Abstract
Prominent column of Bertin (PCB), also known as hypertrophic column of Bertin, are often referred to as pseudo tumors, given that they can mimic the appearance of renal tumors. This case series demonstrates a reverse situation where renal masses mimicked PCB. Two patient cases are presented to demonstrate how renal masses mimicked PCB, and a third comparative case is presented to demonstrate a split sinus sign that represents a PCB, a pseudo tumor confirmed with a computed tomogram (CT). Considering the renal masses mimicking PCB, CT, and histology confirmed the presence of renal cell carcinoma. Renal masses which present sonographically as PCB, are most likely to be overlooked and have negative impacts when undetected. Early detection of renal tumors is vital in improving a patient’s prognosis. This case series is useful in providing further evidence of how renal masses can mimic PCB, with sonography. Careful sonographic examination of PCB should be encouraged, and if sonographic features are uncertain, consider further evaluation by urology, especially for those patients with a background of hematuria and no previous imaging studies.
Prominent columns of Bertin (PCBs), also known as hypertrophic column of Bertin, are areas of normal cortical renal tissues that extend and can surround the renal pyramids.1–3 Prominent column of Bertin can become hypertrophied with incomplete fusion of fetal lobes giving off double thickness, mimicking renal masses.4,5 On a sonogram, PCB shows similar echogenicity compared to the renal cortex and presents as a continuous and clearly defined indentation of the renal cortical tissue, extending into the renal sinus complex. This has become known as the split sinus sign1,2,6 (see Figure 3). A PCB occurs in 50% of the population, and 20% of PCBs can occur bilaterally. 1 This case series demonstrates that although a PCB can be mistaken as a pseudo tumor or renal mass, 7 care should be taken to not overstate a sonographic PCB, as subtle renal masses can also look like a PCB.
Prominent column of Bertin presentations, with sonography, are often considered a normal variant and suggests that no further clinical surveillance is needed. 2 However, with the increasing incidence of renal cell carcinomas (RCCs), late detection of these masses ultimately result in poor prognosis. With adequate clinical evaluation, appropriate laboratory testing (for urine cytology and renal function) and radiologic assessment, early detection of renal masses/malignancy is possible. The significance of early detection influences the impact on prognosis and Improves patient management. 8
The incidence of RCC is on the increase and accounts for 2.4% of cancers affecting the global adult population; 9 since the 1900s, the incidence of renal cancer has increased by 90%. 10 In the United Kingdom, renal cancer is the seventh most common cancer and accounts for 4% of all newly diagnosed cancers. 11 This rate is expected to increase by 26% from 2014 to 2035, especially with the widespread availability of diagnostic cross-sectional imaging. 12 Renal cancers are considered the ninth most common cancers affecting global individuals across the globe, with increasing incidence and is reportedly associated with poor prognosis.1,8,13 A sensitivity of 82% to 83% and specificity of 98% to 99% has been attributed to sonographically detecting RCC; however, the accuracy of sonography to screen for RCC is largely dependent on the size of the mass. 13 Unlike RCC, the prevalence of a PCB in the population is largely unknown; 7 however, Houat et al, 1 posit that a PCB occurs in 50% of the population, and 20% of PCBs occur in both kidneys. Available literature would indicate that knowledge of a PCB entity and its characteristics were not well known by radiologists until the 1960s, and efforts toward its understanding are continuing. 7
While sonography has proven to be a good diagnostic imaging tool in the screening of renal masses, it does have some pitfalls in practice that must be acknowledged. A particularly important pitfall is that the sonographic presentations of PCB could mask co-existing or actual renal masses, which can only be uncovered with complimentary imaging, such as contrast-enhanced ultrasound (CEUS), computed tomography (CT), or magnetic resonance imaging (MRI).1,2,4 Routine renal sonograms are prone to technical, interpretative, and imaging pitfalls, and subtle renal lesions presenting as a PCB can be missed.7,8,10 Some renal masses can be isoechoic to the surrounding renal cortex on a sonogram, and mimic a normal PCB, therefore, differentiating between PCB and the presence of a subtle renal mass can be challenging, even for experienced sonographers. The current practice in some health care settings, is that a PCB presentation with sonography is often considered a normal variant, 7 suggesting no diagnostic surveillance is required. This course of action could lead to poor prognosis if masked masses are not initially detected.
