Abstract
Objective:
This study aimed to investigate sonographic parameters that could predict 1-month mortality in patients with acute kidney injury (AKI).
Materials and Methods:
This study was a post-hoc analysis of multi-organ sonographic examinations conducted between August 2020 and July 2021. Adult patients who presented to the emergency department (ED) with AKI were recruited. Multi-organ (lung, cardiac, inferior vena cava, kidney, and bladder) sonographic examinations were performed by emergency physicians and evaluated by a radiologist, cardiologist, and another emergency physician, while being anonymized to the results.
Results:
There were 165 patients with AKI enrolled in the study, and 40 (24.2%) of them experienced 1-month mortality. Following univariate analyses of 23 sonographic parameters, multivariate regression analysis revealed that diffuse ascites (odds ratio [OR], 3.67; 95% confidence interval [CI]: 1.27–10.61) and left ventricular basal diameter (OR, 0.93; 95% CI: 0.87–0.99) were related with 1-month mortality in patients with AKI.
Conclusion:
In this cohort of AKI patients, the most powerful sonographic parameter, as a predictor of 1-month mortality, was diffuse ascites. Further clinical studies are needed to validate and refine these diagnostic findings.
Acute kidney injury (AKI) is a clinical syndrome characterized by a sudden impairment in renal function, often triggered by causes such as decreased renal perfusion, toxic effects, or obstruction. It is typically defined by an increase in serum creatinine levels and a decrease in urine output. This serious medical condition has a substantial risk of mortality. The hospital inpatient mortality rate is approximately 13%, 1 approximately 20%–28% in the emergency department (ED),2 –4 as well as in intensive care unit (ICU) settings.5,6 Complications of AKI may include electrolyte disturbances, metabolic acidosis, fluid overload, and the development of chronic kidney disease. These risks are particularly heightened in patients with comorbidities. Therefore, early identification and appropriate management of AKI are essential to mitigate these complications and improve patient outcomes.
The prediction of patient mortality, associated with AKI, is a frequent concern for clinicians and researchers. Numerous laboratory and clinical parameters have been recently investigated for this purpose. Various diagnostic scoring systems have also been developed to assess patients. Advanced age, cirrhosis, sepsis, malignancy, and later-stage AKI are commonly used mortality predictors.7,8 However, sonographic parameters have rarely been studied, and only a few diagnostic tests, such as renal resistive index, have been explored in the literature as diagnostic predictors. 9 Rarely has previous research focused on the investigation of multi-organ sonographic parameters to predict short-term mortality in AKI. This study aims to determine which predictive sonographic parameters would indicate 1-month mortality in those AKI patients reporting to the ED.
Materials and Methods
Study Design and Participant Sample
In a previous study, the diagnostic accuracies of multi-organ point-of-care ultrasonography (POCUS) was conducted in an academic setting, between August 2020 and July 2021, to detect AKI subtypes (IRB: NCT04470661). 4 This study represents a post-hoc analysis of that prospective multi-organ POCUS study. Ethical approval was given for the study (No. 2020-07/693), and the patients had already signed consent forms.
The participants enrolled in the study were all adult patients diagnosed with AKI. According to the Kidney Disease: Improving Global Outcomes (KDIGO) criteria, AKI was defined as a known rise (≥50%) in serum creatinine or a rise (≥0.3 mg/dL) in serum creatinine, within 2 days. Patients who had a history of renal intervention, end-stage renal illness, pregnancy, and polycystic renal disorders; had received a single kidney transplant; or had refused the consent process were all excluded.
Sonographic Assessment: Predictive Parameters
All multi-organ (e.g., lung, cardiac, inferior vena cava [IVC], kidney, and urinary bladder) sonograms were performed and recorded on ultrasound equipment used by four emergency physicians, with at least 2 years of POCUS experience. These examinations were performed before patients received excess fluid or diuretic therapy (e.g., 500 cc intravenous [IV] fluid or 40 mg iv diuretics) since this might have affected the sonographic measurements recorded for analysis. An experienced radiologist, cardiologist, and an additional emergency physician were anonymized to patient findings, assessed the appropriateness of the imaging data, and performed measurements needed for analysis. The Venue ultrasound equipment system (GE Medical Systems, WI) was used for the sonographic examinations. Every POCUS examination was conducted with the head of the bed tilted from 0 to 45°. The details of the sonographic examinations are provided in Figures 1 and 2.
