Abstract
A cholangiocarcinoma is a cancerous growth that originates within the biliary tree and spans from the bile ductules, in the intrahepatic region, to the ampulla of Vater. Most of these tumors are adenocarcinomas, with squamous cell carcinoma (SCC) of the bile duct and are considered a rare finding. Published literature on these types of malignancies is limited. This case report provides an example of cholangiocarcinoma in SSC type with liver metastasis. Computed tomography (CT), ultrasonography, magnetic resonance imaging (MRI) with contrast, and magnetic resonance cholangiopancreatography (MRCP) were performed, and the results showed a necrotic mass throughout the central liver. Finally, a liver biopsy was completed and yielding a diagnosis of an SSC of the bile duct.
Cholangiocarcinoma (CCA) is a form of cancer that develops in the biliary tree and can affect any segment of the bile duct. Many of these tumors are typically identified as adenocarcinomas.1,2 In contrast, squamous cell carcinoma (SCC) is a rare occurrence, accounting for only 1.4% of all malignant biliary tumors. 3 Tumor progression is characterized by rapid yet asymptomatic development, frequently resulting in an advanced stage diagnosis, thereby attributing to an unfavorable prognosis. Few patients survive more than 12 months, even after surgical resection.2,4 Diagnosing CCA poses a diagnostic challenge regardless of the various imaging techniques. The similarity between its common clinical symptoms, such as nausea, vomiting, jaundice, and the typical indication of classic cholecystitis, increases the risk of a misdiagnosis. 5
Case Report
A woman in her early eighties arrived at the hospital through the emergency department and was complaining of fever and persistent abdominal pain, which had lasted four days. She also exhibited a lack of appetite and general weakness that had been persisted over four months. An elevated white blood cell count (WBC), as part of her lab results, raised concerns of sepsis. Her aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels were within the normal range. Tumor marker, CA19-9, also yielded normal results. Further investigation through an abdominal and pelvic computed tomogram (CT), without contrast, revealed multiple infiltrative masses in the liver, adjacent to an anomalous-looking gallbladder (See Figure 1). In addition, a necrotic periportal lymph node was identified, as part of that examination (See Figure 2). An abdominal sonogram was performed the same day and showed a diffusely heterogeneous liver with multiple necrotic appearing masses and an indistinct gallbladder (See Figures 3 and 4). A possible calcification, within the gallbladder wall, was also visualized (See Figure 5). The porta hepatis was surrounded by necrotic soft tissue, resulting in poor visualization of the common bile duct (CBD) (See Figures 6–8). Later the next day, a magnetic resonance imaging (MRI) study, with contrast, and a magnetic resonance cholangiopancreatography (MRCP) revealed centrally necrotic masses, throughout the central liver (See Figure 9). It also demonstrated mild to moderate intrahepatic biliary dilatation in both the right and left liver lobes. The gallbladder was grossly normal in appearance (See Figure 10). The CBD was measured, and a 3.0-mm stone was found in the distal portion of the duct. Following the MRCP, a liver biopsy was performed which confirmed a malignancy. Histological examination verified the existence of SSC of the bile ducts. Brain and lung metastases were excluded due to the results of a head and chest CT examination. Unfortunately, the liver tumors were determined to be unresectable. After managing the patient’s pain and reducing her fever, she was discharged. The patient was referred to palliative care, due to her declining strength, as well as a decision against aggressive chemotherapy. Subsequently, the patient stopped attending appointments for medical surveillance.
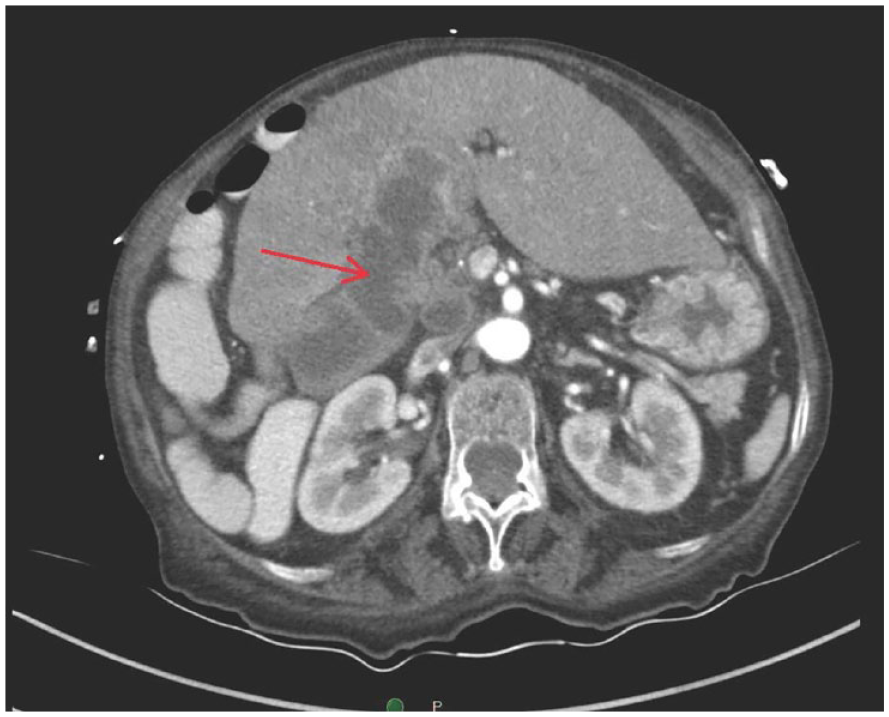
A computed tomographic axial image demonstrated multiple infiltrative masses in the liver (red arrow). The gallbladder was ill-defined in this view.
