Abstract
Megavesicles are an uncommon diagnostic finding during sonography of the bladder, especially when the examination is performed transabdominally. Although megavesicles are more likely seen with transrectal ultrasonography, computed tomogram (CT), vesiculography, or magnetic resonance imaging (MRI), it has been noted during transabdominal sonography for patients suffering from autosomal dominant polycystic kidney disease (ADPKD). When seminal vesicles become dilated, they are often visualized during transabdominal sonography. Two patient cases are provided of seminal megavesicles, associated with ADPKD and have documented sonographic findings. Both patient cases of megavesicles were discovered incidentally during the course of a renal sonogram. The importance of these diagnostic findings and the possible pathogenesis are provided.
Keywords
Autosomal dominant polycystic kidney disease (ADPKD) is a prevalent genetic disorder that causes multiple cysts of varying size in both kidneys. Approximately 0.125% of the population is affected by this disease. 1 Seminal vesicle enlargement is most often seen in the presence of men with the polycystic kidney disease 1 mutation. Seminal megavesicles are known to be associated with ADPKD, however are rarely noted during transabdominal sonography. This series review explores the sonographic findings of two male patients with ADPKD and concurrent megavesicles, incidentally noted during transabdominal sonography.
Case 1
A 52-year-old man was referred for a renal transplant sonogram, due to elevated creatinine levels, high blood urea nitrogen, and a low estimated glomerular filtration rate (eGFR). One month prior to this sonogram, the patient’s metabolic panel demonstrated an eGFR level of 45 mL/min. This eGFR value was lower than the normal range of greater than or equal to 60 mL/min and indicative of renal dysfunction. His additional patient history indicated ADPKD, infertility, and a renal transplant. The patient’s mother had also been diagnosed with ADPKD.
A sonogram of the renal transplant and bladder was performed, utilizing a Phillips iU-22 ultrasound system (Phillips Medical, Bothell, WA) and a 5 MHZ curved linear transducer. The native kidneys were not assessed at that time. The renal transplant located in the right iliac fossa was assessed for visible structural defects and included color and spectral Doppler of the renal arteries and veins.
