Abstract
Objective:
The Centers for Disease Control and Prevention (CDC) has documented that the leading cause of death in the United States is cardiovascular disease (CVD). Specifically, CVD is responsible for the death of one out of every five women in the United States. Therefore, the research question was, “What specific diagnostic tests, including a novel approach to detecting abdominal aortic calcifications (AAC), could be added to screening women for CVD?”
Materials and Methods:
A prospective cohort of 14 female volunteers, between the ages of 40 and 60 years, were consented to sonography of the aorta, collection of demographic data, as well as contributing biometric data. Each volunteer gave informed consent in compliance with the university’s Institutional Review Board. Volunteers also were provided with a Health Insurance Portability and Accountability Act (HIPAA) release that was signed prior to the collection of any data. All sonographic images were analyzed post-examination and aortic calcifications were graded using a modified scoring system derived from a dual-energy x-ray absorptiometry (DXA)-based grading system.
Results:
In this cohort of 14 females, their AAC category scores ranged from I to IIIB (grades: normal to multiple calcified atheroma). Descriptive statistics demonstrate that it is possible to document AAC on this cohort of women using sonography. Correlational statistics showed a statistically significant association between a volunteer’s posterior aortic-intimal media thickness (a-IMT) and their atherosclerotic cardiovascular disease (ASCVD) 10-year risk score.
Conclusion:
This pilot study suggests the need for further research on screening techniques to determine women’s specific CVD risk in this age group. Although the preliminary results allude to sonographic measures of a-IMT being a possible CVD screening method in the future, more research data are needed.
The Centers for Disease Control and Prevention (CDC) has documented that the leading cause of death in the United States is cardiovascular disease (CVD). This would include conditions such as coronary artery disease, heart attack, and other varied heart complications. 1 Specifically, CVD is responsible for the death of one out of every five women in the United States. 2 Despite increases in awareness over the past decades, only about half (56%) of women recognize that heart disease is a primary mortality risk. 3 To address this major health crisis, the CDC has launched a specific preventive health initiative called the well-integrated screening and evaluation for women across the nation (WISEWOMAN). 4 The purpose of this initiative is to provide women 40 to 64 years of age with screening tests that will assist in determining their CVD risk. Patients undergo cardiovascular screenings (i.e., blood pressure, cholesterol, diabetes, body mass index [BMI], and smoking), a complete a health risk assessment (e.g., Framingham Risk Assessment tool), and a review of their documented medical history to assess whether they may be at risk for CVD. 4
These data were used to determine the level of risk, so that health counseling and coaching can be provided to address a suspected risk of CVD. Grant funding has been provided to several state health departments to support this initiative in an attempt to reduce the female mortality due to CVD. 5 There are several underlying difficulties with this state-wide program and those are that data being collected may not be sufficient to properly assess CVD risk for women of this age group. It also may not properly reflect the CVD risk for women who are in lower social-economic situations.
A potential solution to addressing this disparity, between CVD incidence and risk assessment, is to implement asymptomatic screening of women. This could be easily accomplished during their annual physical examinations by their primary care physician (PCP). 6 A current screening method that has been implemented in the primary care setting that addresses CVD risk is the calculation of the atherosclerotic cardiovascular disease (ASCVD) risk score. The ASCVD risk assessment is a tool used to establish the percent chance of an individual experiencing a CVD event over the next decade. Although ASCVD is widely used, Rana et al 7 completed a study (n = 307 591) that demonstrated this risk assessment did not capture the holistic variables that contribute to a patient’s CVD risk. That study indicated a tendency to overestimate and provide inaccurate CVD risk predictions. 7
The objective of the current research project was to determine what specific screening diagnostic tests and any novel approaches for detecting abdominal aortic calcifications (AAC) could be added to gauge CVD risk. This research had the potential to add the assessment of AAC as an additional factor for CVD risk assessment.