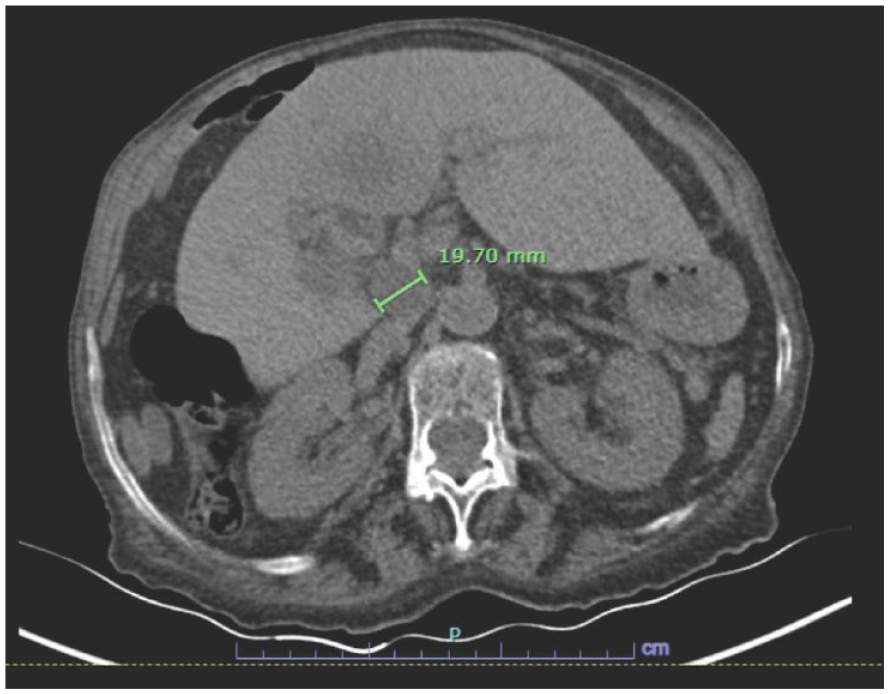
A computed tomographic axial image demonstrated a necrotic periportal lymph node that measured 19.7 mm (note green calipers).
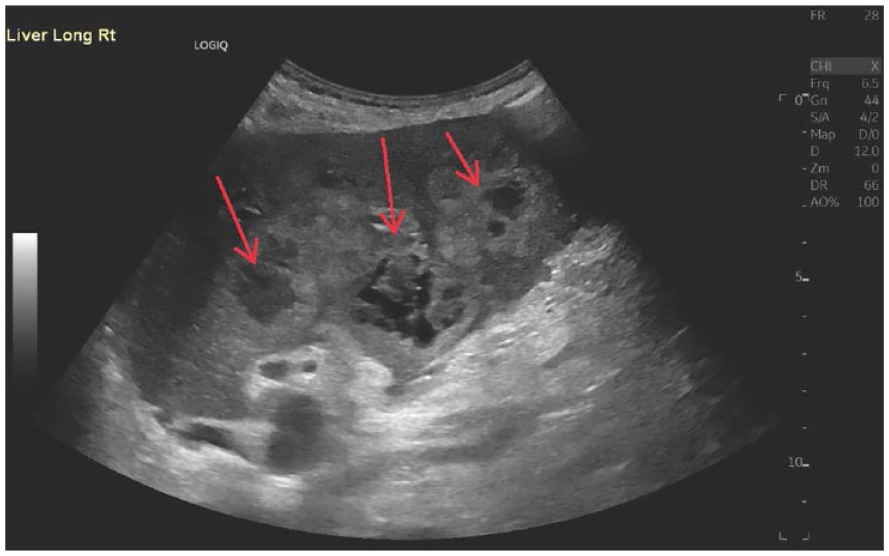
This sagittal sonogram of the liver demonstrated a diffusely heterogeneous texture, which featured numerous heterogeneous liver masses (red arrows) emanating from the porta hepatis, and the gallbladder was not clearly delineated in this view.
