Abstract
Surgical pelvic mesh is a net-like material used to support weakened pelvic muscle walls and the urethra or urinary bladder. It is used to treat symptomatic pelvic organ prolapse (POP) and stress urinary incontinence (SUI), among female patients. However, its use has become controversial due to higher relapse rates and mesh-related complications. Moreover, removing the mesh as a whole may not be possible, and the symptoms may persist. Several surgical pelvic mesh techniques are available; familiarity with these techniques, along with knowledge of normal and abnormal appearances, is necessary to successfully complete diagnostic imaging for these patients. This case report presents a female patient who had undergone revision surgery for retrieval of surgical pelvic mesh implanted for POP. The patient was unaware of its incomplete removal. The patient presented with urinary complaints and was initially diagnosed with a urinary bladder calculus on sonography, due to the lack of disclosed surgical history. During the pre-operative diagnostics for calculus removal, cystoscopy revealed encrusted soft tissue, which was found to be residual mesh and sutures in the urinary bladder wall. This case highlights the importance of a comprehensive clinical and diagnostic imaging history, along with a review of previous operative notes, to provide a complete evaluation of these patients.
Pelvic organ prolapse (POP) is characterized by the descent of the uterus, bladder, rectum, or bowel into the vaginal canal. The accompanying symptoms such as urinary incontinence, voiding dysfunction, pelvic discomfort, sensation of bladder, vaginal or rectal bulge, and difficulty in defecation prompt the need for either conservative management or surgical intervention. The primary approach to treatment is surgical, with the objective of restoring the anatomical integrity of the structures that support the pelvic organs. Such surgical repair techniques can be classified into two categories: the first involves the use of synthetic materials, such as mesh, whereas the second employs only native pelvic support tissues to reinforce the compromised support system, referred to as “native tissue repair.” 1 A mesh implant is a net-like material employed in the reconstruction of weakened pelvic muscle walls, providing support to the urethra or urinary bladder. The aim of this surgical intervention is to address symptomatic POP and stress urinary incontinence (SUI) in female patients. A cumulative incidence of undergoing surgery for POP or SUI or both is 11.1% to 11.8% by age 80 years, and 29.2% of these patients will require repeat surgery for POP or SUI.2–4 With primary native tissue repair, there is a recurrent prolapse rate of up to 30%, 3 while with surgical pelvic mesh reconstruction, the complications reported are up to 12%. 4 However, the use of pelvic mesh for prolapse surgery has become a highly controversial subject, due to the high failure rate and serious, well-documented mesh-related complications, such as mesh erosion into adjacent organs, pain, fistula formation and infections. 5 In addition, complete retrieval of the mesh is not possible in every case and may even exacerbate the symptoms. Hence, mesh removal surgery may not be the right treatment choice for mesh failure as a first approach. 6 The patient case provided describes a diagnostic scenario where a residual pelvic mesh had eroded into the urinary bladder, mimicking adherent bladder calculus on sonography.
Case Report
A 39-year-old woman, with a history of two live births (P2+0), presented with repeated episodes of fever, burning micturition, and urge incontinence for the past 2 years. Her routine blood tests were normal. The urinalysis revealed pyuria and Escherichia coli that was detected in the urine culture. On pelvic sonography, an echogenic, wavy, linear area measuring 2.45 cm in length, producing strong posterior acoustic shadowing, was observed along the posterior wall of the urinary bladder (See Figure 1a). The echogenic area remained stationary when the patient changed positions. A sonographic diagnosis of adherent urinary bladder calculus was determined. Cystoscopy was performed, revealing an encrusted area with sutures in the supratrigone region of the urinary bladder. After taking an elaborate history, it was discovered that the patient had undergone abdominal pelvic mesh repair surgery for uterine prolapse 5 years ago. Two days post-operation, the mesh was removed due to severe pelvic pain. The prior operative notes regarding the type of mesh used and the surgery performed were unavailable. The patient was unaware of the incomplete removal of the mesh implant. Upon repeating the sonogram with knowledge of cystoscopic findings, consideration was given to the possibility of a residual mesh implant, within the urinary bladder wall. The mesh appeared hypoechoic and was located anterior to the echogenic, encrusted area (See Figure 1b). The patient underwent a computed tomogram (CT) of the pelvis, which showed a linear, irregular, hyperattenuating area, measuring 2.47 cm × 1.1 cm along the posterior urinary bladder wall (See Figure 2) with isodense soft tissue (See Figure 2b and d) extending up to the anterior uterine wall, consistent with surgical mesh with encrustation. Magnetic resonance imaging (MRI) was performed to precisely delineate the size and location of the residual mesh, in relation to the pelvic anatomy. MRI views showed a curvilinear, altered signal intensity area abutting the posterior bladder wall and anterior vaginal wall, which was hypointense in the periphery on all the pulse sequences (See Figure 3a and b). There was no evidence of a pelvic fluid collection, abscess, sinus tract or fistula formation. The patient underwent a repeat cystoscopy (See Figure 4a), and the retained mesh was removed entirely (See Figure 4b). The postoperative period was uneventful. At a 3-month visit after surgery, the patient was relieved of her urinary complaints. However, she was still experiencing pelvic pain on and off, to which she responded well to analgesics.