Prior to executing this research, it was important to determine what evidence existed and the various cardiovascular screening methods, which have been tested and implemented. One of the first important CVD risk screening tools were individual measures of high-density lipoprotein (HDL) and low-density lipoprotein (LDL). These have been shown as effective indicators of CVD risk over several decades.8,9 The cohort study that confirmed the efficacy of HDL and LDL provided a significant step in developing this useful CVD screening tool. Unfortunately, in 1998, the data collected were primarily based on the measures from a cohort of male participants.8,9 The lack of gender-specific information has been a persistent issue in the development of CVD screening tools and urgently needs to be addressed. To reduce CVD risk among women, it is important to consider a set of diagnostic tools that are not exclusively based on data collected from men. 10
Besides using HDL and LDL as CVD risk assessment tools, AAC assessment is an additional method that has demonstrated high efficacy in determining individual risk. Currently, the assessment of AAC through imaging has been shown to be a strong predictor of CVD events, based on data gleaned from dual-energy x-ray absorptiometry (DXA), computed tomography (CT), or a radiograph. The DXA has been shown as a viable method in measuring AAC. 11 This method has demonstrated reproducibility, with multiple evaluators finding similar plaque counts, on similar images. The DXA was shown to yield similar results, as data obtained from a radiograph. 11 The AAC has been shown as an independent risk factor of CVD and actually outperforms the Framingham risk assessment. Framingham risk score (FRS) is a personal estimate of the 10-year risk for CVD risk. 12 O’Connor et al 13 conducted a multiyear, large cohort study that found a statistically significant advantage to determine CVD risk from a nonenhanced CT, of the abdominal aorta, compared with the FRS. This retrospective study was composed of an asymptomatic cohort of 451 women and 378 men (N = 829). The individuals in this cohort all received nonenhanced CT of part of the aorta. These aortic images were graded for AAC and a chart analysis was completed to obtain individual FRS. These data were then compared with the individuals who experienced a CVD event and with those who did not, for the years between the patients’ aortic CT and the follow-up (11.2 years with a standard deviation of 2.8 years). Using the AAC values demonstrated a statistically significant difference between the scores of the CVD event group and the nonevent group, compared with the FRS.
This may make AAC seem like a perfect diagnostic replacement for assessing individual CVD risk, but this screening method comes with a major pitfall of gauging AAC, given a patient’s ionizing radiation dose due to the imaging techniques, as well as the cost of this imaging equipment. Ionizing radiation has repeatedly shown a deleterious risk due to the biological effects of ionizing radiation.14–16 Lauer 15 argued that most radiologic imaging tests do more harm than good due to a patient’s exposure to ionizing energy. Through statistical analysis and modeling, Brenner and Hall 16 estimated that between 1.5% and 2% of cancer cases within the United States are due to diagnostic ionizing radiation imaging examinations. The combined issues of radiation dose and expense make these imaging techniques not an optimal choice for CVD screening. Additional research is needed to find noninvasive and low-cost diagnostic tools that can be used during a PCP appointment. 17
Similar to an AAC assessment, it was important to review alternative CVD screening methods to detect coronary artery calcium (CAC), such as a contrast-enhanced CT. Arad et al 18 performed a prospective study (N = 4613), in which asymptomatic individuals underwent a CT and a subsequent follow-up screening, 4.3 years later. In that large cohort, 119 individuals had a CVD event that occurred during that time period. These individuals had a statistically significant higher baseline coronary calcium CT score than those who did not experience a CVD event. 18 This method of screening is a reliable CVD screening tool that directly measures the calcium in the coronary vessels. 19 Coronary artery calcium screening comes with a similar risk as AAC, given that it relies on a CT or CT coronary angiography, as well as the associated ionizing radiation.19,20 Both of these diagnostic imaging techniques add the risks of ionizing radiation and an added expense for screening patients.
An alternative method of CVD screening was the diagnostic imaging of individuals to detect a life-threatening abdominal aortic aneurysm (AAA). This screening set of images has been proven to be an effective tool in decreasing mortality from CVD events and is a promising diagnostic tool.21–23 Guirguis-Blake et al 23 performed a review of four randomized controlled trials (RCTs; N = 137 214) and demonstrated that screening men aged 65 years and older for AAA decreased aneurysmal rupture and related mortality. The screening for AAA is another diagnostic strategy that has been conducted for over a decade, but the empirical evidence has some important flaws. Each of the referenced studies highlighted the effectiveness of this technique, but underscored how this technique was only completed for men aged 65 years and older.21–23 Guirguis-Blake et al 23 also reviewed an RCT that involved a cohort of 9324 women which indicated no benefit to screening women for AAA. Again, the gender bias that exists in determining women’s risk for CVD, using varied techniques, is highly influenced by the empirical evidence generated from cohorts of men. In addition, there appears to be a tendency for these screening techniques to be applied to patients above the age of 65. Interestingly, disconfirming evidence exists due to a review of 18 cohort studies and a total sample size of 257 384 individuals. 24 In that meta-analysis of individuals, the risk for a CVD event was detected among those as young as 45 years. 24 Due to this review, it is apparent that a set of screening techniques for detecting CVD is still needed for women 40 to 64 years of age. This gap in evidence could be addressed by conducting an analysis of specific diagnostic techniques which use nonionizing radiation and are low cost. Given these important parameters for implementing CVD screening, diagnostic medical sonography and biomarkers are possible methods for generating risk data. Therefore, the research question was as follows: Can a current modified AAC grading system be used to assess the abdominal aorta of women 40 to 64 years of age with sonography? Can sonography-derived measurement of AAC and biomarkers be used to detect women at risk for CVD?
