Abstract
Contrast-enhanced ultrasound is a valuable tool to evaluate liver lesions in a pediatric patient cohort. Unusual progressive hepatic echogenicity (UPHE) is a self-limiting phenomenon noted in a cohort of children after administration of ultrasonographic contrast agents (UCAs). UPHE appears initially as heterogeneous branching similar to portal venous gas, culminating in a diffuse globular appearance. The etiology of UPHE is unclear and may be from underlying hepatic microvascular disease or altered gut permeability. This case series describes pediatric patients with UPHE following UCA bolus and discusses its potential mechanisms in children.
Keywords
An unusual progressive hepatic echogenicity (UPHE) is a self-limiting phenomenon noted in a cohort of children, after administration of ultrasonographic contrast agents (UCAs). Similar findings have been described in adults as prolonged heterogeneous liver enhancement (PHLE). UPHE appears initially as heterogeneous branching similar to portal venous gas, culminating in a diffuse globular appearance. Generally considered benign in adults, its cause and significance in children are unknown. The etiology of UPHE is unclear and may be from underlying hepatic microvascular disease or altered gut permeability. In this case series, an UPHE following UCA bolus is described as well as its potential mechanisms in children.
Patient Case 1
A 5-month-old Hispanic infant with major histocompatibility complex (MHC) class II deficiency was admitted for bone marrow transplant. Less than two weeks following transplant, the patient developed fevers, rising bilirubin, and thrombocytopenia. This prompted a liver Doppler to evaluate for sinusoidal obstruction syndrome (SOS; formerly known as hepatic veno-occlusive disease [VOD]) or infection. At the time of the sonogram, the patient’s total bilirubin had risen from 0.2 to 0.8 mg/dL (normal lab values are <0.8 mg/dL). The patient had been on low bacteria enteral feedings. Grayscale and duplex Doppler demonstrated heterogeneity of the liver parenchyma and patent hepatic vasculature, with appropriately directed vascular flow. The main portal vein velocity was approximately 30 cm/s. The spleen was normal in sonographic appearance.
Contrast-enhanced ultrasound (CEUS) was performed at age 7 months using a LOGIQ E9 ultrasound equipment system (GE Healthcare; Milwaukee, WI), to evaluate for hepatic and splenic micro-abscesses. One intravenous injection of 0.5 mL Lumason (Bracco; Monroe Township, NJ) UCA was administered using a three-way stopcock technique. According to the manufacturer-recommended weight-based dosing of 0.03 mL/kg, the patient’s recommended dose would be 0.2 mL; however, a higher dose was selected based on the use of a small-caliber central line for injection, the presence of needle-free neutral displacement connectors in the injection system, and prior experience at the host institution with difficult visualization of such small doses of sonographic contrast. Static cine loops and representative images were captured using a curvilinear 2 to 9 MHz transducer, at the manufacturer’s low mechanical index (MI) contrast-specific mode.
Contrast-enhanced ultrasound demonstrated heterogeneous parenchymal enhancement, a finding that has been described in association with SOS/VOD.1,2 At approximately 1:15 (minutes: seconds) following injection, linear branching hyperechogenicity was visible in the liver parenchyma on the grayscale image in low MI dual-screen mode (see Figure 1A and B). This became progressively more conspicuous over 5 minutes, assuming a globular heterogeneous appearance (see Figure 1C). Comparison images of the spleen showed no such phenomenon (see Figure 1D). The abnormal liver echogenicity pattern was diminished, but still present 50 minutes post-injection despite attempts to rupture the UCA (see Figure 1E and F). Microbubbles could still be visualized within the main portal vein at this time; spectral Doppler revealed spikes in the main portal vein waveform characteristic of portal venous gas (see Figure 1G). There was no change in main portal vein flow velocity compared with pre-contrast imaging.
