Abstract
Objective:
The aim of this study was to investigate the usefulness of recording sonographic fetal scalp thickness (FST) to predict the estimated fetal weight (EFW), in third trimester of pregnancy.
Materials and Methods:
A cohort of 175 uncomplicated singleton pregnancies, at 27 to 40 gestational weeks, was included in this study. The FST was measured with sonography and defined as the distance between the fetal skin and the outer table, on a cross-section of the skull, along the parietal aspect of the fetal head. Measurements were taken twice, and the average of the measurements was recorded. The estimated fetal weight (EFW) was derived from the Hadlock’s formula. Simple linear and multiple linear regression models were performed to predict the EFW, based on the FST measures.
Results:
A strong positive correlation was demonstrated between FST and the EFW (r = 0.65,
Conclusion:
A potential may exist to use FST as a viable indicator of EFW and could be used in conjunction with current fetal biometric parameters to improve EFW prediction.
In the third trimester of pregnancy, there is heightened fat accumulation around specific fetal anatomic sites, such as the head, trunk, arms, face, and subcutaneous tissues.1 –3 During this time, there is marked acceleration of fetal weight, especially between the 26th and 40th weeks of gestation, at an increased rate of nearly 4-fold. 4 Any fluctuation in this process could result in a significant change in fetal growth trajectory, weight, and the overall nutritional score,4,5 which may indicate some underpinning maternal factors or pathologic process.
In Nigeria, about 241 000 newborns suffer avoidable demises on a yearly basis, giving a perinatal mortality rate of 39 to 130 per 1000 live births. 6 A large majority of these fetal deaths are linked with diminished estimated fetal weight (EFW), which is considered an important parameter in determining neonatal survival. 7 An estimated 16% of live-born infants have a low birth weight, which is associated with high perinatal morbidity and mortality. Low birth weight pregnancies are largely associated with fetal prematurity or intrauterine growth restriction, 5 while excessive birth weight is associated with maternal morbidity, shoulder dystocia, birth asphyxia, and trauma. Precise EFW can help to reduce the risk of the complications associated with growth-restricted and macrosomia, as well as improving perinatal outcomes.3,4
Sonography offers a simple and accurate method of calculating EFW, and this metric has the potential to transform obstetric care and reducing perinatal mortality and morbidity, through early detection of diminished fetal growth 7 Regardless of conflicting opinions, from its inception, this set of measurements show higher accuracies than clinical methods for EFW, 2 that includes fetal biparietal diameter (BPD), head circumference (HC), femur length (FL), and abdominal circumference (AC). While the fetal AC has been shown to be a strong determinant of EFW, albeit some studies have shown that the AC may be misleading in assessing EFW, for example, in diabetic patients. 8 Abramowicz et al 8 stated that sonography could be unreliable in the EFW where there are irregularities in anatomic changes with fetal growth and can be influenced by disproportionate increases in biometric parameters, as pregnancy advances. According to Owen et al, 9 less than 3% of births occur at 40 weeks gestation. However, the normal range of term EFW is referenced to the mean EFW, for those patients delivered at 32 to 42 weeks gestation. The mean EFW, during the third trimester, varies considerably and depends on many factors, and include maternal race, size, parity, pregnancy, weight gain, hematocrit, and ambient elevation. 10 One study has outlined several factors that may affect traditional sonographic parameters for EFW; these include oligohydramnios, polyhydramnios, anterior placentation, fetal presentation, and crowning. 11
Correct determination of EFW, prior to delivery, is important in obstetric care and can influence pregnancy outcome and survival of the newborn. Documented evidence shows that many fetal biometric parameters can be inaccurate in some scenarios, 12 which gives room for other sonographic parameters to be explored, in calculating EFW. A few researchers have reported that sonographic EFW can fluctuate by as much as 15%, compared to actual birth weight, and more deviations can be attributed to macrosomia and fetal growth restriction.13,14 This argument is hinged on the fact that EFW is derived from indirect measures of fat mass and shows poor correlation with newborn adiposity.13,15 This understanding has since triggered a few studies that have modeled fetal fat mass deposits (majorly subcutaneous) into the EFW. Since the fetal head represents one of the anatomic fatty deposition mass sites, during fetal development, scalp thickness could be used as a single fetal metric, as well as combined with other fetal biometry measures, to calculate EFW in the third trimester.
Materials and Methods
This research was a prospective analytical study of a cohort of uncomplicated third-trimester pregnancies, assessed from July and September 2022 in Calabar, Nigeria. Patients with singleton pregnancies, at 27 to 40 weeks gestation, were recruited into this study. Pregnancies presenting with the following conditions were excluded from the cohort: polyhydramnios, oligohydramnios, suspected fetal intrauterine growth restriction, chronic alcoholic mothers, fetus below gestational age of 27 weeks, and multiple gestation, known abnormal karyotypes, congenital malformation, hypertensive patients, and gestational diabetes mellitus. A cohort of 175 patients was recruited into the study, based on a statistical power analysis. Ethical approval for the study was obtained from the Ethical Research Committee within the Department of Radiography and Radiological Science, at the University of Calabar (IRB# UC/ECRA/22/015).
