Abstract
The antenatal diagnosis of fetal congenital heart disease (CHD) allows for identification of potentially life-threatening conditions that may occur in the early neonatal period. A suspicion of congenital heart disease often occurs during the routine fetal anatomical survey. Subsequently, fetal echocardiography allows for a more detailed assessment of the cardiac lesion that is necessary in order to plan for delivery and postnatal surgical management. To enhance detection of congenital cardiac anomalies, and in particular ones that are potentially serious, it is important for the obstetrical sonographer to have an understanding of potential teratogenic agents associated with an increased risk of congenital heart disease.
Introduction
The incidence of congenital heart disease (CHD) is approximately 6 to 12 per 1000 live births. 1 There are some data, although not definitive, that prenatal diagnosis has a positive effect on infant morbidity and mortality, particularly of ductal-dependent lesions that require early postnatal administration of prostaglandins to maintain patency of the ductus arteriosus. 2
For the obstetrical sonographer, congenital heart disease is often detected in the setting of a pregnancy at increased risk of aneuploidy, such as trisomy 21, 18, and 13. However, maternal exposure to certain teratogens may also increase the risk of CHD. A teratogen is defined as “any agent that acts during embryonic or fetal development to produce an alteration of form or function.” 3 Drugs are commonly thought of as teratogens; however, other disease processes or external influences can also have teratogenic effects on the fetus, such as diabetes or obesity.
Many teratogens have been associated specifically with fetal cardiac malformation. Certain classes of medications, including antibiotics, antiepileptic drugs (AEDs), antidepressants, nonprescription drugs, as well as certain maternal conditions may increase the risk of CHD. It is important to keep in mind that reported associations do not necessarily imply true cause and effect, as data are often conflicting.