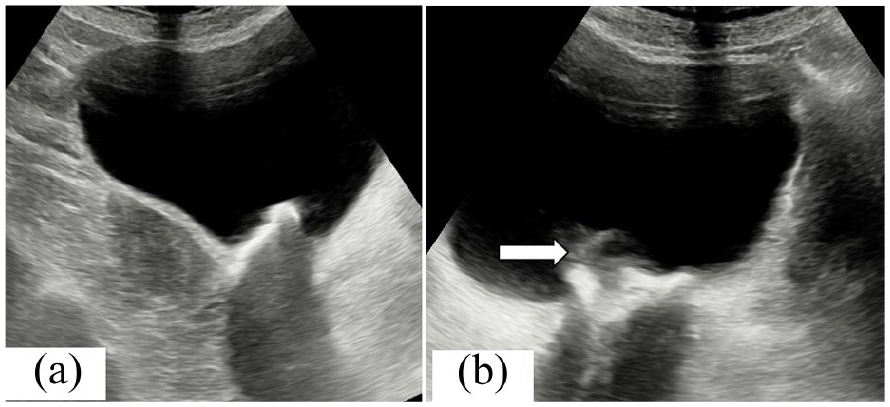
Grayscale sonographic sagittal (a) and (b) axial views demonstrate an echogenic area, with posterior acoustic shadowing, in the urinary bladder, as well as a partially hypoechoic component, adjoining this hyperdense area (marked with an arrow). This area is noted in (b), which was suggestive of residual uterine mesh, with encrustation.
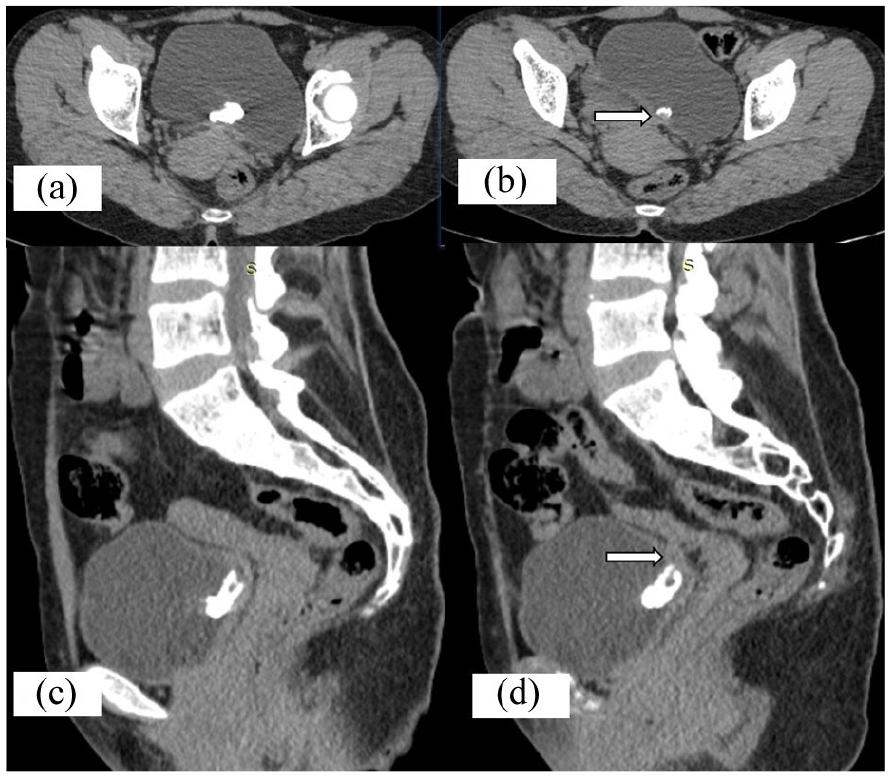
Computed tomographic axial (a, b) and (c, d) sagittal views show a calcified area in the urinary bladder with soft tissue component, extending to the anterior uterine wall (marked with an arrow). This calcific area (noted in b, d) represents residual uterine mesh with encrustation.
