Abstract
Objective
Macrosomia is associated with the risk of mortality and morbidity in neonates and their mothers. Despite the considerable public health effect of macrosomia, evidence on the determinants of macrosomia is limited in Northwest Ethiopia in general and in Amhara region in particular. Therefore, this study aimed to identify determinants of macrosomia among newborns delivered in referral hospitals in 2020 in Amhara region, Northwest Ethiopia.
Methods
A facility-based unmatched case-control study was conducted among 279 mothers and their newborns in Amhara region referral hospitals. Newborns weighing 4000 g and above and between 2500 and 3999 g were considered cases and controls, respectively. Bivariable and multivariable binary logistic regression were used to identify the determinants of macrosomia.
Results
In total, 273 of 279 mothers and their newborns (97.8% response rate) were included. The mean birth weights of cases and controls were 4312.97 ± 357.53 g and 3161.92 ± 452.12 g, respectively. Weight gain over pregnancy, antenatal follow up, physical activity during pregnancy, and neonate sex were the main determinants of macrosomia.
Conclusion
The main determinants of macrosomia were determined in this study. Government should place special emphasis on reducing the modifiable factors of macrosomia.
Introduction
The term macrosomia can be defined in a variety of ways; considerable variation is observed in the minimum weight that defines macrosomia.1–3 Although absolute consensus on the definition has not been reached, most researchers from low- and middle-income countries define macrosomia as a birth weight of 4000 g or more that harms maternal and neonatal health outcomes.4–6
Worldwide, macrosomia affects 10% of pregnancies annually.7,8 In developed countries, the prevalence of fetal macrosomia ranges from 15% to 20% 9 whereas the prevalence is between 2% and 9% in developing countries.5,9 Prevalence rates of 7.6% and 8% have been observed in Chad and Nigeria, respectively.10,11 In the Ethiopian context, studies in Mekelle and Hawassa indicate macrosomia rates of 6.6% and 11%, respectively.12,13
Maternal risk factors for macrosomia include high monthly income, full-term pregnancies, history of macrosomic newborns, pre-gestational body mass index (BMI), gestational weight gain (GWG), pre-existing diabetes mellitus, gestational diabetes mellitus, and multiple pregnancies.1,9,12,13 In addition, newborn sex and gestational age are recognized as independent neonatal factors for macrosomia. 14 Studies conducted in Ethiopia identified gestational age, gestational diabetes mellitus, neonate sex, pre-gestational overweight, maternal age, and parity as notable factors.13,15,16
