Abstract
Objective:
The aim of this study was to examine whether high-resolution ultrasonography (HRUS) can provide information, on signs of inflammation and destruction, due to rheumatoid arthritis (RA). The focus was on the wrist joint, during early and late disease and compared the findings to those with magnetic resonance imaging (MRI).
Materials and Methods:
The study was based on 31 RA patients, with joint involvements, who were selected from those attending the outpatient rheumatology clinic. The patients completed both HRUS and MRI examinations, and their results were compared.
Results:
The HRUS and MRI images had near-perfect agreement, for detecting joint anomalies, in those RA patients. While comparing the overall performance of the HRUS and MRI studies, it was observed that HRUS was equal or nearly equal, demonstrating 90.83%, 100.0% and 94.93%, diagnostic sensitivity, specificity, and accuracy, respectively, for diagnosing RA pathologies.
Conclusion:
The early diagnosis of RA with HRUS and MRI examinations is very important, in the early treatment of RA. The HRUS examinations demonstrated a sensitive and reliable diagnostic method for assessing inflammatory activity and destructive changes in the joints of these RA patients, with HRUS findings comparable to those of MRI.
Rheumatoid arthritis (RA) is a chronic autoimmune systemic disease that can cause inflammatory changes and it may affect many tissues and organs, but predominantly involves the synovial joints. The cause of the disease is unspecified and frequently affects 1%–2% of world population.1,2
The hallmark of the disease process often leads to the destruction of synovium, articular cartilage, bony erosions, and ankylosis of the affected joint. Early disease activity in RA significantly affects articular and periarticular soft tissues and if left untreated leads to disorganization and deterioration of the joints.3,4 Even though RA is not a fatal disease, but comorbidities, such as cardiac and respiratory along with RA can lead to transience. 1
Early diagnosis of RA is important because early aggressive treatment can reduce long term disability. For early detection of RA conventional radiography, clinical examination, and laboratory tests have limited usefulness. 5
As modern diagnostic imaging tools, such as ultrasonography (USG) and magnetic resonance imaging (MRI) become increasingly accessible, rheumatologists have access to highly sensitive measures to evaluate both the inflammatory and structural damage components underlying various forms of arthritis, over the disease cycle. 6 The gold standard for the assessment of soft tissue abnormalities is MRI. However, in the last few years, USG has become a more popular tool among rheumatologists and radiologists, as the first radiologic investigation, to assess rheumatoid disease, as well as diagnostic follow-up. This has been possible due to the increased availability of USG equipment systems, transducers, and software. Both USG and MRI have associated strengths and weaknesses, and at times they can provide complementary diagnostic information.6–8
Thus, the aim of this present study was to explore the possibility to establish high-resolution ultrasonography (HRUS), as an initial diagnostic tool for the detection and characterization of pathological features, in the joints of RA patients.
Materials and Methods
Study Design
This hospital based descriptive study was carried out in the Department of Radio-diagnosis of the host institute, during January 2020–June 2021. Institutional ethical clearance was obtained and all patients provided their written consent. The aims and objectives for the study were to compare HRUS with MRI in the evaluation of joint abnormalities, for newly diagnosed cases of RA. In addition, it was important to establish the imaging techniques’ role in the early detection of disease. It was also important to compare HRUS with MRI, in evaluating those patient cases with RA, already in treatment.
Subjects
Those consecutive patients, with RA, who attended the outpatient rheumatology clinic, gave their informed consent, as part of the recruitment. Thirty-one patients fulfilling American College of Rheumatology (ACR) 2010 criteria of RA, were selected. For those patients for whom MRI was contraindicated, due to implanted medical devices, uncooperative patients, coexisting joint disease, were excluded from the study.
HRUS and MRI Diagnostic Assessment
HRUS was performed using a Siemens Acuson S2000&3000 and a Siemens Acuson Juniper ultrasound equipment system (Siemens Healthineers, Erlangen, Germany), which included the use of a linear “hockey stick” transducer operating at 6–18 MHz.
All patients were examined with a Siemens 1.5 Tesla MRI Magnetom Avanto Tim and Dot system (Siemens Healthineers, Erlangen, Germany). The usual slice thickness required for this examination is 3 mm, with interslice gap of 0.3 mm, however, thinner slice sections should be taken while examining smaller joints. This is especially true when attempting to image the hands and wrist joints. For the diagnostic assessment of larger joints, such as the ankle, hip, knee, shoulder, a slice thickness of 3 mm can be used. The MRI protocol was as follows:
T1-weighted images give accurate assessment of anatomical details and detects inflamed synovium.
T2-weighted sequences detect both fat and fluid.
Short-TI inversion recovery (STIR) is a fat suppression technique and is sensitive for detecting fluid.
PD is most ideal sequence for detecting joint pathologies. This was important in making a clear cut demarcation between the fibrocartilage, hyaline cartilage, and fluid.
The patients’ images were acquired in the following sequences for joint analysis:
PD: axial, coronal and sagittal
T1: axial
T2: sagittal
STIR: coronal MRI assessment
Later in the day, when the HRUS was performed, MRI examination was completed (Siemens 1.5 Tesla MRI Magnetom Avanto Tim and Dot system (Siemens Healthineers, Erlangen, Germany)) of both hands. Patients were placed in a prone position, with their hands placed above the head. Sand bags were used to avoid any involuntary patient movement. The scanning was completed using a conventional T1-weighted sequences, SE images of repetition time (TR) 600 m-seconds, echo time (TE) 15 m-seconds. T2-weighted sequences, TSE: TR = 2000–4000 m-seconds, TE = 90–130 m-seconds. Conventional STIR sequences were also used. Fluid and pathologic tissues have high signal intensity in comparison to suppressed marrow and fat signal. To increase image quality it required a small field of view (FOV), in this case, a 16–18 cm FOV was used for the examinations of bilateral hands. The coronal, sagittal, and axial views in T2, and fat suppressed STIR-weighted spin-echo magnetic resonance sequences were performed. Continuous axial and coronal pre-Gadolinium (Gd) and post-Gadolinium T1-weighted spin-echo were also used.