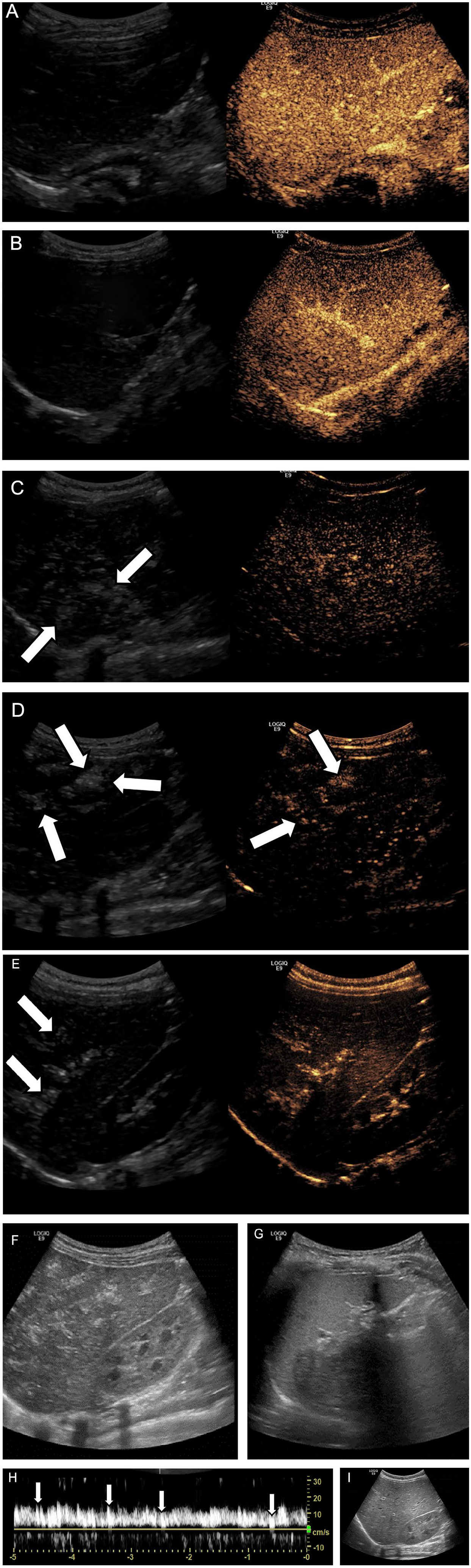
UPHE in a 5-month-old infant with rising bilirubin status post bone marrow transplantation. The grayscale images (left frame) demonstrate heterogeneous arterial (A) and portal venous phase (B) liver parenchymal enhancement, early appearance of UPHE (arrows) during the delayed phase with a linear, branching appearance resembling portal venous gas (C and D), and conglomeration of echogenic foci with a globular configuration at 5 minutes (E). Grayscale sonographic images show diminishing appearance of UPHE within the liver at 50 minutes (F), whereas a comparison image of the spleen obtained concurrently demonstrates normal homogeneous echotexture (G). Spectral waveform depicts spikes in the main portal vein (H). Grayscale sonographic image demonstrates spontaneous resolution of UPHE 12 hours after ultrasonographic contrast agent bolus injection (I). UPHE, unusual progressive hepatic echogenicity.
Grayscale sonography performed 12 hours later demonstrated hepatic parenchymal heterogeneity similar to the pre-contrast examination with resolution of linear branching hyperechogenicity (see Figure 1H). Spectral Doppler demonstrated slow velocity in the main portal vein (approximately 15 cm/s, previously 30 cm/s) without additional abnormality. There were no significant changes in the laboratory values in the 24 hours following the examination.
The patient experienced a prolonged hospital stay due to concerns for graft failure and rejection. Grayscale and duplex Doppler showed reversal of flow in the portal veins, diminutive caliber of the hepatic veins and ascites, concerning for SOS.
The patient subsequently developed shock with multi-organ failure in the setting of adenoviremia, graft failure, and engraftment syndrome. The patient died in hospital seven weeks following bone marrow transplant and approximately six weeks following CEUS.
