Abstract
A list of gynecologic diagnoses is provided, which shows the use of sonography in significantly assisting the practice of modern clinical gynecology. The clinical value of this technology to assist in formulating diagnoses for the presenting patients is discussed. Sonography has become a necessary tool for today’s practice of gynecology, and women’s health, in general.
While there may be many medical specialties to which sonography has significantly contributed, gynecology (GYN) has particularly benefited from the clinical tools that have been offered, over the past few decades. It should be noted that the clinical role of sonography, and its related clinical applications, has dramatically expanded within the practice of women’s health care over recent times. This includes the use of transvaginal sonography (TVS) with Doppler vascular imaging, three-dimensional enhancement, and the use of contrast infusions (e.g., sonohysterography). The outpatient practice of GYN can include a variety of presentations for which the ultimate diagnosis can best be made with the use of sonography. Many examples can be found in gynecology and are described below.
In gynecologic practice, there are a host of presentations warranting the use of sonographic assessment. This includes verification of an intrauterine gestational sac when considering a possible ectopic pregnancy (EP). This may even be needed multiple times, until the so-called discriminatory zone is reached—the range of human chorionic gonadotropin in which an intrauterine gestational sac could generally be identified. 1 The sonogram provided (see Figure 1) reveals the sonographic ability to detect an unusual type of EP, an intramural variant, which may be difficult to identify without the use of sonography.
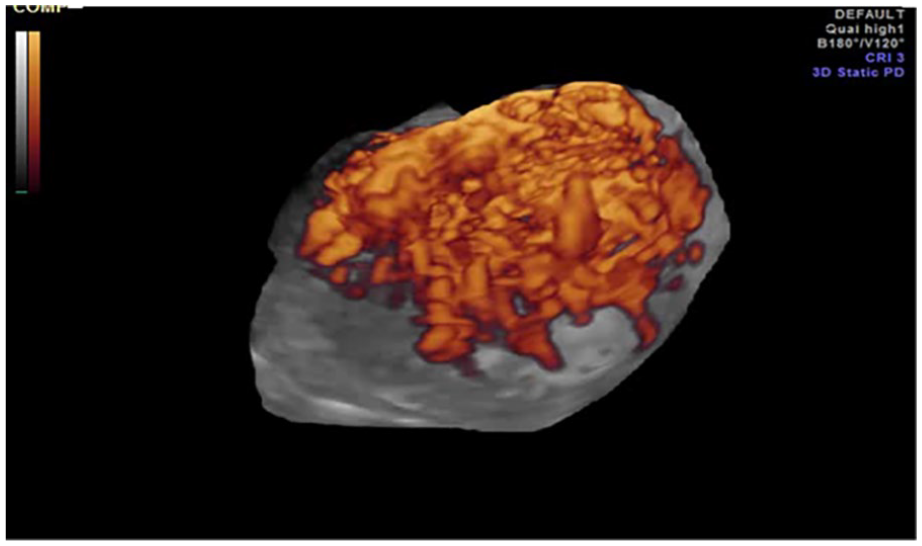
A three-dimensional glass body–rendered image of an intramural ectopic pregnancy, which reveals a hyperechogenic/heterogeneous mass, within the uterine wall, with abundant vascularity.