Statistical Analysis
At the end of the study, after acquisition of the images, the interpretation and diagnostic findings were recorded on a predefined pro forma. The acquired data were entered into a Microsoft Excel spread sheet and subjected to statistical analysis. Considering MRI as the diagnostic gold standard, the sensitivity, specificity, positive predictive value, and negative predictive value of HRUS were calculated. The main aim was to determine the presence or absence of pathology without quantifying its severity, therefore, scoring based on the OMERACT RA imaging studies system was not performed.
Results
The age of the study cohort ranged from 18 to 74 years, with mean age ± SD of 43.48 ± 15.57 years. The study cohort showed that 64% of the patients were female (20 patients), whereas 35.5% were male (11 patients). The wrist joint (54.8%) was found as a most affected joint, among this cohort, while others were found in the following order: knee (32.3%), ankle (9.7%), and shoulder (3.2%) (see Figure 1). Among the age distribution of this study cohort, the maximum number of patients belonged to the age group of > 61 years (19.4%). Figures 2–8 help to illustrate the findings of synovial effusion, synovial proliferation, cartilage changes, erosions, tenosynovitis, and bursitis.
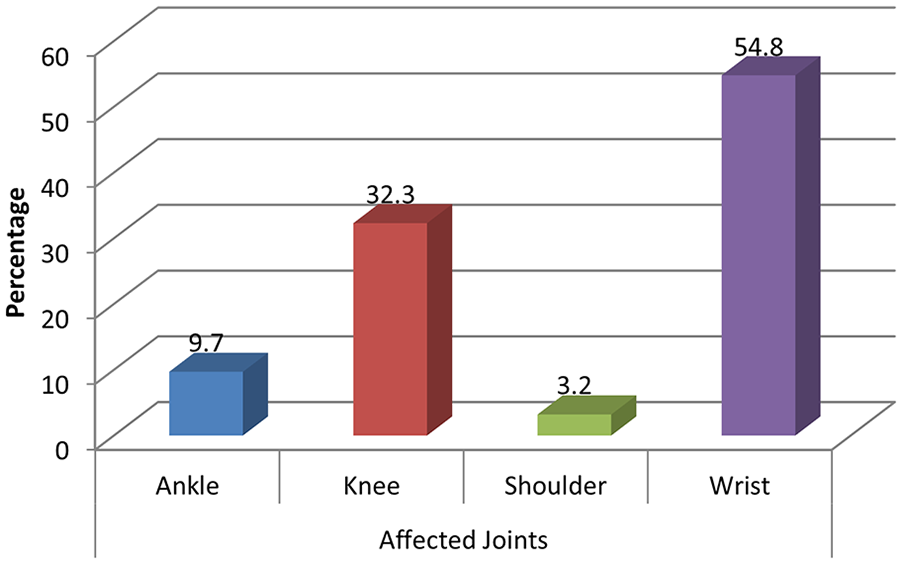
The distribution of affected joints, among the study cohort.
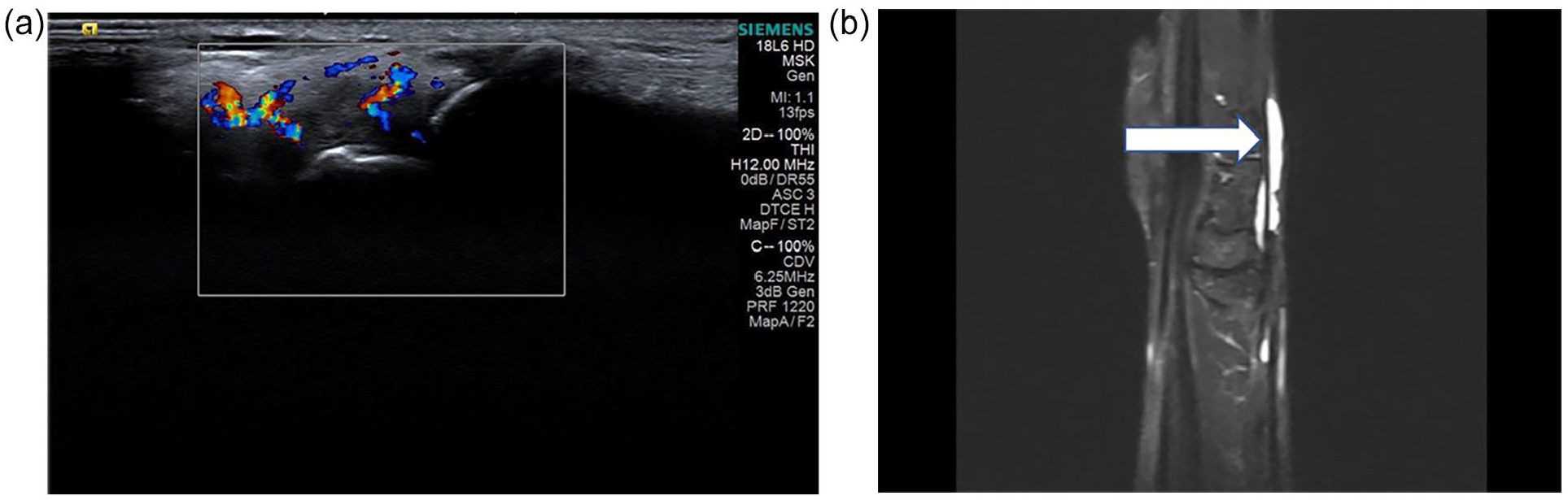
(a) A high-resolution ultrasonography (HRUS) sagittal images showing increased vascularity along tenosynovium (arrow). (b) A short-TI inversion recovery magnetic resonance imaging (STIR MRI) sagittal image showing fluid along the tendon sheath.