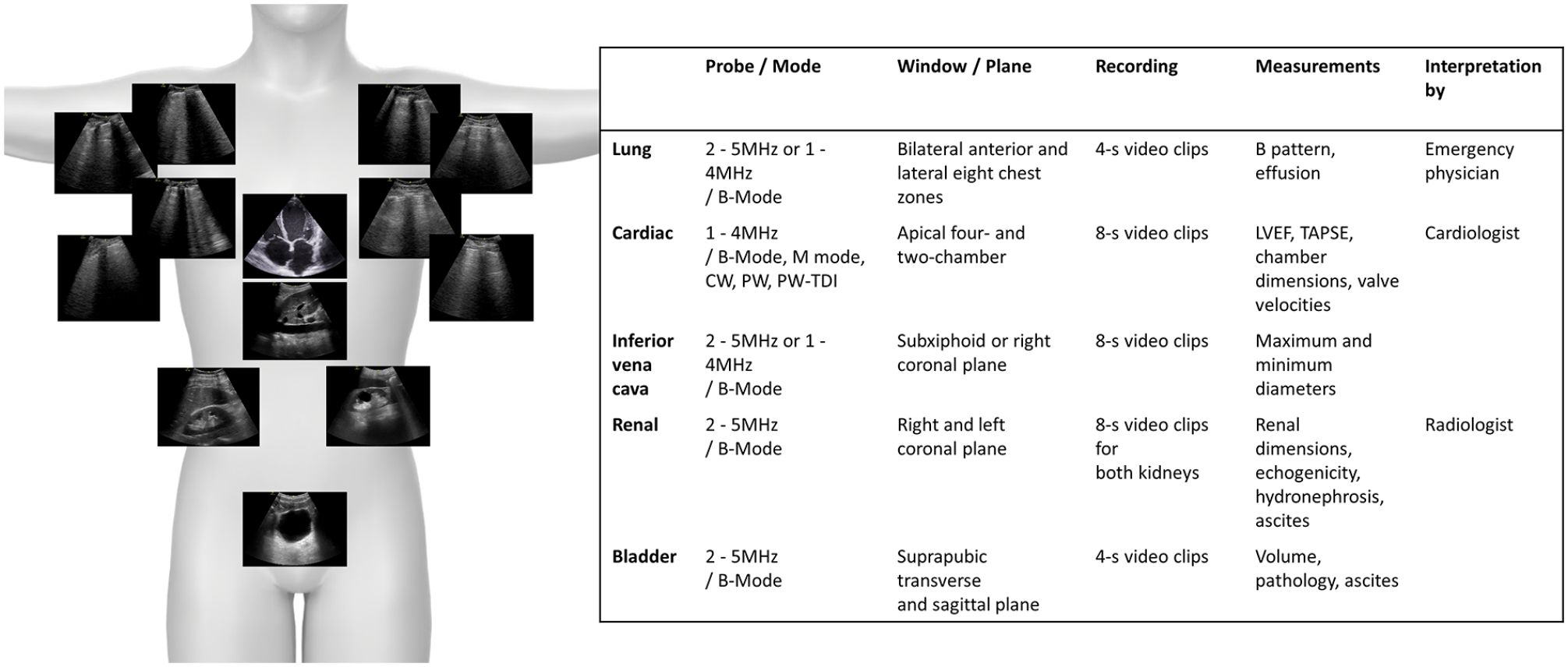
The details of the methods used to conduct the sonographic examinations in this cohort of patients.