Materials and Methods
A prospective sample of 14 female volunteers between the ages of 40 and 60 years was subjected to aortic sonographic imaging, demographic data collection, and biometric data collection. Each volunteer was given written informed consent as to comply with the Institutional Review Board. Volunteers also were provided with a Health Insurance Portability and Accountability Act (HIPAA) release that was signed before any data were collected.
For collection and analysis of volunteer images, a GE Logiq I (Waukesha, Wisconsin) ultrasound equipment system, with a 2 to 5 MgHz curvilinear transducer, was used. Sonography was completed on the infra-renal aorta of each volunteer. The aorta was divided into three regions and was evaluated on three calcification metrics: count, morphology, and thickness. Aortic intima-media thickness (a-IMT) was also noted for each of the regions. Volunteer vertebral bodies were used as guidelines to delineate each region of the aorta, with the bifurcation of the aorta being used as a guide to find the L4 vertebrae (see Figure 1). The regions are described using the vertebral bodies as follows: (1) region 1 was defined as the area between the bifurcation of the aorta and the superior part of the L3 vertebrae, (2) region 2 was defined as the area between the superior part of the L3 vertebrae and the inferior part of the L1 vertebrae, and (3) region 3 was defined as the area above the inferior part of the L1 vertebrae. Analysis of the images was performed after the sonographic examination was completed. The AAC was graded using a modified scoring system derived from a DXA-based grading system. 25 This measurement scale is based on the thickness of the calcification, which is given a grade from I to V. This AAC scoring system can be further detailed by reviewing the methods used by Stigall-Weikle et al. 26
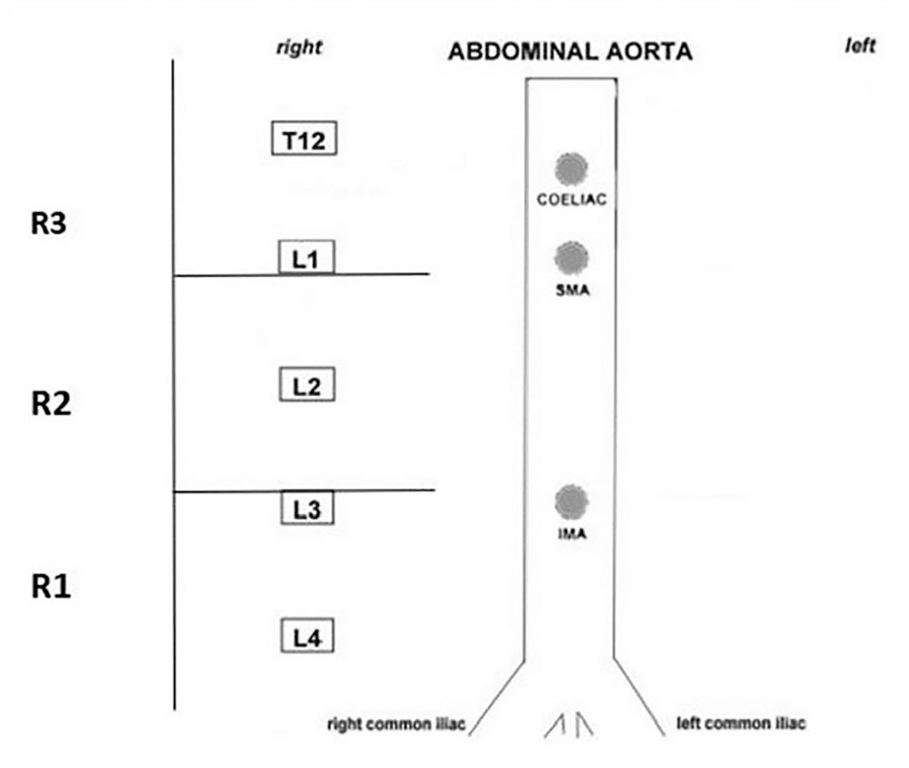
Adapted abdominal aortic calcification scoring by region, based on dual-energy x-ray absorptiometry. SMA, superior mesenteric artery; IMA, inferior mesenteric artery.