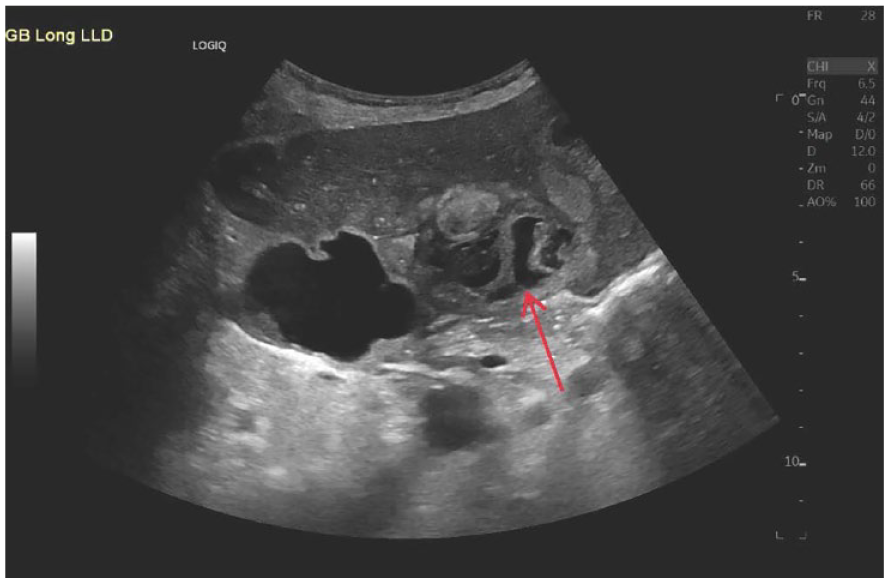
This sagittal sonogram of the liver demonstrated a multilocular cystic structure (red arrow), within the ill-defined gallbladder.
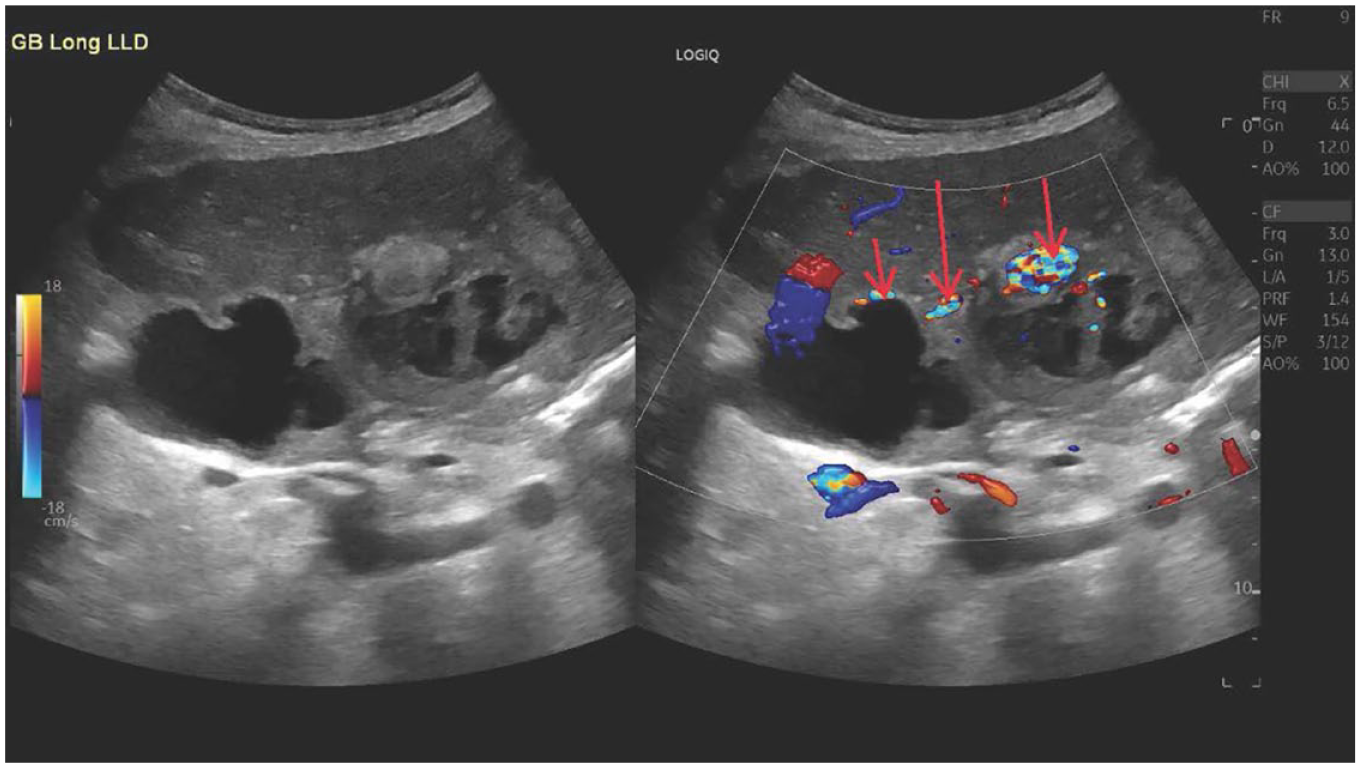
In these dual sonographic views of the liver, color Doppler demonstrated calcifications at certain areas (red arrows) within the gallbladder wall.