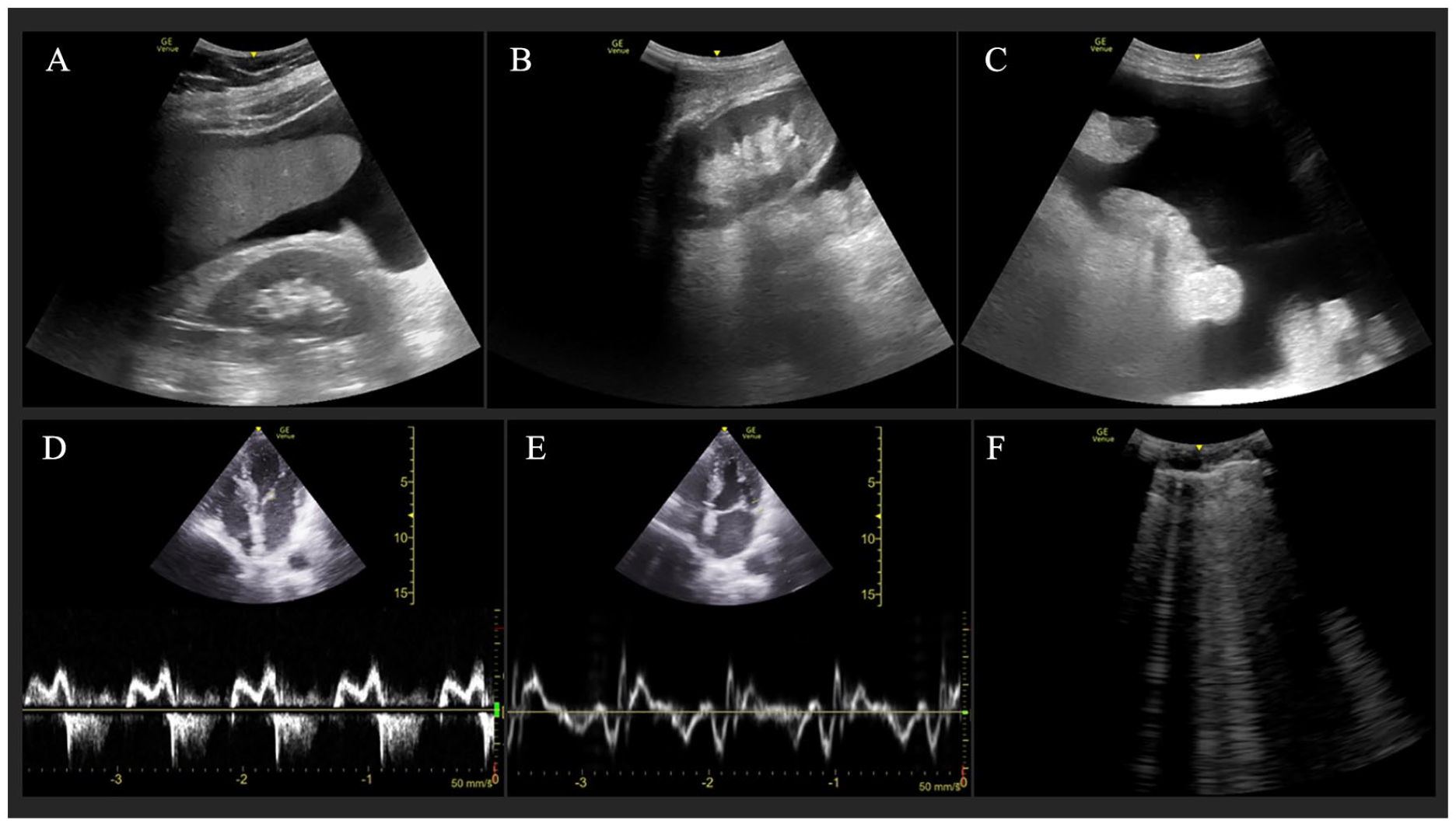
A 68-year-old male patient, with malignancy, succumbed during hospitalization, after presenting to the emergency department with dyspnea and AKI. The multi-organ point-of-care ultrasonography (POCUS) examination demonstrated diffuse ascites in the right kidney (A), left kidney (B), and suprapubic (C) regions. The echocardiogram revealed a mitral E/A ratio of 0.8 (D) and a mitral E/e ratio of 11 (E). The bilateral lung sonogram demonstrated the presence of multiple B-line artifacts (F).