When considering the possibility of pelvic inflammatory disease (PID), sonographic visualization of the pelvis may be helpful for its diagnosis, in lieu of always performing laparoscopy for confirming its presence. The identification of a possible associated tubo-ovarian abscess (TOA) may be useful to direct the most appropriate treatment of a patient who is presenting with this condition. Recognizing the sonographic presence of hydrosalpinx can usually confirm the condition of PID, which causes its painful associated symptoms (see Figure 2). 2
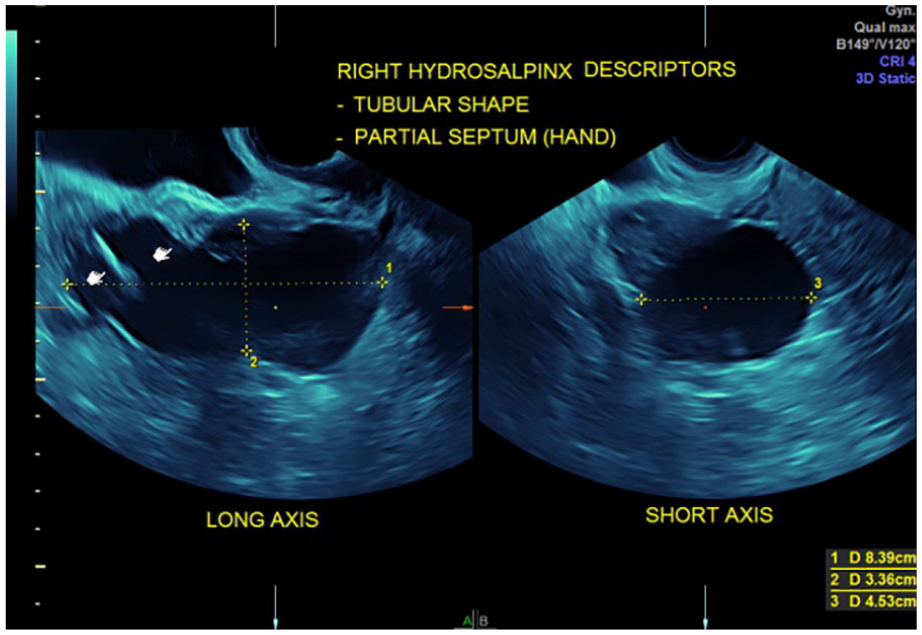
A case of right hydrosalpinx in a patient with right-sided pelvic pain. The transvaginal sonogram demonstrated a 8.4 × 3.4 × 4.5 cm right hydrosalpinx. A laparoscopic salpingectomy was performed, and the pathology was confirmed.
If an ovarian enlargement is detected by a bimanual physical examination or if a patient’s symptoms might suggest the presence of a functional or neoplastic ovarian mass, sonography can be helpful in identifying ovarian cysts, solid tumors, and/or ovarian malignancies, especially when combining the visualization with various serum-detected biomarkers and using the algorithms described by the International Ovarian Tumor Assessment (IOTA) group for comparison with sonographically obtained images. 3 Now, there is even the availability of computer applications that can be downloaded to one’s device to analyze the identified sonographic features for assessing the risk of malignancy for the imaged adnexal structure. One example of this is ACR Guidance®, which is an application created by the American College of Radiology that can be downloaded to android and iOS phones.
Endometriosis is an especially painful condition that can affect women, for which numerous treatments are available once a diagnosis is made. Identification of endometriomas with two-dimensional TVS (2D TVS) has been well established, although the “gold standard” that has been used for years is laparoscopy. However, much evidence has been gained recently emphasizing that the sonographic detection can be entirely relied upon. 4 In fact, a subtype has been described, deep infiltrating endometriosis (DIE), which cannot always be identified laparoscopically via the visualization of the sometimes-seen vascular markings on the peritoneal surface. The use of three-dimensional (3D) sonography, however, can sometimes reveal the presence of DIE. 5 This has been repeatedly demonstrated in the medical literature.6,7 The sonogram provided (see Figure 3) demonstrates an example of this pathology.
