Abstract
Objective:
The study was conducted to determine the value of various lung ultrasonography (LUS) findings in diagnosing neonatal respiratory distress syndrome (RDS) and to assess the ability of ultrasonography to predict the need for surfactant treatment.
Materials and Methods:
This cohort study was conducted on 164 neonates with RDS with a gestational age of over 28 weeks. Transthoracic sonography was performed in all patients in the infant isolet immediately after admission and stabilization of the infant and during the first 6 hours before surfactant treatment. Immediately after the sonogram, the neonates underwent an anterior–posterior view chest radiograph, before and after surfactant treatment.
Results:
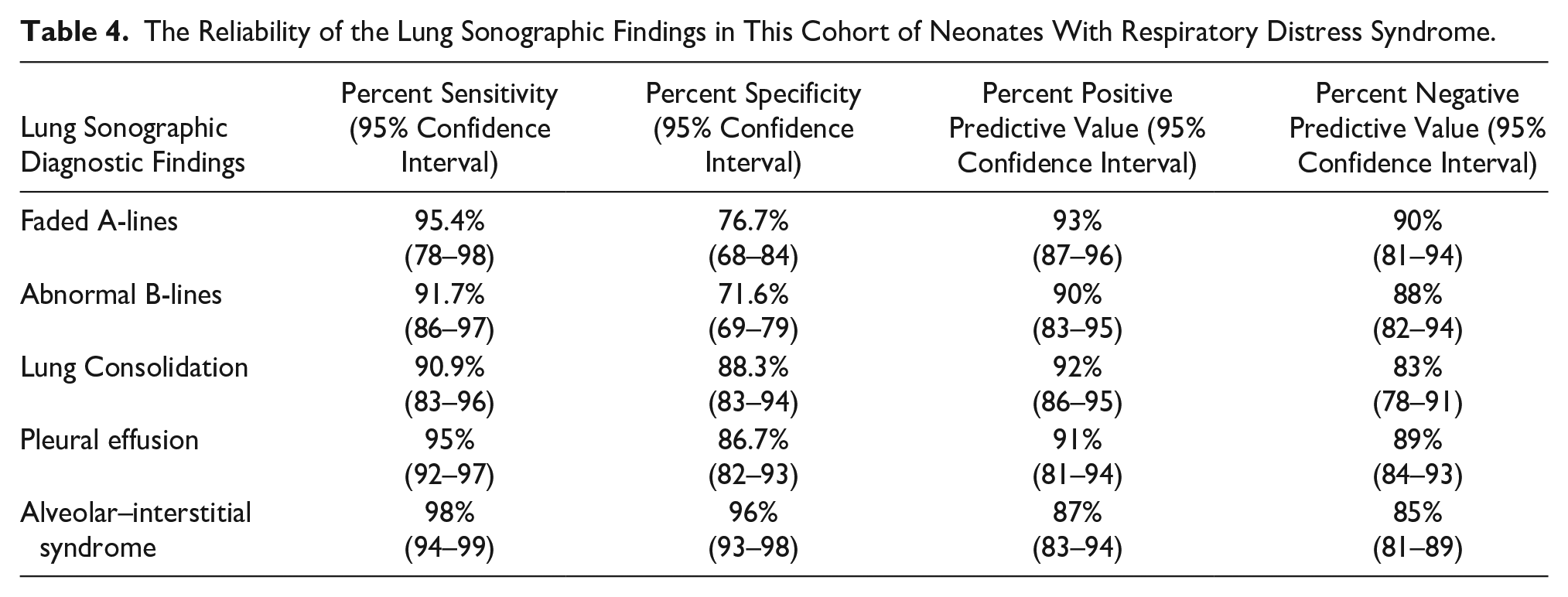
In comparison with clinical manifestations and radiographic findings as the references, the sensitivity and specificity of each of the LUS-related parameters for detecting RDS were as follows: Faded A-lines (95.4% and 76.7%), abnormal B-lines (91.7% and 71.6%), the presence of consolidation (90.9% and 88.3%), the presence of pleural effusion (95% and 86.7%), and alveolar–interstitial syndrome (AIS) (98% and 96%).
Conclusion:
LUS findings were valuable in assessing and grading the severity of neonatal RDS alongside clinical evaluation. LUS may assist in selecting infants for surfactant therapy and monitoring treatment response.