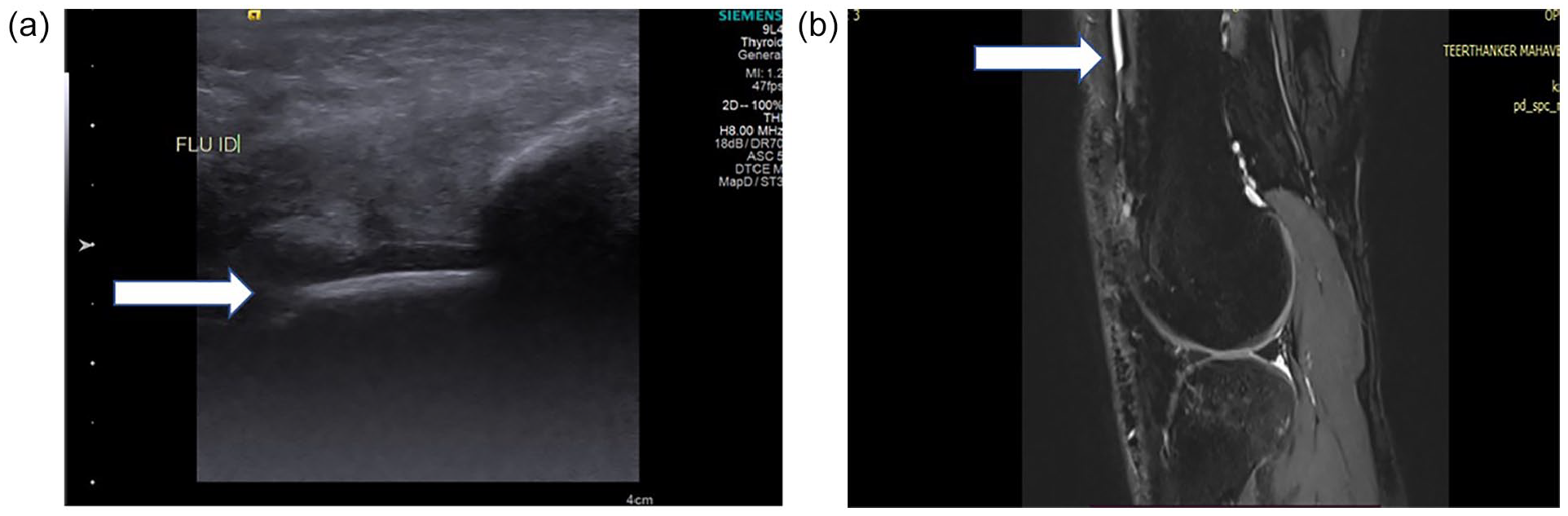
(a) A high-resolution ultrasonography (HRUS) demonstrating fluid that is noted along the quadriceps tendon in a positive rheumatoid arthritis (RA) patient, (b) A Proton Density (PD) magnetic resonance imaging (MRI) coronal image that shows hyperintensity along quadriceps tendon of a positive RA patient suggestive of synovial effusion.
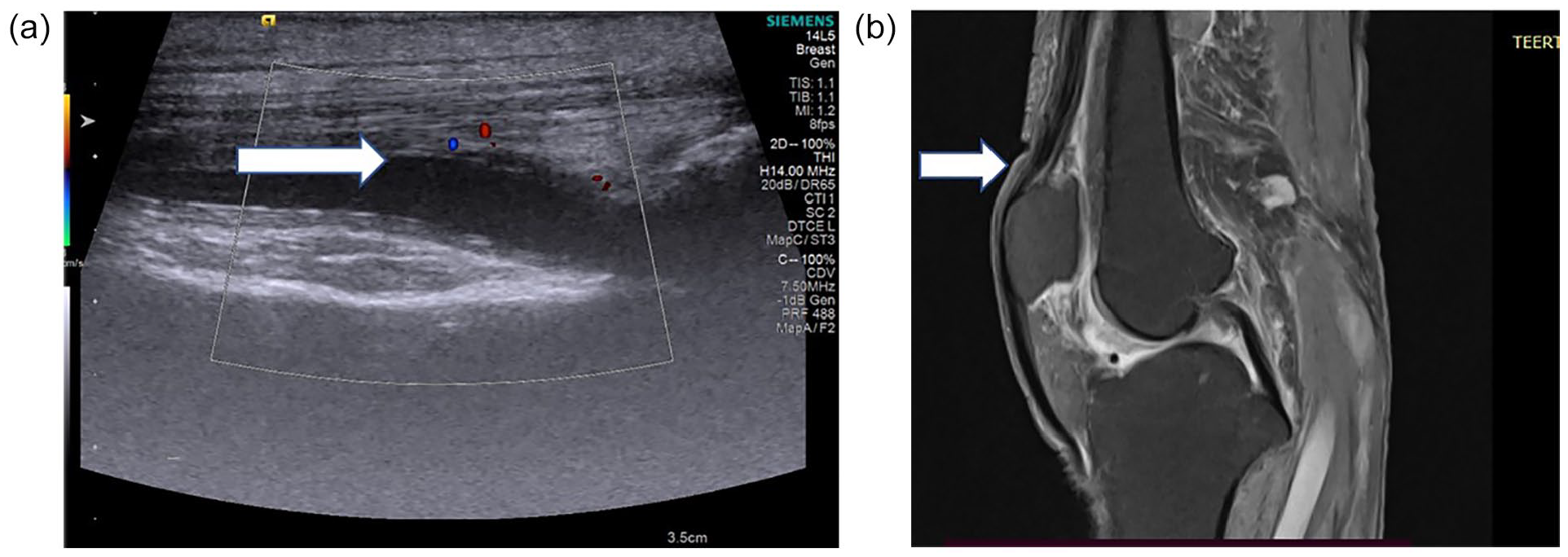
(a) A high-resolution ultrasonography (HRUS) showing inflammation of quadriceps tendon with internal vascularity in a positive rheumatoid arthritis (RA) patient suggestive of tenosynovitis and (b) A short-TI inversion recovery magnetic resonance imaging (STIR MRI) image showing PD hyperintensities are noted along the quadriceps tendon in a positive RA patient.