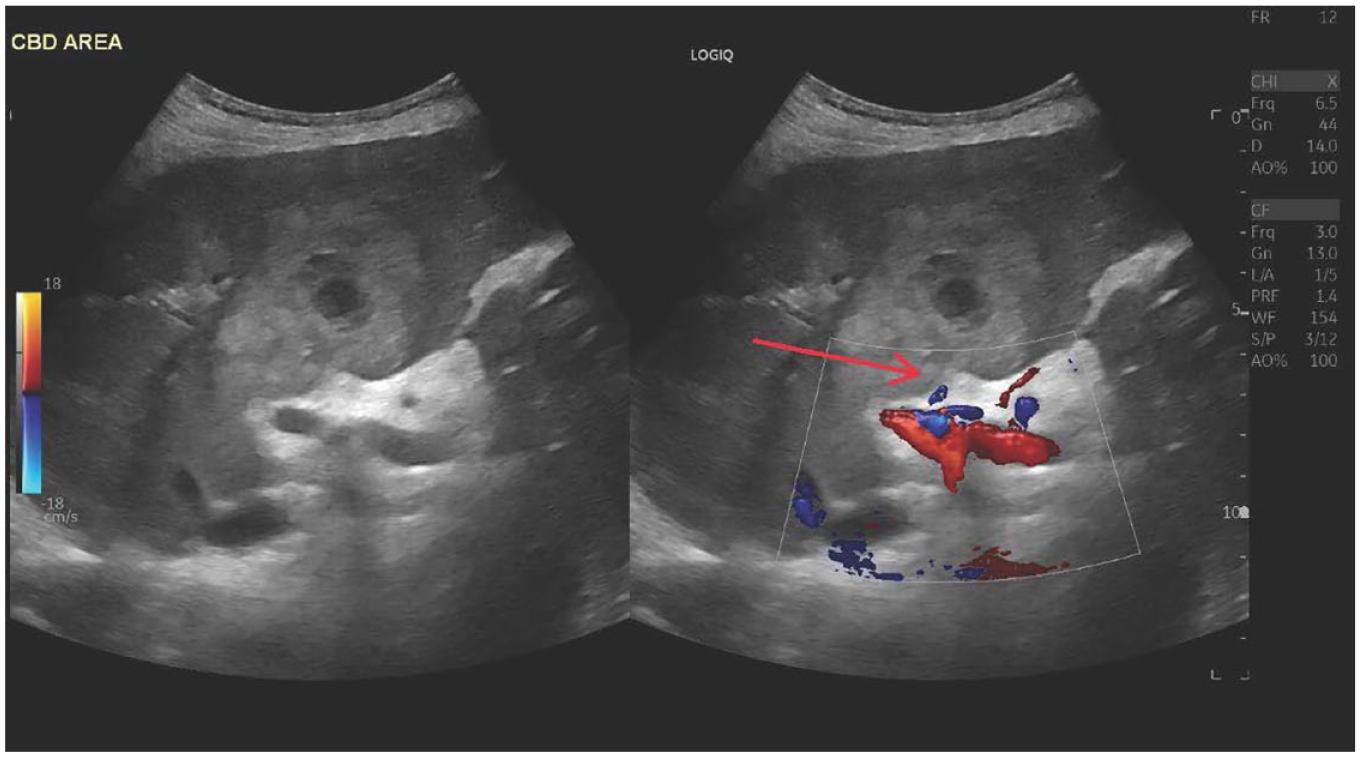
These dual sonographic views of the liver, color Doppler indicated necrotic soft tissue around the porta hepatis and extended into the liver (red arrow). The common bile duct was not well visualized.