This case series presents two patient’s cases where sonographically appearing PCBs were confirmed to be renal masses when imaged with CT (see Figures 1 and 2). Histology also confirmed these renal masses to be RCCs. This case series is useful in providing further evidence of renal masses mimicking PCB, with sonography. An additional case (see Figure 3) is included as a comparison and demonstrates a CT-proven PCB which although it had a sonographic appearance, like the described cases of renal masses mimicking PCB, it was a hypertrophic normal renal tissue. This would further buttress the fact that the similar appearance in both categories of cases can prove diagnostically challenging and creates a great chance for a missed diagnosis. The careful examination of a PCB, to exclude concerning sonographic features, is encouraged. If a PCB presents with uncertain features, the urology team should be alerted for further consideration of investigative management, especially those with a history of hematuria and no previous imaging studies.
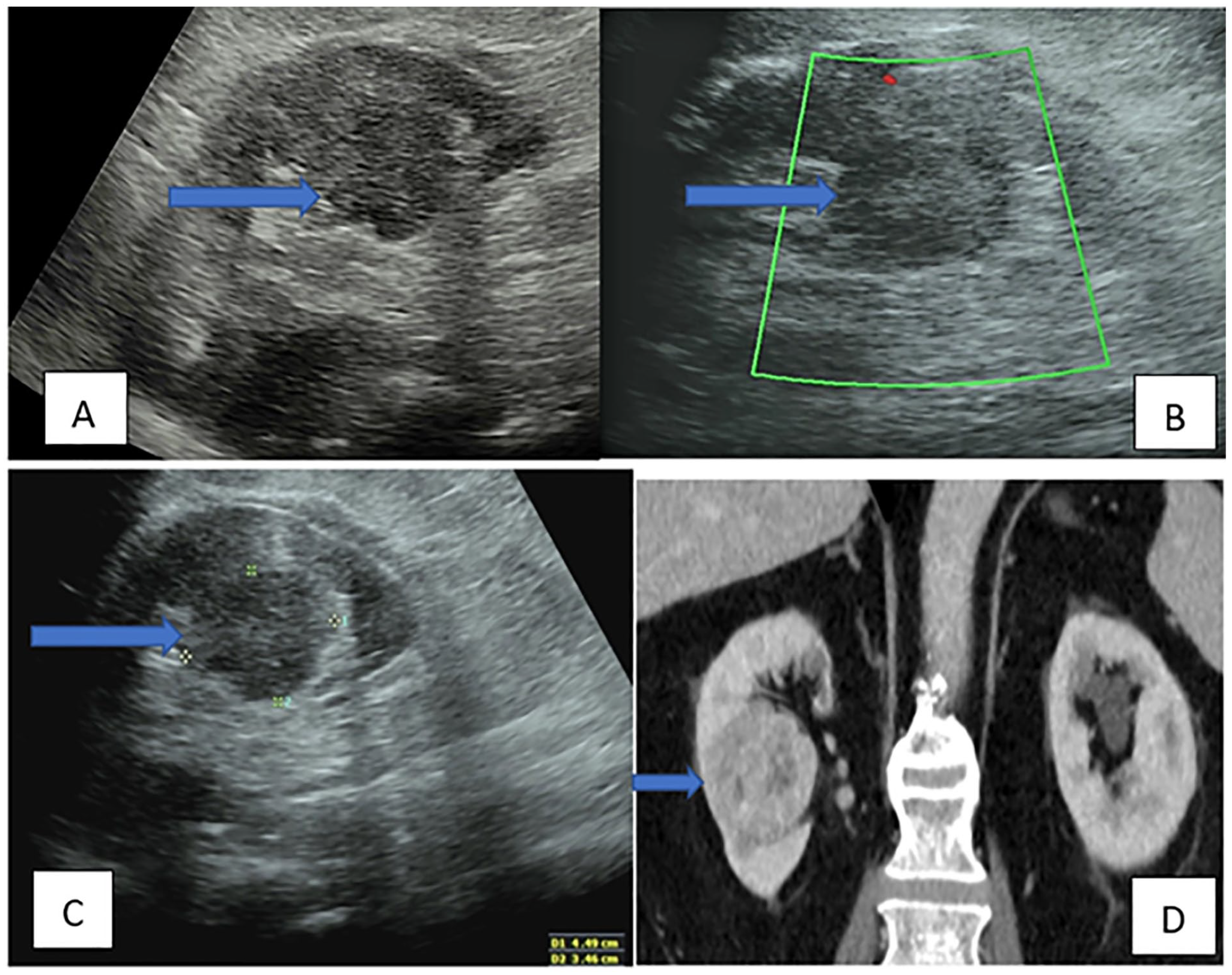
Case 1: Selective sonographic and CT images of a renal mass, which mimic a PCB. In image (A), a longitudinal view of the right kidney, with a renal mass at the mid-lower pole, mimics a PCB on the sonogram (blue right arrow). In image (B), the longitudinal view of right kidney lesion demonstrated no detectable vascularity, with color Doppler (blue right arrow). Image (C) provides a longitudinal view of the right renal lesion with measurements of 4.44 cm by 3.46 cm (blue right arrow); the CT image, in (D), shows the right renal mass on coronal CT with heterogeneous enhancement (blue right arrow). CT, computed tomogram; PCB, prominent column of Bertin.