Macrosomia is associated with an increased risk of complications for both mothers and their neonates. 4 For pregnant women, delivery of macrosomia is associated with significantly elevated risks of dysfunctional uterine contractions, prolonged labor, cesarean section, uterine rupture, postpartum hemorrhage, chorioamnionitis, and lower genital tract lacerations. 17 For newborns, macrosomia increases the risks of shoulder dystocia, birth trauma, respiratory distress, meconium aspiration, birth asphyxia, perinatal infection, hypoglycemia, polycythemia, and hypoxic-ischemic encephalopathy and increases the need for admission to the neonatal intensive care unit.2,5 In addition, macrosomia increases the risk of developing cardiovascular disorders, obesity, and insulin resistance in adulthood.17,18 Lastly, macrosomia may produce a higher risk of psychiatric problems for babies in adulthood and mothers. 19
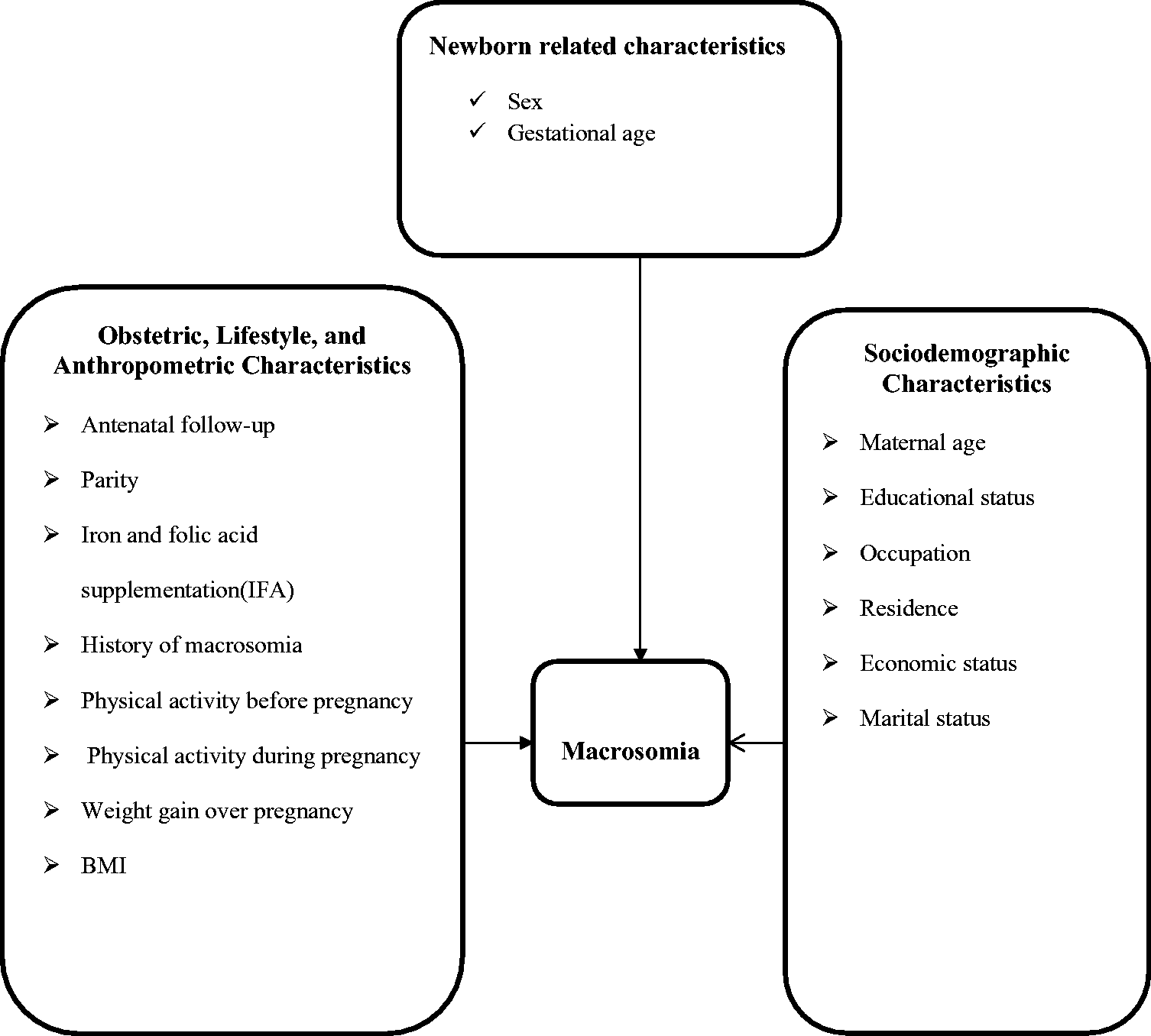
The Ethiopian federal Minister of Health has implemented various strategies to enhance maternal and neonatal health, including expanding healthcare facilities and increasing the availability of supplies and skilled health professionals. 20 Despite these actions, maternal and neonatal mortality rates remain at 412 per 100,000 and 29 per 1000 live births, respectively. 21 Macrosomia remains a significant factor in maternal and neonatal mortality and morbidity in Ethiopia.5,15,22 Although the several risk factors of macrosomia in Ethiopia have been assessed, the evidence on the determinants of macrosomia—especially that of the modifiable risk factors at the country level in general and in the study area in particular—remains limited. Physical activity before and during pregnancy, BMI, GWG, and iron–folic acid supplementation are modifiable risk factors that have not been addressed by prior research in Ethiopia. Therefore, the study aimed to identify the determinants of macrosomia among newborns delivered in referral hospitals in Amhara region (Figure 1).
Methods
Study design
A facility-based unmatched case-control study was conducted.
Study setting
The study was conducted at referral hospitals in Amhara regional state, Ethiopia from February 2020 to May 2020. According to a 2017 Ethiopian central statistical agency estimate, the regional state has a population of over 20 million, with equal representation of the sexes. Urban residents represent 12% of the population (below the national average) and roughly 91.4% of the regional state population is Amhara in ethnicity. 26 Bahir Dar, the capital city of the regional state, is located 563 km from Northwest Addis Ababa, the capital city of Ethiopia.