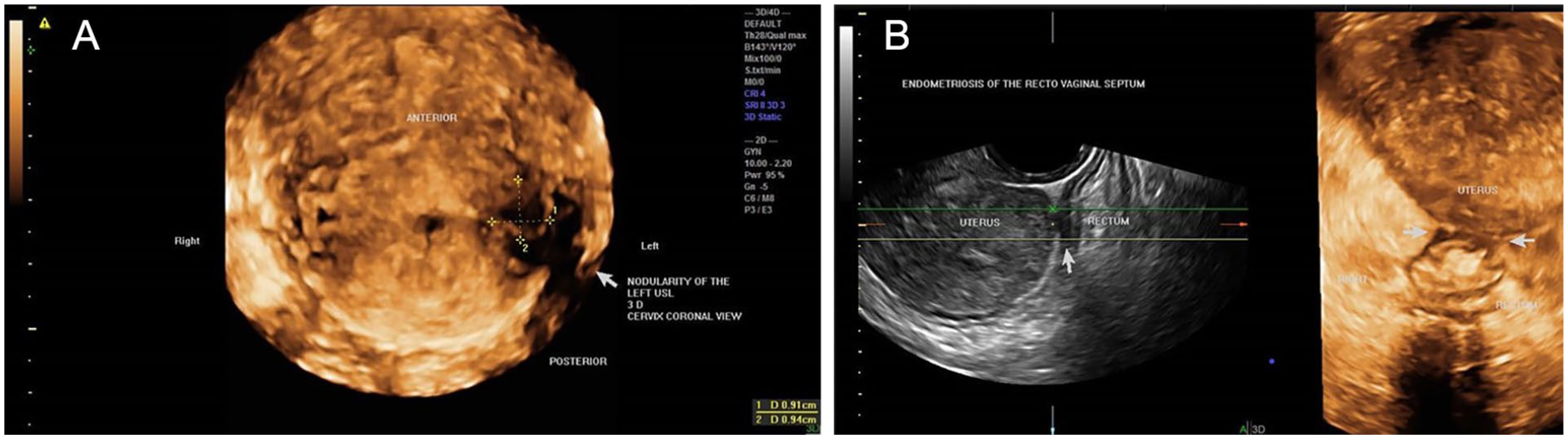
(A) A case of deep infiltrating endometriosis in a woman with pelvic pain and dysmenorrhea. (B) The cervix is displaced to the left with tender nodularity of the utero-sacral ligament and rectovaginal septumx.
Uterine leiomyomas (fibroids) are yet another problem that can cause multiple disturbing symptoms such as menorrhagia and abdominal/pelvic pain, for which women commonly present to the gynecologist. Sonography is often used for fibroid mapping and sizing, to be able to follow the patient during medical treatment, although surgical therapy may be ultimately necessary. 8 A sonogram (see Figure 4) that reveals an example of such clinically useful sonographic imaging is provided. It should be mentioned that one of the available surgical treatments that can be offered for uterine fibroids (Acessa™) uses laparoscopically directed ultrasound during the application of radiofrequency volumetric thermal ablation to the visualized fibroid. 9 Sonographic imaging had been previously compared with computed tomography (CT) and magnetic resonance imaging (MRI) for the identification of fibroids, but such comparisons have simply resulted in the substantial use of sonography with or without saline infusion sonohysterography (SIS) for this purpose in current clinical practice. 10
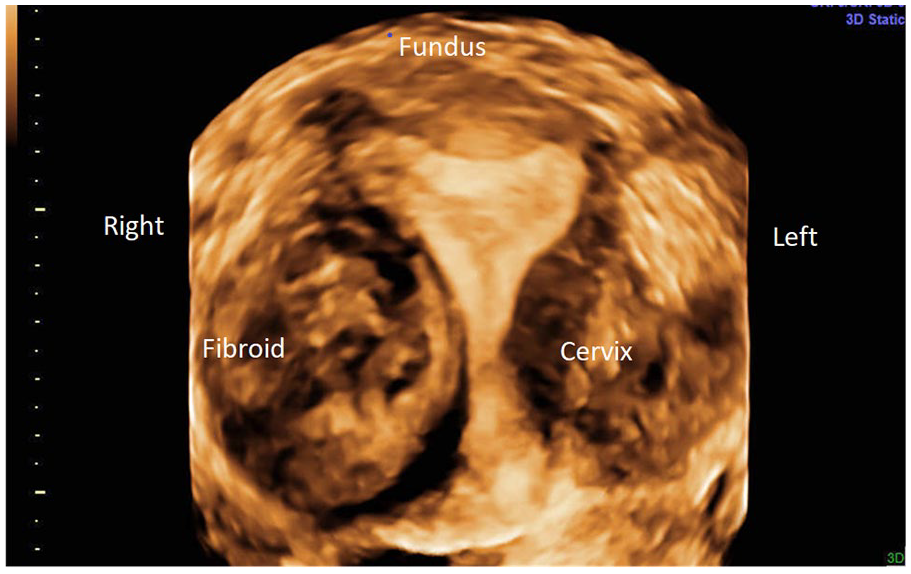
A 34-year-old G1 P1001 patient presented with menorrhagia and dysmenorrhea. A three-dimensional sonographic coronal view of the uterus, in rendering mode, demonstrated a right/lateral grade 2–5 submucosal fibroid. The fibroid’s position resulted in minimal left bowing of the endometrial echo and protrusion through the right uterine serosa.