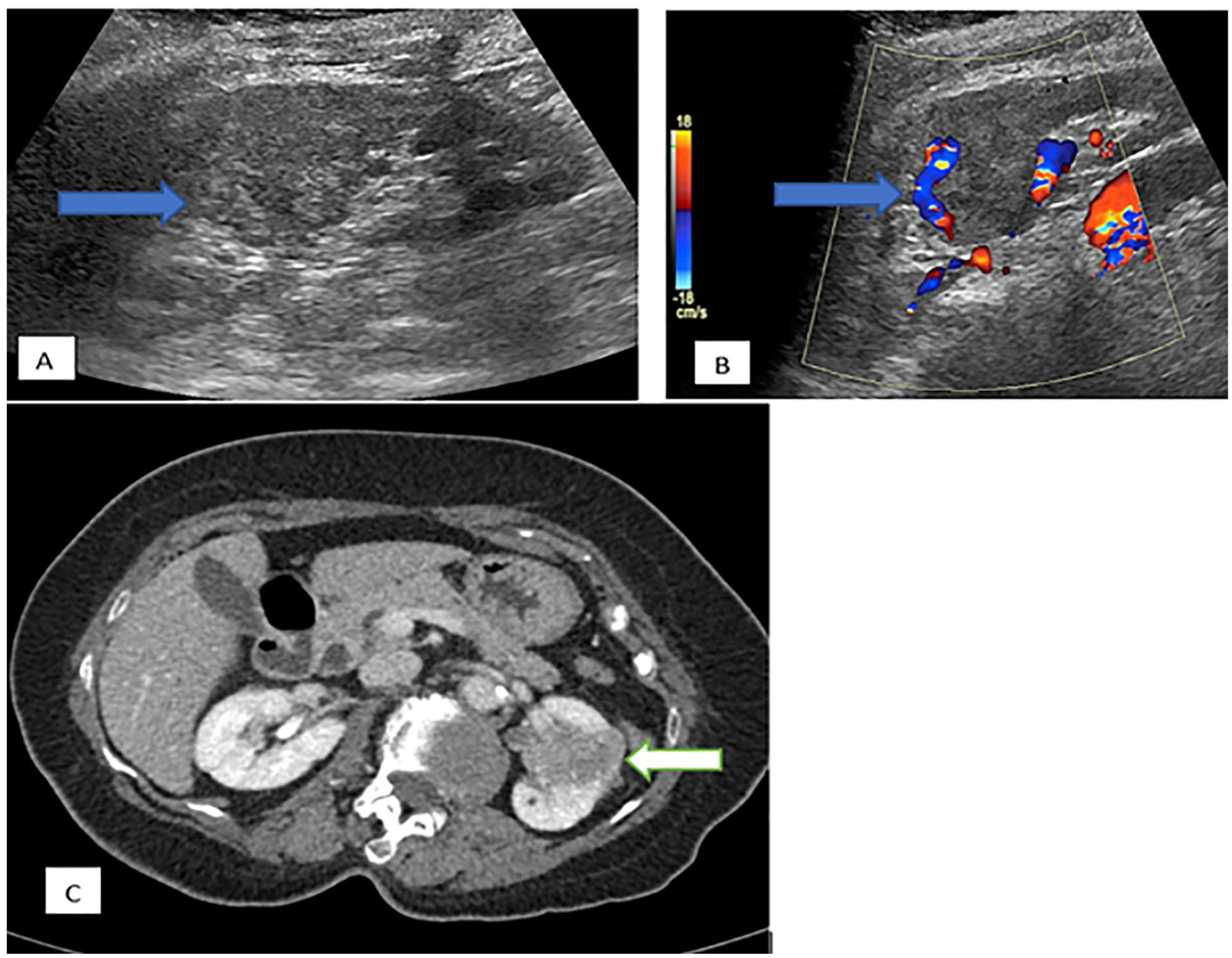
Case 2: This series of sonograms and CT images demonstrates a renal mass mimicking PCB. In image (A), a longitudinal view of the left kidney demonstrates a renal mass, at the mid pole mimicking PCB on the sonogram (blue right arrow). The (B) image shows the same area within the longitudinal view of the left renal mass mimicking PCB with peripheral vascularity on color Doppler (blue right arrow). The (C) image is a CT (axial image) confirming the presence of a left renal mass with heterogeneous enhancement (white left arrow). CT, computed tomogram; PCB, prominent column of Bertin.