The cortical and parenchymal thickness, length of the kidneys, renal echogenicity, the presence of hydronephrosis, and initial urine volumes were assessed based on the sonographic images of the kidneys and bladder. 10 Echogenicity was categorized into four grades, from 0 to 3, based on the evaluation of the ED physicians. The criteria used were as follows: hypoechoic in comparison to the liver, isoechoic in comparison to the liver, hyperechoic in comparison to the liver, and isoechoic in relation to the renal sinus. The thicknesses of the parenchyma and cortex were measured bilaterally from the middle, lower, and upper renal poles. These measurement means were noted, initially for each kidney, as well as bilaterally. At any grade, hydronephrosis was categorized as bilateral, unilateral, or absent. Urine volumes, at initial imaging, were calculated using both sagittal and transverse views of the urinary bladder. Diffuse ascites was noted when free fluid was present in two of the three following areas: right peritoneal space, left peritoneal space, and pelvic cavity (See Figure 2A–2C).
Based on the echocardiogram, left ventricular ejection fraction (LVEF), tricuspid annular plane systolic excursion (TAPSE), left atrium (LA) diameter and length, and right (RV) and left ventricle (LV) basal dimensions were determined based on the apical four- and two-chamber sonographic views. From the same apical four-chamber acoustic window, Doppler measurements were obtained for the mean mitral annular e′ velocity, tricuspid regurgitation peak velocity, and mitral inflow peak velocities (E and A). The IVC diameters were taken longitudinally and 3 cm from the right atrium, with the subcostal acoustic window.
Finally, eight anterior-lateral chest zones, bilaterally, were evaluated and reviewed for lung sonographic B-line artifacts. The B-line artifacts were chosen as indicators of interstitial syndrome and required three or more B-lines, in at least two bilateral areas, intercostally. Furthermore, from the posterior axillary line, the lungs were evaluated in the coronal plane for pleural effusion. Unilateral, bilateral, or nonexistent pleural effusions were noted for each patient.
Data Analysis
SPSS software (IBM Corp., Armonk, NY) was used for the statistical analyses. The continuous variables were shown using the interquartile ranges and median values. Categorical variables were summed using percentages and frequencies. The normality of the continuous variable distribution was tested using the Kolmogorov-Smirnov test and Q-Q plots. The student’s t-test or the Mann-Whitney U test was used to evaluate the continuous variables that differed between 1-month mortality and non-mortality groups. The Pearson chi-square test or the Fisher test was employed for categorical variables. A statistically significant α value of 0.05 was chosen a priori.
The univariate model contained 23 multi-organ sonographic parameters. Variables with a significance level below 0.01 in univariate analysis were included in a multivariate regression model to determine their predictive value for 1-month mortality. Only parameters with completion rates exceeding 90% were included in the statistical model. 4
Results
Data from 165 patients with AKI, from the previous study, were included in this analysis. 4 The patients’ baseline demographics are presented in Table 1. The 1-month mortality rate among these patients was 24.2% (n = 40). The median age was higher in the mortality group than in the non-mortality group (n = 78 and 70, respectively; P = .04). The malignancy rate was also higher in the mortality group than in the non-mortality group (45 and 23.2%, respectively; P = .01). The diagnoses of sepsis, septic shock, and the systemic vasodilation/renal vasomodulation subtypes were more common in the mortality group (P < .05). No statistical differences existed in the other patients’ demographics, except for admission and discharge rates.
A Comparison of Demographic Factors Collected Within the Mortality and Non-mortality Patient Groups.