Patient Case 2
A 36.4-week gestational age Hispanic boy with intrauterine growth restriction and gastroschisis was delivered vaginally following induction of labor for poor biophysical profile score (2/8). Birth weight was 1680 g. Gastroschisis reduction was achieved with silo placement starting on the first day of life (DOL 0) and surgical closure by DOL 6. The patient was subsequently treated medically for necrotizing enterocolitis (NEC) with pneumatosis and was placed on total parenteral nutrition (TPN). The patient developed cholestasis, with total bilirubin 7.4 and direct bilirubin 4.6. Aspartate transaminase (AST) and alanine transaminase (ALT) were mildly elevated at <2× normal limits. The hepatobiliary scintigraphy was negative for biliary atresia.
On DOL 77, an abdominal sonogram revealed a solitary hypervascular liver mass measuring 2.1 × 1.8 × 2.3 cm3. The serum alpha-fetoprotein (AFP) levels went from 11 115 IU/mL (normal laboratory values are 40–19 953 IU/mL) to 26 692 IU/mL over the following 9 days. The patient was clinically stable and tolerating oral feeding of fortified breast milk. There was no concern for NEC at that time.
Contrast-enhanced ultrasound was conducted with a GE LOGIQ E9 ultrasound system for further characterization of the liver lesion. Two injections of Lumason sonographic contrast (0.6 and 0.8 mL) were performed and cine images were obtained with a curvilinear 2 to 9 MHz transducer. Delayed static images of the liver mass were obtained using the linear 9 MHz and matrix linear 6 to 15 MHz transducers. The patient’s weight was 2.6 kg; therefore, the weight-based Lumason dose per the manufacturer’s package insert was calculated to be 0.08 mL (0.03 mL/g). However, as described previously, the higher doses were selected based on the use of a small-caliber peripherally inserted central line for injection, the presence of needle-free neutral displacement connectors in the injection system, and prior experience at the host institution with difficult visualization of such small doses of sonographic contrast. Three-way stopcock injection technique was utilized. The vendor-specific “flash” mode and application of color Doppler were applied to increase the MI and rupture the residual Lumason microbubbles after the first injection.