Respiratory distress syndrome (RDS), formerly known as hyaline membrane disease, is a common cause of death and complications in premature neonates. Preterm labor accounts for about 8% to 10% of all deliveries, and preterm birth accounts for about 60 to 80% of worldwide deaths and morbidity in neonates without congenital anomalies.1 –5 One of the most important morbidities due to prematurity is respiratory problems, headed by RDS.6 –8 RDS is a type of pulmonary developmental defect commonly seen with preterm labor and caused by a surfactant deficiency. Pulmonary surfactant deficiency leads to extensive atelectasis, residual lung capacity loss, and ventilation-to-perfusion ratio disturbance. The consequences of this condition are the weakness of the respiratory muscles and decreased pulmonary compliance, which is associated with decreased oxygenation, cyanosis, respiratory and metabolic acidosis, increased pulmonary vascular resistance, and right-to-left shunt through the ductus arteriosus. The prominent radiological findings include pronounced hypoaeration, bilateral fine granular opacities in the pulmonary parenchyma, and peripherally extending air bronchograms. In severe cases, extensive atelectasis and complete lung opacity (white lung) are seen.9 –14
In the last two decades, significant progress has been made in using ultrasonography in pediatrics, especially in assessing the developmental status of the fetus and infant. 15 This method has been very popular because it is less expensive, easily accessible, and noninvasive. However, based on previous studies, different sensitivities and specificities have been reported for sonography in the evaluation of neonatal lungs, which might be caused by various physiological and pathological factors.16 –19 This role has been particularly prominent in the assessment of neonatal respiratory disorders. Accordingly, various criteria have been proposed and interpreted to assess the rate of neonatal lung development.20,21 In general, pulmonary sonography has made it possible to assess the natural alveolar bed of the lung quickly and accurately and to distinguish between the alveolar and interstitial patterns of involvement of the lung.22 –25 Ultrasonography can aid in describing a neonates’ respiratory problems, including evaluation of meconium aspiration syndrome, RDS, TTN (transient tachypnea of neonates), and pneumothorax. Given that patients with RDS can benefit from treatment with continuous positive airway pressure (CPAP) and administration of selective surfactant, it is obvious that the changes in pulmonary function resulting from these treatments would be consistent with ultrasonography findings.26,27 There has also been evidence of the use of ultrasonic-based scoring systems to assess the need for surfactant administration in infants with RDS. 28 Based on the evaluations, the neonatal lung ultrasonography score correlated with pulmonary function indicators such as oxygenation. However, it has been understood that lung ultrasonography scores’ ability to predict response to treatment and the need for surfactant administration in infants with RDS has been highly dependent on various factors, such as gestational age.29 –31
Various studies have been performed regarding the value of ultrasonography in diagnosing neonatal RDS, and the diagnostic value has been reported as high for this purpose.32 –34 In a recent meta-analysis, the total evidence indicated a sensitivity of 92% (in the range of 89 to 94%) and a specificity of 95% (in the range of 93 to 97%) for ultrasonography in the diagnosis of RDS. 8 However, few studies have individually assessed the diagnostic value of each lung ultrasonography’s findings in neonates with RDS.
In this regard, the aim was to determine the sensitivity and specificity of each ultrasonography-related parameter for detecting RDS and the need for receiving surfactant considering the clinical manifestations and radiographic findings, as the references.
Materials and Methods
Patients
This cohort study was conducted on 164 neonates with RDS who were admitted to a teaching hospital’s neonatal intensive care unit in Tehran from April 2016 to July 2022. The study has received ethical approval from the Ethics Committee of Shahid Beheshti University of Medical Sciences with the IRB number (IR.SBMU.MSP.REC.1398.357). Before enrolling the neonates into the study, the study was discussed with the parents, and written informed consent was obtained from them. The inclusion criteria were (1) gestational age of at least 28 weeks at birth, (2) infants with the following symptoms during the first 24 hours after birth: high respiratory rate 60 times per minute, intercostal or sub-rib retraction, granting, or nasal flaring.
In this regard, the neonates who were intubated in the operating room immediately after birth due to respiratory distress and underwent surfactant therapy, those with congenital or cardiovascular abnormalities, or congenital lung disease, patients who were discharged or died before completing both chest radiography and ultrasonography, patients who developed dyspnea solely due to severe infectious agents and meconium aspiration syndrome were all excluded.
Considering the fraction of inspiratory oxygen (FIO2), neonates with RDS were classified into two groups with and without the need for surfactant, the ones who required assisted ventilation with a fraction of FIO2 higher than 0.3 were categorized in the group with the need for receiving surfactant (200 mg/kg of poractant, through the intubation–surfactant–extubation technique) and others were categorized in the group without the need for surfactant therapy. The study relied on the European Consensus Guidelines on the Management of Respiratory Distress Syndrome: 2022 Update for guidance and reference. 35
Lung Ultrasonography Protocol
Transthoracic ultrasonography was performed in all patients with a SonoSite M-turbo portable ultrasound device with a linear array transducer at a frequency of 7.5 MHz. Patients were examined by a radiologist with 10 years of experience in doing pediatric ultrasonography. Ultrasonography was completed on the neonate bedside immediately after admission and stabilization of the infant and during the first 6 hours before surfactant treatment.4,14
Transthoracic lung ultrasonography (LUS) was performed with longitudinal views on the anterior and posterior walls of the chest. Bilateral upper anterior, lower anterior, upper posterior, and lower posterior chest images were obtained. LUS images were stored digitally.
Suggested findings of RDS on LUS were as follows (see Figures 1–4):
A gradual fading to disappearance of the normal A-lines, which are unrealistic echogenic images of the pleural line seen in parallel and at equal distances from the top of the examination site to the edge of the site.
Appearance of the unusual B-lines, which are unrealistic hyperechoic images that look like a column from the pleural line to the edge of the examination site.
Lung consolidation is defined as the similarity of lung tissue to liver tissue, sometimes referred to as hepatization, and contains air bronchogram images.
B-line pattern or alveolar–interstitial syndrome (AIS) during an LUS as more than 3 B-lines are seen in the images, or a white lung is seen in the examined area. It is also known as a severe B-pattern if it is large and compacted.
Pleural effusion (fluid accumulation in the space between the diaphragm and the pleura).
