Abstract
Teratomas arise from totipotent primordial cells and unveil a mixture of tissues of tri-dermal or bi-dermal origin. They are the most common ovarian germ cell tumors. The extragonadal teratomas may be seen in the sacrococcygeal region, mediastinum, retroperitoneum, and intracranially. These can be either mature, which are considered benign, or immature and classified as malignant. Mature cystic teratomas have characteristic imaging features and a pre-operative diagnosis that can be reliably made. The case provided was of a 48-year-old female who was diagnosed with three separate intraabdominal masses that had typical imaging features of teratomas. An extragonadal mesenteric teratoma is itself a rare entity and its coexistence with bilateral ovarian teratomas, has rarely been described in medical literature. Hence, common pathologies in an uncommon location should always be considered while providing a list of diagnostic differentials, in such a patient scenario.
Keywords
Teratomas are germ cell tumors (GCTs) arising from totipotent primordial cells that are present in midline and consist of two or three embryonic germ cell tissues that are foreign to anatomic site.1,2 These are the most common ovarian GCTs. However, they can also be in extra-ovarian sites. In order of decreasing frequency, the most common extragonadal GCTs can be located in the anterior mediastinum, retroperitoneum, as well as the pineal and suprasellar regions in adults. 2 In infants and young children, the most common sites for GCTs are the sacrococcygeal and intracranial regions. 2 This case study presents an unusual trio of intraabdominal mature cystic teratomas, in an adult female, one in each ovary and a third in mesentery, and is rarely described in the published literature.
Case Report
A 48-year-old, P 2+0, presented to the emergency department with a chief complaint of severe lower abdominal pain and vomiting. Her sonogram revealed an endometrial polyp and two adnexal masses in the pelvis. A third echogenic mass was seen in the lower abdomen (See Figure 1). As the masses were very echogenic with ill-defined borders, along with non-delineation of posterior margin, a possible diagnosis of a bilateral ovarian and one abdominal teratomas was given and magnetic resonance imaging (MRI) was advised for further characterization and anatomical delineation. The MRI examination (See Figure 2) showed bilateral ovarian cystic masses filled with fat and small specks of calcification, along their posterior walls. Both the ovarian masses showed diffusion restriction and no post-contrast enhancement. No septae, mural nodule, or soft tissue component was seen in either of these adnexal masses, so on MRI, they were considered as mature cystic teratomas. The third, separate cystic mass in the abdominal cavity demonstrated similar signal characteristic imaging features. A mural nodule was seen along its inferior aspect suggestive of a Rokitansky nodule. However, in addition, it showed areas of hemorrhage, thin enhancing septae, and post-contrast enhancement, more so, along its right anterolateral wall. Compiling these extra imaging features in the abdominal teratoma, a diagnosis of teratoma with a possibility of malignant degeneration was given. As the specks of calcification were small, a computed tomogram (CT) was further done, as an adjunct imaging technique, for better delineation of the calcifications, within the masses (See Figure 3).