Sonographic Imaging and Measurements
A Sonoscape P50 Elite (Shanghai Heyi Medical Instrument Co, Ltd) ultrasound equipment system was used for this study. The obstetric sonograms and measurements were performed by a single sonographer, with over 18 years of experience, in diagnostic sonography. The quality assessment of images and a repetition of sonographic measurements were performed as part of the equipment’s quality control measures. The mechanical index (MI) was kept below 0.3.
Subjects were examined supine on the cart and the sonographic images were completed in different scan planes until a good ovoid hyperechoic structure (bony margin), of the fetal skull, was obtained. The landmark for measurement was recognized by the presence of the
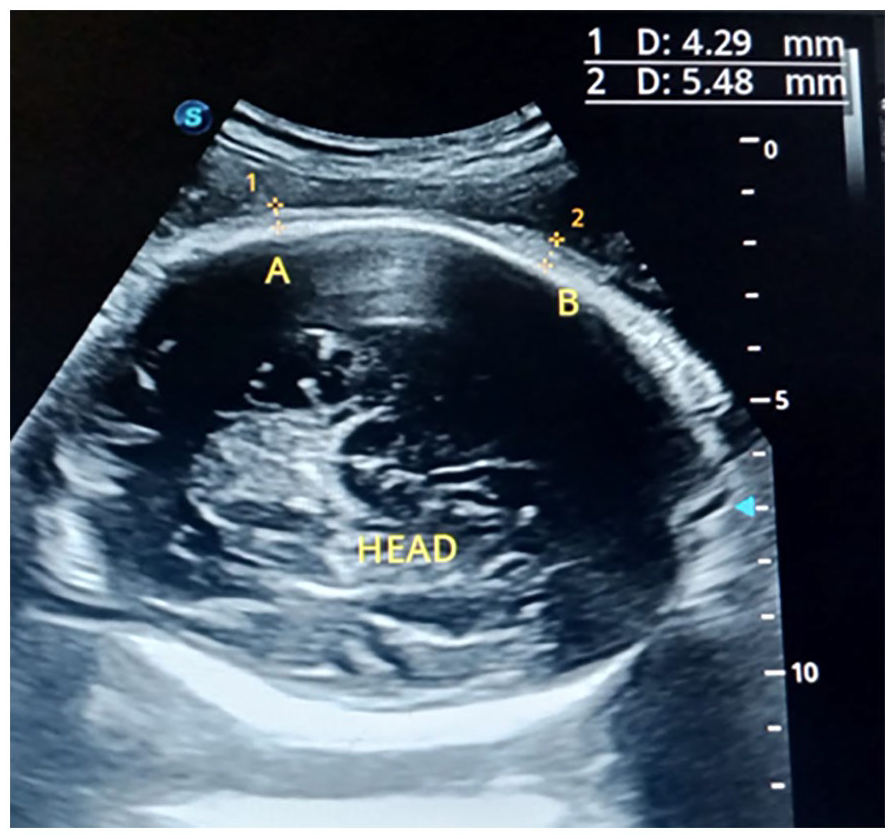
A sample sonogram of the fetal head, with electronic measurement calipers is provided. This transverse sonographic plane shows the fetal scalp thickness (FST), for this fetus, at 33 weeks gestation.
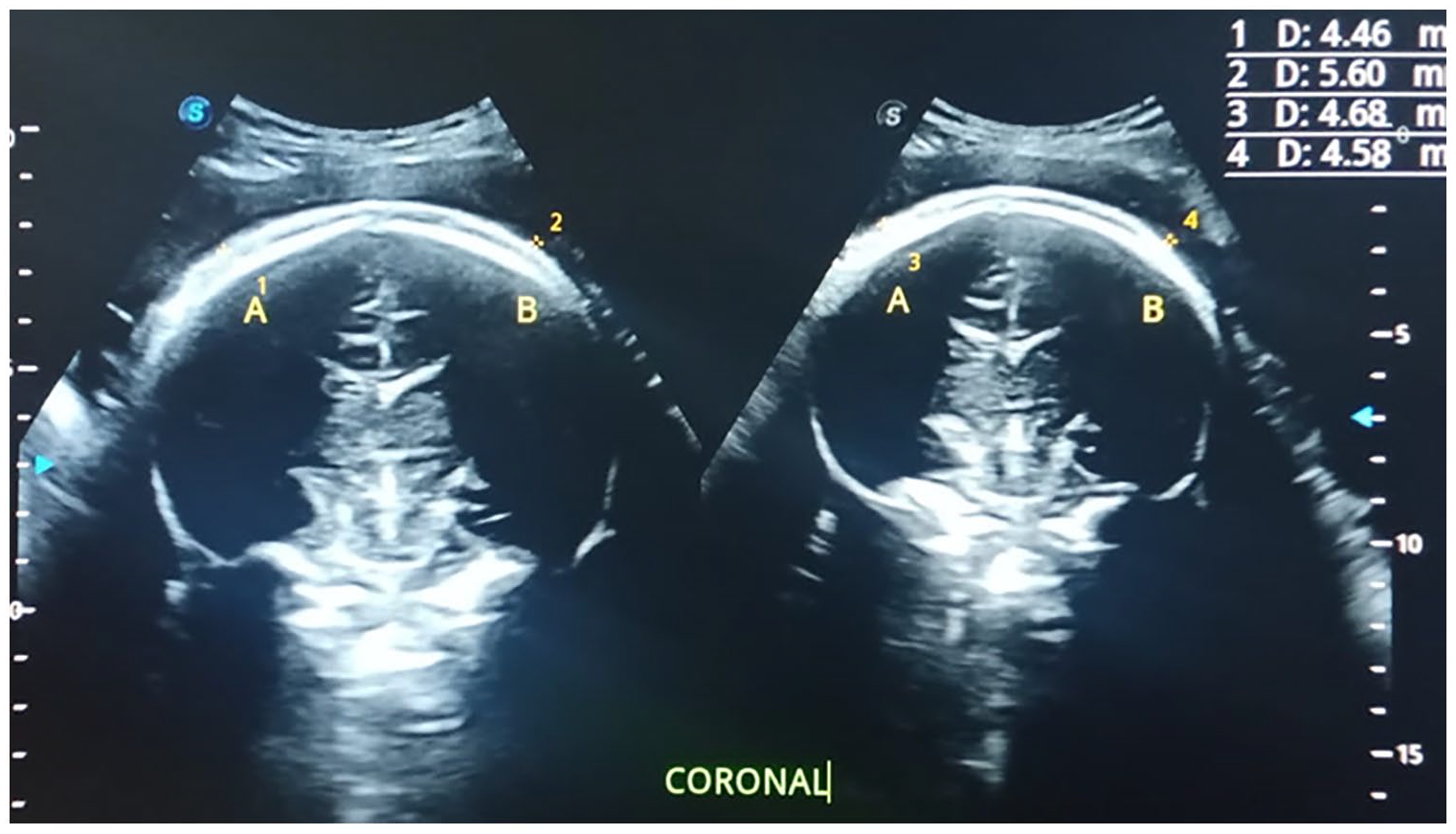
A sample dual sonogram of the fetal head, with electronic measurement calipers is provided. This coronal view of the fetal brain allowed for an additional fetal scalp thickness (FST) to be taken at 38 weeks gestation.
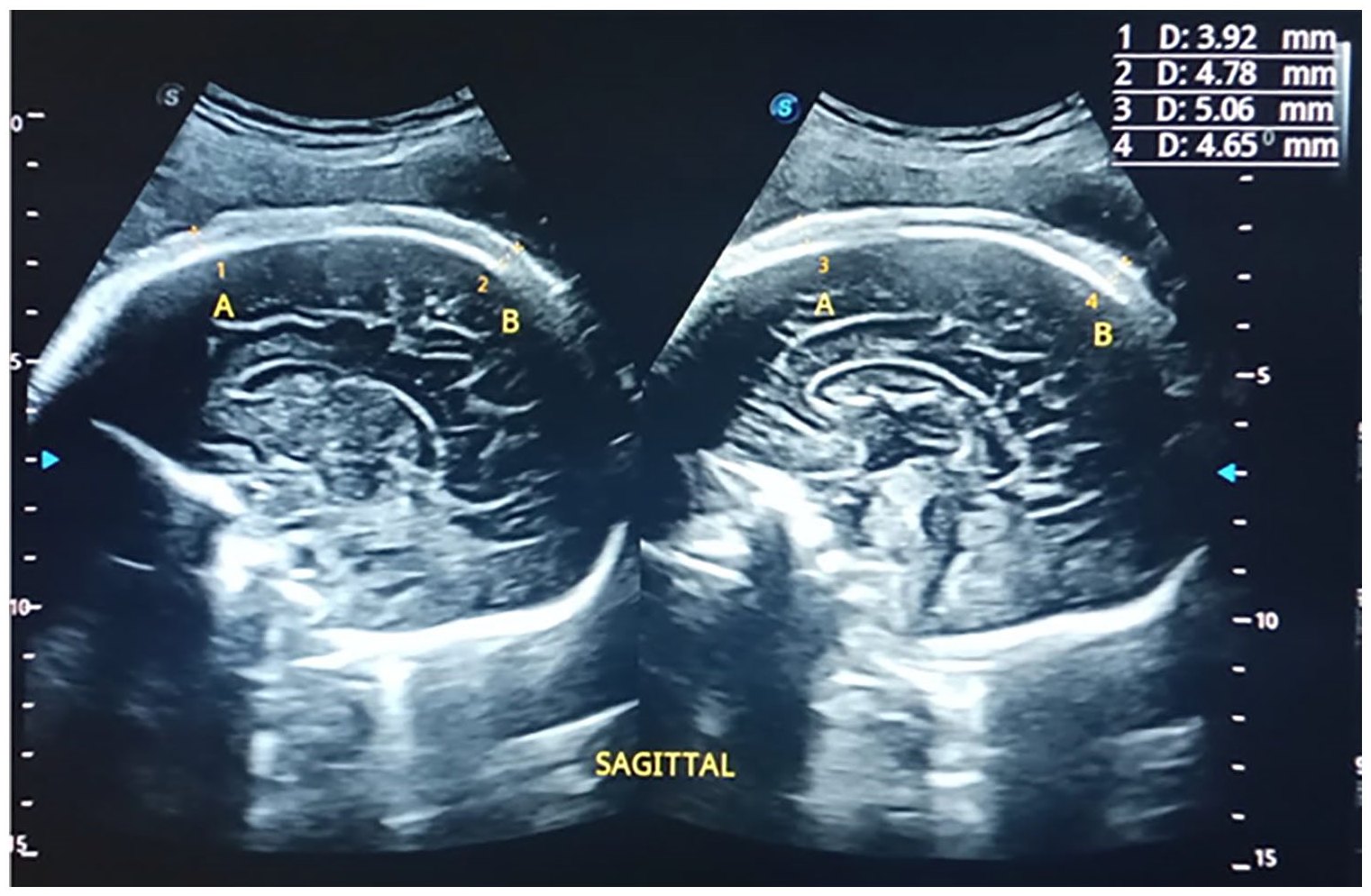
A sample dual sonogram of the fetal head with electronic measurement calipers is provided. This sagittal view of the fetal brain demonstrates an additional method of measuring the fetal scalp thickness (FST), at 38 weeks gestation.