Embryology
The heart is the first organ to develop in the fetus. In the primitive heart, plates fuse and give rise to one single heart tube by 22 days. The ends of the primitive heart tube are fixed, and therefore during rapid growth, loops form. The cavities then divide with formation of endocardial cushions, which give rise to the four chambers of the heart. There are several signals that are essential to the development of the various portions of the heart. Divisions between the chambers occur simultaneously and give rise to the valves and septa.
The most crucial period for fetal heart development ranges from weeks two through seven of gestation. 4 Thus, fetal exposure during this critical period and the three months prior to pregnancy are thought to be the times that a teratogen is most likely to affect development. Figure 1 illustrates a timeline for embryological development of the fetal heart.

Timeline of fetal heart development.
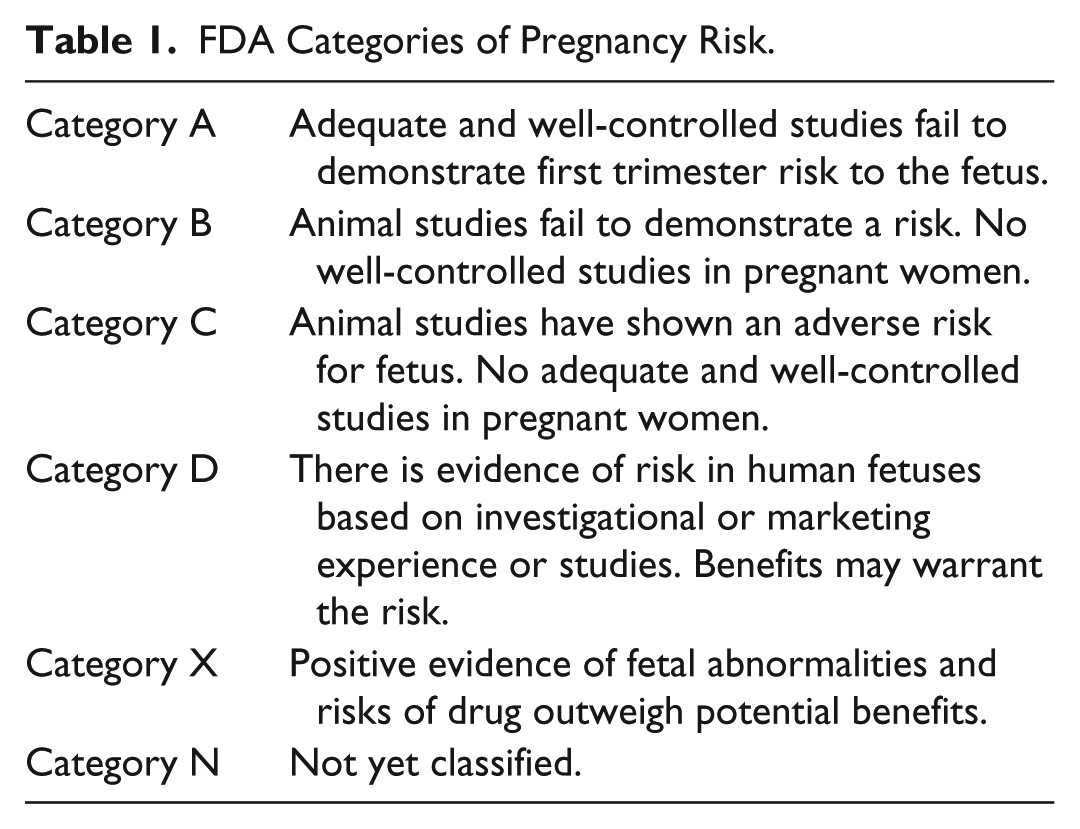
Since 1979, clinicians in the United States have derived information about the safety of medication use during pregnancy from the US Food and Drug Administration (FDA) classification system. This system classifies medications into five categories (Table 1) based on the level of available data and risk-benefit ratio. 5 The FDA is planning to release a new pregnancy category system that will better reflect the existing data. However, for purposes of this article, we will use the current classification system when discussing the various medications. Table 2 offers a comprehensive list of exposures with the corresponding pregnancy category. Table 3 lists both exposures and drugs with the specific cardiac lesions they may be associated with.
FDA Categories of Pregnancy Risk.
Overview of Exposures and Corresponding Pregnancy Category.
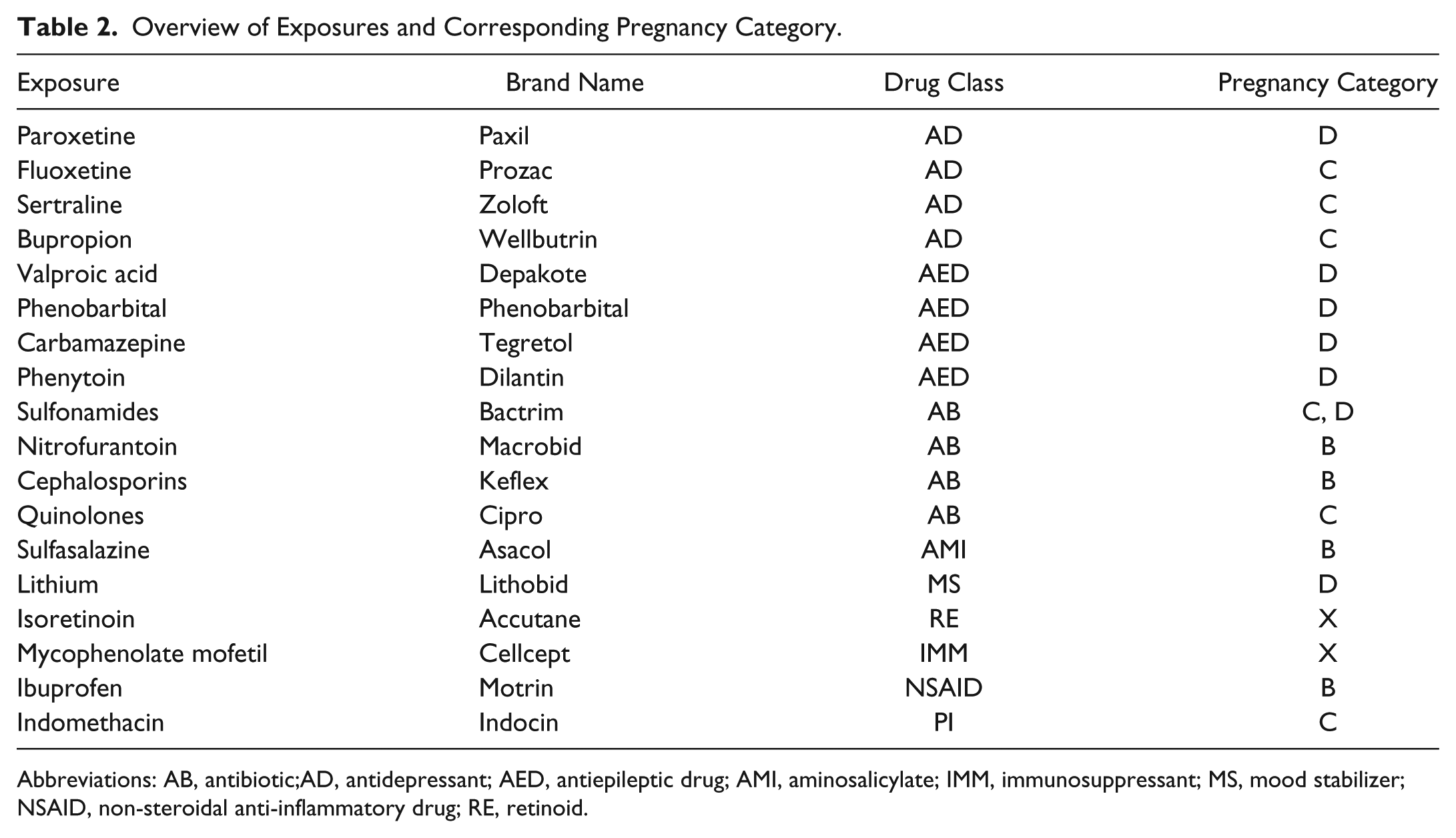
Abbreviations: AB, antibiotic;AD, antidepressant; AED, antiepileptic drug; AMI, aminosalicylate; IMM, immunosuppressant; MS, mood stabilizer; NSAID, non-steroidal anti-inflammatory drug; RE, retinoid.