Posterior to the bladder, two rounded mostly anechoic structures with areas of small linear echogenic reflectors were visualized (Figure 1A). The right structure measured 2.2 cm long by 2.1 cm wide (Figure 1C), and the left structure measured 2.1 cm long by 2.1 cm wide (Figure 1D). No vascularity was visualized in or around the structures. The patient did not have complaints of pain or tenderness in this region. The location and size of the structures were consistent with the appearance of seminal megavesicles, as seen on a previous computed tomogram (CT) (Figure 1B).
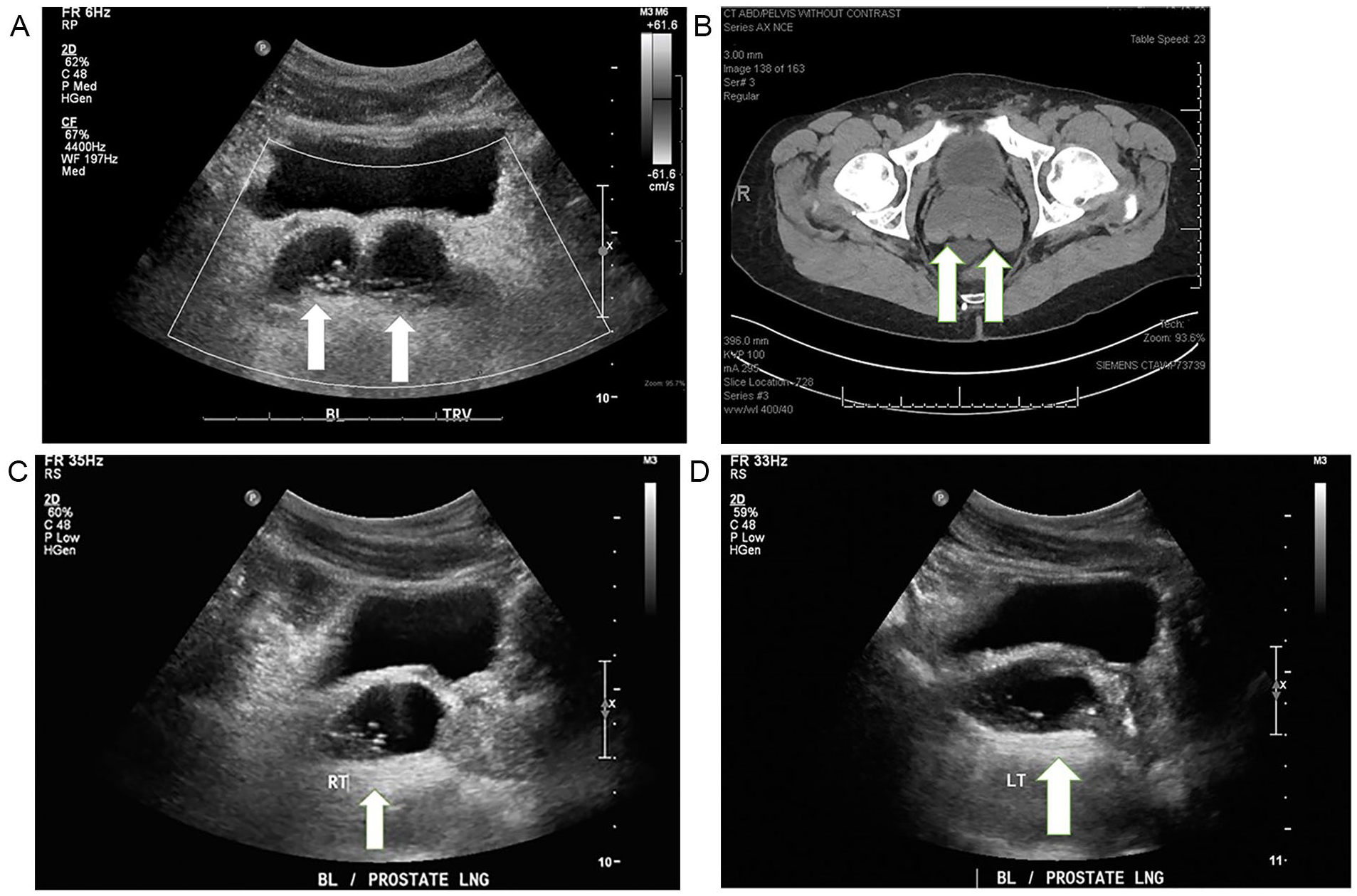
Case 1: (A) Transverse sonographic image of bilateral megavesicles creating a “bowtie” appearance. Arrows show location of megavesicles. (B) A transverse computed tomographic image of megavesicles. Arrows show location of megavesicles. (C) Longitudinal sonographic image of right megavesicle. Arrows show location of megavesicles. (D) Longitudinal sonographic image of left megavesicle. Arrows show location of megavesicles.
The renal transplant sonogram was interpreted as normal appearing without evidence of hydronephrosis. The renal vessels appeared patent, had slightly increased resistive indices, measured by spectral Doppler, but were noted as a nonspecific finding. A repeat annual sonogram was suggested to follow the condition of the renal transplant and a specific follow-up or treatment of the megavesicles was not required.
Case 2
A 38-year-old man presented to the hospital with a complaint of hematuria. The patient was diagnosed with ADPKD, 9 years earlier. This patient also suffered from various complications of his disease which included ruptured cysts, aortic dissection, and the comorbidity of Marfan syndrome. One week prior to the sonogram, the patient’s metabolic panel indicated an eGFR level of 44 mL/min. This eGFR value is lower than the normal range of greater than or equal to 60 mL/min and is indicative of renal dysfunction. The patient reported that his father and brother also suffered from ADPKD. A sonogram of the patient’s kidneys and bladder was performed, utilizing a Phillips iU-22 ultrasound system (Phillips Medical, Bothell, WA) and a 5 MHZ curvilinear transducer. This study revealed bilaterally enlarged kidneys replaced with numerous cysts of varied size. The right kidney measured 20.6 × 8.8 × 8.2 cm. There were multiple calcifications noted, throughout the right kidney, with an associated twinkle artifact, consistent with nephrolithiasis (Figure 2D).