Six referral hospitals are located in the regional state: the University of Gondar Referral Hospital (UGRH), Felege Hiwot Referral Hospital (FHRH), Tibebe Ghion Referral Hospital, Dessie Referral Hospital (DRH), Debre Markos Referral Hospital, and Debre Birhan Referral Hospital. The study was conducted simultaneously at three (FHRH, UGRH, and DRH) of these referral hospitals using the simple random sampling (lottery) method. These hospitals provide emergency obstetric care, maternal and neonatal health infrastructures, and a full complement of skilled health professionals including obstetricians, anesthesiologists, radiologists, neonatologists, nurses, and midwives. A total of 1960 health professionals (730 in FHRH, 670 in UGRH, and 560 in DRH) work in the selected hospitals.
A proportional sample allocation was applied to each hospital based on delivery data from the previous year. A total of 279 mothers and their newborns (93, 102, and 84 from FHRH, UGRH, and DRH, respectively) participated in the study. Participants were recruited within 12 hours of delivery in a labor or delivery room and were classified as cases or controls within one hour of delivery. Two controls were recruited for each case until the required sample size was reached at each hospital.
Participants
The study population consisted of mothers and their newborn babies who were delivered at selected Amhara region referral hospitals in the study period. Cases and controls were newborn babies who weighed 4000 g or more and between 2500 and 3999 g, respectively. Newborns with congenital anomalies, multiple births, newborns who were not weighed within the first hour of life, mothers with diabetes mellitus, and mothers who were too critically ill were excluded from the study. Permission and a supporting letter were obtained from Bahir Dar University, Ethiopia before data collection. The study was approved by an ethics review committee at Bahir Dar University. Confidentiality was assured and written consent/assent was obtained from all respondents.
Variables
Dependent variable
Macrosomia.
Independent variables
Case definitions
Sample size determination
The sample size was determined using a double population proportion formula in EpiInfo Version 7 statistical software by considering a 95% confidence interval (CI) and assuming a power of 80% and a case-to-control ratio of 1:2. Given that a previous study reported that newborn sex, gestational age, and a history of macrosomic newborns were significantly associated with macrosomia, 13 all of these variables were considered when calculating sample size. Based on the adjusted odds ratio (AOR) of 2.2 for newborn sex and assuming that 54.7% and 74.6% of controls and cases, respectively, were exposed, the maximum sample size was estimated. After adding 10% for the non-response rate, 93 cases and 186 controls (i.e., a total sample size of 279) were included in the study.
The formula for the sample size was
where
Data sources and measurement
The structured questionnaire and extraction checklist were adapted from previous literature.5,12,13,15,16,23–25 Data were collected by interviewing the mother, reviewing medical records, and measuring the anthropometry of the mother and newborn baby.
An interviewer-administered questionnaire was used to collect information on the wealth index, maternal age, religion, marital status, occupation, educational level, and place of residence. Physical activity before and during pregnancy was reported by the mother using an interviewer-administered questionnaire.
Data on iron–folic acid supplements, parity, antenatal follow up, first-trimester weight, third-trimester weight, and history of macrosomia were extracted from the antenatal follow-up record using an extraction checklist. Data on gestational age at delivery were further collected using an extraction checklist from the antenatal follow-up chart. When information could not be ascertained, evidence from first or second-trimester ultrasound was used and respondents were then classified into pre-term, term, and post-term.