Bilateral white lung is the presence of many B-lines in 6 lung regions without horizontal reverberation. 36
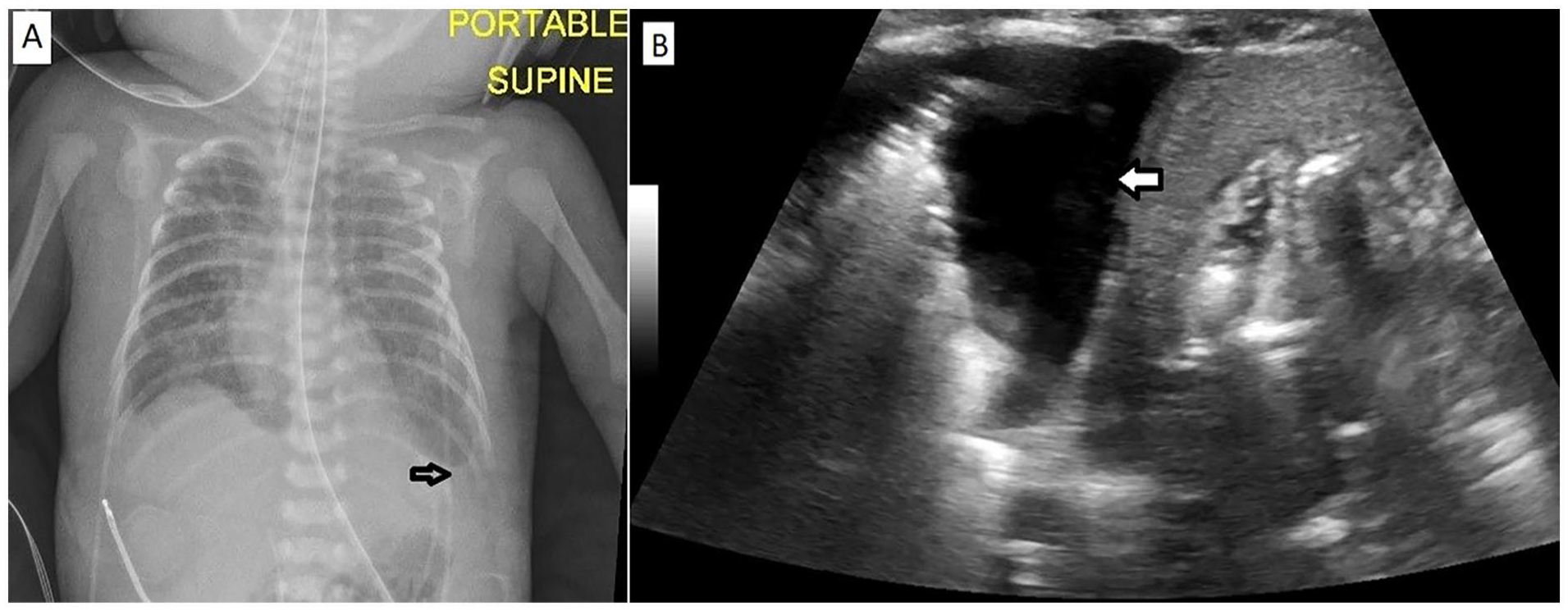
(A) A chest radiograph of a 28-week gestational age neonate demonstrated the diffuse ground glass opacities bilaterally accompanied by blunt left costophrenic angle (see white arrow) consistent with respiratory distress syndrome and left pleural effusion. (B) The lung sonographic image, of the same neonate, shows pleural effusion (see white arrow) and atelectasis of the lung.
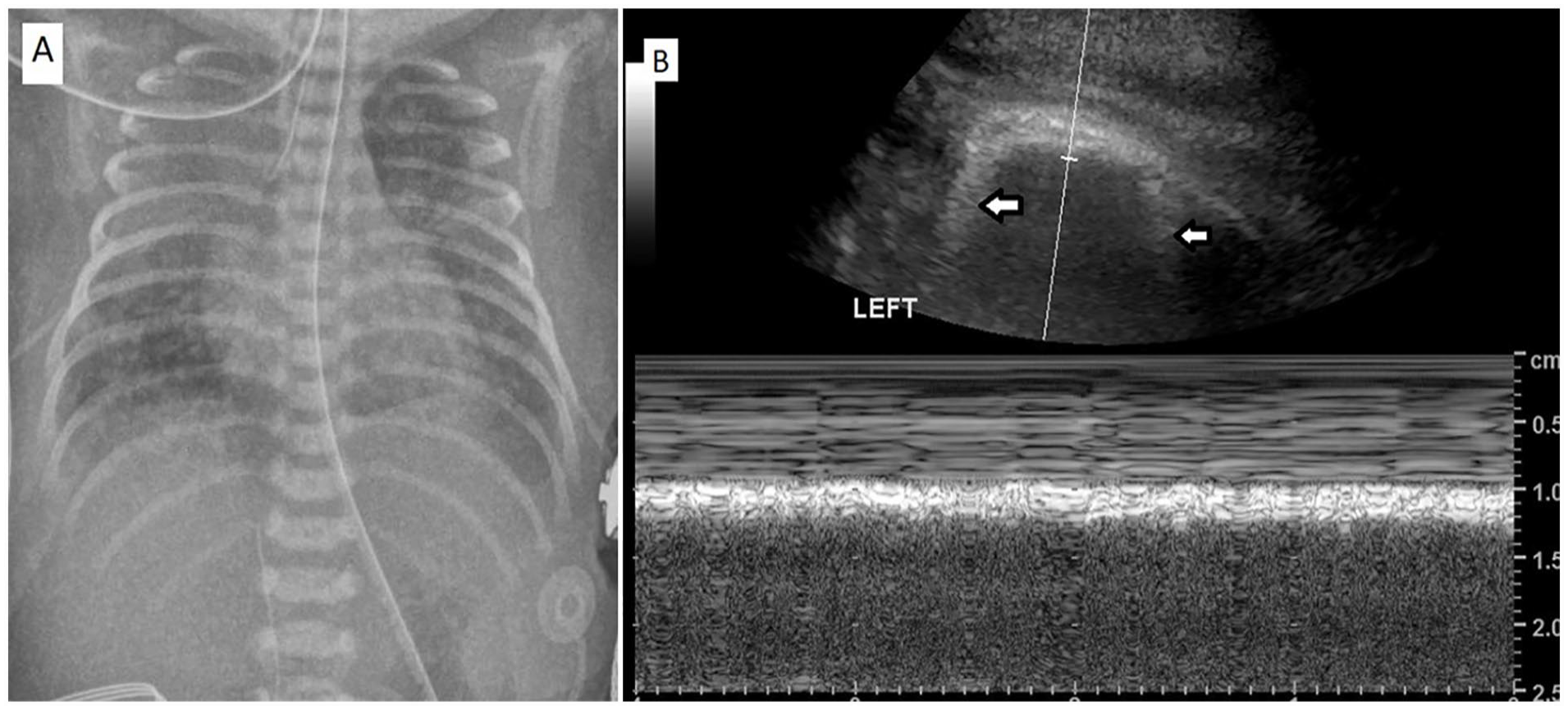
(A) A chest radiograph of a 30-week gestational age neonate demonstrated the diffuse ground glass opacities bilaterally and the appearance of the white lung in the upper zone of the right lung consistent with respiratory distress syndrome. (B) The lung sonographic image, of the same neonate, shows the disappearance of A-lines and the presence of B-lines (see white arrows).