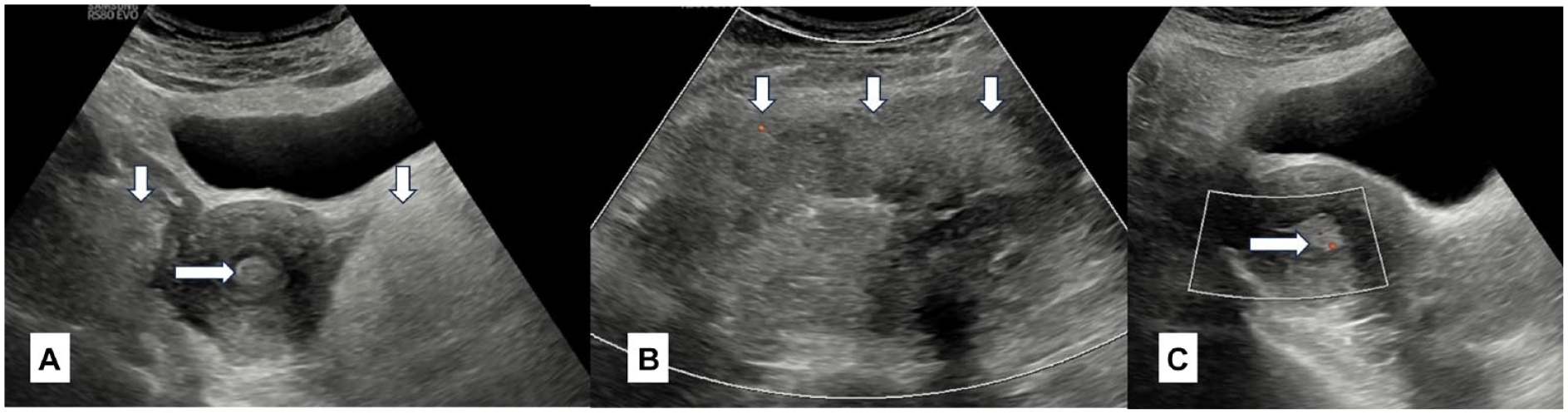
A 48-year-old, P 2+0 with pain lower abdomen. A grayscale sonographic axial image (A) shows two ill-defined echogenic masses in bilateral adnexae (vertical arrows) and an echogenic polyp within the endometrial cavity (horizontal arrow). (B) An echogenic mass in abdomen is provided (vertical arrows). (C) A sagittal color Doppler image of the uterus shows endometrial polyp (horizontal arrow) with a speck of vascularity indicating vascular pedicle of the polyp.
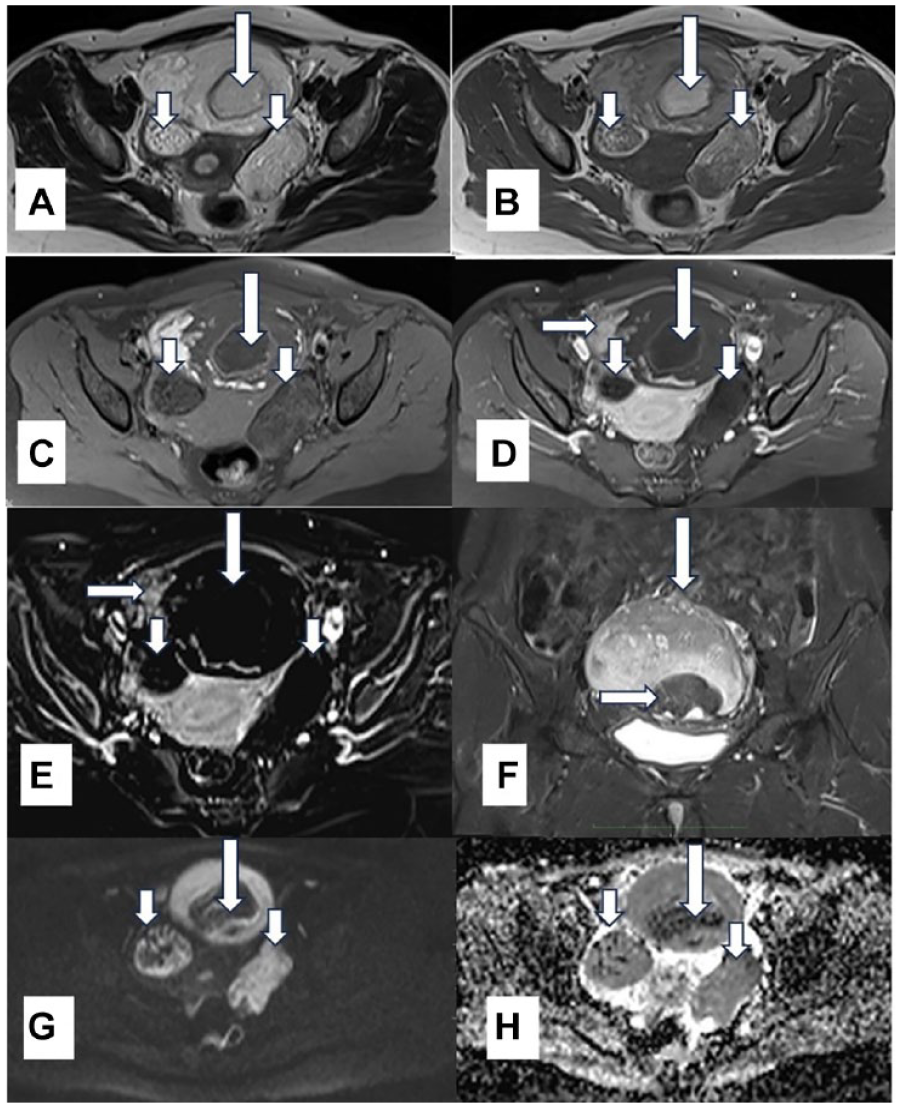
A magnetic resonance image of the same patient is provided. The mesenteric teratoma (long vertical arrow), bilateral adnexal teratomas (short vertical arrows) are heterogeneously hyperintense on T2 weighted (WI) axial image (A) and T1 WI (B) and show suppression on T1 fat saturated axial image (C). No enhancement