The liver mass demonstrated heterogeneous mild early arterial hyperenhancement relative to background liver parenchyma and delayed washout, an enhancement pattern suspicious for hepatoblastoma (see Figure 2A). The initial injection proceeded without incident. However, prior to the second Lumason injection and despite adequate clearance of echoes from the background liver parenchyma, globular, branching hyperechogenicity was apparent in the liver appearing as portal venous gas. This was also observed at 3:00 minutes following the second Lumason injection and as late as 16:45 minutes following injection (see Figure 2B). Further delayed imaging was not performed.
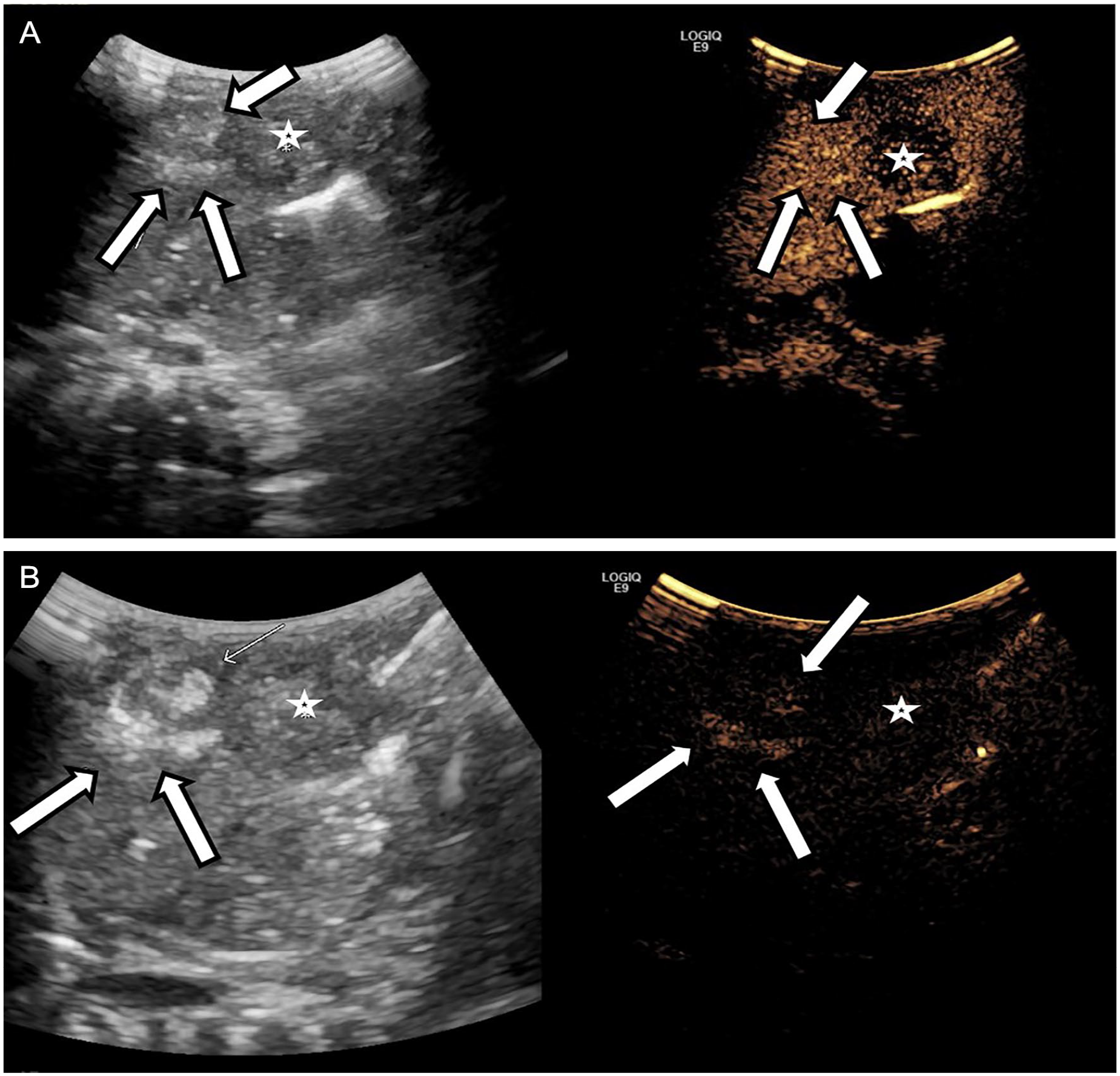
UPHE in a 3-month-old premature infant with gastroschisis and hepatoblastoma. Grayscale and CEUS images demonstrate a delayed washout of a solid liver mass consistent with hepatoblastoma (*) and UPHE (arrows) at 5 minutes (A). UPHE at 16:45 minutes despite washout of the background liver parenchyma (B). CEUS, contrast-enhanced ultrasound; UPHE, unusual progressive hepatic echogenicity.
The liver mass was resected on DOL 105 and confirmed the histologic diagnosis of hepatoblastoma. The adjacent liver specimen that was not involved by tumor demonstrated portal inflammation, fibrosis, and cholestasis. The patient underwent two cycles of chemotherapy with cisplatin, 5-fluorouracil, and vincristine, and the post-therapy magnetic resonance imaging was negative for disease.
Patient Case 3
A 5-year-old Hispanic boy with a history of cerebral palsy, complex partial epilepsy secondary to hemimegalencephaly, chronic lung disease, and Nissen fundoplication was admitted with vomiting and pancreatitis. The patient’s lipase was elevated at 1042 U/L (normal 127–209 U/L) and gamma-glutamyl transferase (GGT) elevated at 42 U/L (normal 5–17 U/L); AST, ALT, and total bilirubin values were normal.The patient was receiving electrolyte solution via percutaneous gastrostomy tube during the acute illness and was chronically on polyethylene glycol for constipation.