During menopause and the peri-menopausal timeframe, abnormal bleeding may prompt sonographic imaging to consider the possibility that it may be caused by endometrial carcinoma (EC) or any related problem of endometrial hyperplasia. In considering this, it is common to sonographically measure the endometrial thickness, which may indicate the need for histologic sampling (e.g., with an endometrial biopsy or dilatation and curettage [D&C]). 11 Again, sonography is clinically used to direct its appropriate management once the diagnosis is made with the help of this imaging. Moreover, sonography has not only been used for the detection of EC and the continuum of associated endometrial neoplastic disease, but the use of 3D sonography has also been demonstrated to be useful for the staging of EC, which has been helpful in determining the optimal surgical treatment for EC, whether to possibly include a pelvic lymphadenectomy along with hysterectomy prescribed.12–14 An example sonogram is provided (see Figure 5).
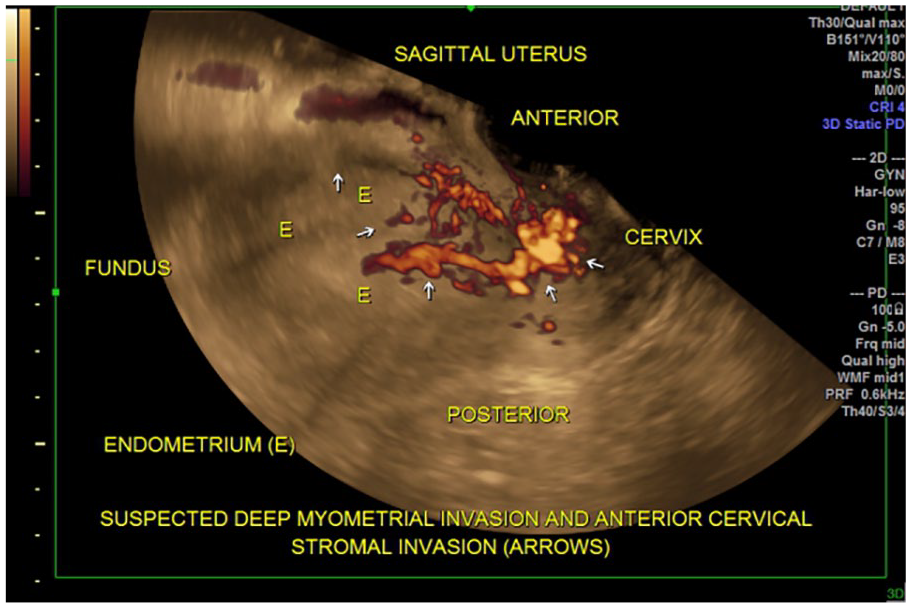
A 27-year-old G0 patient was imaged, with the three-dimensional glass body rendering image demonstrating endometrial cancer, with deep myometrial invasion, in the anterior uterine wall and anterior cervical stroma. The patient underwent a Da Vinci robotic–assisted hysterectomy. The final pathology result showed well-differentiated endometrioid adenocarcinoma International Federation of Gynecology and Obstetrics (FIGO) grade 1, with deep myometrial invasion and cervical stromal invasion.
Additional gynecologic sonographic applications include the detection of the presence or location of intrauterine device (IUD), as may be needed for the management of some IUD symptomatic complications (e.g., perforation [or migration as it is sometimes called], embedment, or malposition), as these problems are sometimes known to occur. 15 Representative sonograms demonstrate the use of IUD imaging (see Figures 6 and 7). Sonography may also be diagnostically useful for other gynecologic conditions that can present to the gynecologist, such as for identifying intrauterine polyps, sometimes presenting with abnormal uterine bleeding (AUB), and then using sonohysterography for their delineation. 16 The sonogram provided (see Figure 8) demonstrates this identification of a polyp, which can be the source of abnormal bleeding or malignancy.