Data Analysis
This cohort’s data were described with means, standard deviations (SD), as well as ranges were estimated for FST and the EFW. Additional traditional biometry measures were also recorded (e.g., BPD, HC, AC, FL). A Pearson’s correlation was performed to determine the strength of the relationship between the FST, EFW, and other biometry parameters (BPD, HC, AC, and FL). Simple and multiple linear regression models, with scatter plots, were utilized to provide an EFW, using FST and the other biometric measures. All statistical analyses were performed using the Statistical Package for the Social Sciences (SPSS Inc, Chicago, Illinois) version 21.0. The statistical significance was determined a priori at
Results
The FST ranged between 0.21 and 0.68 cm, with a mean of 0.36 cm and SD 0.078, while EFW ranged between 620 and 3860 g, with an estimated mean of 2270 g and SD 813. The BPD ranged between 5.28 cm and 9.81, HC 19.64 cm and 38.77 cm, AC 21.79 cm and 36.55 cm, and FL 4.13 cm and 8.00 cm (see Table 1). The FST showed a strong positive correlation with EFW (r = 0.65;
The Descriptives Statistics of Fetal Biometry Measures in This Cohort of Patients.
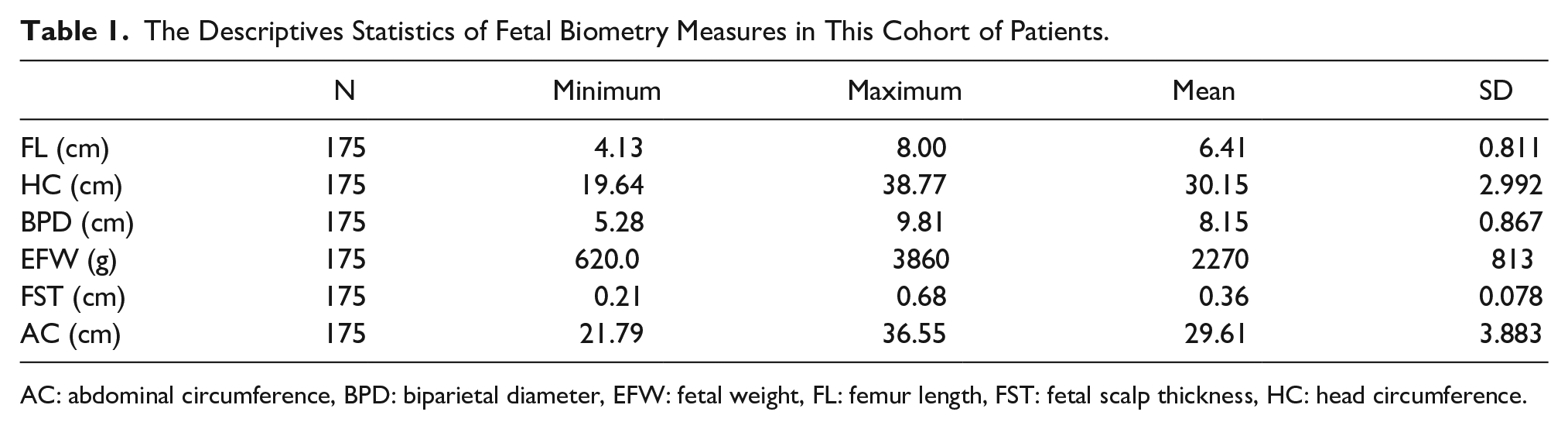
AC: abdominal circumference, BPD: biparietal diameter, EFW: fetal weight, FL: femur length, FST: fetal scalp thickness, HC: head circumference.