Newborns were weighed within 1 hour of birth using a digital scale to the nearest 1 g. Maternal height was calculated to the nearest 0.01 m using a wall height measurement. Mothers were asked to maintain an upright position with their feet together and a horizontal headpiece was lowered onto their heads. Pre-pregnancy BMI was calculated using first-trimester weight in kilograms divided by height in meters squared (kg/m2) then divided into three categories based on Centers for Disease Control guidelines: underweight (<18.5 kg/m2), normal (18.5 to <25 kg/m2), and overweight (>25 kg/m2 and above). 28 GWG was calculated by subtracting first-trimester weight from the last third-trimester weight measurement and was categorized as low weight gain, normal weight gain, and excessive weight gain based on Institute of Medicine guidelines, which are accepted by the American College of Obstetricians and Gynecologists. 29
Data quality control
Data were collected using questionnaires and checklists that were written in English and translated into Amharic. Six registered midwives (i.e., with a Bachelor of Science in Midwifery) were trained on techniques for data collection. The principal researchers were also involved in data collection. The pretest was conducted outside of the study area and the questionnaire and checklist were improved to meet the research objective. The birth weight of every newborn was measured within 1 hour of delivery using a similar, pre-calibrated digital scale balance. The study is reported according to the relevant Equator network guideline. 30
Data processing and analysis
The data were cleaned, coded, and entered into EpiData version 3.2 (EpiData, Buenos Aires, Argentina) and transferred to SPSS, Version 25.0 (IBM Corp., Armonk, NY, USA) for analysis. The study variable was summarized using descriptive statistics such as frequency and percentage. The differences in the proportions of the characteristics between cases and controls were compared using the χ2 test. Bivariable and multivariable logistic regression models were used to test the associations between dependent and independent variables. Variables with a p-value ≤0.25 in the binary logistic regression were considered candidate variables for the multivariable logistic regression model. The main assumptions of the logistic regression model (i.e., the absence of outliers and multicollinearity) were checked. Accordingly, none of the interaction terms showed statistical significance, indicating the absence of a significant modification effect. Multicollinearity between the independent