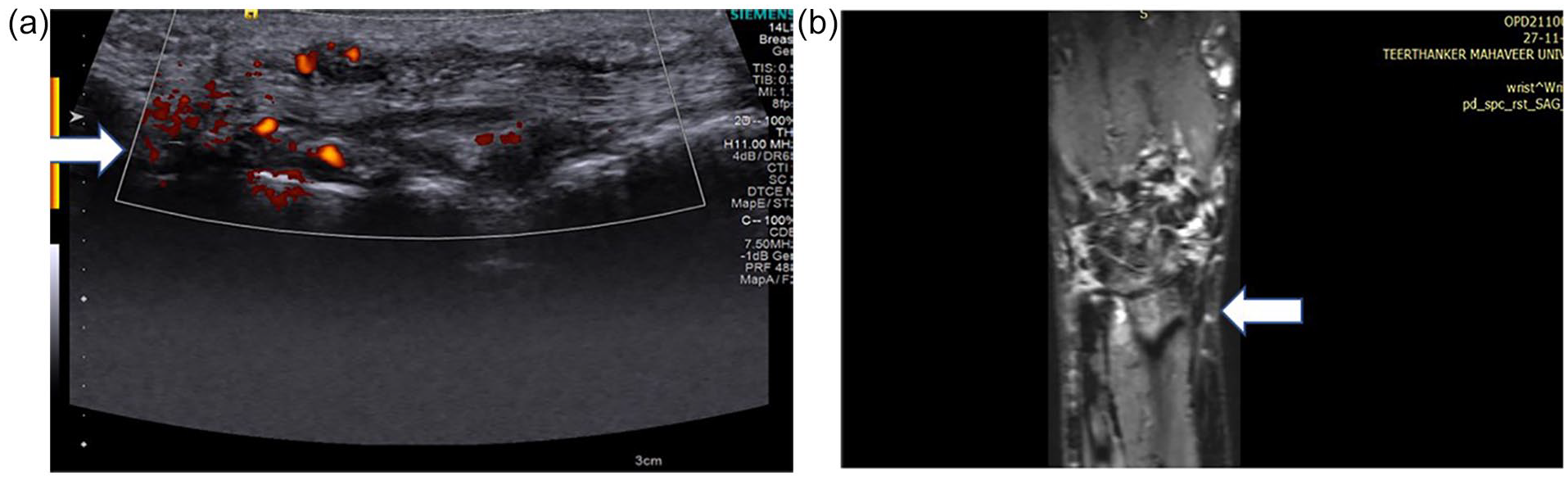
(a) A high-resolution ultrasonography (HRUS) showing increased vascularity along extensor carpi ulnaris tendon and thickening of tendon in a positive rheumatoid arthritis (RA) patient, suggestive of tenosynovitis and (b) A magnetic resonance imaging (MRI) PD coronal image showing hyperintensity along the extensor carpi ulnaris tendon in a positive rheumatoid arthritis (RA) patient, suggesting tenosynovitis.