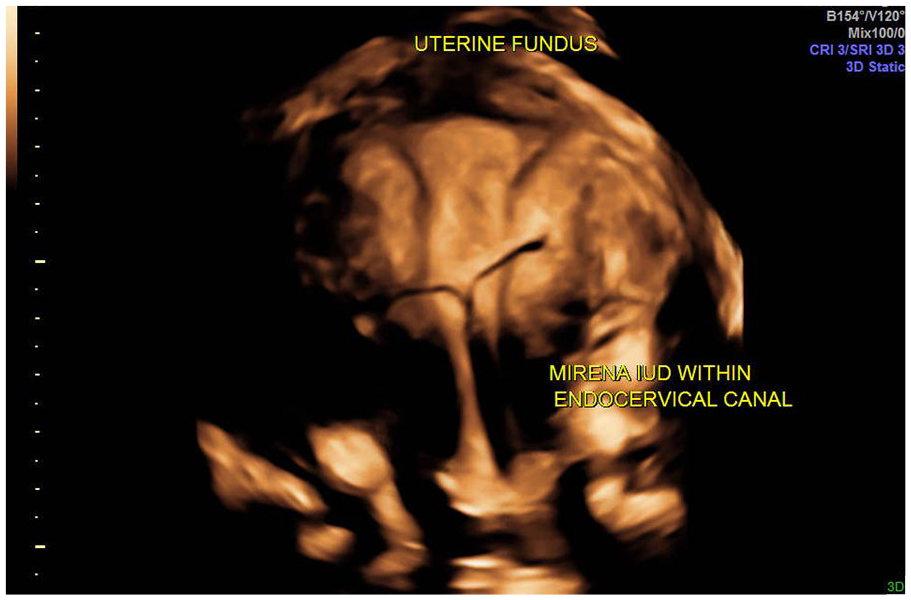
A Mirena intrauterine device is visualized in the endocervical canal, rather than in the expected intrauterine placement.
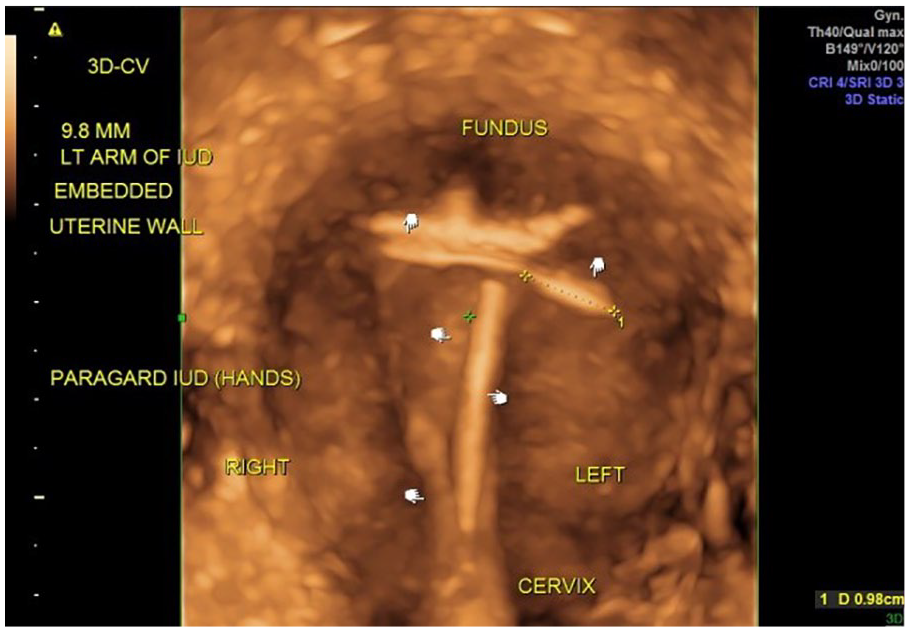
A 27-year-old G0 female patient presented complaining of noncyclic left lower quadrant pain, dyspareunia, dysmenorrhea, and increased menstrual flow for several months. A Paragard intrauterine device placement (IUD) had occurred 2.5 years previously. The three-dimensional transvaginal sonogram of uterus, with sono-hysterogram, demonstrated a malpositioned Paragard IUD, with 9.8 mm left arm embedment.