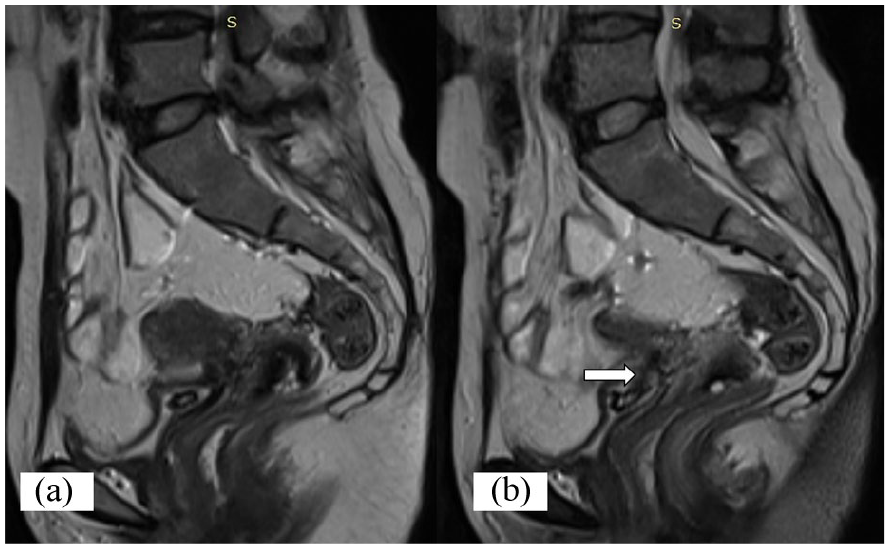
Magnetic resonance imaging provided sagittal views that were T2-weighted images (a, b) demonstrated the hypointense mesh extending from the urinary bladder to the anterior uterine wall (marked with an arrow).
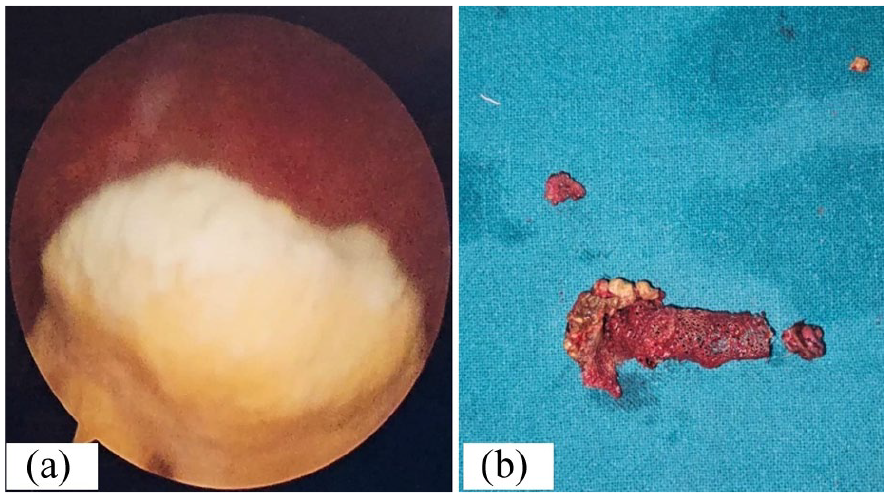
An intraoperative cystoscopic image is provided (a) and a postoperative image (b) shows the residual mesh with encrustation.