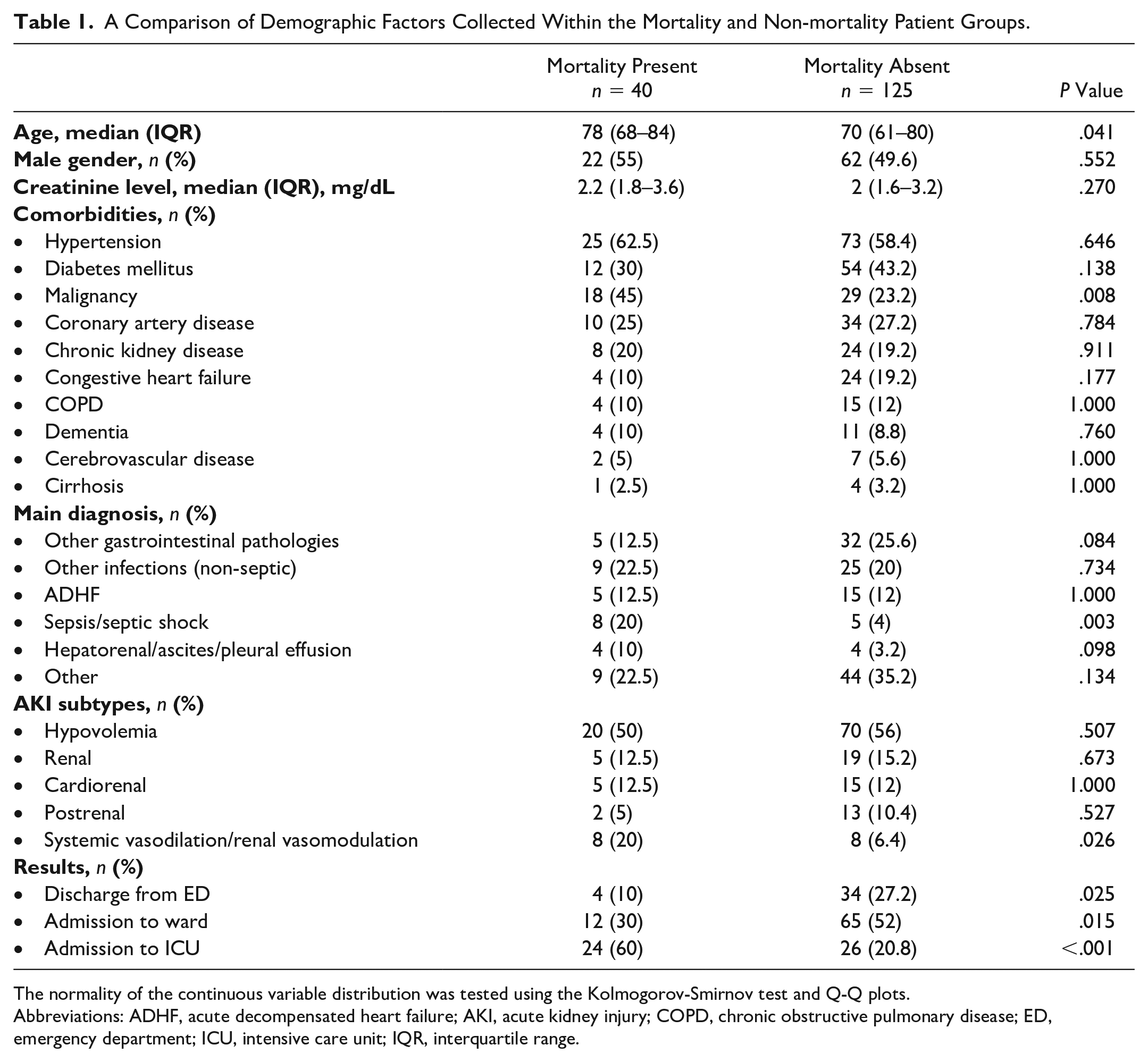
The normality of the continuous variable distribution was tested using the Kolmogorov-Smirnov test and Q-Q plots.