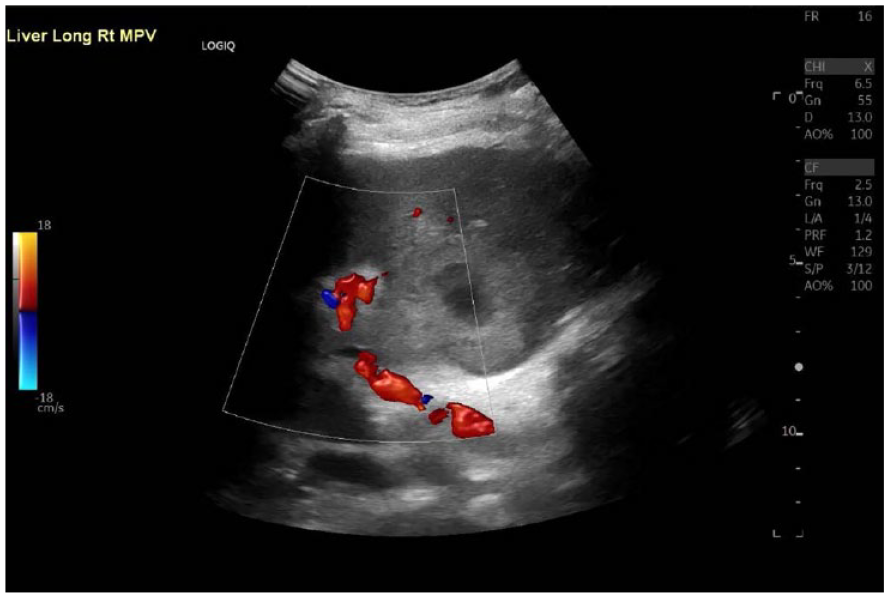
This sagittal sonogram of the liver, with color Doppler, provided improved visualization of the porta hepatis.
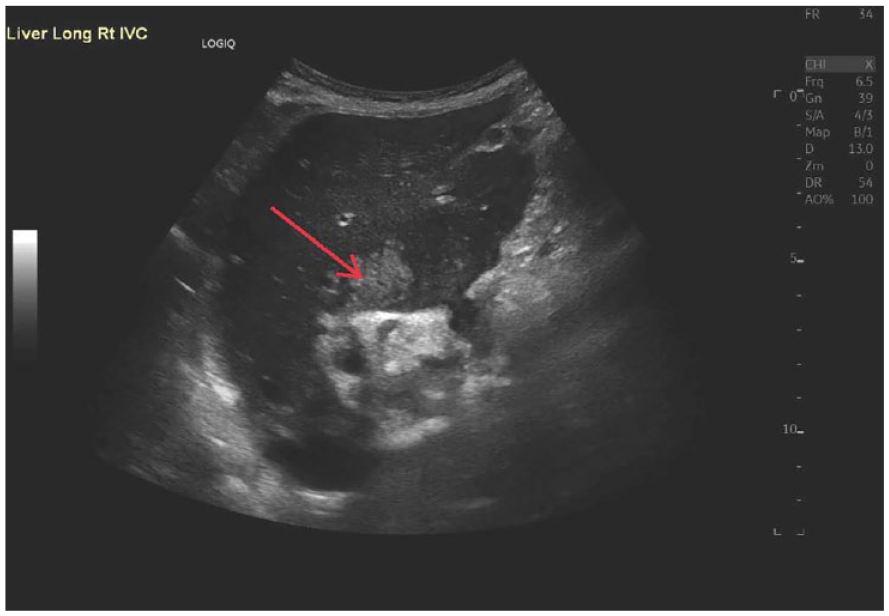
This sagittal sonogram of the liver shows the central region of the liver, which displays heterogeneous tissue (red arrow).