Discussion
The reported prevalence of POP, as stated, varies widely. The global incidence of POP in 2019 was estimated to be 13 million new cases. 7 POP can be compartmentalized as anterior (e.g., cystocele or urethral hypermobility), apical (e.g., vaginal apex or uterine prolapse), or posterior (e.g., rectocele, rectal descent, or cul de sac hernia), with frequent concomitant multiple compartmental involvement. 8 SUI is the leakage of urine that occurs when laughing, coughing, or sneezing, which increases the pressure in the urinary bladder. It has a global prevalence of around six percent in women. 9 The risk factors for POP and SUI include vaginal childbirth, connective tissue abnormalities, weakness of the pelvic floor, hysterectomy, aging, menopause, and chronically raised intra-abdominal pressure. Both POP and SUI are often comorbid disorders that can greatly affect quality of life and can be treated conservatively or surgically. However, surgical repair is considered the primary treatment for POP. Native tissue repair procedures exhibit lower costs, shorter operating times, and a reduced risk of morbidity. However, these procedures are correlated with a heightened risk of recurrent prolapse over the long term. Conversely, surgeries utilizing mesh for POP are characterized by a lower prolapse recurrence rate but extended operating durations, increased length of hospital stay, and greater blood loss. Furthermore, the morbidity with these mesh procedures is elevated due to complications such as mesh erosion, ileus or small bowel obstruction, thromboembolism, and bladder injuries incurred during surgery.10–14
Although pelvic surgical mesh implants have been used since the late 1990s for POP and SUI treatment, 3 the US Food and Drug Administration (FDA) has banned the use of transvaginal mesh products for POP repair, due to the high prevalence of relapse rates and mesh-related complications, except for mid-urethral slings in 2019.6,7,15
Weak pelvic muscles can result in the prolapse of the urinary bladder, uterus, or rectum, leading to a condition in which these organs descend from their normal positions. These mesh implants act as hammocks underneath these pelvic organs, preventing or limiting their sagging. An exhaustive list is available mentioning the ideal properties of a mesh, which include non-toxicity, biocompatibility, chemical inertness, lightweight with low density, large pores and high porosity, mechanical strength, non-degenerative, non-carcinogenic, non-allergenic, sterile, and resistant to mesh shrinkage. However, out of these, two critical properties are essential: ideal flexibility that allows for easier handling and positioning during surgery, and sufficient strength to provide robust long-term structural support for the pelvic organs, preventing them from prolapsing again. The materials used for fabricating the mesh implant can be broadly divided into two main types: synthetic and biological. Biological grafts can either be autologous or heterologous.5,7 The selection of mesh subtype is individualized, based on a case-by-case basis, taking into account the preferences of both surgeons and patients, as well as risk analysis. 16
The various surgical options available for POP and SUI are abdominal surgery, laparoscopic surgery, robotic surgery, and vaginal surgery. Proper patient selection is the most critical prerequisite for mesh surgery. The surgery should be reserved for high-risk individuals in whom the benefit of mesh placement may justify the risk, such as individuals with recurrent prolapse. Such patients can be identified through clinical examination and diagnostic pelvic floor imaging. 17 To address the growing medico-legal aspects, written informed consent should be obtained from the patient after explaining the risks and benefits of the procedure and discussing alternative treatments.
The complications encountered during mesh surgery can be categorized into intraoperative and postoperative. The intraoperative complications include vaginal or pelvic hematoma, bleeding, rectal injury and bladder injury. These are related to technique rather than the direct use of mesh and are usually low-risk complications. 16 In contrast, the postoperative complications are more frequent, severe and are mainly related to the mesh. The most common postoperative complications include mesh erosion with vaginal extrusion or erosion into the urethra or bladder, mesh failure, pain, and infections.