Demographic data were collected on each of the volunteers to perform statistical analyses and to compute the ASCVD 10-year risk score. The demographic data were collected via a self-reporting questionnaire that each volunteer filled out. The data collected in this questionnaire included race, personal diabetes history, tobacco smoking history, hypertension treatment status, statin usage, aspirin treatment, and a personal health rating from poor to excellent. Volunteer height, weight, age, and sex were all obtained from the volunteer’s medical record; height and weight measurements were taken the same day as the sonographic images. The ASCVD 10-year risk score was conducted on each volunteer by entering volunteer age, race, sex, blood pressure, total cholesterol, HDL cholesterol, diabetes status, smoking status, statin use, and aspirin therapy into the American College of Cardiology ASCVD Risk Estimator tool. The ASCVD 10-year risk scores are as follows: Low risk (<5%), Borderline risk (5%–7.4%), Intermediate risk (7.5%–19.9%), and High risk (≥20%).
Biometric data were collected from each volunteer via blood draw and blood pressure cuff on the day of their sonographic examinations. The information collected included volunteer systolic and diastolic blood pressure, total cholesterol, LDLs, and HDLs. These values were used for individual statistical comparisons with aortic calcification score and were used in computing each volunteer ASCVD 10-year risk score.
Results
Descriptive Statistics
The 14 sample volunteers in this pilot study were women in the age range of 41 to 57 years old, with a mean age of 48. Each of these volunteers had ASCVD 10-year risk scores completed as part of the study. The volunteer’s risk scores ranged from 0.3% to 2.5% and indicated a chance for a cardiovascular event over the next 10 years of life. One volunteer’s ASCVD 10-year risk score was impossible to calculate due to her total cholesterol being too low to use in the assessment. This cohort’s descriptive statistics are provided in Table 1.
The Descriptive Data Generated From the Cohort of Females 40 to 60 Years of Age.
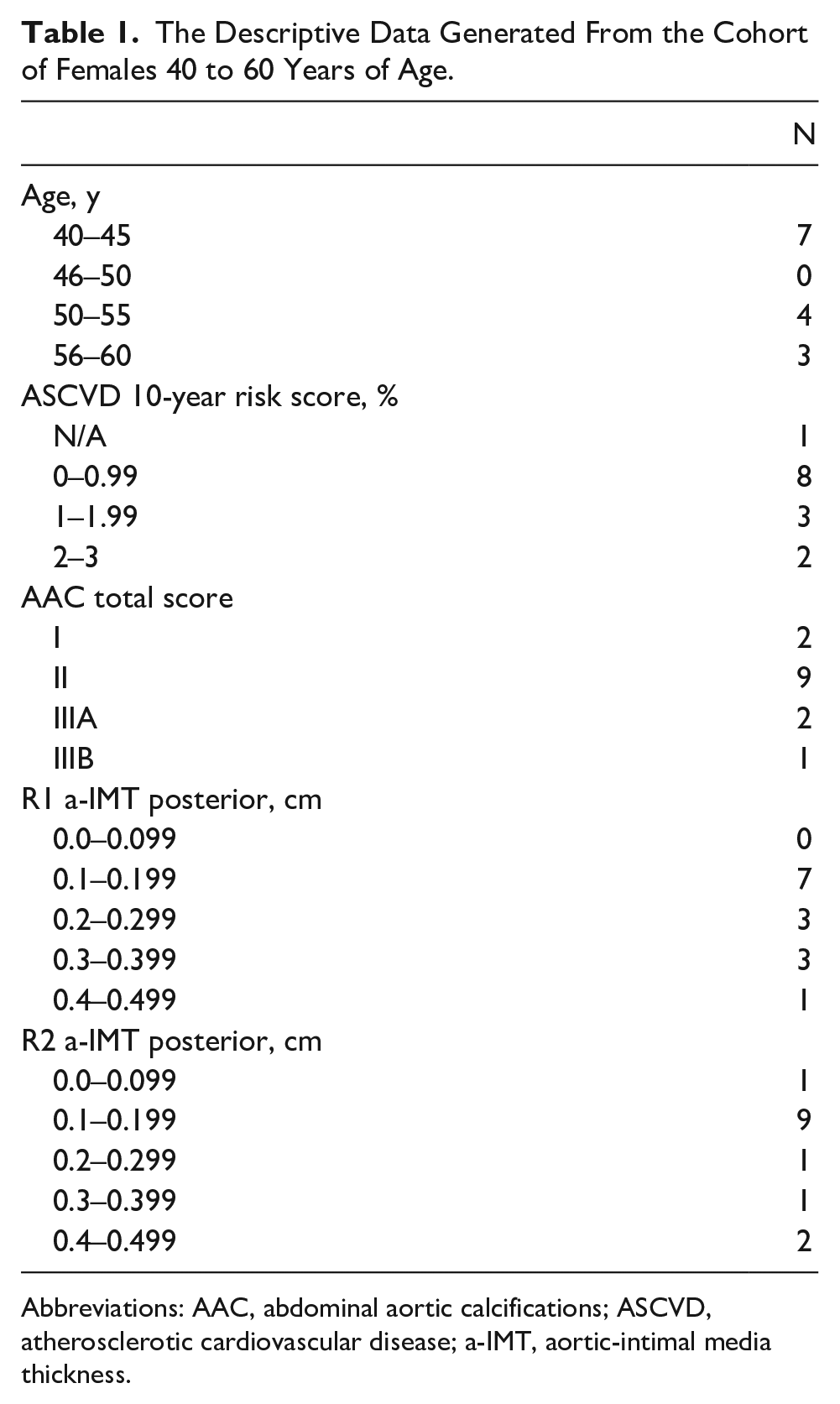
Abbreviations: AAC, abdominal aortic calcifications; ASCVD, atherosclerotic cardiovascular disease; a-IMT, aortic-intimal media thickness.