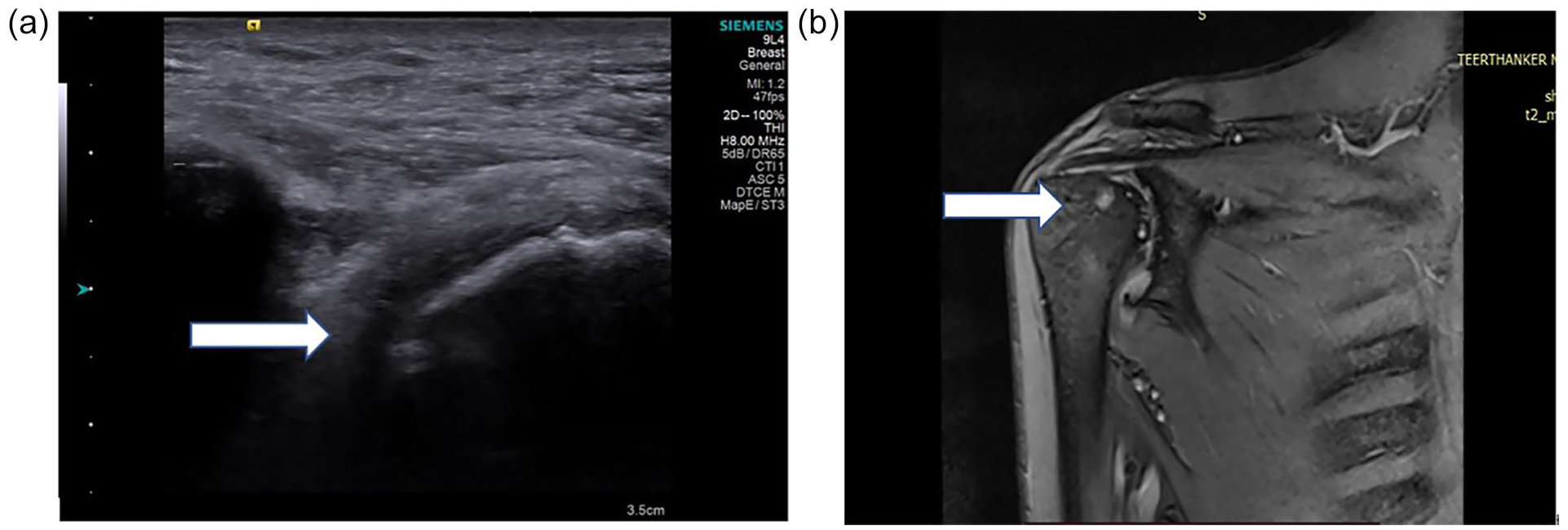
(a) A high-resolution ultrasonography (HRUS) showing cortical irregularity that is noted at head of the humerus, of a positive rheumatoid arthritis (RA) patient, suggestive of bony erosions. (b) A magnetic resonance imaging (MRI) T2 coronal image that illustrates a cortical irregularity at head of humerus in a positive RA patient, suggesting bony erosions and subcortical cysts are also noted.
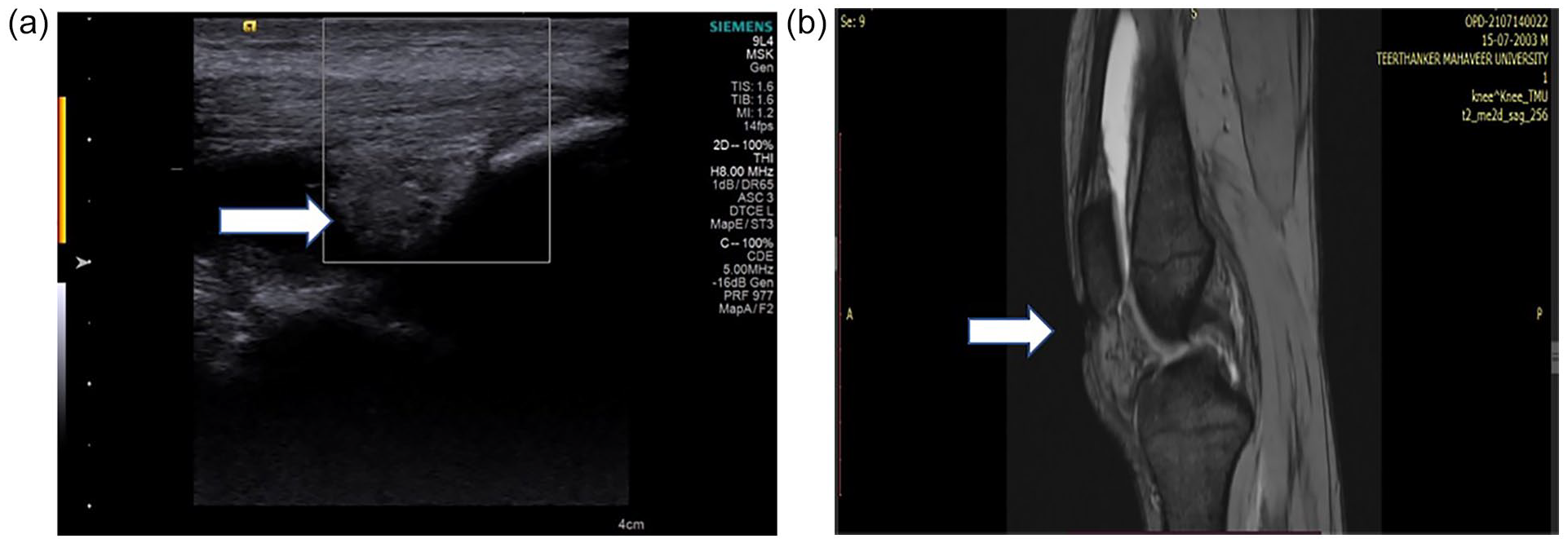
(a) A high-resolution ultrasonography (HRUS) image showing a mixed echogenicity area, with an increase vascularity in the knee joint of a positive rheumatoid arthritis (RA) patient, suggesting synovial proliferation. (b) A magnetic resonance imaging (MRI) T2 sagittal image showing synovial proliferation and T2 hyperintensity in the knee joint of a positive RA patient, suggesting joint effusion.