Possible Association of Congenital Heart Disease With Particular Drugs/Exposures.

Abbreviations: AO, coarctation of the aorta; ASD, atrial septal defect; AVSD, atrioventricular septal defect; CM, cardiomyopathy; HPLHS, hypoplastic left heart syndrome; HPRH, hypoplastic right heart; PA, pulmonary atresia; PDA, patent ductus arteriosus; PS, pulmonary stenosis; TAPVR, total anomalous pulmonary venous return; TGA, transposition of the great vessels; TOF, tetralogy of Fallot; VSD, ventricular septal defect.
Antidepressants
Paroxetine, also known as Paxil, is a selective serotonin reuptake inhibitor (SSRI) that is used for the treatment of depression. There have been some data supporting an increased risk of cardiac malformations with an even higher risk if the exposure occurred during the first-trimester. This includes ventricular outflow defects and atrial/ventricular septal defects6,7 (Figures 2–4). Fluoxetine (Prozac) and sertraline (Zoloft) are other SSRIs that may be associated with cardiac malformations.
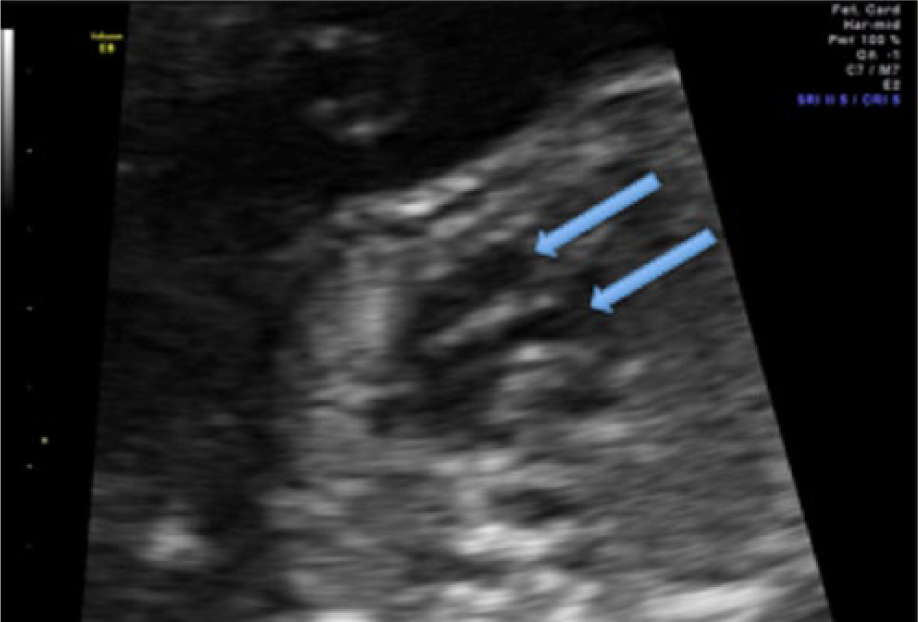
Gray-scale image of transposition of the great vessels (TGV). The cardiac outflow tracts appear to arise in parallel (arrows), suggestive of TGV.