is seen in bilateral adnexal masses as seen on post-contrast T1 WI (D) and in subtraction image (E). However, enhancement is obvious in the right anterolateral aspect of mesenteric teratoma (horizontal arrow) as seen on post-contrast T1 WI (D) and in subtraction image (E). Fat saturated, T2 W coronal image (F) shows the mesenteric teratoma (vertical arrow) and Rokitansky nodule (horizontal arrow) on its inferior aspect. Diffusion WI (G) and corresponding apparent diffusion coefficient values (H) show diffusion restriction in all three teratomas.
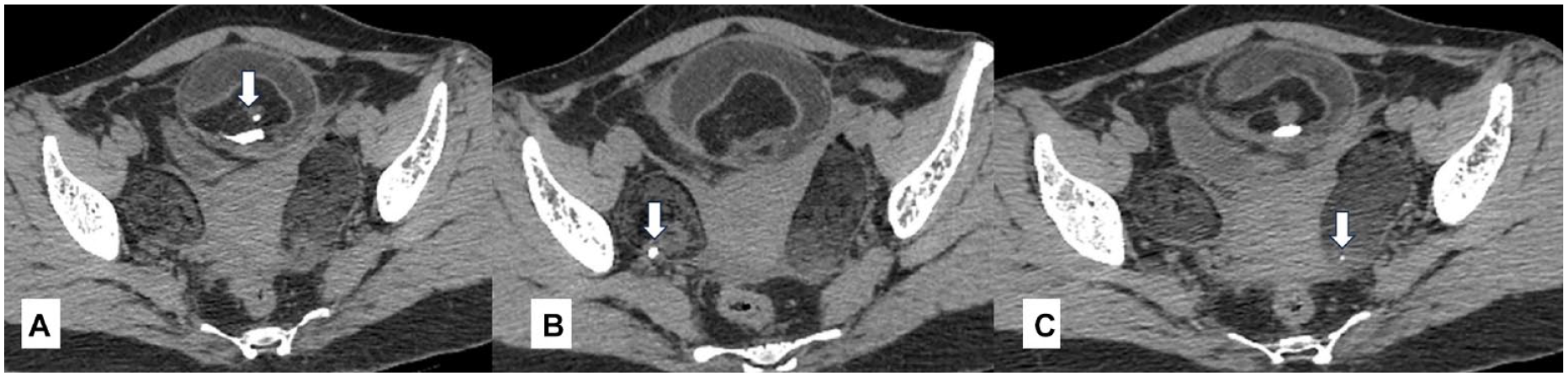
A non-contrast computed tomogram is provided with axial images that show (A) calcifications in mesenteric teratoma (vertical arrow), (B) calcification in right adnexal teratoma (vertical arrow), and (C) calcification in left adnexal teratoma (vertical arrow).