Grayscale sonography revealed an echogenic 9 mm focus at the porta hepatis of unclear etiology (see Figure 3A). However, portal venous and hepatic arterial flow were normal by color and spectral Doppler. A request for CEUS was completed 2 days later to evaluate for portal vein thrombosis versus artifact. One intravenous 0.5 mL injection of Lumason contrast was performed, consistent with the manufacturer-recommended weight-based dosing (patient weight 17 kg at the time of the examination). Imaging was performed with a GE LOGIQ E9 ultrasound system and a curvilinear 2 to 9 MHz transducer. Following intravenous contrast administration, the portal and hepatic vasculature was demonstrated to be patent without filling defect to suggest thrombus. Globular heterogeneous liver parenchymal enhancement was appreciated as early as 1-minute post-injection on the grayscale images and linear branching liver echogenicity persisted to at least 5:21 minutes post-injection time (see Figure 3B). Subsequent delayed images were not performed, as repeat contrast injection was not planned, and this was felt to be a benign and self-limited phenomenon. The patient’s pancreatitis improved with supportive care.
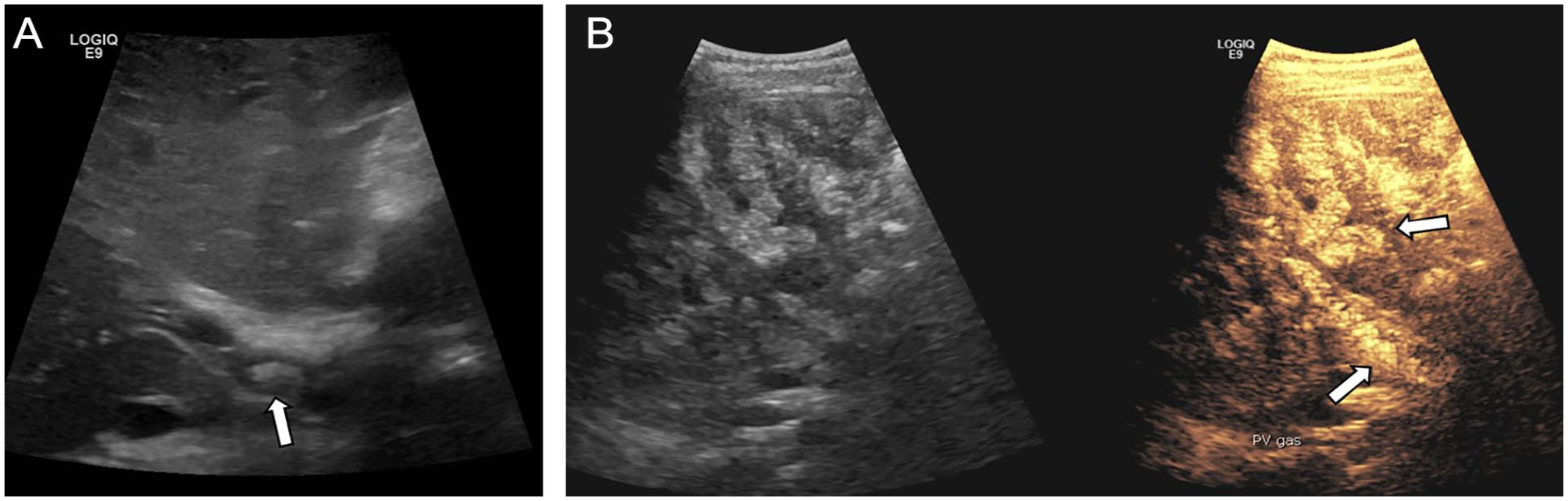
A 5-year-old boy with pancreatitis and suspected portal vein thrombosis. Grayscale sonography revealed an echogenic 9 mm focus at the porta hepatis of unclear etiology (A). Unusual progressive hepatic echogenicity (UPHE) is demonstrated at 5 minutes in grayscale and contrast-specific maximum intensity projection mode with full expression of the globular heterogenicity and portal venous gas (B).
Discussion
Prolonged heterogeneous liver enhancement following UCA administration has been described in the adult literature since 20023 and is characterized as having a “cloudy” or “wool-like” appearance.4,5 Based on a limited review of the literature, this series may be the first description of a similar finding in the pediatric population and its significance is uncertain. As discussed below, the etiology of this phenomenon is unlikely to be related to prolonged liver enhancement; therefore, the term “unusual progressive hepatic echogenicity” (UPHE) is proposed. Although the phenomenon in adults is felt by most authors to be benign in nature with no implications regarding underlying liver disease, it has been observed in patients with liver disease3–7 and in one patient with lactose intolerance. 8 These authors’ experience with three medically complex pediatric patients raises the possibility of a pathologic mechanism underlying this phenomenon.