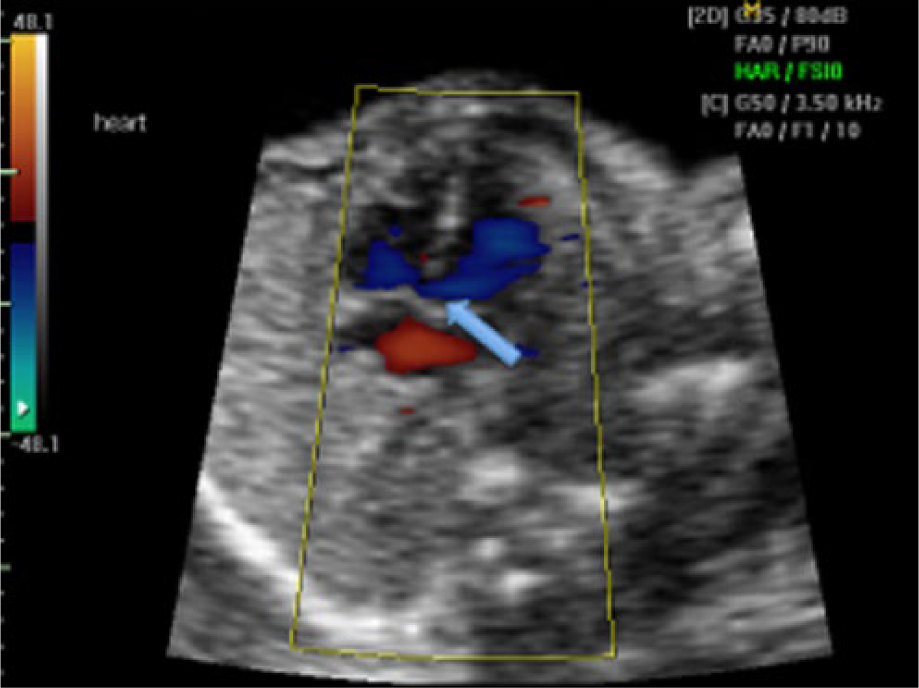
Peri-membranous ventricular septal defect (VSD). In this apical four-chamber view, color flow suggests a defect in the peri-membranous portion of the ventricular septum (arrow).
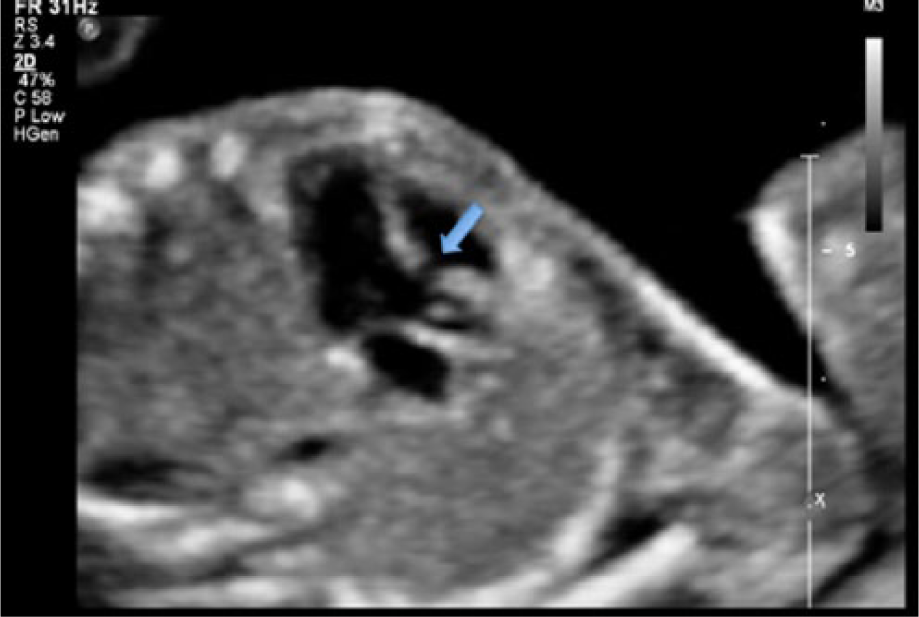
Muscular ventricular septal defect (VSD). Long-axis view of the left outflow tract shows a defect in the ventricular septum (arrow).
Buproprion, also known as Wellbutrin, is primarily used for smoking cessation and mild depression. In small studies, it was found to be associated with left outflow tract defects. 7 Importantly, data for all antidepressants are conflicting. There has not been a single antidepressant that has been definitively associated with CHD. Thus, the absolute risk remains low.