Mesh erosion describes the exposure, extrusion, or protrusion of the mesh exteriorly at the surgery scar or found perforating an organ after surgery, such as the vagina, the urinary tract or the bowel. Mesh exposure has been reported in 4%–19% of cases for transvaginal mesh and approximately 3% of cases for sacrocolpopexy mesh.8,18 Mesh erosion can be classified into two main types according to the location of erosion. The first type is vaginal extrusion, the more common form in which mesh is visible or palpable through the vaginal mucosa. The patient usually presents with vaginal discomfort due to mucosal irritation, abnormal vaginal discharge, pelvic pain or dyspareunia. This occurs in two to four percent of women after a mid-urethral sling and in 5%–10% of women, after POP surgery. The risk factors include vaginal atrophy, extremes of age, concurrent hysterectomy, diabetes, steroid use, and smoking. The amount of mesh implanted, implant design, the extent of dissection, the amount of tension on the mesh, the twisting of the mesh, and surgical expertise are also relevant factors affecting the outcome. The second type of mesh erosion or suture exposure is erosion into the urethra or urinary bladder, as seen in the present case (See Figure 2d). The patients typically show urge incontinence, SUI, urinary retention, hematuria, or dysuria and the potential to progress to more serious complications such as fistula formation or pelvic sepsis. The incidence of this complication is two percent after mesh procedures for SUI and four percent for POP. In the case of sacrocolpopexy or rectopexy, the placement of sutures to anchor the mesh may inadvertently result in the placement of permanent sutures into the bladder lumen, leading to a similar presentation as mesh exposure in the bladder.17,19 Cystoscopy is necessary for a thorough examination of the urethra and bladder to accurately diagnose this complication. Mesh within the urinary tract should be surgically removed as it serves as a nidus for infection and stone formation. 9
Mechanical mesh failure is mesh laxity whereby it follows an indirect route along the pelvic sidewall from the sacrum to the vagina or cervix, instead of a direct course through the pelvis, likely due to its breakage or detachment from the vagina, cervix or sacral promontory. 20 The incidence of chronic pain with mesh is variable and has been reported in up to 30% and appears to be the most common reason for mesh removal. Postoperative pain usually occurs in the pelvic, groin, and thigh regions or during sexual intercourse. It can be caused by mesh erosion or subclinical infection. The inflammatory response in the postoperative period induces the shrinkage of mesh, leading to vaginal tightening; this is one of the postulated hypotheses for the etiopathogenesis of mesh-related pain. A fistula may develop between the vagina and either the urinary system (i.e., vesicovaginal or urethra-vaginal fistula) or the gastrointestinal system (i.e., rectal fistula or colo/recto-vaginal fistula), after the placement of mesh. These complications, although rare, are technically challenging to address and necessitate operative intervention. The cause may be inadvertent injuries during surgery or slow extrusion into adjacent organs. Pelvic infection, although rare, is still an inherent risk for any surgical implantation. Mesh infection has been reported to occur in up to eight percent of women, after transvaginal mesh placement. 19 The mesh characteristics (e.g., smaller pore size) and coexisting mesh exposure in the vaginal are relevant risk factors. Rarely, it can progress to pelvic abscess, necrotising infections, and distant infectious complications. A complete mesh excision is highly recommended for severe infections that do not respond to conservative treatment. 19
Diagnostic imaging can be used to evaluate the success of native tissue repair procedures by visualizing the restored anatomy and assessing the degree of prolapse reduction. It can help identify early signs of prolapse recurrence. Sonography enables the dynamic evaluation of the pelvic floor during straining, which helps assess the functional aspects of prolapse and the effectiveness of repair. Diagnostic imaging plays a fundamental role in identifying prior surgical interventions and can also characterize implant-related complications. Multiple imaging techniques are complementary to document such complex patient cases. Sonography offers the advantage of dynamic assessment and effective visualization of the periurethral space. MRI provides better soft tissue contrast and enables a more comprehensive evaluation of the retropubic space and the more peripheral pelvic floor, which is outside the sonographic field of view. If the surgical mesh is radiopaque, micturating cystourethrography (MCU) and CT may also be beneficial. 8