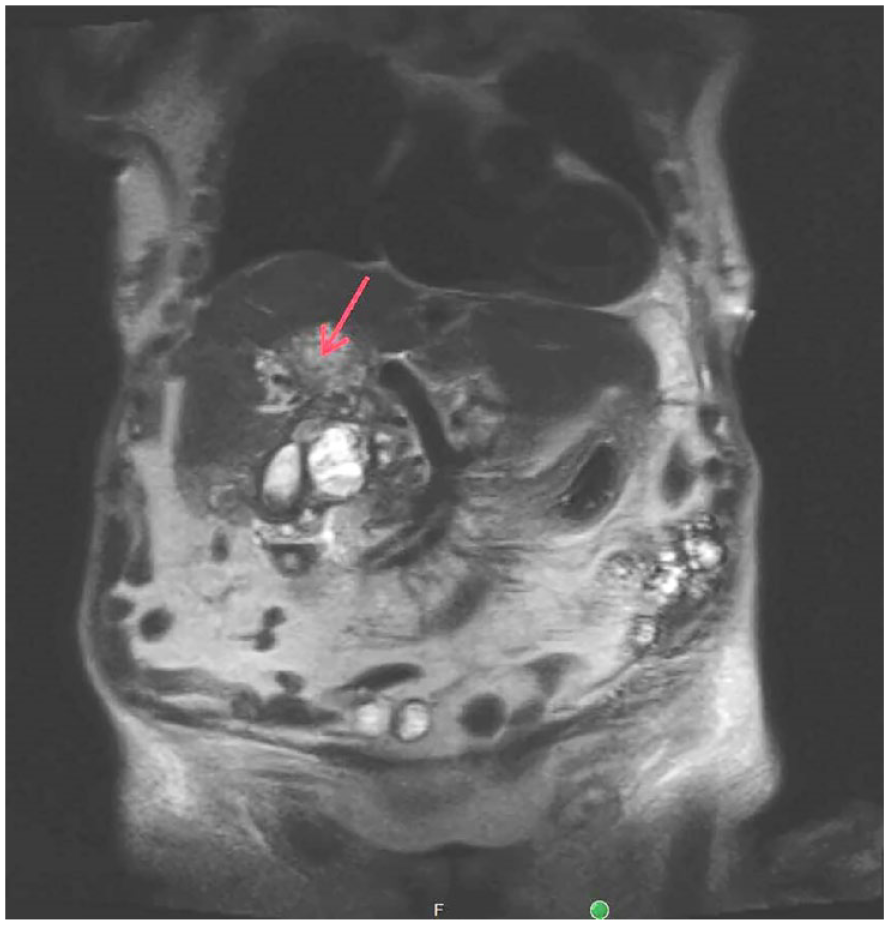
This coronal magnetic resonance imaging view, with contrast, demonstrates the centrally necrotic masses (red arrow) throughout the central liver.
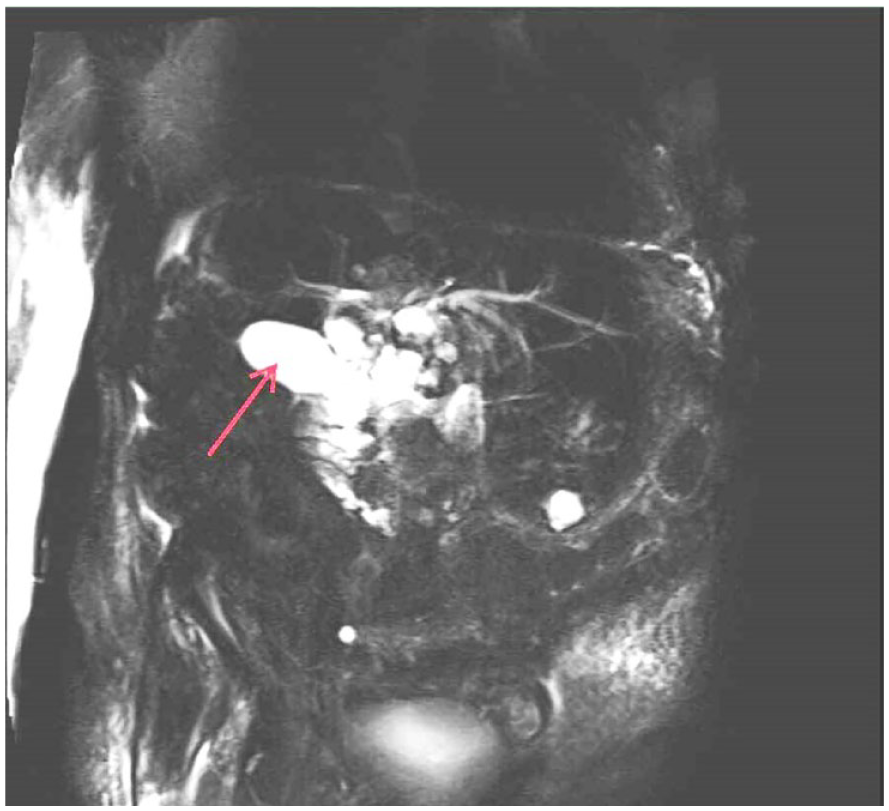
A coronal view captured during the magnetic resonance (MR) cholangiopancreatography revealed a gallbladder (red arrow) with a grossly normal MR appearance.