All the volunteers’ sonographic images were used to obtain a score for possible AAC and the a-IMT across the three regions of the aorta outlined in the methods. All volunteers were successfully imaged via sonography and the images scored for a-IMT and AAC in regions 1 and 2 for 13 of 14 volunteers in this cohort; the sonographic images obtained in region 3 only yielded 7 of the 14 volunteers with data that could be successfully obtained in that area.
A total AAC score was given to each volunteer based on the highest score for a particular region. The AAC scores ranged from grade I (normal intima) to IIIB (multiple plaques ≤3 mm). Two individuals had an AAC grade of I, nine were given the grade of II, two were given the grade of IIIA, and one was given the grade of IIIB. Posterior measurements of a-IMT were more reliably acquired from volunteer images and are used in place of anterior a-IMT for measuring the a-IMT. In region 1, the a-IMT ranged from 0.1 to 0.45 cm, with a mean of 0.22 cm. In region 2, the a-IMT ranged from 0 to 0.42 cm, with a mean of 0.21 cm. The full set of descriptive statistics for this cohort are included in Table 1.
Correlational Statistics
Inferential correlational statistical analyses were based on the data that were obtained from the variables of ASCVD 10-year risk score, AAC total score, R1 posterior a-IMT, and R2 posterior a-IMT. A Spearman rho test was used to determine the strength of association between each variable and the two-tailed statistical significance of the analysis. Spearman rho is a nonparametric test used to measure the strength of association between two variables, where the value r = 1 means a perfect positive correlation and the value r = −1 means a perfect negative correlation. 27 The complete correlational analysis and statistical significance are provided in Table 2. The only correlation to demonstrate a strong association and high statistical significance was between the ASCVD 10-year risk score and R1 a-IMT. The strength of association was measured by the correlation coefficient of .806 with a P value of .001.
The Strength of Association, Utilizing Spearman rho, Between Variables Generated by a Cohort of Women 40 to 60 Years of Age.