The patient’s routine blood parameters and tumor markers were normal. The patient underwent exploratory laparotomy to remove the bilateral adnexal masses. A third mesenteric mass was also excised, which had thick adhesions to the omentum (See Figure 4). Total abdominal hysterectomy with bilateral salpingo-oopherectomy with peritoneal toileting was done. On histopathological evaluation, all three masses were diagnosed as benign mature cystic teratomas with no malignant change despite the MRI indications of malignant degeneration in the larger mesenteric teratoma.
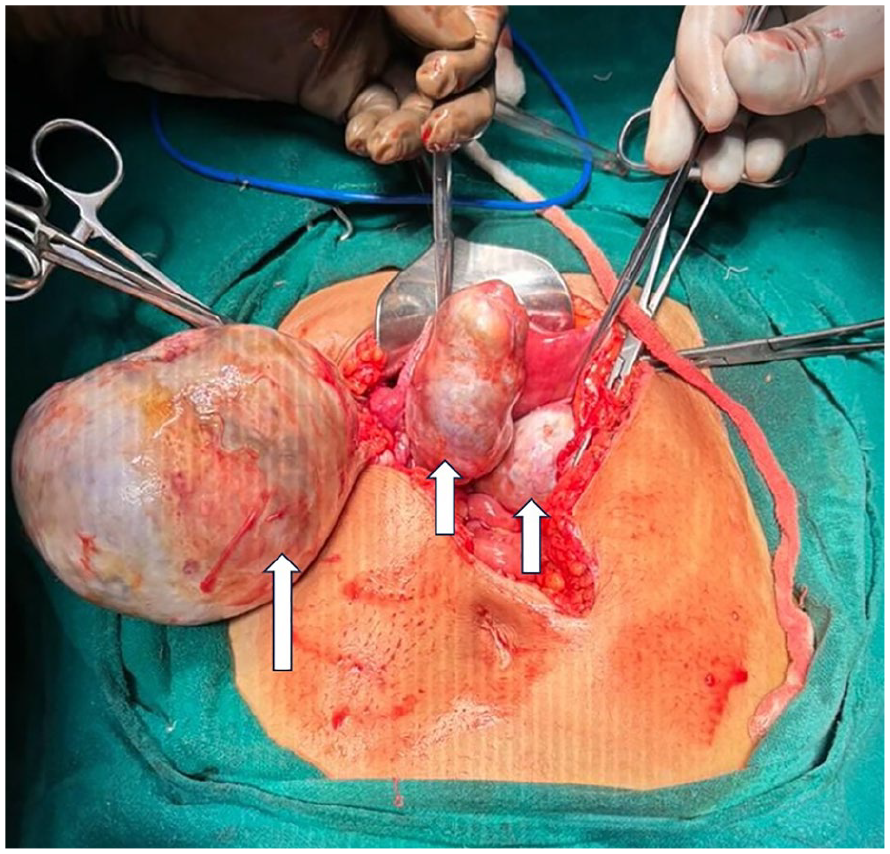
An intra-operative image shows mesenteric mass (long vertical arrow) and bilateral adnexal masses (short vertical arrows).