Discussion
Epidemiology and Etiology
Cholangiocarcinoma, the primary cancer of the bile ducts, accounts for only 10% to 15% of hepatobiliary neoplasms, yet its incidence is increasing. 6 An estimated 3000 to 4000 new cases of carcinoma, in the bile ducts, are diagnosed annually in the United States. 7 The peak incidence of CCA is observed during the 6th to 7th decade with a slight male predominance. 8 Two thirds of CCA affect the extrahepatic bile ducts, whereas the remaining one third impacts the intrahepatic biliary tree.8,9 The most often seen risk factor for CCA is primary sclerosing cholangitis (PSC). 5 Hepatolithiasis, recurrent pyogenic cholangitis may be predisposing factors. 1 The most common malignant tumor of the bile duct are adenocarcinomas. Squamous cell carcinomas are very rare with an incidence of less than 1.4%. 3 Many theories have been proposed on its etiology. Histologically, the biliary mucosa is composed of a single layered cuboidal/columnar epithelium without squamous epithelial cells. Persistent irritation from an inflammatory stimulus is believed to be the primary mechanism, triggering carcinomatous transformations in the normal cuboidal/columnar epithelium, accomplished via squamous metaplasia, of the biliary epithelium.1,2,10
Pathophysiology and Presentation
According to the primary onset location in the biliary tree, CCA is classified into extrahepatic (eCCA) and intrahepatic (iCCA) types. 9 Although the majority of the literature centers on adenocarcinoma-type CCA, the clinical features of SCC-type CCA bear resemblance to those of adenocarcinoma-type, potentially indicating heightened aggressiveness. 2 Given the advanced stage of diagnosis in this particular case, discerning whether it pertains to eCCA or iCCA poses a challenge. Current research would indicate that when a patient exhibits a primary liver mass without discernible evidence of extrahepatic disease upon examination, laboratory tests, and imaging studies, the likelihood of iCCA origin is increased. 9 Given that the current patient case mirrors these aforementioned characteristics, the focus of discussion pertains to iCCA pathology and presentation.
In contrast to eCCA, which spreads along the bile ducts in a longitudinal and transmural manner, iCCA can infiltrate the adjacent hepatic parenchyma directly, reaching the portal pedicle and bile ducts through contiguous spread. Vascular invasion, particularly involving branches of the portal vein or hepatic artery, is commonly seen, along with lymph node extension in the hepatic pedicle. 8 The iCCA is classified into four growth types: Mass Forming, Periductal Infiltrating, Mass Forming with Periductal Infiltrating, and Intraductal. 9
Detecting iCCA, in its early stages, poses a diagnostic challenge, as its clinical symptoms, such as nausea, vomiting, elevated liver function tests, and jaundice, often mimic those of classic cholecystitis. Later stages may present chronic symptoms like diminished appetite, weight loss, malaise, and night sweats. Laboratory tests usually show an increased alkaline phosphatase with a normal bilirubin. Serum tumor markers, such as CA 19-9, may be increased. 9
The current patient case revealed unusual laboratory findings, with an exceptionally high WBC (18.8 × 109/L), normal liver function tests, and a CA19-9 tumor marker within the normal range (uncommon in CCA cases, whether it is adenocarcinoma or SCC). In addition, jaundice, a typical symptom of biliary/liver disease, was absent in this patient. The atypical disease presentation remains unexplained. Given the nonspecific symptoms and blood lab results, diagnosis commonly occurs incidentally through imaging, sonography, CT, and/or MRI. Although diagnostic imaging features can be helpful, a liver mass biopsy is the most reliable way to confirm the diagnosis. Liver biopsy is considered the diagnostic gold standard as the results yield malignant cells with squamous cell differentiation.