The Strength of Association Between Fetal Scalp Thickness and Estimated Fetal Weight, as Well as the Traditional Fetal Biometry Measurements.
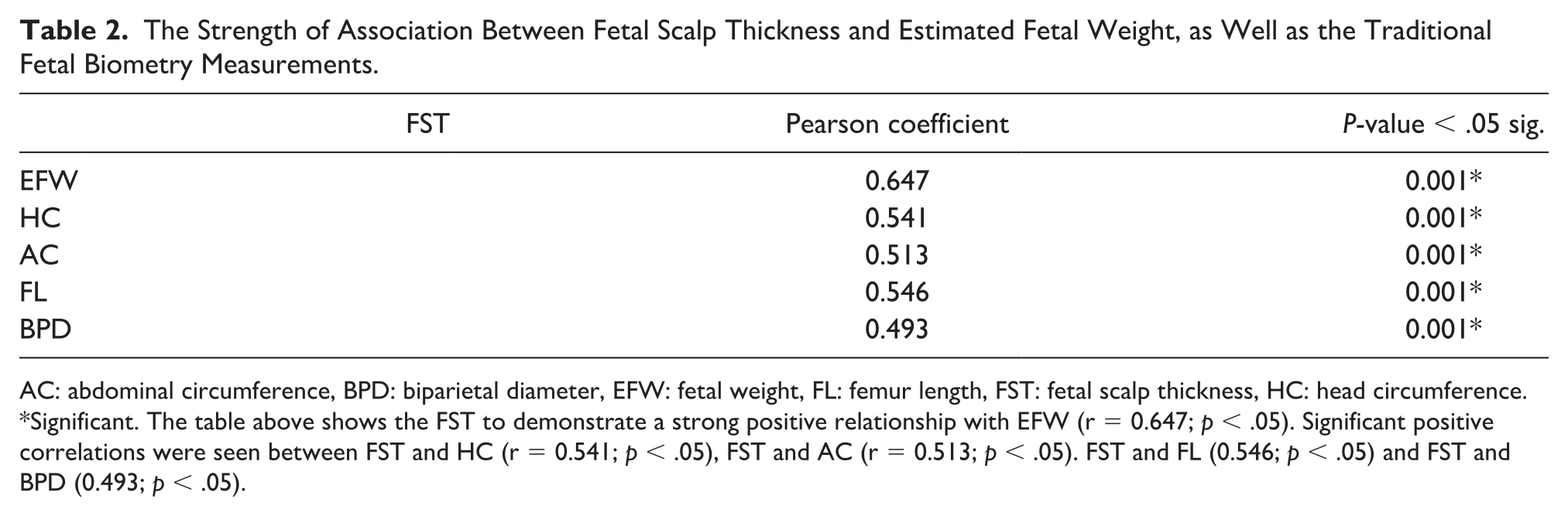
AC: abdominal circumference, BPD: biparietal diameter, EFW: fetal weight, FL: femur length, FST: fetal scalp thickness, HC: head circumference.
Significant. The table above shows the FST to demonstrate a strong positive relationship with EFW (r = 0.647;
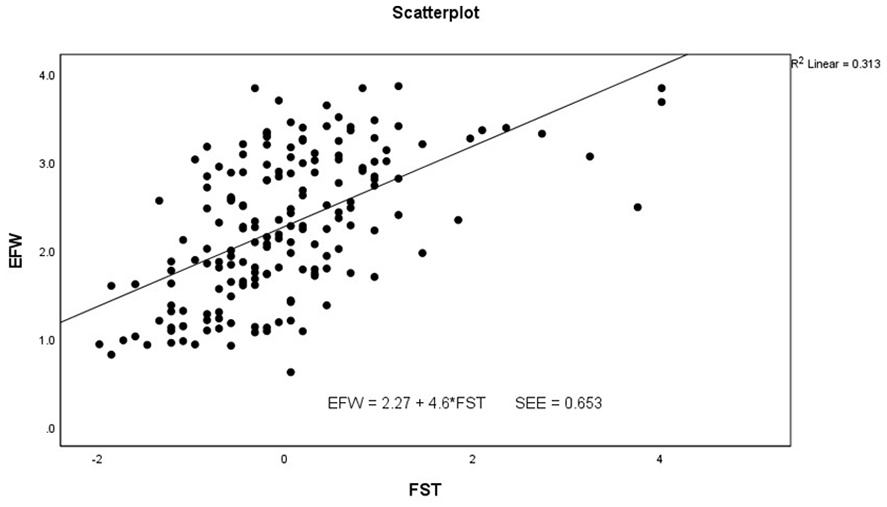
The scatter plot and regression analysis indicating the relationship between fetal scalp thickness and estimated fetal weight.