Abbreviations: ADHF, acute decompensated heart failure; AKI, acute kidney injury; COPD, chronic obstructive pulmonary disease; ED, emergency department; ICU, intensive care unit; IQR, interquartile range.
In univariate analysis, including predictive sonographic parameters for mortality, the rate of diffuse ascites was higher in the mortality group than in the non-mortality group (30.8% and 9.6%, respectively; χ2; P = .01). The rate of bilateral effusion was also higher in the mortality group (27.5% and 12.8%, respectively; χ2; P = .03) (See Table 2).
The Univariate Analysis of the Sonographic Parameters Collected Within the Mortality and Non-mortality Patient Groups.
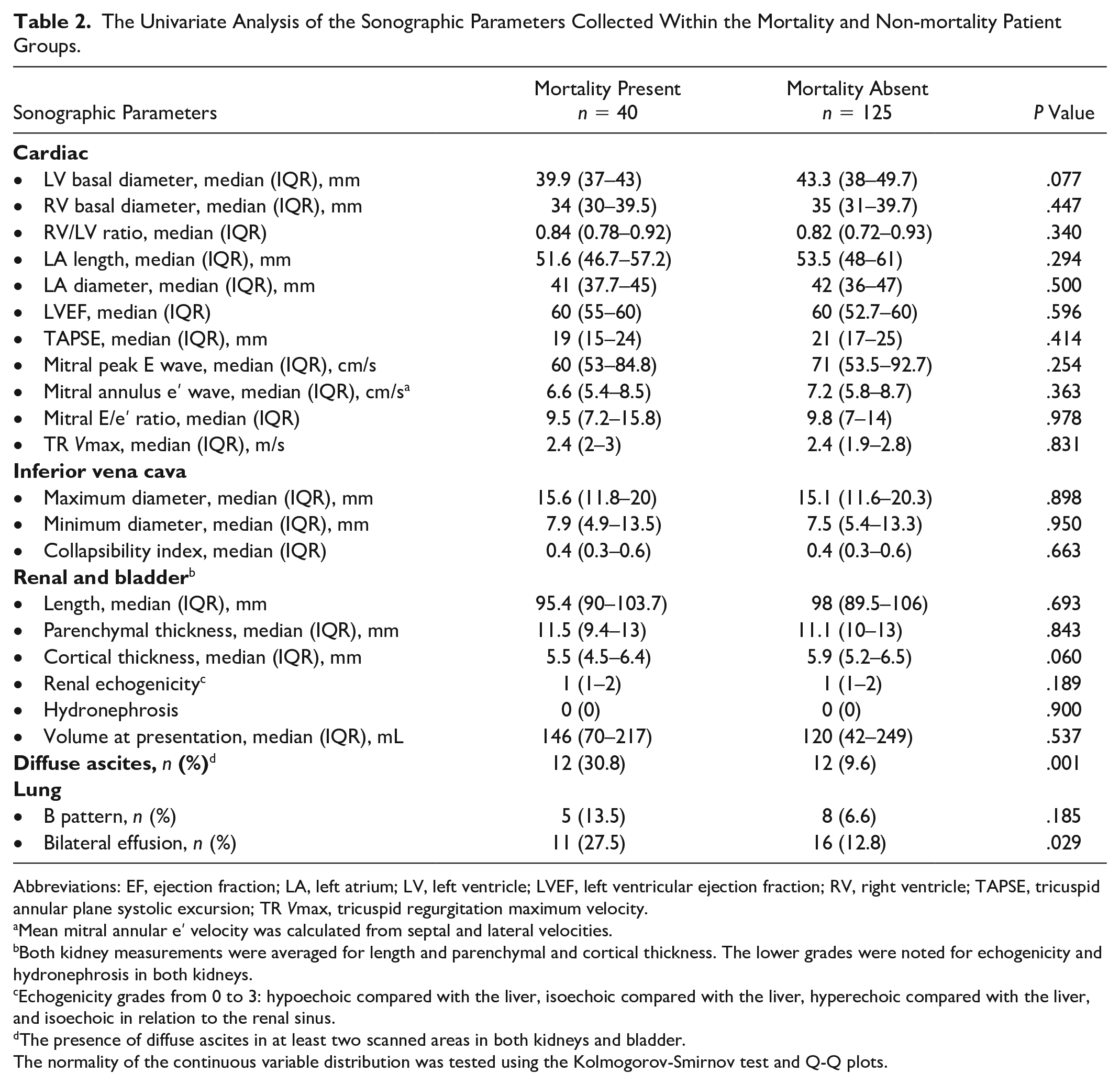
Abbreviations: EF, ejection fraction; LA, left atrium; LV, left ventricle; LVEF, left ventricular ejection fraction; RV, right ventricle; TAPSE, tricuspid annular plane systolic excursion; TR Vmax, tricuspid regurgitation maximum velocity.