Sonography
Sonography is often the initial imaging study, in patients presenting with obstructive jaundice. Usually, the first visible sonographic finding suggestive of CCA is intrahepatic ductal dilatation. 5 Sonography serves as a valuable diagnostic tool for excluding benign causes of bile duct obstruction, such as choledocholithiasis. It also reliably visualizes the intrahepatic ductal anatomy, along with pinpointing the proximal obstruction site. It is common for sonography to not visualize small strictures and masses, yet it can demonstrate lesions forming masses and invading the surrounding liver tissue or affecting portal vessels. 11 Sonographically, iCCA has variable echogenicity but tends to be hyperechoic. The internal architecture of the tumor is usually homogeneous, but it can be heterogeneous, depending on the amount of fibrous tissue, mucin, and calcification. 11 If the mass is located beyond the cystic duct, biliary stasis will occur and result in gallbladder distention and stagnant bile within the intrahepatic ducts. 5
Computed Tomography
On CT, iCCA can present as well-defined or infiltrative lesions, distinct from hepatocellular carcinoma (HCC) due to the absence of fibrous capsules. The iCCA tumors typically appear hypo- or iso-attenuating compared with normal liver tissue. Notably, the degree of enhancement can vary among tumors, particularly with necrotic or mucin-producing types. In addition, some small mass-forming iCCAs may show arterial enhancement, resembling HCC. 11 Following intravenous contrast administration, the periphery of these masses, where active tumor tissue is concentrated, may exhibit rapid and intense enhancement that subsequently diminishes to isodensity or hypodensity during the portal venous phase. Due to the fibrous nature of CCA, the central region of the tumor typically does not enhance during the arterial or portal venous phases but becomes hyperdense in delayed images acquired 5 to 10 minutes post-injection. However, the central area of necrotic or mucin-producing tumors tends to remain hypodense. 12
Magnetic Resonance Imaging
The MRI features of iCCA differ according to its pattern of growth. The iCCA is typically hypo- to iso-intense on T1-weighted (T1W) and variably hyperintense on T2-weighted (T2W) imaging.11,12 The heterogeneous appearance of these lesions on T2W images can explain the low signal intensity of fibrous tissue and the higher signal intensity of mucous and myxoid degeneration. 12 Similar to CT, contrast enhancement may occur early in the periphery of the lesion during MRI, whereas the fibrous components located in the center of the tumor tend to exhibit enhancement at a later stage. 12 Distinguishing between iCCA and HCC poses challenges, particularly as iCCA can manifest in cirrhotic livers. 11 Laboratory tests play a crucial role in diagnosis, as alpha-fetoprotein (AFP) levels are typically within normal range or only slightly elevated in iCCA compared with HCC. 11
Magnetic Resonance Cholangiopancreatography
Magnetic resonance cholangiopancreatography is an accurate method for anatomically mapping the biliary tree. In addition, acquisition of 3D data sets provides information useful for preoperative management and surgical planning. 11 Magnetic resonance cholangiopancreatography images can reveal various features associated with iCCA, such as the displacement of bile ducts away from the tumor, obstruction within intrahepatic ducts, or the presence of a polypoid mass within the ductal system. 12
In summary, among gastrointestinal tumors, CCA is the most difficult to detect and diagnose. 13 The diagnosis of CCA presents difficulties when employing different imaging techniques. The iCCA typically present as a poorly marginated mass on cross-sectional imaging studies. 12 In the case study patient, the gallbladder’s appearance proved challenging to delineate with sonography and CT, whereas the MRI indicated a grossly normal appearance of the gallbladder. It is plausible that the proximity of intrahepatic tumors to the gallbladder, coupled with extensive overlapping necrotic tissue, contributed to the diagnostic imaging challenges. The use of contrast-enhanced MRCP, however, successfully revealed the grossly normal appearance of the gallbladder. Irrespective of the gallbladder’s visual characteristics, imaging techniques consistently revealed liver masses containing centrally necrotic tissue, which extended into gallbladder fossa.
Treatment
Cholangiocarcinoma is not only challenging to diagnose and visualize through imaging but also carries a grim prognosis, with a 5-year survival rate of less than 5%. 13 Due to the extremely rare incidence of biliary SCCs, no standardized therapeutic strategies have been established. Opting for radical resection remains the sole choice for achieving long-term survival, as patients without surgical intervention experience a mean survival time of less than 12 months.2,10 Resectability depends on the biliary extent of the tumor and involvement of the portal vein and hepatic artery. 2 In cases where resection is possible, the next step is chemotherapy and/or radiation. However, unlike lesions at other sites, SSC in the bile duct is resistant to radiation therapy. 2 The recommended chemotherapy is GEMOX (gemcitabine plus oxaliplatin) or GP (gemcitabine plus cisplatin), as in bile duct adenocarcinomas. Yet, research indicates that there is no statistically significant difference in prognosis between patients who receive chemotherapy and those who do not. 10 When resection and liver transplantation are not options, palliative care is offered, and a biliary stent is often placed, to bypass the mass.5,14
Conclusion
Cholangiocarcinoma is frequently diagnosed in advanced stages due to both non-specific symptoms and challenges in visualizing early-stage bile duct cancer on imaging. Squamous cell carcinoma, of the bile duct, is an uncommon occurrence, sharing clinical features with adenocarcinoma but possibly exhibiting increased aggressiveness. In this patient case, the treatment plan was conservative because metastasis was already observed within the liver, as well as portacaval lymphadenopathy. This stage of the disease excluded surgical resection, as a treatment option. Multimodality imaging can play an integral role in the management of the biliary tract malignancies.
Footnotes
Acknowledgements
The author extends her gratitude to Cindi Reber-Bonhall and Tim Gibbs at Orange Coast College, as well as Mimi Reise at Providence St. Joseph Hospital Orange, for their insightful discussions regarding this case, which greatly contributed to the development of this paper.
Ethics Approval
Ethical approval was not sought for the present study because this case study is a retrospective study with images all de-identified.
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