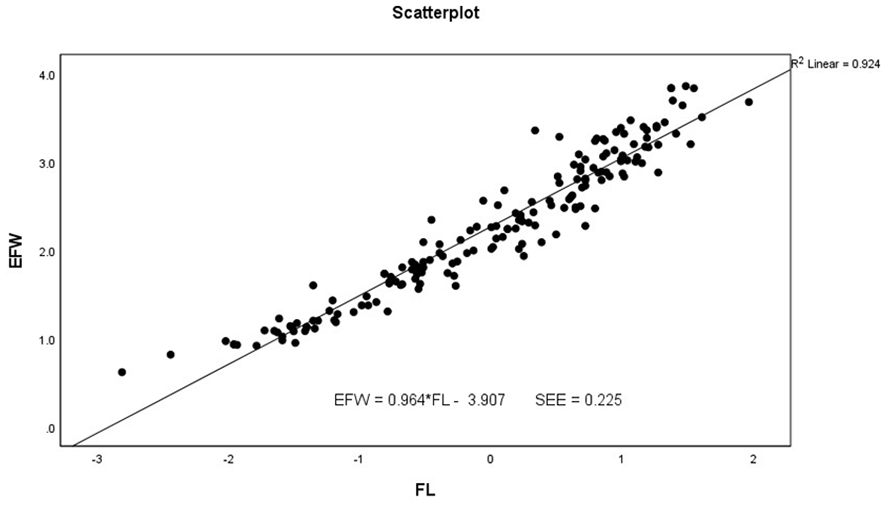
The scatter plot and linear regression analysis of femur length (FL), as an independent predictor of estimated fetal weight.
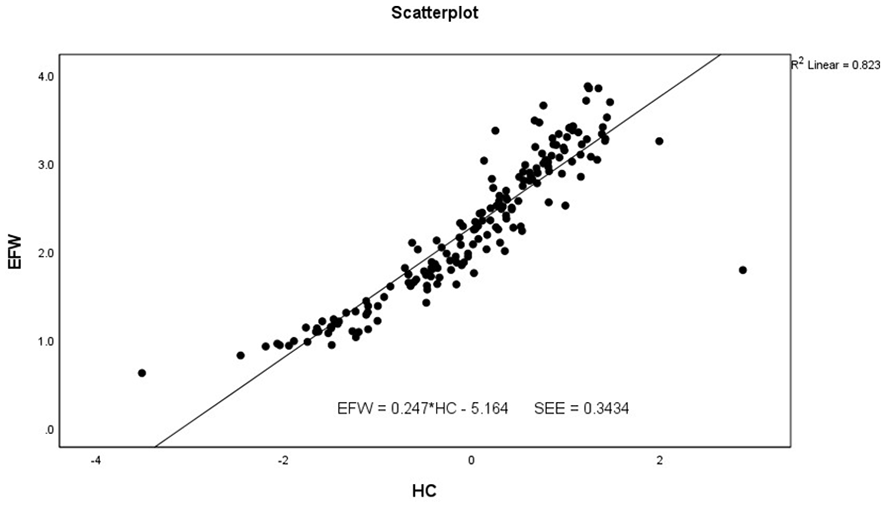
The scatter plot and linear regression analysis using fetal head circumference, as an independent predictor of estimated fetal weight.
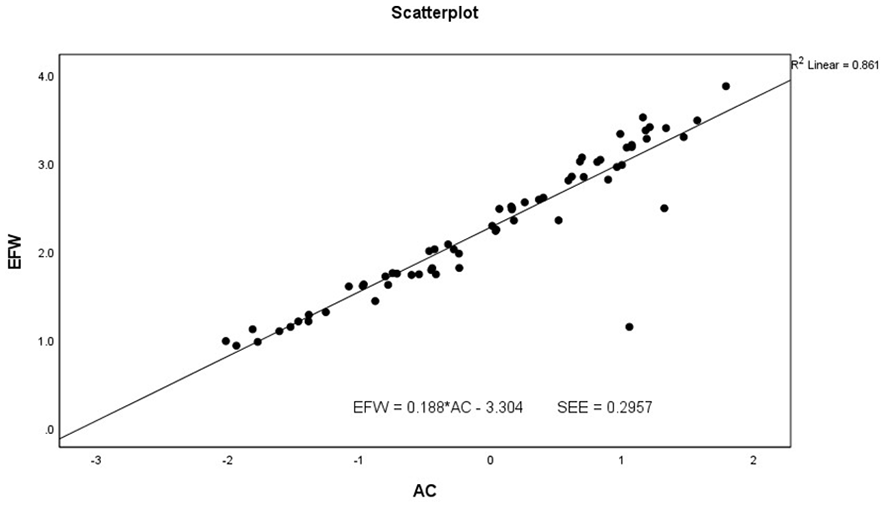
The scatter plot and linear regression analysis using abdominal circumference, as an independent predictor of estimated fetal weight.