Mean mitral annular e′ velocity was calculated from septal and lateral velocities.
Both kidney measurements were averaged for length and parenchymal and cortical thickness. The lower grades were noted for echogenicity and hydronephrosis in both kidneys.
Echogenicity grades from 0 to 3: hypoechoic compared with the liver, isoechoic compared with the liver, hyperechoic compared with the liver, and isoechoic in relation to the renal sinus.
The presence of diffuse ascites in at least two scanned areas in both kidneys and bladder.
The normality of the continuous variable distribution was tested using the Kolmogorov-Smirnov test and Q-Q plots.
Multivariate analysis (Hosmer-Lemeshow test, P = .056) identified diffuse ascites (odds ratio [OR], 3.67; 95% confidence interval [CI]: 1.27–10.61) and LV basal diameter (OR, 0.93; 95% CI: 0.87–0.99) as being associated with 1-month mortality in patients presenting to the ED, with AKI (See Table 3).
The Results of a Logistic Regression Analysis, Based on the Multi-organ Sonographic Parameters, to Predict 1-Month Mortality for This Cohort of AKI Patients.
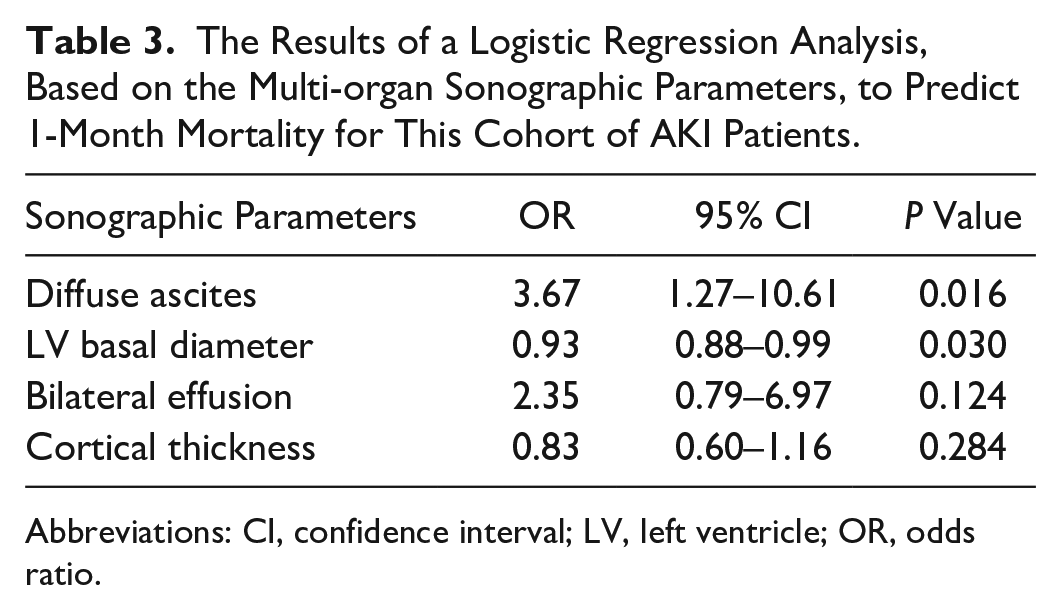
Abbreviations: CI, confidence interval; LV, left ventricle; OR, odds ratio.