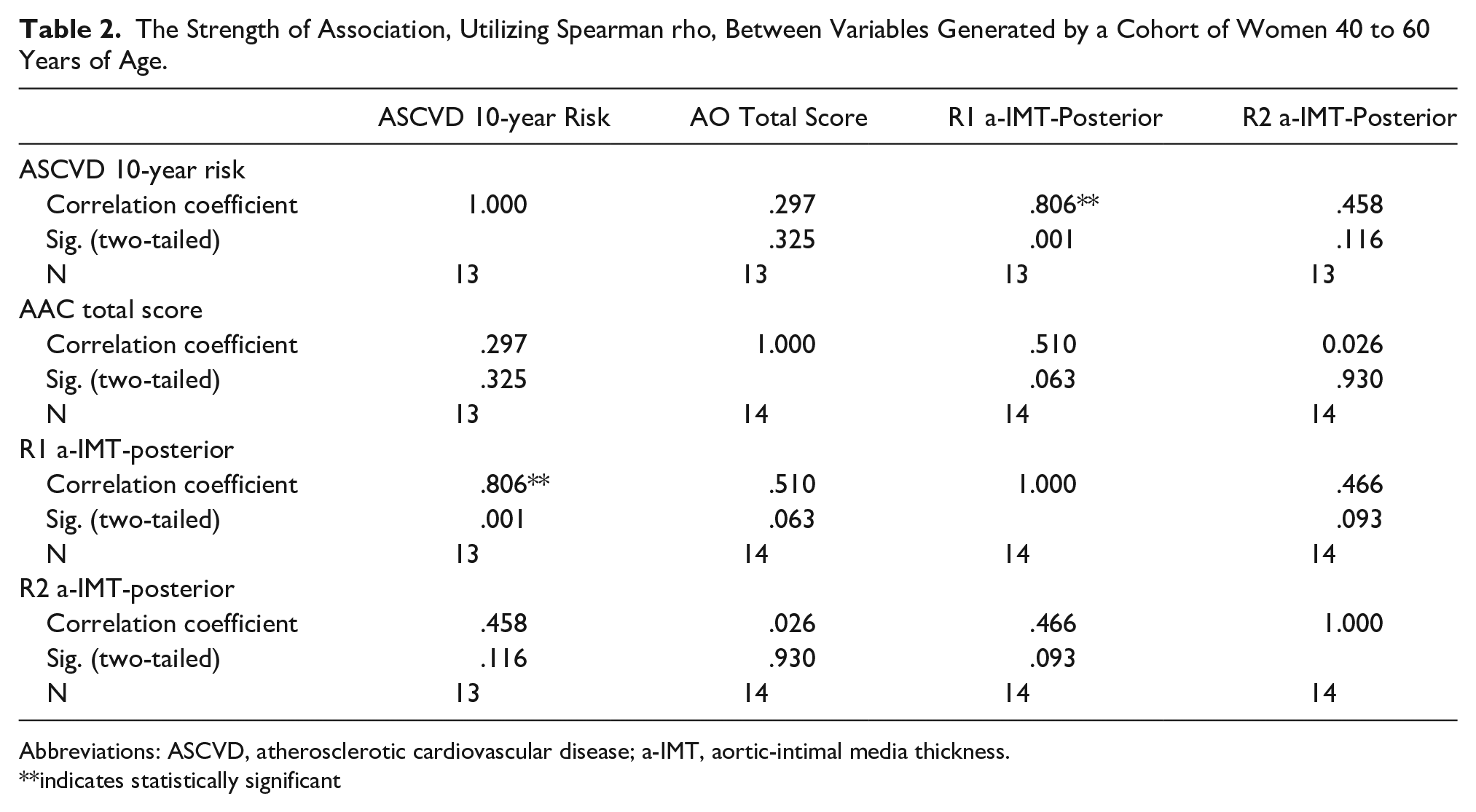
Abbreviations: ASCVD, atherosclerotic cardiovascular disease; a-IMT, aortic-intimal media thickness.
**indicates statistically significant
Discussion
Descriptive statistics were used to answer the first research question seeking if a current modified AAC grading system can be used to assess the abdominal aorta of women 40 to 64 years of age, with sonography. This was found to be feasible as trained sonographers were able to obtain sonographic images of the abdominal aorta for each of the 14 female volunteers. Each of these volunteers were also able to have their images scored for calcification and receive a total aortic calcification score. The AAC grades I (n = 2), II (n = 9), IIIA (n = 2), and IIIB (n = 1) were observed in the volunteer population.
Each volunteer was able to be reliably imaged in region 1 of their aorta. In region 2, only 1 volunteer was not able to be accurately imaged, and in region 3, 7 of the 14 volunteers were properly imaged and scored for calcification. It was found that scanning further superior in the abdominal aorta became increasingly difficult due to the volunteer’s intestinal gas and the sternum blocking the ultrasound transmission. Although the scanning of region 3 was part of the methods described for imaging, the experience of the sonographers was that this area was not consistently imaged with quality. Due to compromised image quality, the subsequent aortic measurements could not be discretely obtained.