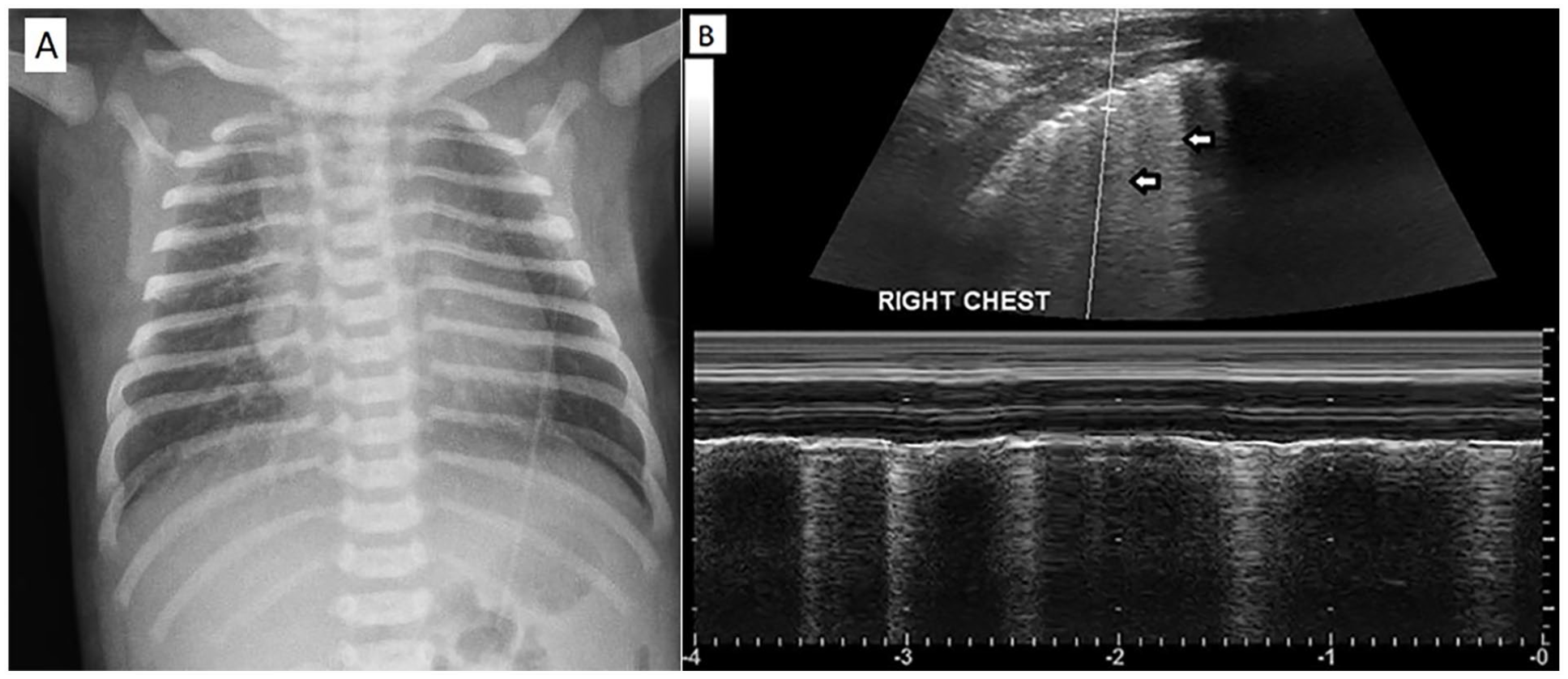
(A) A chest radiograph of a 29-week gestational age neonate exhibited diffuse fine reticular opacities consistent with respiratory distress syndrome. (B) The lung sonographic image, of the same neonate, shows the fading of A-lines along with the presence of B-lines (see white arrows).