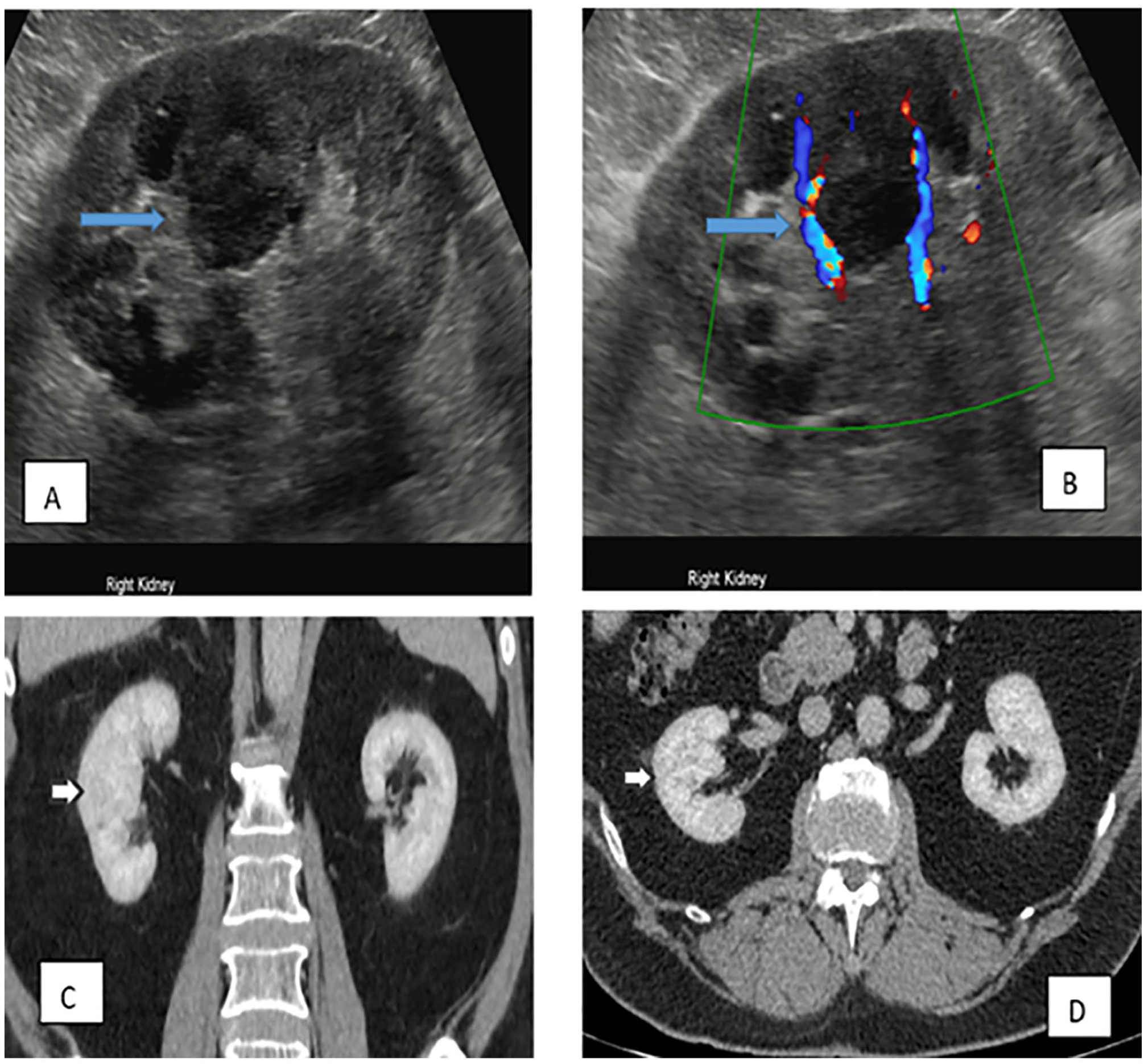
A selection of sonograms and CT images demonstrate the appearances of PCB. In image (A), a longitudinal view of the right kidney demonstrates the presentation of PCB mimicking a pseudo tumor (blue right arrow); The image (B) shows a longitudinal view of the right kidney, with peripheral vascularity around PCB (blue right arrow); The (C) image shows both kidneys on a coronal computed tomogram (CT white right arrow) with no abnormally enhancing lesion. Image (D) shows axial view of both kidneys on the CT image (white right arrow), with no abnormally enhancing lesion. CT, computed tomogram; PCB, prominent column of Bertin.
