capable of detecting a broader spectrum of cardiac and non-cardiac pathologies (i.e., valvular disease or pleural effusions) could further expand diagnostic utility, usability, and adoption in primary care.
Educational Perspectives
The current study findings, compared to previous studies, may suggest that AI could act as an effective real-time tutor, facilitating rapid skill acquisition in echocardiography. This aligns with the broader vision of POCUS as an extension of the physical examination and a tool for focused diagnostic and procedural applications.14,15 Structured training programs, potentially incorporating digital platforms with AI-based feedback, should be developed to enhance proficiency in POCUS among GPs. A critical component of this approach is ensuring the appropriate acquisition, documentation, interpretation, and limitations inherent in POCUS findings. 16
Finally, not all handheld POCUS devices are created equal, and performance can vary across clinical applications. In a comparative study by Perez-Sanchez et al six portable ultrasound systems were assessed and the Vscan Air™, Lumify™, and Mindray TE Air™ emerged as top performers for acquiring key views such asapical four-chamber, right upper quadrant, and superficial lung imaging. The Vscan Air™ also ranked highest for ease of use and was the preferred device among expert users in this study from 2024. 17 However, this line of technology advances fast, and continued evaluation of device performance will be important to inform procurement and implementation decisions in clinical practice, as well as taught in education sessions
Limitations
The main limitation to this feasibility study is the research design, which has threats to internal and external validity. The patients represent a sample recruited by convenience that further limits the generalizability of these results. In addition, this was a small patient sample, and there was a lack of inter-rater reliability assessment. There results reported were from only a single device and software platform. As such, the findings of this feasibility study are promising, but these results are dependent on a replication of this study with more patients and conducted on a larger scale. The strengths of this work include real-world implementation in primary care, use by an untrained operator for POCUS, and a diverse patient cohort.
Conclusion
This study suggests that AI-guided handheld cardiac ultrasound is feasible in general practice and also provides early evidence on its diagnostic performance for assessing LVEF based on a three-view AI-based estimate. It shows that real-time AI support enabled successful image acquisition in the majority of patients and produced LVEF estimates in these cases that strongly correlated with standard echocardiography. These findings support the potential of this technology to enable earlier detection and triage of cardiac dysfunction in primary care. However, given the limitations of this study, further validation through larger, multicenter studies is warranted.
Footnotes
Acknowledgements
Thanks to the patients and staff at Holendrecht Health Center for their support.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was waived by the Medical Ethics Committee of the Amsterdam UMC because it involved the use of routine primary care data collected as part of a routine care improvement project (reference: W23_074, MEC-AMC, Amsterdam).
Informed Consent
Verbal informed consent (verbal assent) was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