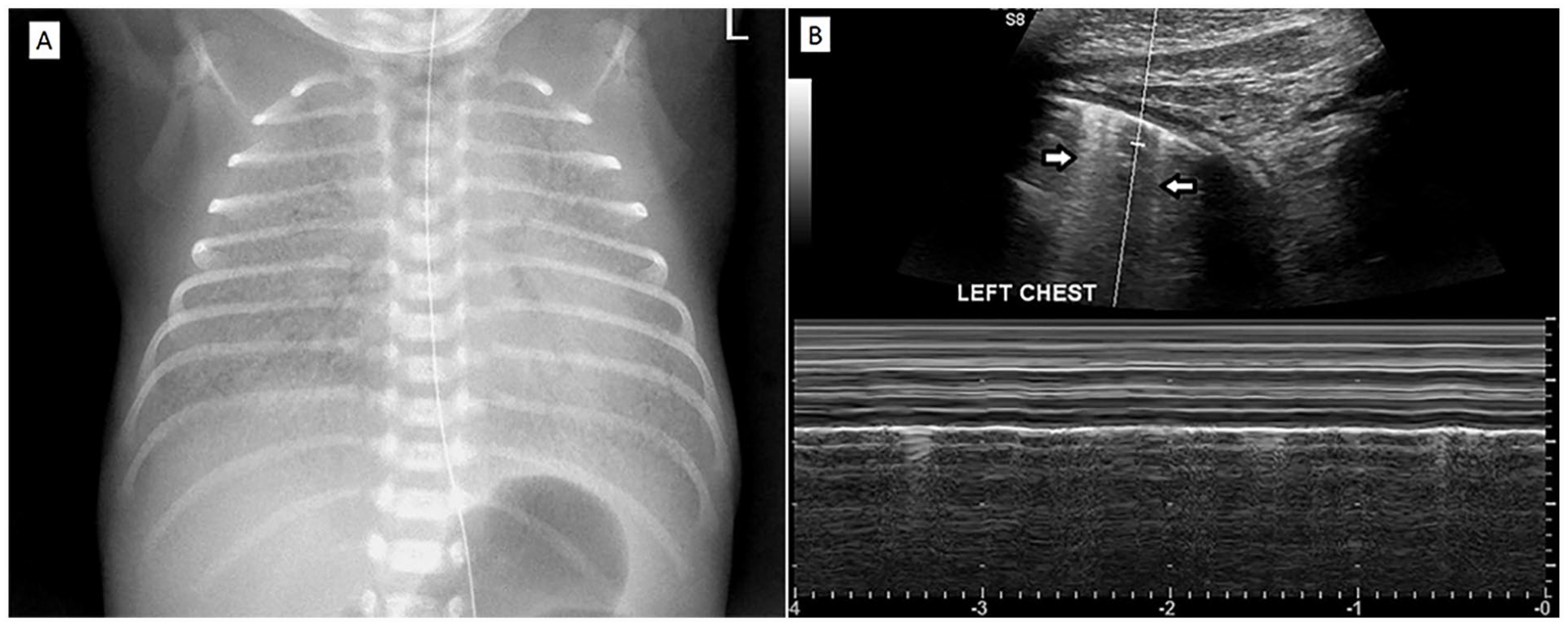
(A) A chest radiograph of a 31-week gestational age neonate demonstrated the diffuse ground glass opacities and air bronchogram bilaterally in favor of respiratory distress syndrome. (B) The lung sonographic image of the same neonate shows the fading of A-lines along with the presence of B-lines (see white arrows).
Chest Radiography Protocol
Immediately after the LUS, the neonates underwent an anterior–posterior view chest radiograph (CXR), before and after the possible need for surfactant treatment. Before including the CXR images in the study, a second radiologist, with 5 years of experience reporting pediatric chest imaging, conducted a thorough assessment. Only the radiographic images of high quality were included. Then the CXRs were interpreted by a third radiologist with 8 years of experience in reporting pediatric chest imaging. This radiologist was asked to review the two groups of neonates with and without the need for receiving surfactant, after anonymization. The typical CXR diagnostic findings that suggest RDS are hypoexpansion, fine and scattered granular densities, air bronchogram, ground glass opacity, or white lungs.
Statistical Analysis
For data analysis, results were presented as mean ± standard deviation (SD) for quantitative variables and were summarized by frequency (percentage) for categorical variables. Continuous variables were compared using the t-test or Mann-Whitney U test whenever the data did not appear to have normal distribution or when the assumption of equal variances was violated across the study groups. In this regard, the best cut-off value and the sensitivity and specificity of this cut-off point were also determined. P-values of ≤.05 were considered statistically significant. The statistical software SPSS version 23.0 for Windows (IBM, Armonk, New York) was used for the statistical analysis.
Results
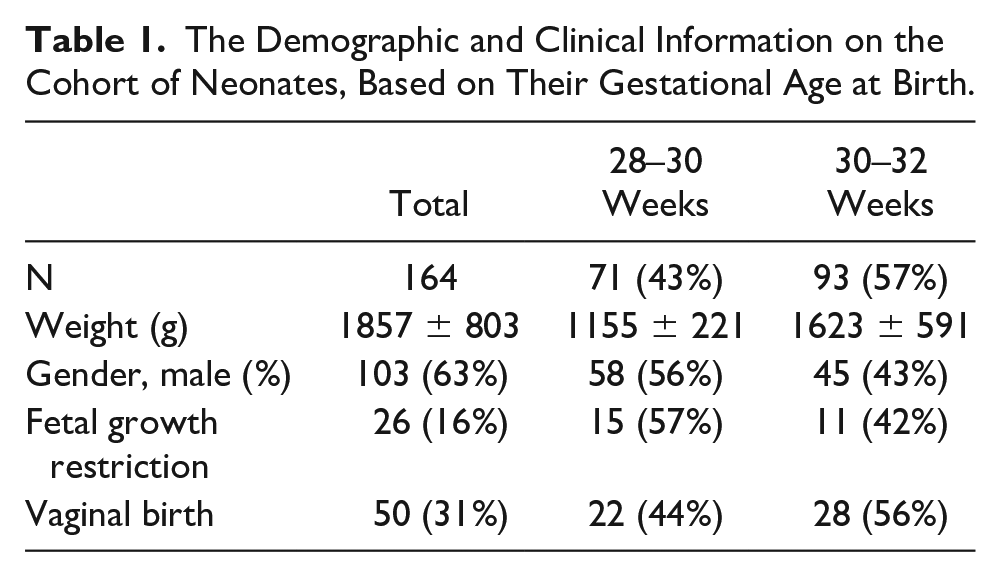
In this study, 66 neonates with RDS needed surfactant administration and 98 neonates with RDS, without the requirement of surfactant administration, were included. Demographic and clinical details are shown in Table 1. 71(43%) neonates were at the gestational age of 28 to 30 weeks, and 93(57%) were at 30 to 32 weeks of age. The average birth weight of newborns was estimated to be 1857 ± 803 g. 103 (63%) of neonates were male. 26(16%) of cases had a history of fetal growth retardation. 50(31%) of neonates were given birth vaginally (see Table 1).