Correlational statistics were used to answer the second research question seeking if sonography-derived measurement of AAC/a-IMT can be used to detect women at risk for CVD. With a limited cohort size, it is hard to make definitive statements regarding this research question. However, the limited results may suggest that region 1 posterior aortic-IMT is strongly and statistically significantly correlated to an individual’s ASCVD 10-year risk score. These variables were obtained on 13 of the 14 volunteers and showed a correlation coefficient of .806 with a two-tailed P < .001. These IMT measurements are more classically used for assessment of the carotid arteries but this pilot study demonstrated that these same measurements could be used to assess the anterior layers of the aorta. Region 1 posterior IMT would need to be further investigated to assess its clinical significance as a potential CVD risk measure. Due to the ultrasound physics constraints, the posterior IMT measurement was affected by diminished spatial resolution due to attenuation of power. Current ultrasound imaging equipment and transducers have imaging limitations that must be improved in order for these measures to be recorded with greater diagnostic confidence.
In this cohort, AAC measures were not statistically significantly associated with the ASCVD 10-year risk score. Due to the limitations of the sample size, this metric cannot be dismissed as a risk assessment tool, based on this limited data collection. Also, this was a correlation between AAC and only one other measure of CVD risk assessment. It would be beneficial to compare AAC with more metrics such as a coronary calcium score to get a more complete determination of the role that AAC may contribute to the assessment of CVD risk in the future.
Few studies have been completed with data that are derived from females in this age group of 40 to 60.8,9 It is important to push this investigation further so that women’s risk assessment is based on female data, rather than male cohort risk assessments. 10 This pilot study aimed to gather female-specific data to begin to combat the disparity that may exist between CVD research on women and men. Further studies that incorporate larger sample sizes of diverse female cohorts are paramount to a more accurate measure of CVD risk in asymptomatic women.
Sonographic measurement of AAC has been previously shown to be feasible in a cohort consisting of five females and six males. 26 The methodology used for obtaining 20 images was repeated in this cohort of 14 females and was again found to be feasible. These studies in conjunction provide a solid base supporting usability of ultrasound imaging as a means to measure AAC in females.
Limitations
The present pilot study was limited by the research design and purposive recruitment of volunteers. The sample size was limited for number and gender. The recruitment of volunteers was somewhat hampered by only using two outpatient settings, within 20 miles from each other.
Conclusion
These data were collected during a global pandemic, underscoring these volunteers’ willingness and confidence in obtaining a CVD risk assessment. Post-pandemic efforts should be renewed and execution of this diagnostic method could be even more successful in various medical practices/venues. A statistically significant correlation between posterior a-IMT and the ASCVD 10-year risk score was observed in this cohort, but that does not ensure that posterior a-IMT is going to work as a measure of CVD risk in all settings. There were not significant correlations between other metrics but that could be due to the limitations of this study. A similar study with a larger cohort, completed at multiple sites, would be beneficial in being more determinant of the true significance of the measures of AAC and a-IMT as CVD risk assessment tools. Should this future study be carried out, having volunteers fast before undergoing the sonographic assessment of the abdominal aorta may reduce internal gas and allow for better imaging of regions 2 and 3.
Footnotes
Acknowledgements
The research team is also indebted to Randell K. Wexler, MD, Vice Chair of Clinical Affairs, who provided physician oversight of this work.
Correction (October 2023):
The Ethics Approval section has been updated with the IRB number.
Ethics Approval
This study was covered under the ethical guidelines of The Ohio State University’s IRB (#2014H0467).
Informed Consent
Informed consent was not sought for the present study because all case data were de-identified and/or aggregated and followed ethics committee or IRB guidelines (also referred to as the Honest Broker System). Participants all signed informed consent through IRB protocols for the study.
Animal Welfare
Not applicable.
Trial Registration
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially supported by a grant from the American Society of Radiologic Technology Foundation research grant. Mr Bloom was also supported in his research efforts by funding from The Ohio State University’s Honors and Scholars program.
Peer Reviewer Guarantee Statement
The Editor/Associate Editor of JDMS is an author of this article; therefore, the peer review process was managed by alternative members of the Board and the submitting Editor/Associate Editor had no involvement in the decision-making process.