variables was evaluated using multiple linear regressions. No evidence of multicollinearity was identified given that the variance inflation factor for all variables was less than 10 and the tolerance statistic was greater than 0.1. The Hosmer–Lemeshow test was used to evaluate the fitness of the logistic regression model; a value of χ2 = 2.21, df = 8, (p = 0.974) indicated the model’s fitness. The model explained that 78.6% of the variation in the outcome and 78.4% of the variables were correctly classified. The strength of association between macrosomia and the determinants was assessed using AORs with 95% CIs. A statistically significant association was declared when the p-value was less than 0.05 in the multivariable regression analysis.
Results
Sociodemographic, obstetric, neonatal, anthropometric, and lifestyle characteristics
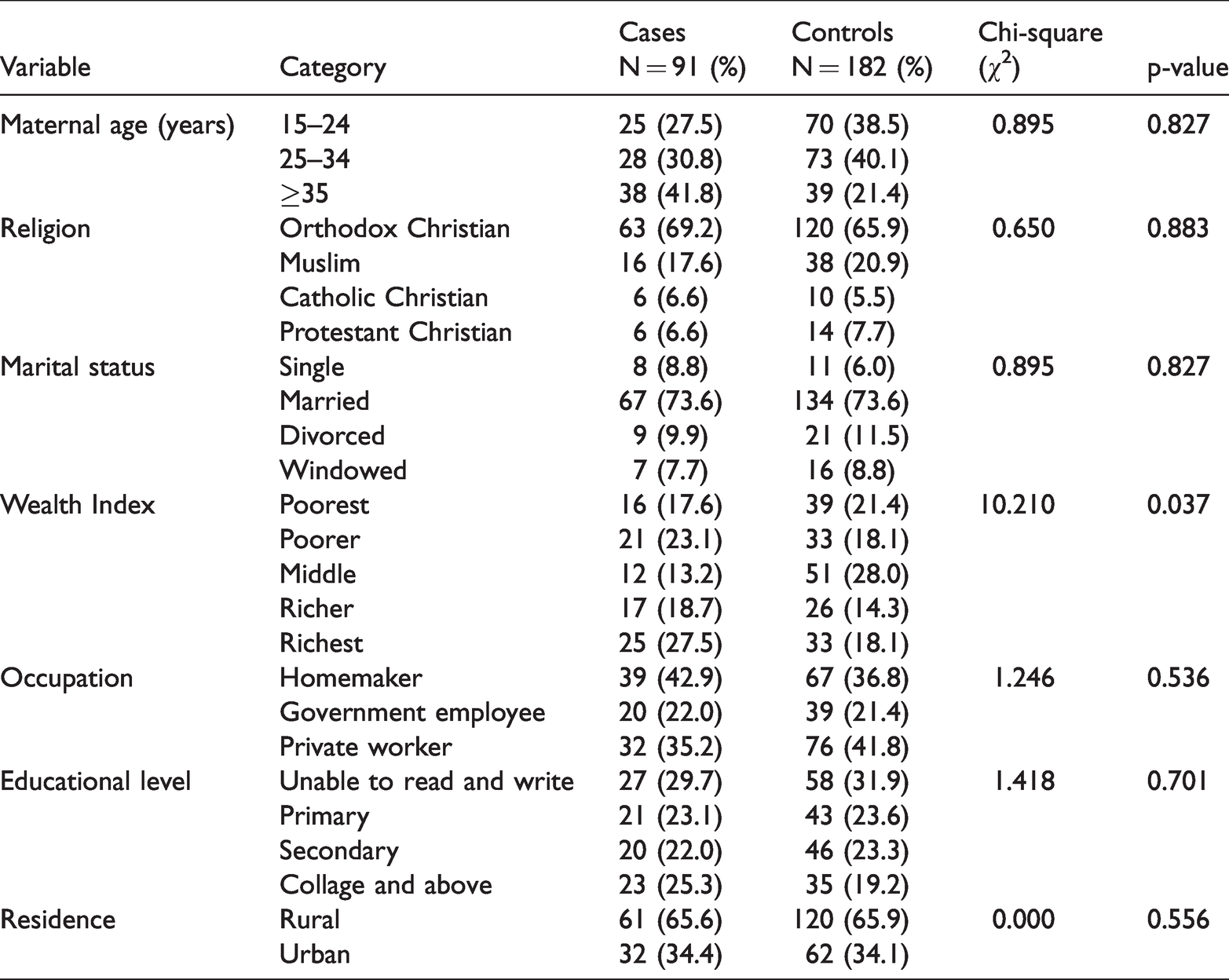
A total of 273 mothers and their newborns (91 cases and 182 controls) were included in the study and represented a response rate of 97.8%. The mean birth weight of cases was 4312.97 ± 357.53 g and that of controls was 4312.97 ± 357.53 g (p < 0.001). The mean gestational age at birth was 38.8 ± 2.54 weeks (p < 0.001). The proportion of mothers aged 35 years and above tended to be higher among cases (41.8%) compared with controls (21.4%; p = 0.002). In addition, the proportion of mothers with a middle wealth index tended to be lower among cases (13.2%) compared with that of controls (28.0%; p = 0.037). However, no significant differences were observed between case and control groups for maternal religious affiliation, marital status, level of educational), residence, and occupation (Table 1).
Sociodemographic characteristics in macrosomia and controls.
The proportion of mothers with a history of macrosomia tended to be higher among cases (38.5%) compared with controls (12.1%; p < 0.001) whereas the proportion of mothers with antenatal follow up tended to be lower among cases (80.2%) compared with controls (94.0%; p = 0.001). Moreover, the proportion of primipara mothers tended to be lower among cases (18.7%) compared with controls (35.2%; p = 0.012). Post-term pregnancy also tended to be higher for cases (26.4%) than for controls (12.4%; p = 0.009). Furthermore, female neonate sex was significantly lower for cases (47.3%) than for controls (72.0%; p < 0.001). However, significant differences were not observed between case and control groups in iron–folic acid supplementation and the frequency of antenatal follow up (Table 2).
Obstetric and neonatal factors in macrosomia and controls.