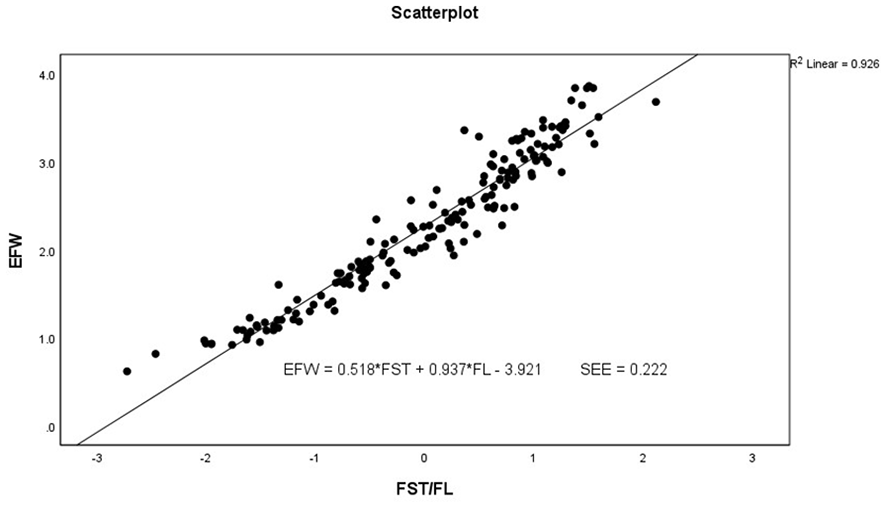
A scatter plot and multiple linear regression analysis based on fetal femur length and scalp thickness, as predictors of estimated fetal weight.
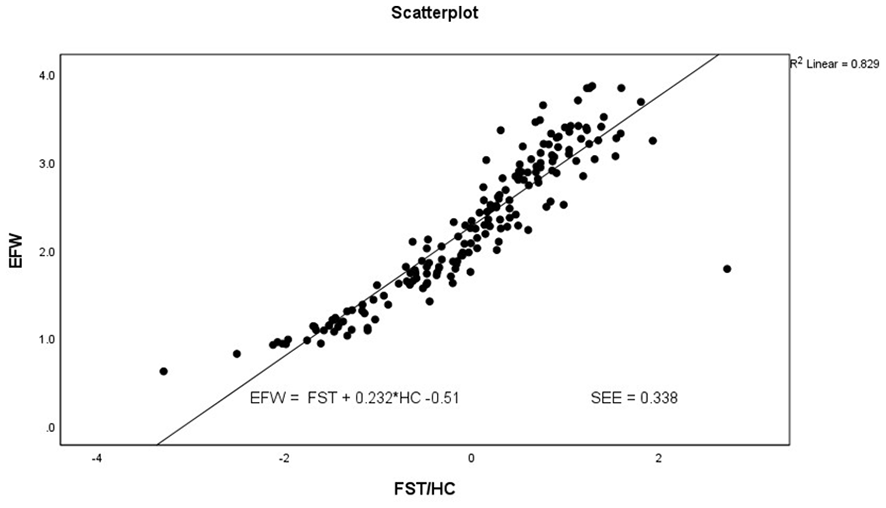
The scatter plot and multiple linear regression analysis of fetal head circumference and scalp thickness, as predictors of estimated fetal weight.
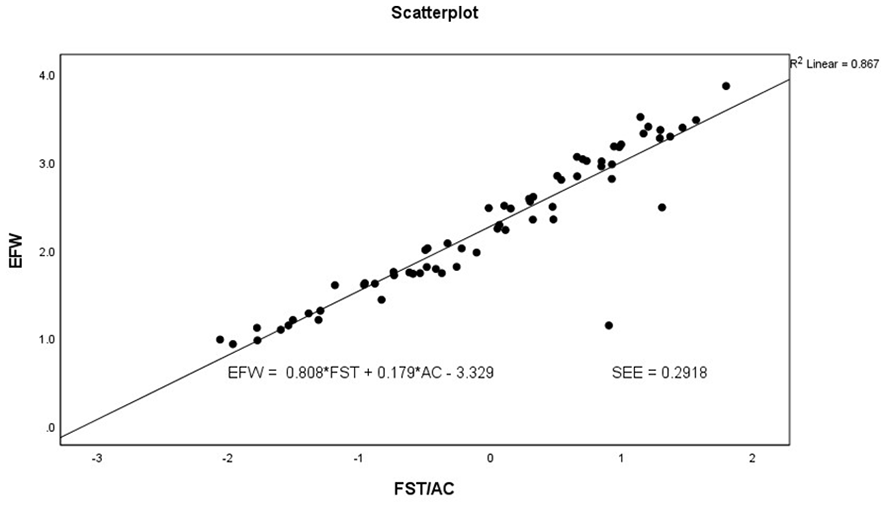
A scatter plot and multiple regression analysis of fetal abdominal circumference and scalp thickness, as predictors of estimated fetal weight.
Discussion
Sonographic surveillance of fetal growth is an integral part of modern-day obstetrics, which is targeted at monitoring fetal well-being and improving perinatal outcomes. The EFW is an important parameter in determining fetal well-being and requires highly accurate measures of estimation. There is evidence that the accuracy EFW with sonography can improve by incorporating multiple fetal biometric parameters. 16 This understanding has since stimulated research toward integrating more fetal parameters to better determine EFW. A few research studies have been tailored to a more direct,17,18 measure of EFW, by integrating fetal soft tissue fat mass in its determination, with very reliable outcomes. This study may be the first to look at the predictability of EFW using FST independently and in combination with some fetal biometry parameters. In this study, the EFW was seen to increase proportionately with FST and accounted for about 31% variability in EFW, implying its viability to indicate sonographic fetal weight, which is a strong determinant of fetal well-being and development. The FST also demonstrated significant moderate positive correlation with AC, which confirms the result of a study carried out by Moore et al. 19 In their study, AC appeared to have a positive relationship with fetal soft tissue fat mass, weight, and growth trajectory. In another study, it was reported that fetal and neonatal soft tissue fat accounted for a considerable percentage of the variance in EFW and neonatal weight.