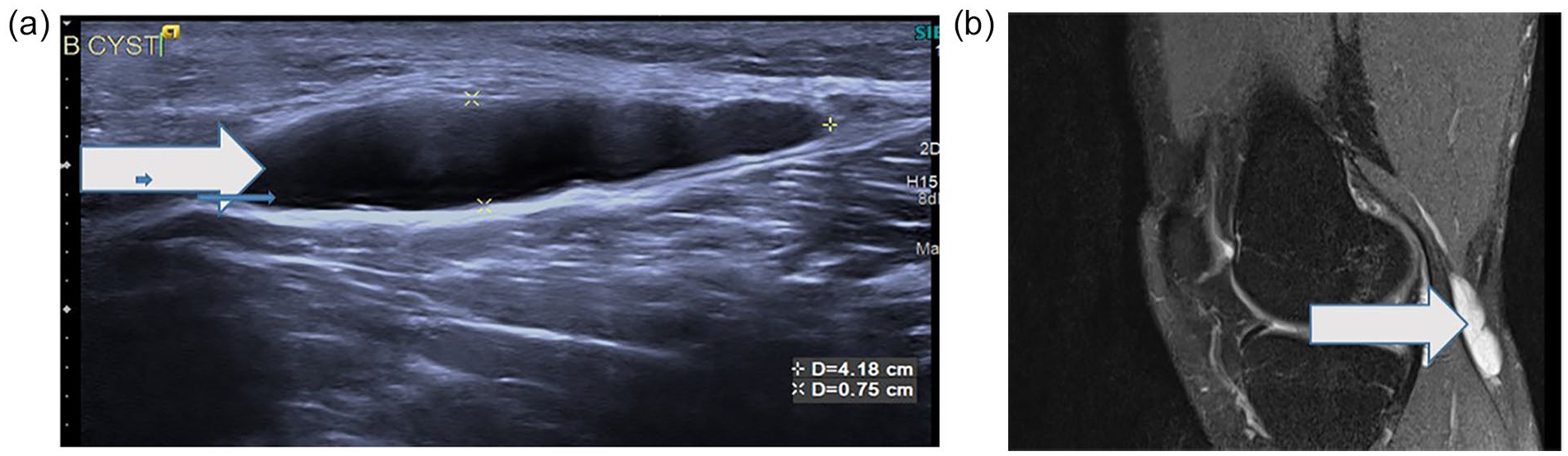
(a) A high-resolution ultrasonography (HRUS) sagittal image showing an anechoic cystic lesion in a positive rheumatoid arthritis (RA) patient, suggesting a Baker’s cyst. (b) A magnetic resonance imaging (MRI) PD fat sat sagittal image showing hyperintensity adjacent to lateral head of gastrocnemius, suggesting a Baker’s cyst.
Synovial effusion, synovial proliferation, erosions, and tenosynovitis were observed slightly higher (96.8%, 90.3%, 83.9%, and 93.5%) in MRI as compared to HRUS (93.5%, 87.1%, 77.4%, and 90.3%), respectively. The cartilage change was absent (100%) in all cases in HRUS, while in case of MRI, it was absent in 83.9% cases. The bursitis was also absent in 93.5% and 96.8% cases in MRI and HRUS, respectively. Thirty patients had synovial effusion on MRI, and sensitivity and specificity of HRUS in the identification of synovial effusion were 96.67% (95% CI, 83.33%–99.41%) and 100% (95% CI, 20.65%–100%), respectively, with positive and negative predictive values of 100% (95% CI, 88.3%, 100%) and 50% (95% CI, 9.453%, 90.55%), respectively.
Synovial proliferation were observed in 28 patients on MRI, and sensitivity and specificity of HRUS in the identification of synovial effusion were 96.43% (95% CI, 82.29%–99.37%) and 100% (95% CI, 43.85%–100%), respectively, with positive and negative predictive values of 100% (95% CI, 87.54%–100%) and 75% (95% CI, 30.06%–95.44%), respectively.
Similarly, erosions and tenosynovitis were observed in 26 and 29 patients, respectively, at MRI and sensitivity and specificity of HRUS in the identification of erosions were 92.31% (95% CI, 75.86%, 97.86%) and 100% (95% CI, 56.55%, 100%), while in case of tenosynovitis, these were 96.55% (95% CI, 82.82%, 99.39%) and 100% (95% CI, 34.24%–100%) with the positive and negative predictive values of 100% (95% CI, 86.2%–100%) and 71.43% (95% CI, 35.89%–91.78%), and 100% (95% CI, 87.94%–100%) 66.67% and (95% CI, 20.77%–93.85%), respectively.
Bursitis was observed in two patients on MRI and HRUS with a sensitivity and specificity for identification at 50% (95% CI, 9.453%–90.55%) and 100% (95% CI, 88.3%–100%) with the positive and negative predictive values of 100% (95% CI, 20.65%–100%) and 96.67% (95% CI, 83.33%–99.41%), respectively. Enthesitis-related arthritis was absent in all the patient cases, with both imaging methods (see Table 2).
Discussion
RA is a chronic autoimmune systemic inflammatory disease that mostly affects the articular joints. Globally, this disease commonly affects elderly people. The hallmark feature of the disease is demolition of cartilage and cortical irregularity, which leads to difficulty in doing routine work and permanent irreversible changes, in the joint. 9
The synovium of joints is particularly affected by RA. The symptoms of RA are similar to those of many other illnesses and conditions, making the diagnosis difficult. If RA is not treated, it causes joint damage and deformity and further chronic illness causes diseases including synovial enlargement and angiogenesis.10,11
Early diagnosis of RA is an important determinant factor for early treatment and improvement of the long term disease outcome. Moreover, rapid development of powerful and expensive therapeutic agents in the last decade can achieve favorable clinical outcomes, if an early intervention has been provided.12,13
Radiographs have been the diagnostic tool of choice in rheumatology for generations. However, in recent years, USG has become more popular as a diagnosing rheumatoid imaging modality, identifying subclinical inflammation and predicting joint deterioration development. Both USG and MRI can detect RA. In addition, each of these imaging modalities is capable of depicting anomalies.14,15
Thus, in concordant with literature, this study was focused on comparing the effectiveness of HRUS and MRI, in the early detection and extent of severity of RA. This prospective observational study was conducted with 31 patients (over the age of 18 years), diagnosed with clinically and biochemically with RA. This cohort was evaluated with both HRUS and MRI, and these imaging studies were conducted on the same day, followed by a radiologist’s interpretation, in a single anonymized manner.