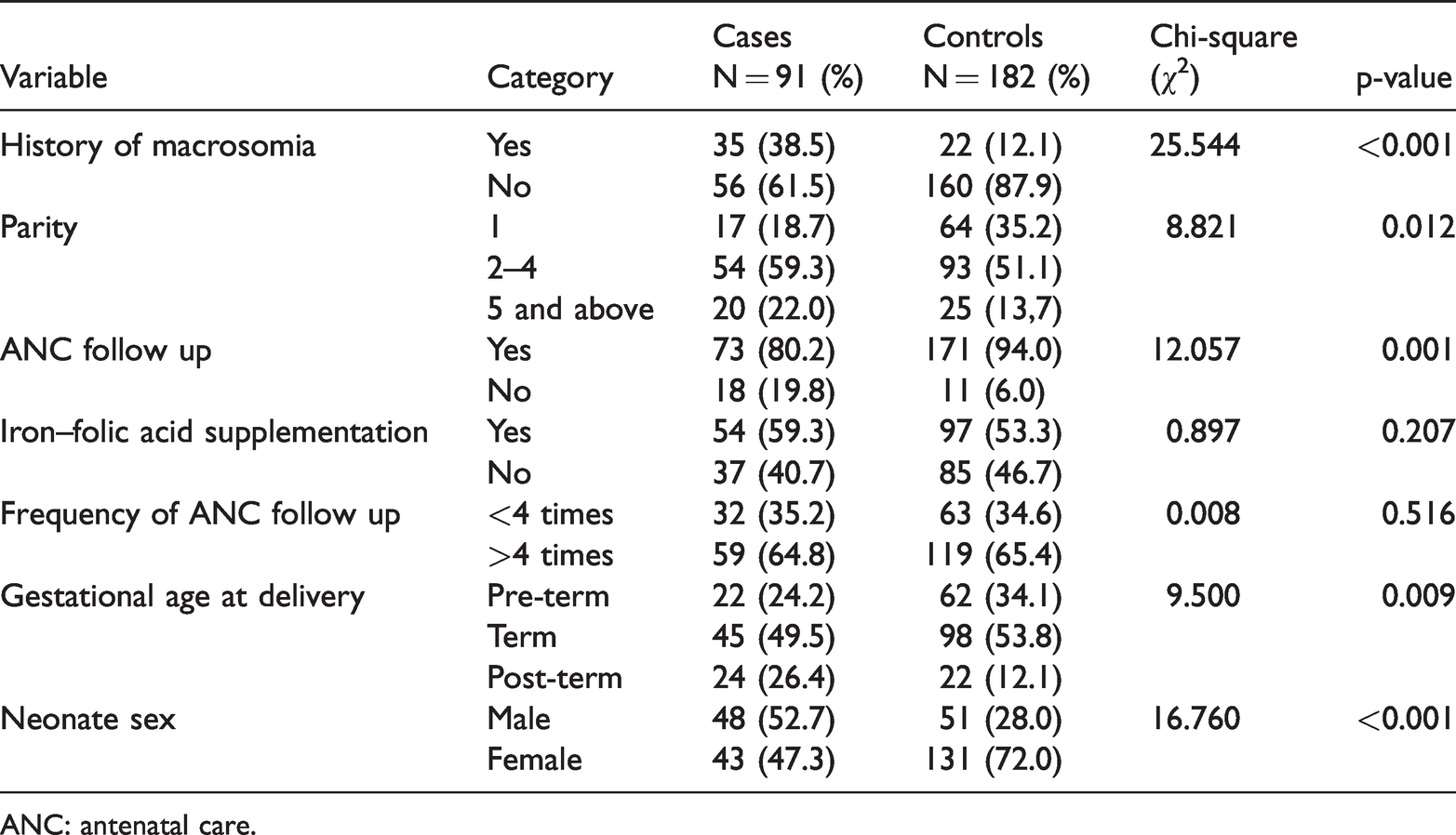
ANC: antenatal care.
The proportion of mothers who practiced pre-pregnancy physical activity tended to be lower among cases (69.2%) compared with controls (90.1%; p < 0.001) and the proportion of mothers who practiced physical activity during pregnancy tended to be lower among cases (51.6%) compared with controls (81.9%; p < 0.001). Moreover, the proportion of mothers who were overweight or obese based on pre-pregnancy BMI tended to be higher among cases (26.4%) compared with controls (8.8%; p < 0.001). Finally, the proportion of mothers with excessive GWG tended to be higher for cases (54.9%) than for controls (29.1%; p < 0.001; Table 3).
Anthropometric and lifestyle characteristics in macrosomia and controls.

Determinants of macrosomia
In the bivariable logistic regression model, maternal age, wealth index, parity, history of macrosomia, pre-pregnancy physical activity, physical activity during pregnancy, antenatal follow up, GWG, pre-pregnancy BMI, gestational age at delivery, and neonate sex were the main determinants of macrosomia. After adjusting for confounding variables in a multivariable logistic regression model, GWG, antenatal follow up, physical activity during pregnancy, and neonate sex were independent determinants of macrosomia.