Unusual progressive hepatic echogenicity in this series of pediatric patients was noted as early as 1 minute following contrast injection, whereas prior accounts from the adult literature describe PHLE as occurring at 2 minutes or later.3–8 This phenomenon was observed in three children over a 2-year time period out of approximately 35 CEUS abdominal examinations (~9%) compared to the much lower rates described in the adult literature, which range from 0.001% to 0.4%.3,4,6,7 These clinically fragile and younger patients may be more susceptible to UPHE, and the phenomenon may be observed earlier in children than in adults due to rapid contrast circulation in small patients. The finding was observed at doses higher than the weight-based manufacturer recommendation in two of the three patients, which may also play a role in its occurrence.
Unusual progressive hepatic echogenicity resembles the sonographic finding of portal venous gas, a finding associated with several clinical conditions, including bowel pneumatosis and inflammatory bowel disease. Several theories regarding the underlying mechanism of hepatic portal venous gas have been proposed: increased intraluminal gas production by colonic bacteria (i.e., abscess, intestinal dysbiosis); increased intraluminal pressure (i.e., secondary to obstruction); compromised intestinal mucosal barrier (i.e. inflammatory bowel disease, graft vs host disease); and gaseous dissection through the mediastinum and across the gastric wall (i.e., pulmonary disease).3–6
Similarly, proposed mechanisms for UPHE include intravasation of intestinal luminal gas resulting in enterohepatic circulation. It has also been proposed that microbubble expansion or aggregation related to the CEUS procedure itself may lead to sinusoidal retention or microvascular occlusion.3,4,6,9
Only a low percentage of phagocytosis by hepatic Kupffer cells has been demonstrated with sulfur hexafluoride UCA relative to other UCAs, 10 making the latter mechanism unlikely. Intravascular bubbles with diameter >50 μm, much larger than the average UCA microbubble size (1.5–2.5 μm), have been observed in the cecum following UCA administration based on animal studies. 11 In addition, because the UPHE phenomenon persists far beyond the expected circulating lifespan of UCAs and this phenomenon is not eliminated by the high MI insonation that would typically rupture sonographic microbubble contrast agents, it is likely that the portal venous gas seen with UPHE is enteric in origin. Therefore, this phenomenon is not related to prolonged microbubble circulation.
This theory is further supported by reports of sonographic visualization of gas bubbles in the portal vein and superior mesenteric vein, but not the Inferior vena cava (IVC), hepatic veins, splenic vein, or aorta.4,6 The phenomenon in the present cases may reflect some combination of sinusoidal pooling and intestinal microcirculation of gas emboli to the liver, given both the early appearance of heterogeneous liver enhancement and its persistence over time.
Bone marrow transplant, TPN, NEC, and gastroschisis, as in this case series, are well-established risk factors for impaired intestinal mucosal integrity and function.2,12 Portal venous gas has also been described in severe pancreatitis associated with bowel necrosis. However, the UPHE seen in the current patient with pancreatitis (patient case 3) is more likely explained by poor intestinal motility related to his neurological status.
Conclusion
This patient case series extends the hypothesis that underlying gut disturbances are risk factors for transient portal venous gas as UPHE following CEUS. More importantly, perhaps, these cases raise the question of whether there is a particular feature of the intestinal milieu of children that predisposes them to a pathophysiologic process underlying UPHE. Studies have shown differences in the intestinal microbiota of infants, children, and adults, though the clinical implications are unclear. 13 Of the cases mentioned, each patient shares a Hispanic, Latino, or Spanish origin. It is unclear whether this finding is due to small sample size, patient population, or has clinical implications. Differences in race and ethnicity, possibly attributable to differences in diet and other cultural factors, may be linked to variation in the gut microbiome.14–17 This possible risk factor warrants further attention in future studies.