Females appeared to be predominately affected in the study cohort and these findings were also reported by Singh et al., 16 Salama, 17 Malla et al., 18 Bao et al. 19 and El-Shereef et al. 20 when they compared HRUS to MRI, among RA patients. The mean age of the current study cohort was also found to be comparable with a study reported by Malla et al., 18 in which the ages varied from 19 to 65 years.
As many previous studies have considered MRI the gold standard diagnostic modality, there is much evidence in the literature documenting that inflammatory changes found on MRI can mirror other pathologic findings.6,21,22 The various parameters that were evaluated in the current study, comparing HRUS and MRI, included synovial effusion, synovial proliferation, cartilage changes, erosion, tenosynovitis, bursitis, and enthesitis-related arthritis.
The sensitivity, specificity, and diagnostic accuracy of HRUS for diagnosing synovial effusion and proliferation showed an approximate perfect agreement between HRUS and MRI
This matched with the results described by Szkudlarek et al. 22 who compared USG with MRI, as the reference modality. They suggested that the visualized inflammatory changes are similar using both imaging modalities. Furthermore, later, Szkudlarek et al. 23 revealed that the sensitivity, specificity, and accuracy of USG as compared to indications of inflammation as MRI as a reference were 70%, 78%, and 76%, respectively. The current study would strongly confirm their observation. The intermediate signal intensity of synovial proliferation can be easily depicted against the background of bright marrow signal and correlated with its appearance on USG.
The current study results revealed that 87.1% of patients showed effusion by HRUS which were lower numbered those seen by MRI (90.3%) (see Table 1). Similarly, Szkudlarek et al. 23 and Scheel et al. 24 reported that USG was sensitive for the detection of very small amounts of fluid accumulation and proved to be better than MRI. They explained that in contrast to MRI, USG is a dynamic imaging modality, which permits the evaluation of target joints during motion and under transducer compression. In addition, better resolution and higher magnification of joints with USG can efficiently increase its diagnostic sensitivity (see Figures 2 and 3).
The Comparison of HRUS and MRI to Detect Joint Abnormalities.

Abbreviations: HRUS, high-resolution ultrasonography; MRI, magnetic resonance imaging.
The number of cases diagnosed as tenosynovitis by HRUS (90.3%) was also comparable to that diagnosed by MRI (93.5%) with matched diagnostic sensitivity, specificity, and accuracy of HRUS, as confirmed with MRI (see Table 2). In another study reported by Hampole et al. 25 who compared USG and MRI, for wrist joint pain, they also found USG equally matched with MRI with 100% of sensitivity, specificity, and accuracy (see Figures 4 and 5).
The Diagnostic Performance of High Resolution Ultrasonography (HRUS) to Detect Abnormalities Compared to Magnetic Resonance Imaging (MRI).
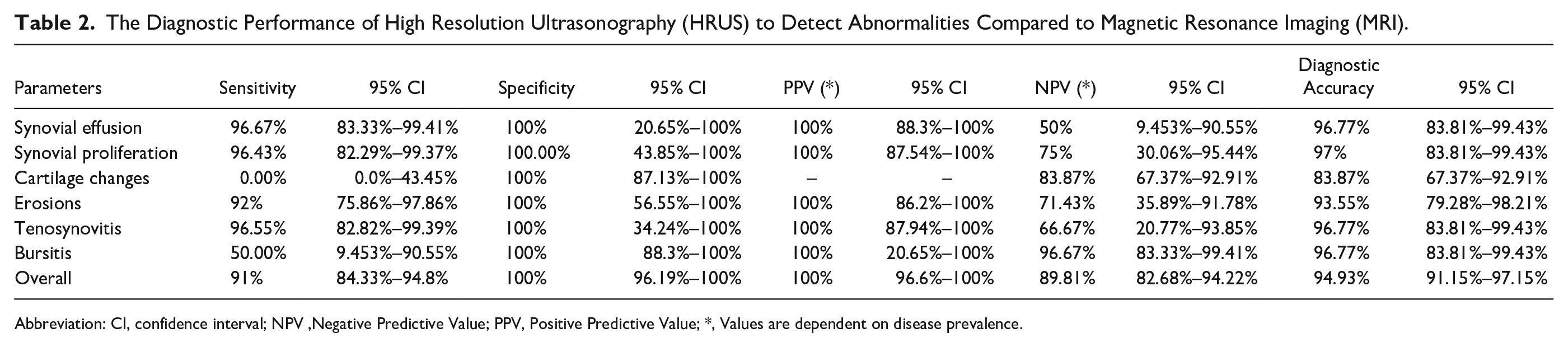
Abbreviation: CI, confidence interval; NPV, Negative Predictive Value; PPV, Positive Predictive Value; *, Values are dependent on disease prevalence.
Tenosynovitis is considered the hallmark of early tendon affection and in some cases with early RA, tenosynovitis predominates over joint synovitis. 26 It is crucial because tendon sheath synovitis and synovial proliferation can lead to tendon rupture owing to fraying of the tendon sheath along with eroding bone edges.