Neonates born from mothers who had excessive GWG were 3.5 times more likely to be macrosomic than their counterparts (AOR = 3.54; 95% CI: 1.52–8.23). Newborns of mothers who had no history of antenatal visits were at 3.2 times greater risk of having macrosomia compared with mothers who attended at least one antenatal visit (AOR = 3.21; 95% CI: 1.13–9.12). The risk of having a macrosomic newborn was higher among physically inactive mothers (<30 minutes of exercise/day) compared with their counterparts (AOR = 2.17; 95% CI: 1.09–4.70). The odds of experiencing macrosomia were higher among male neonates compared with female neonates (AOR = 2.07; 95% CI: 1.06–4.03; Table 4).
Bivariable and multivariable analysis of the determinants of macrosomia.

*Significant at ≤5%. COR: crude odds ratio, ANC: antenatal care, AOR: adjusted odds ratio 1: Reference.
Discussion
A commitment to protecting maternal and neonatal health is necessary for ensuring Ethiopia achieves the United Nations’ Sustainable Development Goals. 31 Macrosomia and its consequences are factors that contribute to neonatal and maternal morbidity and mortality. Our study explored the determinants of macrosomia among newborns delivered in Amhara region referral hospitals in Northwest Ethiopia.
In the study, GWG was observed to have a significant effect on macrosomia. Mothers with excessive GWG were 3.5 times more likely to deliver macrosomic newborns than mothers with low GWG. This finding is consistent with those of studies in Nigeria, China, and Brazil.23,32,33 A possible explanation could be that excessive GWG is associated with decreased insulin sensitivity, which facilitates the transfer of glucose via the placenta to the growing fetus and leads to increased birth weight. 34 Additionally, a 1-kg gain in maternal pregnancy weight is expected to increase neonatal birth weight by 94 g.35,36
The study further indicated that antenatal follow ups have a significant protective effect against macrosomia. Mothers who did not have any antenatal visits were 3.3 times more likely to have macrosomic neonates than mothers who attended at least one antenatal visit. This finding is similar to that of a study in the United States. 37 A possible reason is that antenatal follow up is a cost-effective health promotion and preventive healthcare service that offers early detection and the treatment of pre-existing diseases and complications, ensuring healthy outcomes for the mother and newborn. 37
A lack of physical activity during pregnancy had a significant association with macrosomia in the study. Newborns from physically inactive mothers (<30 min of exercise/day) were 2.1 times more likely to be macrosomic than their counterparts. This finding is supported by studies conducted in Spain and Canada.38,39 A low level of physical activity during pregnancy may result in GWG and may increase the risk of macrosomia. Physical activity during pregnancy has been shown to reduce maternal storage adiposity and fetal fat; therefore, it may prevent excessive GWG and promote healthy birth weight. 40 Furthermore, exercise “steals” cardiac output from the womb and placenta and decreases the risk of macrosomia without increasing birth weight. 41
Finally, a key determinant of macrosomia was newborn sex. Male newborns were twice more likely to be macrosomic than female neonates. This result is consistent with those of studies conducted in Gondar, Ethiopia; Hawassa, Ethiopia; Cameroon; Iran; and China.12,15,42–44 and may be attributable to the greater weight and length of male newborns compared with female neonates.45,46 However, this finding was inconsistent with that of a Saudi Arabian study, which concluded that the rate of macrosomia was higher in female than in male neonates. 47 This may be due to methodological differences given that the latter was a descriptive study.
One strength of this study is that given that the study included primary data, secondary data, and anthropometry measurements, several variables were included that had not been previously investigated in a study setting. In addition, cases and controls were enrolled by accurately measuring newborn weight within 1 hour of delivery to avoid discrepancies.
This study had some limitations. Our research might have been subject to recall bias; however, we used strategies—careful selection of the study question and employment of local data collectors familiar with the community culture—that might have reduced recall bias. Another study limitation was the use of first-trimester weight information recorded on the antenatal chart as a proxy for pre-pregnancy weight, which might have led to misclassification bias. However, we believe this bias was negligible given that GWG occurs largely in the third trimester. 48
Conclusion
Determinants of macrosomia were identified in this study. GWG, physical activity during pregnancy, and antenatal follow up were the modifiable determinants of macrosomia, whereas newborn sex was a non-modifiable factor of macrosomia. Accordingly, the government must place special emphasis on reducing the modifiable factors of macrosomia.
Footnotes
Acknowledgements
We would like to thank hospital managers, study participants, data collectors, and supervisors for their support of the overall research procedure.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and publication of this study.