Antiepileptic Drugs
Valproic acid (Depakote) has been linked to an increased risk of neural tube defects, with fairly consistent data. Other anomalies that have been linked to the use of valproic acid include atrial septal defects (ASD), ventricular septal defect (VSD), tetralogy of Fallot, pulmonary valve atresia, and hypoplastic right heart. The increased risk is usually seen with first-trimester exposure.8,9 Other AEDs that may be linked to CHD, but with less supportive data, include phenobarbital (a barbituate), carbamazepine (Tegretol), and phenytoin (Dilantin). 9
Antibiotics
Nitrofurantoin, a commonly used antibiotic, has been associated with fetal heart abnormalities including ASD and hypoplastic left heart (Figure 5). In addition, sulfonamides, such as trimethoprim-sulfamethaxole (Bactrim), have been associated with hypoplastic left heart and coarctation of the aorta. In one study, the cephalosporin and quinolone classes of antibiotics suggested an increased risk of CHD, 10 although again, data are conflicting.
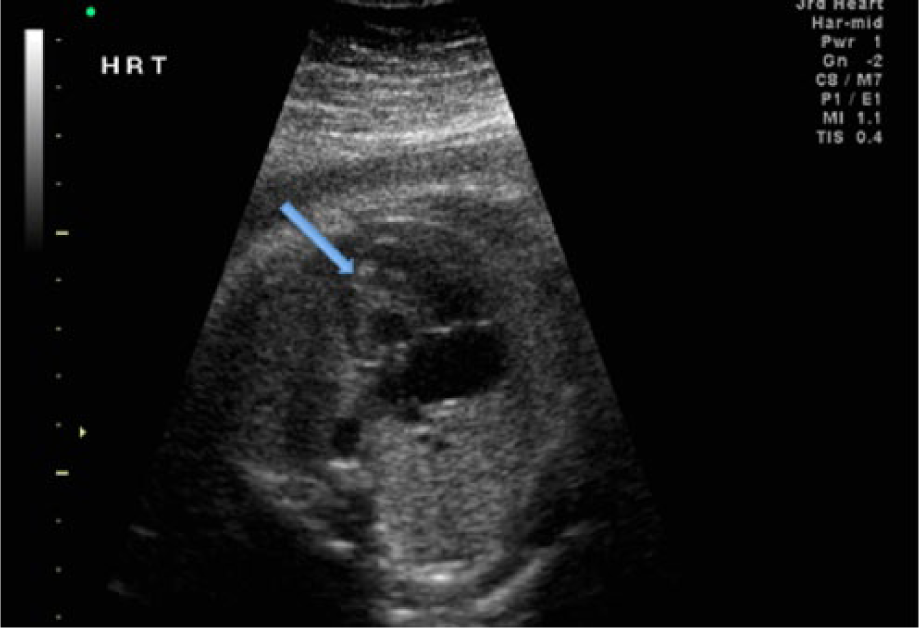
Hypoplastic left heart. In this apical four-chamber view of the fetal heart, the left side (arrow) is significantly smaller than the right.
Other Drugs
Sulfasalazine (Asacol) is an amniosalicylate that can be used for the treatment of ulcerative colitis, rheumatoid arthritis, and other rheumatologic diseases. Case reports have demonstrated a possible association with coarctation of the aorta and VSD.11,12
Lithium, a mood stabilizer used for the treatment of bipolar disease, has been associated with Ebstein’s anomaly. In this condition, the tricuspid valve is displaced toward the apex of the heart, often resulting in “atrialization” of the right ventricle. This condition is also associated with functional or anatomic pulmonary atresia and can progress to right atrial enlargement, severe tricuspid regurgitation, and fetal hydrops. 13 Although the risk of CHD is increased with lithium use, this risk is often overestimated; the actual risk is approximately 1 in 2000.
Isoretinoin, commonly used for acne treatment, is a pregnancy category X drug, which is associated with a myriad of fetal anomalies, including congenital heart disease.4,13,14 It is important to note that the topical form (Retin-A) has not been shown to increase the risk of CHD.