The Demographic and Clinical Information on the Cohort of Neonates, Based on Their Gestational Age at Birth.
In the evaluation of the CXR, in total, 152 cases (93%) had an abnormality in their radiographs, including 122 cases (74.3%) with fine reticular opacity, 33 cases (20%) with ground glass opacity, and 9 cases (5%) had white lungs (see Table 2).
Comparison of Chest Radiographic Findings, in Neonates of a GA of 28–30 Weeks and 30–32 Weeks.
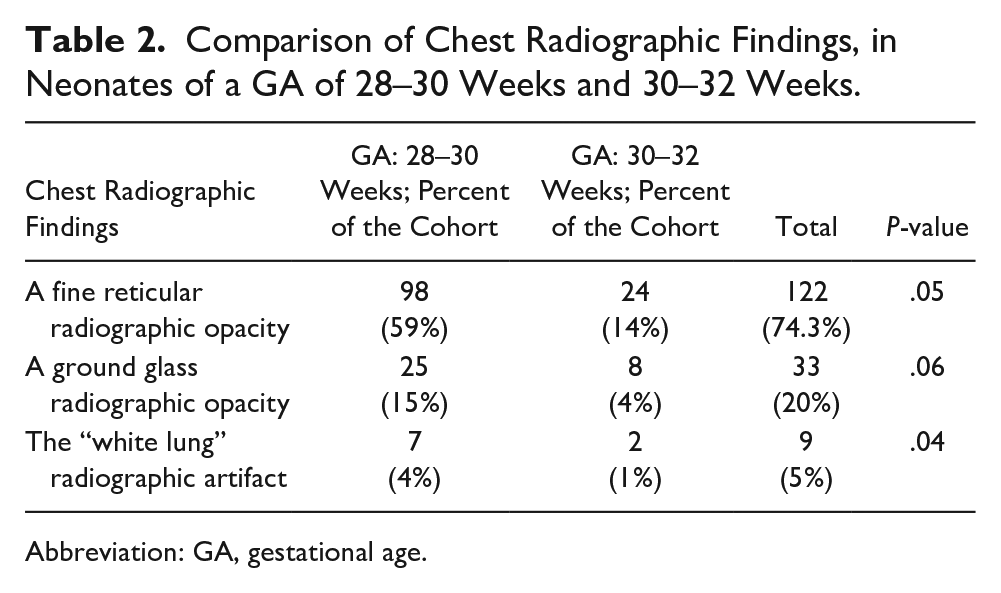
Abbreviation: GA, gestational age.
For the neonatal LUS, the diagnostic findings were as follows:
The disappearance of the A-line in 66 cases (40.2%) in the right lung and 41 cases (25.6%) in the left lung.
Unusual B-lines in the right lung in 67 cases (41.5%) and the left lung in 63 cases (39%).
A consolidation in the right lung in 39 cases (24.4%) and the left lung in 37 cases (23.2%).
The presence of a pleural effusion, in the right lung, in 5 cases (3%) and the left lung in 4 cases (2.4%).
AIS was reported in the right lung in 16 cases (9.8%) and the left lung in 23 cases (14.6%) (see Table 3).
A Comparison of Lung Sonographic Findings in Neonates in Neonates of a GA of 28–30 Weeks and 30–32 WEEKS.