Furthermore, UPHE may potentially be a novel finding associated with hepatic microvascular disease including SOS/VOD, given the observation of this finding in one patient who eventually died from SOS/VOD (patient case 1). In this patient there may have been early UCA microbubble accumulation in damaged sinusoids or extravasation into the space of Disse in the context of underlying sinusoidal endothelial cell damage, 18 with subsequent UPHE due to intestinal gas supersaturation and further expansion and trapping of gas in the sinusoids or space of Disse. This mechanism does not explain early appearance of the findings in the third patient case, however. No adverse effects have been reported in the presence of UPHE/PHLE,4–6 with the exception of the case of a 28-year-old woman who experienced “general discomfort and a transient skin rash.” 8 The patients presented suffered no observable clinical changes from the CEUS examination, although it is important to exercise caution in neonates and unstable patients who may be more susceptible to microvascular effects. Animal studies have demonstrated various bioeffects of UCAs, albeit at up to 50 times clinical dosing and with high MI insonation. These bioeffects include cell membrane and endothelial damage, microvessel rupture, inflammation, necrosis and ulceration in the cecum and proximal colon, and midzonal hepatocyte coagulative necrosis in the liver.9,11,19 A proposed mechanism for these lesions is gas supersaturation in the cecum and microbubble expansion causing microvascular ischemia and necrosis in the presence of CO2, similar to the intestinal gas bubble expansion hypothesis discussed above.9,11
Interestingly, the liver and cecal lesions were not observed in mice fed a diet containing no cellulose or starch, and the lesion incidence was reduced on a diet low in cellulose and starch compared with a standard diet containing cellulose and starch. 11 This may implicate some role of the intestinal microbiome environment and gas production in lesion formation and, by extension, potentially in UPHE. 11 Additional data demonstrate the absence of intestinal and hepatic lesions in mice maintained on a fiber-free diet as well as in germ-free mice 11 further supporting the potential role of the gut microbiome and gas formation in intestinal and liver lesions and UPHE. This hypothesis may be supported clinically by the case of UPHE/PHLE in a lactose-intolerant patient reported by Tana et al. 8 That report also proposed some role of altered carbohydrate metabolism in UPHE/PHLE. 8 It may be that the finding of UPHE in the second patient case provided, which occurred only after the application of high MI flash mode and color Doppler, resulted from microbubble inertial cavitation somehow precipitating additional bubble aggregation.
The liver and colonic lesions described in animal models have not been histopathologically observed in humans. Furthermore, these bioeffects may be reversible and are of unknown clinical significance. The safety of UCAs in clinical use has been demonstrated for decades, and the American Institute of Ultrasound in Medicine (AIUM), European Federation of Societies for Ultrasound in Medicine and Biology (EFSUMB), and the World Federation for Ultrasound in Medicine and Biology (WFUMB) in collaboration with others have addressed the safe use of UCAs in order to minimize the potential for aforementioned bioeffects. 20 Their recommendations include using a low MI (below 0.4) and minimizing exposure times. 11 The results of the present study would reiterate the importance of low MI contrast mode imaging in concordance with above recommendations.
It does, however, raise the question of whether children are at greater risk for UPHE in general, as was reported as a 20-fold increase in prevalence in this pediatric cohort compared to that reported in the adult literature, which may be related to unique features of the pediatric gut itself.
Further elucidation of the mechanism and clinical significance of UPHE will require case-controlled studies comparing UPHE in children with underlying medical conditions versus healthy controls. Particular attention to patients’ feeding status, use of antibiotics, and history of NEC or inflammatory bowel disease may warrant consideration in imaging practice, such as the use of low MI insonation and attention to contrast agent dosing.
Footnotes
Acknowledgements
The authors thank Dorothy Bulas, MD, FACR, FAIUM, FSRU, for review, editing, and mentorship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was exempt from approval by the institution’s review board due to the retrospective nature of this study of three or fewer patients. The information obtained by the investigators in such a manner that the identity of the Human Subjects cannot readily be ascertained, directly or indirectly, through identifiers linked to the subjects.
Informed Consent
Informed consent was not sought for the present study because all case data was de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