Cartilage changes were also compared among selected imaging modalities. In the current study cartilage changes occurred in 0.0% and 83.87% patients with HRUS and MRI, respectively. HRUS demonstrated a poor diagnostic sensitivity, specificity, and accuracy, compared to MRI (see Table 2). The current study findings can be approximately matched with Bedi and Bagga’s 8 statement that “the initial radiologic evidence is generally cartilage thinning, albeit in very early stages of arthritis,” USG may reveal thickening due to edema. Furthermore, nonvisualization of the cartilage is not necessarily a poor sign, because the cartilage may not always be visible depending on the location, but inconsistent reduction or enlargement of the cartilage is usually irregular.
In this investigation, erosion was found in 77.4% and 83.9% of patients, respectively, in HRUS and MRI, as well as diagnostic performance (i.e., sensitivity, specificity, and accuracy of HRUS) were matched more than 90% when compared to MRI (see Table 2). Although the number of patients in the trial who had this result was small, it was possible to demonstrate excellent diagnostic results between HRUS and MRI, for detecting erosions. This is comparable to the study undertaken by Hoving et al. 27 in all aspects (see Figure 7).
It is critical since tendon sheath synovitis and synovial proliferation can induce tendon rupture due to frayed tendon sheaths and eroding bone margins. Døhn et al. 28 also observed MRI and USG sensitivities of just 68% and 42%, in identifying erosions, respectively. Ignoring the fact that only 31 patients, with confirmed RA cases, were recruited for this study, the specificity for erosion was considerable and indicated a negative rate that had a close approximation to MRI.
Moreover McQueen and Østergaard 29 considered MRI the reference method for the assessment of erosions as USG results in their study also yielded sensitivity specificity and accuracy as 59%, 96%, and 86%, respectively. Thus, a good significant relationship was found in the current study between erosions by HRUS and MRI.
In addition, the bursitis parameter was compared between the two selected imaging methods (see Table 1). The current study findings were not inconsistent with the observation of Draghi et al. 30 who reported had 100% sensitivity and specificity in detecting deep infrapatellar bursa (see Table 2). The difference in sensitivity was observed in the current case might be due to only two positive case of bursitis in MRI, and further, it was noticed that the cases missed by HRUS showed very minimal bursal effusion (see Figure 8).
Enthesitis-related arthritis has predominantly been diagnosed by pathologic evaluation, based on the appearance of tenderness and edema. It is profoundly clear, nevertheless, that enthesial intervention in spondyloarthropathy may be subclinical. 31 In the identification of peripheral enthesitis, USG is superior to clinical evaluation. 32 This has also been already proven enthesitis is commonly seen in children, and the average age for diagnosis of enthesitis-related arthritis is about 10–15 years.33,34 This finding well matched with the current study results that did not find any case with HRUS or MRI with 100% specificity, given that the patient cohort, with confirmed arthritis, was greater than 18 years old.
While comparing the HRUS and MRI, the findings of the current study would agree with previous studies in terms of the overall performance. It was observed that HRUS is equal or nearly equal to MRI and shows 90.83% (95% CI, 84.33%–94.8%) sensitivity and 100% (95% CI, 96.19%–100%) specificity to overall diagnostic performance with the positive and negative predictive values of 100% (95% CI, 96.6%–100%) and 89.81% (95% CI, 82.68%–94.22%), respectively.
Given that 90.83%, 100.0%, and 94.93%, sensitivity, specificity, and accuracy, respectively, were reported for the diagnosis of RA, this is congruent with the work of Malla et al. 18 In that study, they found an overall concordance between USG and MRI, for RA pathologies, was more than 80%. 18
Limitations
There are limitations to this research, mainly based on the design and purposive sample. The smaller sample size was due to the time bound nature of the study and also attributed to the ongoing pandemic. In addition, the socio-demographic factors have an influence on tenosynovitis and synovitis, however, the present research did not assess the confounding effects of such variables on the diagnostic methods’ findings.
Future research with a bigger patient population will be necessary to identify which of these two imaging modalities is more suited as the proper diagnostic choice. Both have benefits and drawbacks, and as technology and abilities advance, they may continue to change in terms of precision and accessibility. The availability of a imaging technique is frequently a deciding factor based on the availability at a specific medical center. HRUS assessments that allow for the accurate and early detection of disease pathology in RA patients would be extremely beneficial in developing countries, such as India, where the expense and accessibility of MRI centers are restricted. This might allow practitioners evaluate therapy and avoid debilitation triggered by this disease.
Conclusion
From previous literature and the results of this clinical research, HRUS demonstrated a diagnostic suitability for detecting RA pathologies, and in some instances, was inferior to MRI. Results of this study demonstrated that HRUS could be useful in comparison with MRI. HRUS could be considered a preliminary modality for early assessment of RA. It has been suggested that HRUS could also replace MRI in assessing RA pathologies in certain subsets of patients, such as pediatrics, pregnant patients, and for diagnostic follow-ups.
Footnotes
Acknowledgements
We would extend our thanks to Dr. ML Soni, MPCST, Bhopal (M.P.)i for extending his support in data assessment and statistical analysis.
Ethics Approval
This study was approved by the Institutional Ethical Committee of Teerthanker Mahaveer Medical College and Research Centre, Moradabad, India, on December 19, 2019 (TMMC&RC/18-19/IEC).
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System).
Animal Welfare
Guidelines for humane animal treatment did not apply to the study.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