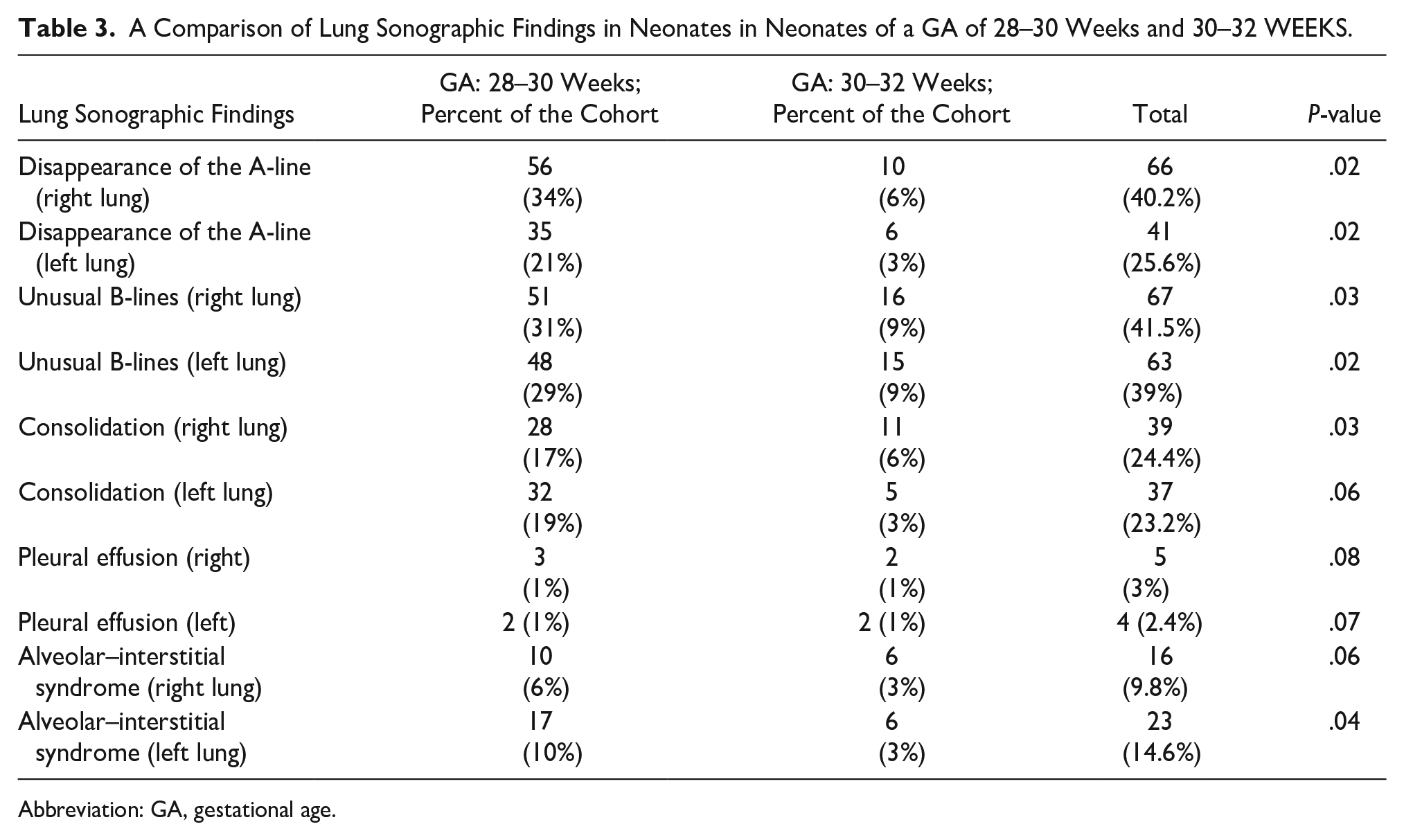
Abbreviation: GA, gestational age.
In comparison with clinical manifestations and radiographic findings as the references, the sensitivity and specificity of each of the parameters related to LUS were as follows: faded A-lines (95.4% and 76.7%), abnormal B-lines (91.7% and 71.6%), the presence of consolidation (90.9% and 88.3%), the presence of pleural effusion (95% and 86.7%), and AIS (98% and 96%) (see Table 4).
The Reliability of the Lung Sonographic Findings in This Cohort of Neonates With Respiratory Distress Syndrome.
A total of 66 cases (40%) underwent surfactant therapy. Comparing the two groups of neonates with and without the need for surfactant treatment (see Figure 5), significantly more prevalence of A-lines (P = .005), abnormal B-lines (P = .009), consolidation (P = .004), pleural effusion (P = .002), and AIS (P = .001) were revealed in those requiring surfactant therapy.
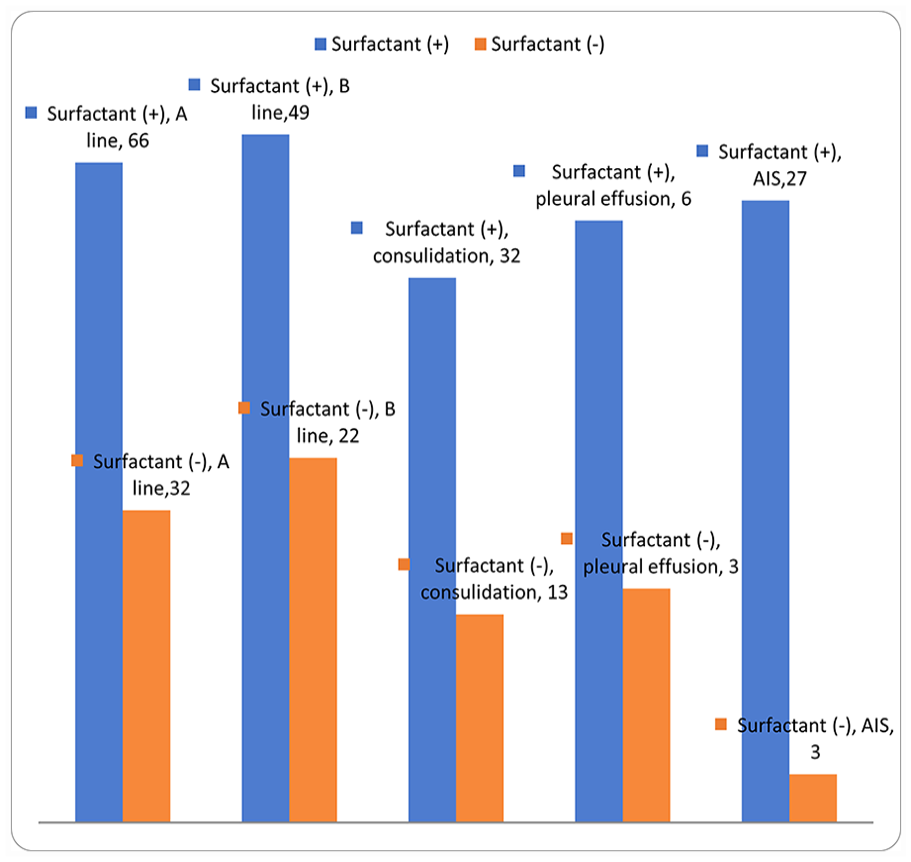
The lung ultrasonographic findings in neonates with respiratory distress syndrome comparing with and without required surfactant therapy. AIS, alveolar–interstitial syndrome.