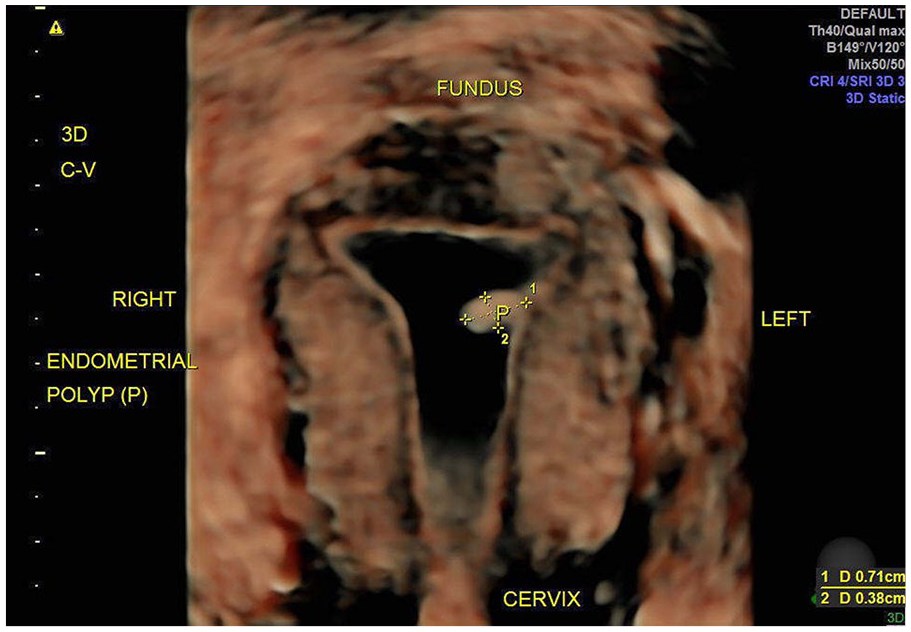
A saline infusion sonohysterography was completed and demonstrated a very small endometrial polyp, which was not visualized on the transvaginal sonogram alone (virtual hysteroscopy).
Polycystic ovarian syndrome (PCOS) is another condition for which sonography can be diagnostically helpful in addition to the various endocrinologic markers that can be used. 17 Another sonographic application in gynecology includes fertility workups (e.g., checking for tubal patency). While hysterosalpingogram, with its use of ionizing radiation, has long been used for this, such practice is changing to the use of SIS.18,19
Transvaginal sonography has often been applied in the Radiology and Emergency Department (ED) settings and at the bedside in office, which can be described as at point-of-care (POC). 20 The focused assessment with sonography in trauma (FAST) has been used in the ED setting to efficiently make a consequential appropriate diagnosis after traumatic injury. This standardized approach for using ultrasound to detect conditions for which surgical intervention is indicated may particularly apply to the gynecologic diagnoses of EP and post-hysterectomy complications. Ultrasound use in gynecology at the POC clearly provides a technological extension of the specialist’s examining hands, enabling an enhanced diagnosing ability. This can excellently identify the position, the dimensions, the sonographic qualities, and the vascularity of targeted organs of the female pelvis. In this way, ultrasound significantly augments the standard clinical practice of obstetrics and gynecology.
While the ease and practicality of sonographic diagnosis in the office setting are notable, so can the similarity in effectiveness with other available imaging modalities (e.g., CT and MRI) which are known to be more costly and often require performance at a different location from the office setting. Sonography may offer a unique benefit (and accuracy) for each of the above-named diagnostic entities when compared with alternative imaging, and this comparison may be important, relevant, and reveal a meaningful best imaging choice.
Conclusion
As has been described, 2D and 3D sonography, with power Doppler angiography, has become an essential part of the everyday clinical practice of GYN, which should be recognized by all providers, patients, and institutions.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