Case Series
Case 1
An 85-year-old male with unexplained visible hematuria and a negative test for urinary tract infection (UTI) presented for a sonographic examination. With sonography, the acoustic windows were limited due to body habitus, a 45-mm (widest diameter) isoechoic renal area was seen in the mid-lower pole of the right kidney, no internal flow was observed with color Doppler, despite Doppler parameter adjustments. The sonographic characteristics showed a clearly defined and continuous renal cortical tissue extending into the sinus-echo-complex of the right kidney (split sinus sign), the echogenicity of this area was also like that of the surrounding normal renal cortex. No previous imaging was available for comparison. The sonographic features appeared typical for PCB (see Figures 1(A) and (B)); however, based on the patient’s symptoms and imaging limitations, the sonographer (with approximately 5 years of experience) contacted the urology team, to discuss the sonographic features. The urology team decided to perform a CT to completely exclude or determine the presence of a renal mass.
The CT showed a heterogeneously enhancing lesion arising from the lower pole of the right kidney with a widest diameter of 49 mm (see Figure 1(C)). There was no invasion of the perinephric fat, renal vein, or adrenal; however, the lesion was concerning for an underlying RCC with a staging of T1b N0 MX. A further note of left lower ureteric transitional cell carcinoma was identified, which was further confirmed with histological assessment to be a left lower ureteric tumor, with high-grade papillary urothelial carcinoma. The CT chest and abdomen showed no further metastatic lesions within the patient. This patient responded well to treatment after a right nephrectomy, which was not performed during the same hospital visit as the sonogram. The patient was then discharged with periodic follow-up and consultation management.
Case 2
An 83-year-old male with dysuria, unexplained visible and nonvisible hematuria and negative for a UTI presented for a sonogram, with no previous imaging study for comparison. The sonographic results described an area of a 30 mm (widest diameter) mimicking a PCB at the left mid renal pole with peripheral vascularity (see Figure 2(A) and (B)). The urology team was alerted that the appearances may represent a PCB, but taking into consideration the patient’s symptoms, the urology team decided to perform a CT to completely exclude or rule-in the presence of a renal mass.
The CT images demonstrated a 40 mm (widest diameter) left renal mass, which was confirmed to be RCC on histology (see Figure 2(C)). No metastatic lesions were seen elsewhere on CT chest and abdomen. The RCC was staged as T1b N0 M0. The patient responded well to treatment after a left nephrectomy, which was not performed during the same hospital visit for the sonogram). The patient was then discharged with periodic surveillance and consultation management.
Case 3
A 78-year-old man with two episodes of unexplained visible, nonvisible hematuria, and negative test for a UTI, presented for a renal sonogram, with no previous comparative diagnostic study. The sonographic assessment demonstrated appearances of a right renal mid pole lesion, with no internal color flow (see Figure 3(A) and (B)). The sonographic appearances demonstrated an extension of renal cortical tissue separating the pyramids; hence, the sonographer’s report suggested this may represent a PCB. The urology team decided to perform a CT to clarify the cause of patient’s symptoms, which revealed no focal renal lesion and confirmed the suspected sonographic renal lesion to be a PCB.