Discussion
The role of imaging in assessing various diseases is well-known.37 –45 As a result of the important clinical consequences of neonatal respiratory distress and the need to evaluate and diagnose its severity and extent, various diagnostic tools, such as radiographic evaluations, have always been considered. Recently, the use of LUS in assessing the extent of pulmonary involvement has been considered, and specific imaging findings have been identified for this syndrome. However, there is still a controversy about whether such diagnostic tools can help clinicians decide on treatment protocols such as surfactant therapy. The aim of the present study was the evaluation of the LUS findings of neonates with RDS and to assess the value of this tool in predicting the extent of the disease. Second, it was important to test the hypothesis of whether using these sonographic parameters could be effective in choosing surfactant as a therapeutic option. In the first phase of the assessment, the LUS findings were examined to determine whether the results were associated with RDS. In this cohort, there was a significant impact but not all of the neonates with RDS had these types of sonographic findings, including A-line blurring, abnormal B-line, a degree of condensation or pleural effusion as well as the appearance of AIS. The second important point was the high agreement between the LUS findings and the CXR findings in neonates with RDS, so each of mentioned sonographic parameters was able to diagnose RDS compared to the radiographic findings with high sensitivity, specificity, and accuracy. Therefore, due to some contraindications in the use of radiography, the use of LUS in estimating the incidence and severity of RDS in neonates could be considered very useful. Also, in the final stage of the study and by comparing the frequency of LUS symptoms in infants with and without the need for surfactant, it was noted that there was a significant difference in the incidence of these sonographic findings between the two groups of infants. In short, these symptoms can also be very helpful in suggesting neonates, with RDS, for surfactant therapy. Of course, judgments about the usefulness of this diagnostic tool in selecting neonates for this treatment protocol will rely on the results and clinical consequences of treatment in infants who are monitored and provided clinical surveillance, which should be evaluated in future studies.
In general, evaluation of these LUS findings in neonates, with suspected RDS and conjunction with clinical findings, can assist physicians with diagnosing RDS and treatment with surfactant. Almost all similar studies have emphasized the accuracy and efficiency of LUS for this purpose. In the study by Lovrenski, 46 out of 47 cases of RDS, 45 of them showed similar results and findings with CXR and LUS, which indicated the reliability of this diagnostic method in preterm neonates with RDS. In the study of Ahuja et al., LUS had a sensitivity of 85.7% and a specificity of 75% for diagnosing RDS, which was very similar to the current study findings. However, they eventually concluded that LUS could be used as a complementary method to CXR in the diagnosis of RDS. It is important to note that this diagnostic choice can be used to predict early bronchopulmonary dysplasia (BPD) and reduce the cumulative radiation dose in these neonates.47,48 In the study of Oktem et al., 49 it was shown that the use of LUS was a safe and accessible diagnostic technique at the patient’s bedside and could determine the response to treatment of patients with RDS to surfactant in the early stages of the disease, as well as determine neonates requiring re-treatment. However, the advantage of their study was the serial evaluation of changes in sonographic symptoms after surfactant therapy. In the study of Liu et al., 50 it was also shown that the simultaneous presentation of pulmonary density, abnormal pleural lines, and the disappearance of A-lines was associated with 100% sensitivity and specificity in diagnosis. In addition, lung pulses had a sensitivity of 80% and a specificity of 100% in diagnosing neonatal RDS, which again emphasized the value of LUS in evaluating RDS. These works underscore the high diagnostic value of LUS, along with its low cost, low side effects, and high diagnostic ability to be performed at the patient’s bedside, which makes it an ideal choice predicting the admission to the neonatal intensive care unit. But there are still many questions that remain unanswered about whether LUS can diagnose RDS alone, whether it can determine RDS severity, and whether it can determine the therapeutic response to the surfactant.
Limitations
This study has limitations and chiefly these are due to the research design and the convenient sample of patients examined. While the study comprises a large cohort and adds scientific evidence, it is important to note that generalizations cannot be made. It is important to further underscore that this study did not include a control group. Therefore, conducting a study with a control group would be beneficial in determining the incidence of LUS abnormalities in neonates without respiratory distress syndrome (RDS). This type of experimental design, conducted as a clinical study, would yield higher levels of evidence. In addition, these results demonstrated the descriptive nature of LUS and CXR findings, but additional studies are needed to employ specific grading systems related to this objective.
Conclusion
In conclusion, this cohort study of LUS findings and their clinical evaluations demonstrate the potential value in evaluating RDS severity, in neonates. Evaluation of LUS findings can also help select neonates for surfactant therapy and assess their response to treatment. Therefore, due to some well-known contraindications with pediatric radiography, LUS could be a useful diagnostic method for detecting RDS and its severity.
Footnotes
Ethics Approval
Ethical approval for this study was obtained from the Ethics Committee of Shahid Beheshti University of Medical Sciences with the IRB number (IR.SBMU.MSP.REC.1398.357).
Informed Consent
Before enrolling the neonates into the study, the study was discussed with the parents, and written informed consent was obtained from them.
Animal Welfare
Guidelines for humane animal treatment did not apply to the present study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