Immunosuppressants are another class of drugs that is also known to have significant fetal effects. Mycophenolate mofetil (CellCept) and thalidomide are category D and X, respectively. Both of these medications are contraindicated during pregnancy. Cellcept has been linked to CHD as well as other anomalies, such as cleft lip/palate, micrognathia, hypertelorism, renal malformations, and agenesis of the corpus callosum.13,15
Maternal Disease/Illness
Phenylketonuria (PKU) is an autosomal recessive disorder that results in an excessive accumulation of phenylalanine. If untreated, PKU may increase the risk of CHD by 6-fold. 4
Pregestational diabetes, defined as diabetes prior to pregnancy, is another well-known risk factor for congenital heart disease, and the risk increases with the degree of hyperglycemia during the period of organogenesis. The pregnant patient who conceives at a time when her glycemic control is poor places her baby at a substantially increased risk of CHD. 4
Maternal febrile illness, including rubella and influenza in the first two months of pregnancy, is associated with fetal heart malformations. Several studies have demonstrated a 2- to 6.5-fold risk of CHD with obesity. The data on obesity are difficult to interpret because of multiple confounding variables (eg, other comorbidities). 4
Tobacco use is associated with many complications during pregnancy. A 2008 study in the journal Pediatrics demonstrated that these complications may include CHD. Specifically, smoking greater than 25 cigarettes a day was associated with an increased risk of septal and right-sided obstructive lesions. 3 Alcohol use has been associated with fetal alcohol syndrome, but evidence for a link to CHD is sparse. Finally, marijuana use has also been linked to an increased risk for VSD and Ebstein’s anomaly. 4 Again, these data are not definitive.
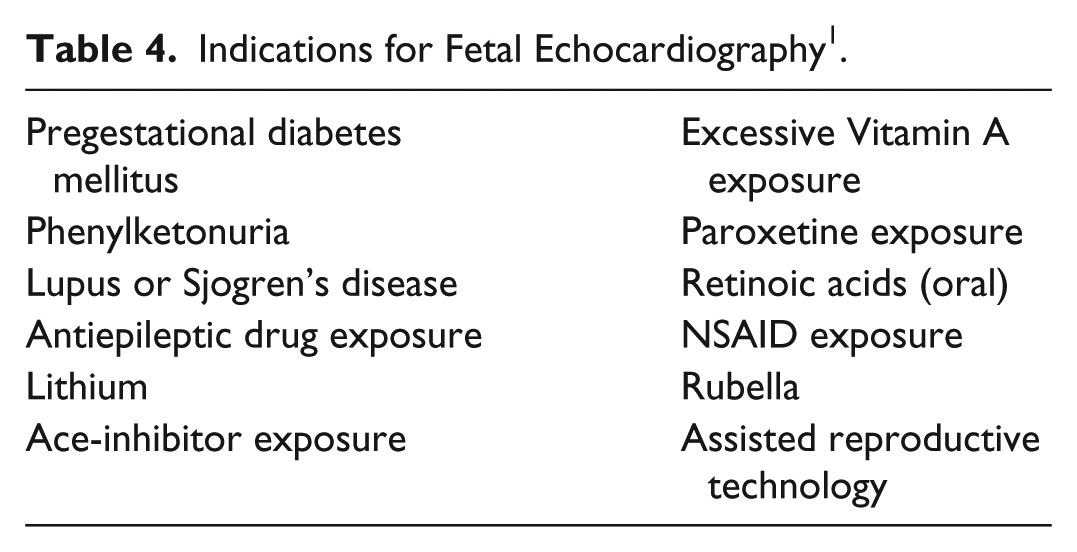
In light of the possible associations between CHD and the drugs/conditions discussed previously, it is important for the sonographer to understand the indications for fetal echocardiography. The American Heart Association in 2014 published guidelines on the diagnosis and treatment of fetal cardiac disease, including the indications for fetal echocardiography (Table 4). 1 These include many of the exposures discussed previously.
Indications for Fetal Echocardiography 1 .
Summary
Obstetric sonographers, and in particular those who work with high-risk pregnancies, should possess knowledge of the possible association between certain medications and CHD. Practitioners should always conduct a thorough discussion regarding a patient’s medication history, including pharmaceutical drugs that were taken both prior to and during pregnancy. However, not all pregnancies at risk for congenital heart diseases are high risk. An estimated 51% of all pregnancies are unintended, 16 and many of these women will have inadvertent medication exposures during fetal organogenesis. Often patients will forget dosages or medication names, and medication adjustments or changes in health care providers can further complicated a patient’s medication history. Therefore, general knowledge of common treatment options and associated anomalies further allows the sonographer to stratify patients’ risk based on exposures. Early detection allows for optimal pregnancy management, and fetal echocardiography is warranted for suspicious lesions noted on the fetal anatomical survey. In summary, this article reviews several teratogens associated with fetal congenital heart disease and provides some examples of these lesions to further the understanding of these associations and enhance early detection.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