The correct EFW is critical to improve pregnancy outcome and for the assessment of growth abnormalities, the importance of EFW in obstetric management and clinical decision-making cannot be overstated, as this single parameter tells a lot about certain physiologic or pathologic processes from maternal or environmental factors which could underlie faltering fetal weight and neonatal outcomes. Other studies have documented even higher positive predictive values using fetal soft tissue fat. This goes to argue that the FST, like other fetal fat regions, could help monitor fetal development and further inform perinatal decision-making, prior to delivery. A significant amount of fetal adipose tissue formation happens in the last 10 weeks of the third trimester. 20 Based on this study’s results, a unit rise in FST yielded a 4.6 increase in EFW with the derived model being significant for the prediction of EFW using FST. However, a much larger SEE was seen using FST in the prediction of EFW. With the FST and FL as combined predictors of EFW, it yielded a lower SEE, with these predictors accounting for 93% variance in EFW. This outcome shows a better performance in terms of prediction with reduced SEE. One unit rise in FST in this model produced a 0.54 increase in EFW in combination with FL, while a unit increase in FL yielded a 0.94 increase in FW, lower than was obtained for both variables as separate independent predictors. This improved relationship was replicated in the combinations between HC and FST and AC and FST: the SEE further reduced, with these predictors accounting for a higher percentage variability in FW, than was achieved with these variables as single independent predictors.
These current study findings demonstrated that the models derived using these traditional biometry measures were further improved with FST, as additional variable in the multiple regression model for EFW. This study results are in concert with reports from related studies. 21 Similar results were also reported in a study by Warska et al, 18 as fetal soft tissue was reported to serve as a viable predictor of EFW, and further improved the contributions of traditional fetal biometrics (BPD, HC, and FL), to the variance in EFW. Some studies, revealed that fetal abdominal subcutaneous tissue thickness in a previous study improved performance in the EFW, and presented a more viable measure of indicating birth weight and neonatal survival than traditional biometric measures, particularly in large for gestational age fetuses.22,23 Hebbar et al 17 reported that the EFW was more reliable in models which included fetal soft tissue and showed even higher positive predictive values than Hadlock’s. 17 Although this study did not consider the impact of FST on EFW in maternal and gestational diabetes, previous studies have documented fetal soft tissue thickening under these conditions, which is evidenced in scalp thickness, and directly indicate large for gestational age. Maternal metabolic state and disease processes can influence fetal growth signaling, 24 leading to fetal growth abnormalities. 25 The increased fat accretion in fetuses of diabetic mothers has been associated with excess energy production from glucose in maternal diabetes, 23 which results in excess fat deposition evident in fetal soft tissues (subcutaneous), including fetal scalp in macrosomia. 3 Some of these maternal factors that affect growth process include the body composition of the mother, placental transport capacity and nutrition.26 –28
From the results of this study, FST might be a potential marker for improving the prediction of EFW, and monitoring fetal development to identify normal, and abnormal growth in fetuses. Most sonographic formulae for EFW are based on multiple fetal measurements: 29 the integration of mid-thigh soft tissue thickness (STT) in the model of EFW improved neonatal weight prediction in a previous study, and the combination of more than two parameters was deemed more reliable than those with one or two measurements. 30
Integrating the FST as a single predictor of EFW, in a third-trimester sonogram, could prove useful, especially as some fetal biometrics may be difficult to obtain. An example might be obtaining the BPD, if the fetal head engages deep into the pelvis. 25 However, such cases represent only a small percentage of all deliveries. 30 The FST being a linear measurement is even more reproducible than circumferences and multiple parameters 31 and can be very useful to novice sonographers in obtaining an EFW.
Limitations
There are a few limitations to this study, and the main issue is the research design. The research design was pre-experimental and has threats to internal and external validity. In addition, the participants were recruited and formed a convenience sample. It was important that the sample size was based on estimated statistical power but will require a randomized allotment of participants to suit a more global audience. Due to these challenges, the findings may not be generalizable beyond this cohort, as the outcomes and data are specific to these patients.
Conclusion
The FST is a viable indicator of fetal and has the potential to improve perinatal outcome if modeled in combination with traditional ultrasound biometric parameters for the prediction of fetal weight. This single parameter (FST) could also prove useful in monitoring fetal growth trajectory.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval for the study was obtained from the Ethical Research Committee within the Department of Radiography and Radiological Science, at the University of Calabar (IRB# UC/ECRA/22/015).
Informed Consent
Verbal informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