Discussion
Numerous studies have investigated the prediction of mortality in AKI. Consistent with these, the mortality group in the present cohort exhibited more advanced age and higher rates of malignancy, distributional type of AKI, and admission to the ICU than the non-mortality group. Based on a limited review of the literature, multi-organ sonographic parameters have not previously been studied for predicting mortality in AKI. The results of this study identified the presence of diffuse ascites and lower LV basal diameter as predictors of 1-month mortality in patients with AKI in the ED.
Cirrhosis was the most prevalent cause of ascites and accounted for about 80% of the cases. 11 Additional causes of ascites include malignancy (10%), heart failure (3%), and other disease factors. 11 The relationship between AKI and ascites is well-established in patients with cirrhosis. 12 However, this study did not find any difference in mortality rates between the group of patients with cirrhosis and those without cirrhosis. This may be attributed to the small number of patients with cirrhosis included in this cohort. Conversely, it has been reported that the presence of malignant ascites is a significant prognostic factor, and patients with this condition have poor survival rates and a median survival of 20 weeks after diagnosis. 13 Almost half of the mortality group had a malignancy, and this rate was twice as high as that of the non-mortality group, in this cohort.
In this study, the other predictor of 1-month mortality, associated with AKI, was a lower LV basal diameter. Only one meta-analysis has reported this finding, and that was in the case of severe sepsis, septic shock, and a mortality rate of 61%; 14 however, in this study’s cohort, the pooled LV dimensions were similar between surviving and non-surviving patients. However, in the meta-analysis, LV dimensions were slightly lower in non-surviving patients. 14 Other studies have reported no difference in baseline cardiac echo findings, in terms of mortality or AKI in the long-term follow-up of cirrhosis.15,16 Since the LV dimensions in the two groups (e.g., median LV dimensions of 40 mm in the mortality and 43 mm in the non-mortality groups) were similar, with a slight statistical difference between them. Based on this cohort’s LV dimensions, it would seem that the clinical utility of this measurement would be limited.
Limitations
The major limitation is the study design and convenient participant sample. There are major threats to internal and external validity. Given the lack of randomization, this is a huge limitation and does not allow for generalization. Another limitation is that the recruitment of patients coincided with the COVID-19 pandemic. The incidence of contracting COVID was not evaluated, since this research was focused on sonographic examinations, but it may have affected the mortality results.
Moreover, this was a secondary data analysis, stemming from a larger study, as outlined, and lacks a power analysis. This situation raises concerns about the adequacy of the sample size to reliably detect statistical differences. The convenient sample of patients reviewed may be underpowered, further compromising the generalizability of these results.
A final limitation involves the rates of completed sonographic examinations. Some were as low as 70%, and univariate analysis was therefore applied to parameters with rates of 90% or more. However, the multivariate analysis was performed in 85.5% of all sonographic cases completed.
Conclusion
This multi-organ sonographic study, including lung, cardiac, IVC, kidney, and urinary bladder, indicated that diffuse ascites was the most powerful diagnostic parameter associated with 1-month mortality, for this cohort of AKI patients. This result is most likely associated with the ED presentations of these patients with malignancy, as well as complications from AKI. These diagnostic findings could provide potential enhancements in risk assessment for AKI patients, presenting to the ED. However, these results must be interpreted with caution due to the cohorts’ recruitment during the COVID-19 pandemic, the purposive sampling approach, and varied sonographic examination completion rates. Further research in this area is needed with more rigorous methodologies that would assist in validating and refining these findings and foster broader clinical application.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The ethical approval for this study was obtained from Gazi University Faculty of Medicine Ethics Committee (IRB: NCT04470661).
Informed Consent
Written informed consent was obtained from all subjects before the study.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study because no animals were used during the study.
Trial Registration
Not applicable.