Discussion
Solid renal masses have a wide range of appearances and can be located anywhere inside or projecting from the kidneys.8,14 Renal cell carcinoma accounts for about 2% to 3% of malignancies in the adult population, and the incidence of occurrence seems to be rising annually. 6 Renal cell carcinoma is considered one of the best masqueraders due to the presence of other paraneoplastic manifestations of nonspecific constitutional symptoms (such as fever, arthralgia, cachexia, and myalgia), which may explain why up to a third of patients already get to the point of advanced disease at the point of diagnosis. 6 In this case series, RCC mimicked PCB (see Figures 1 and 2).
Prominent columns of Bertin are usually seen in the mid renal pole, and about 18% of these cases could be bilateral. 6 On a sonogram, they typically present as an indentation into the renal sinus creating a split-sinus effect, they are often continuous with the rest of the renal cortex, and would normally be isoechoic relative to the renal cortex (see Figure 3). 6 There is sufficient literature to demonstrate that this presentation may sometimes mimic a tumor (pseudo tumor), particularly when further imaging/investigations proved the entity to be of normal renal tissue.1,2,4,7,14,15
Pseudo tumors, such as a PCB, have been reported to mimic renal masses on a sonogram1,3,4 and this has largely influenced the wide acceptance of the sonographic appearance of PCB, as a pseudo tumor, in several clinical settings. It is often considered a normal variant that does not require any diagnostic follow-up, unless it presents with atypical features. 7 The three patient cases presented show similarities (with minimal variations) such as the presence of the split sinus sign in all three cases, the mid-pole lesions are of similar echogenicities as the surrounding cortical tissue, and minimal-to-no internal vascularity in all three cases (see Figures 1–3). This supports the position that it can be very challenging for even the most experienced ultrasound users to differentiate between PCB and the presence of a subtle renal mass, given that renal masses could mimic PCB and vice versa, especially in the absence of previous imaging for comparison.
Renal tumors masked by the appearance of a PCB may be isoechoic in echogenicity with or with no atypical sonographic features or abnormal color flow presentation, making it relatively difficult to diagnose with sonography. Alternatively, CT is considered the diagnostic imaging gold standard for the diagnosis, staging, and surveillance of RCC. 6
This case series should inform and provide evidence to the general body of knowledge that subtle renal masses could sonographically mimic PCB. The careful examination of a PCB is therefore encouraged to exclude concerning features on a sonogram. If PCB presents with uncertain features, the urology team should be alerted for further consideration of investigative management, especially those with a background of hematuria and with no previous imaging. Further cross-sectional imaging (such as CT and MRI) will likely uncover any masked sinister renal lesions (such as PCB-mimics) that would have been challenging to demonstrate on ultrasound1,2,4,7 and significantly improve patient management.
Contrast-enhanced ultrasound is another form of investigative imaging that can be used to rule out PCB.4,5 It has been reported to be safe and a fast method to exclude RCC in cases with atypical features of PCB. 5 A sensitivity of 89%, specificity of 96%, positive predictive value of 89%, and a negative predictive value of 96% have been reported for CEUS in its ability to exclude RCC in developmental pseudo tumors such as PCB. 5 This can serve as a substitute in the diagnostic work-up for developmental pseudo tumors.
On reflection, the recommendation is made for any atypical sonographic features of a PCB to be escalated to the urology team, so that further investigative management can be considered. This would be beneficial and contribute immensely to the early detection of subtle renal masses mimicking PCBs.
Conclusion
Some PCB appearances may mimic renal tumors, which may in fact be normal renal tissue. Similarly, this case series demonstrate that some renal tumors may present as PCBs, therefore increasing the chances of being diagnostically undetected. The possibility of RCC masquerading as a PCB creates the option for uncertainty, given that sonographic PCB presentations can occur. With more emphasis on the increasing prevalence of RCC and the significance of early detection on patient management, it is of utmost importance to escalate any uncertain sonographic features of PCB to the urology team, especially those with a background of hematuria with no previous imaging. It is important to provide a prompt referral to the urology team which can facilitate early detection, better patient management, and improved prognostic outcomes for patients.
Footnotes
Acknowledgements
The authors thank the entire staff of the University Hospitals of Leicester NHS trust for giving them consent to use the ultrasound images for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval was not sought for the present study because informed written consent was obtained from the institution to use the images for publication, together with assurances of complete anonymity.
Informed Consent
Verbal consent was obtained from the patient to use the images for publication. Written permission to use the image for publication was also obtained from the University Hospitals of Leicester NHS trust via the clinical lead.