Discussion
The word “teratoma” is derived from the Greek words “teraton” meaning monster, and “onkoma” meaning swelling. It was coined by Rudolf Virchow in 1869. The current updated definition of teratoma is given by Willis as, “a tumor or neoplasm formed of several foreign tissues arising in an abnormal location, which is different from cases, which contain recognizable organs immaturely developed within the mass, as a result of separation from the fertilized ovum early in pregnancy called fetus in feto.”3,4 These tumors are filled with sebaceous material in 88% of cases and lined by squamous epithelium. 5 A raised protuberance usually projects into these cystic masses known as Rokitansky nodule. Hair, cartilage, teeth, and dermal appendages in different combinations can be found in these masses and are typically found in this protuberance. 5 However, mono-dermal teratomas like struma ovarii and carcinoids have also been described.3,5 Teratomas can have two variants and the first is a mature cystic teratomas that are considered benign. Whereas immature teratomas are usually considered to have a malignant potential. The mature benign teratomas are more common while the immature ones account for 1%–2% of cases. 5 The benign teratomas can transform into squamous cell carcinoma, adenocarcinoma, or carcinoid tumors. 4 There may be an increase in the levels of tumor markers, like CA 125 and CA 19-9, associated with these tumors.6,7
The benign mature cystic teratomas are more common in children, accounting for 50% of all ovarian tumors, than in young adults, in whom 20% of all ovarian tumors are teratomas. 6 Bilateral ovarian teratomas are seen in 8%–15% of patient cases. The origin of these cells are totipotent germ cells, which tend to be migratory. 2 There is a theoretical pathway of ectodermal cell migration, mainly in the midline, from the cranium, anterior mediastinum, retroperitoneum, and sacrococcygeal regions, which explains their extragonadal location. 4 Mesenteric teratomas are quite rare 8 with an incidence of one in 26,794 to 1/250,000 of hospital admissions 3 and one in 1,000,000 live births. 9 The pathogenesis behind mesenteric teratoma is that during embryonic development, germ cells migrate from the yolk sac to the genital ridge through the hind gut (route of mesentery). These germ cells may later give rise to teratomas in unusual anatomical locations. 4
Teratomas are usually asymptomatic for the patient. When present, the symptoms may be non-specific or may reflect their complications like cyst rupture, torsion, or infection. 7 Mostly, the clinical complaint is due to their large size that compresses upon the adjacent organs. The patient may present with acute abdomen, intestinal obstruction, or chemical peritonitis. 2
The mature cystic teratomas have typical imaging features that highlight their composition. With sonography, a varied appearance of mature cystic teratomas may be noted. This can appear as a cystic mass, having an echogenic Rokitansky nodule, to a completely or partially echogenic mass, with ill-defined posterior borders, attributed to fat and calcification. It can also appear to have multiple thin echogenic lines, from the reflecting hair, in the cyst. The fat content may be seen in 50% of cases that may show fluid-fluid level. The calcification may be seen in 50%–75% of patient cases. 10 On CT and MRI, these characteristic imaging features showing fat and calcification are well seen. Computed tomogram demonstrates fatty attenuation areas that can range from −30 to −190 Hounsfield units (HUs), in these masses. Small specks of calcification are better delineated with CT than MRI which shows radiodense area, as CT attenuation can be well above 200 HUs. Magnetic resonance imaging can also easily pick up macroscopic or microscopic fat in tissue, with conventional MRI sequences (T1, T2, and fat saturated) and also on advanced fat detecting sequences (Chemical shift imaging, inversion recovery, Dixon method, etc.). However, the malignant degeneration of benign teratomas is difficult to predict at an early stage unless they have metastasized. Diffusion restriction, on MRI, measures the random Brownian motion of water molecules within a voxel of tissue and helps in tumor characterization. This type of diagnostic information is not a criterion for malignancy, as it may also be seen in mature teratomas. Immature malignant teratomas are displayed as large complex solid-cystic masses with a predominant solid component that may show internal hemorrhage or necrosis. They may show scattered calcification unlike calcification in mature cystic teratomas that is localized to mural nodules. A few foci of fat or absence of fat may be noted. They have a rapid growth and may perforate the capsule, which becomes ill-defined.
The optimum treatment is surgical intervention. Open surgery is preferred over laparoscopic surgery because often the tumor is large and has close proximity to adjacent organs. This also can prevent its rupture that can cause seedling of tumor cells and recurrence, or may cause chemical peritonitis. 7 The tumor should be excised in total, with clear margins, as the pre-operative imaging findings may not be accurate in predicting the benignity of the mass. This result was the example outcome for the current patient case.
Conclusion
Mature cystic teratomas have typical imaging features and do not pose a diagnostic challenge if present in their typical location. However, they should still be considered as a diagnostic possibility if present in an unusual anatomical location.
Footnotes
Ethics Approval
Ethical approval was not sought for the present study because all patient information was de-identified.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