Various sonographic techniques, including transabdominal, 3D transvaginal, transperineal, and translabial imaging, can be used to visualize vaginal mesh and mid-urethral slings. The polypropylene mesh implants are highly echogenic and appear as smooth, flat, linear structures with or without posterior shadowing, situated dorsal to the bladder neck, caudal and dorsal to the trigone, and the posterior bladder wall, or adjacent to either the anterior or posterior vaginal wall, depending on the type of surgery performed. 18 Occasionally, the mesh is wavy due to fibrosis and mesh shrinkage. Assessment of midurethral slings should be performed during a Valsalva maneuver when they tend to rotate in an arc-like manner around the fulcrum of the pubic bone. 6 Endovaginal 3D sonography has the advantage of depicting the extension of mesh into the vaginal wall. With MRI, the vaginal mesh appears as thin, linear, hypointense structures on T2-weighted images (T2 WI), with variable attachment sites. Sometimes, only scarring or fibrosis is observed along the expected course of the mesh, which also appears hypointense on T2-WI. Scar tissue may persist or increase after the removal of mesh material, which serves as a common pitfall that limits the detection of residual mesh tissue. Mesh arms coursing through the fat of the ischiorectal fossa are best visualized on non-fat-suppressed T2-WI as hypointense, non-branching structures. 18 Depending on the density, structure, and thickness of the mesh material, surgical mesh exhibits varied visibility with CT. Polypropylene and plaited polyester mesh are radiolucent and are either invisible or poorly visible on CT, while polytetrafluoroethylene mesh is hyperattenuating, with visibility varying depending on the mesh thickness. A “meshoma” is a scar formation of variable thickness that surrounds the mesh and may be the only indication of its presence when the mesh material is invisible on CT. 15 So, diagnostic imaging can complement a patient’s history by confirming the presence of a synthetic implant and to detect an implant in patients unaware of the specific type of prior pelvic surgery, as noted in the current case report.
The complex nature of mesh removal surgery requires input from multiple professionals in various medical specialities. A multidisciplinary team, including urologists, gynecologists, general surgeons, radiologists, physiotherapists, and pain management specialists, is required for management. It is generally agreed that mesh should be removed only if the patient experiences bothersome symptoms. Conservative treatment is started in case of small vaginal exposure (i.e., less than 0.5–1 cm2). Topical oestrogens and antibiotics, along with intercourse abstinence, are recommended. Nonsteroidal anti-inflammatory drugs (NSAIDs), opioids and/or neuroleptics, and muscle relaxants are given for pain relief. Physiotherapy and nerve block are other options available. The rate of worsening or failure following surgery for pre-existing symptoms is 10%–15%. 9 If conservative treatment fails due to large mesh exposure, partial mesh excision is recommended, with symptoms expected to resolve in most patients. If even partial mesh excision fails, a more invasive complete mesh excision should be considered. Mesh, once fitted, is intended to remain in its original surgical position. This implies that it can be difficult or even impossible to remove it all. The arms of the mesh implant, which are attached to strong ligaments deep in the pelvis, may be difficult to remove in totality, and overzealous surgical exploration may further injure the adjacent normal soft tissue. The risks of mesh removal surgery are different for each woman and depend on factors such as the amount of mesh to be removed, previous mesh removal surgery, the amount of scarring, proximity of the mesh to the urethra, urinary bladder or bowel, if there are more than one mesh implant and the presence of infection around the mesh. There is no guarantee that mesh removal surgery will alleviate long-term pain symptoms. It is essential to counsel the patient regarding the surgical plan, including whether partial or complete excision is feasible. Ultimately, treatment must be individualized based on the patient’s specific symptoms, risks, medical history, and expectations.
Conclusion
Diagnostic imaging is essential for preoperative planning, postoperative assessment, and the management of mesh-related complications. Familiarity with the types of pelvic mesh, their imaging characteristics, various surgical techniques, and mesh-related complications is crucial in managing such complex cases. This patient case underscores that residual mesh should be considered in a differential diagnosis, when encountering echogenic bladder wall findings, as well as those patients who have undergone prior pelvic surgery.
This case report was provided to raise clinician awareness of the well-documented risks of repair-related complications and to strike a balance between the risk and the potential benefit of improved outcomes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for this study was waived by The Ethics Committee of the Institution, IGMC, Shimla, because all case data were de-identified and/or aggregated and followed the ethics committee or IRB guidelines.
Informed Consent
Verbal informed consent (verbal assent) was obtained from the subjects before the release of the report for review.