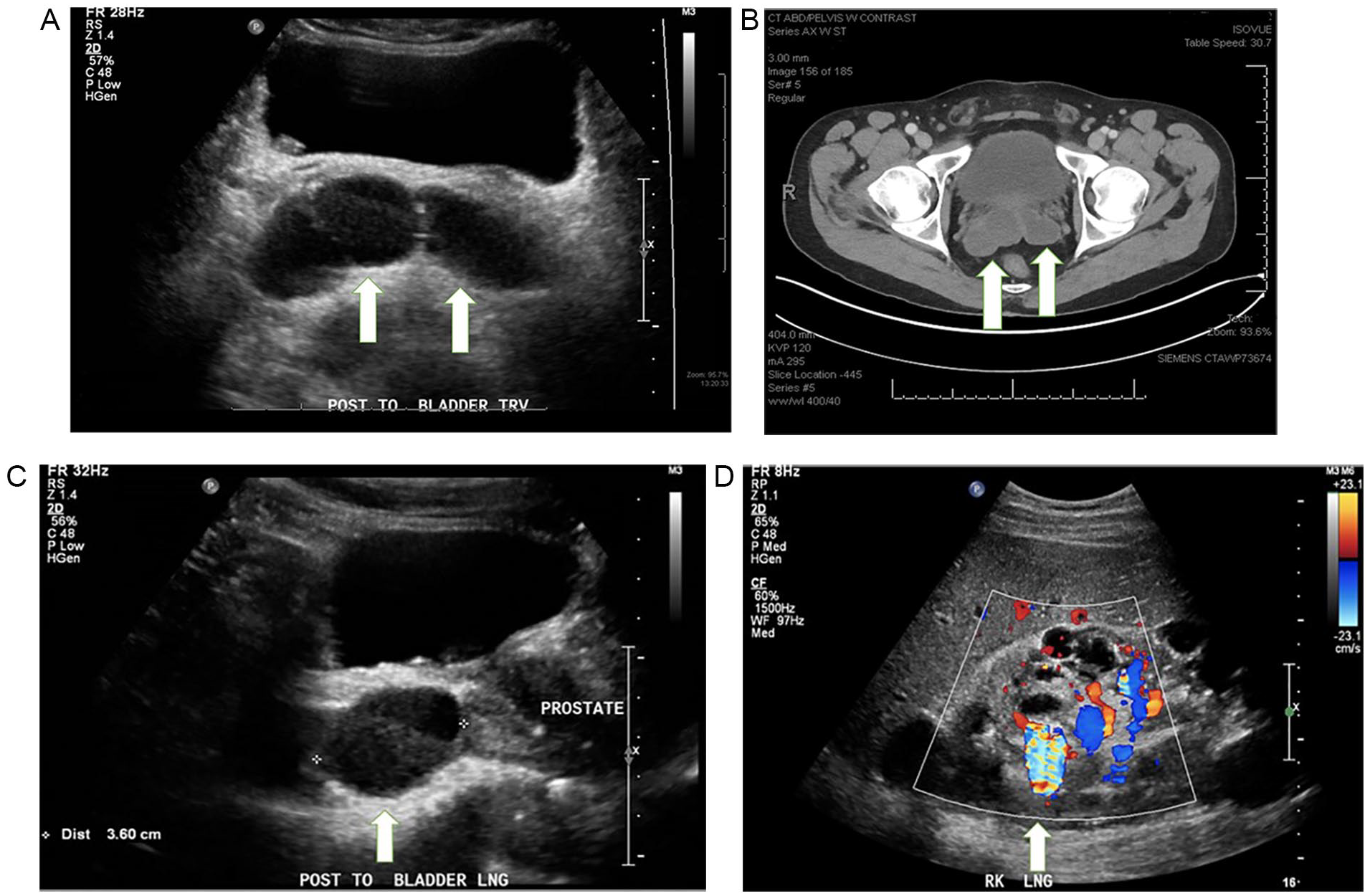
Case 2: (A) Transverse sonographic image of megavesicles posterior to bladder. Arrows show location of megavesicles. Debris visualized within bladder is consistent with blood products. (B) A transverse computed tomographic image of bilateral megavesicles. Arrows show location of megavesicles. (C) Longitudinal sonographic image of right seminal vesicle. Arrow shows location of megavesicle. Debris visualized within bladder is consistent with blood products. (D) Longitudinal sonographic image of right kidney. Cystic areas and twinkle artifact visualized within right kidney. Arrow shows presence of twinkle artifact.
Echogenic foci consistent with calcifications were noted and reported as bilateral non-obstructive nephrolithiasis. The largest cyst measured 1.9 cm in the superior aspect of the right kidney. A complex cyst was visualized in the inferior pole of the right kidney. This cyst had septations and calcifications which also demonstrated a twinkle artifact, with color Doppler. The left kidney measured 23.2 × 10.8 × 10.4 cm. The largest cyst in the left kidney was noted in the superior pole and measured 5.7 cm in diameter. Multiple calcifications were noted throughout the left kidney, consistent with nephrolithiasis, with no evidence of hydronephrosis.
The bladder appeared distended and contained layered debris, consistent with blood products. Posterior to the bladder and separate from the prostate, two anechoic rounded structured were noted. The right structure measured 5.1 cm long by 2.8 cm in diameter (Figure 2C). The left structure measured 4.5 cm long by 1.7 mm in diameter. The paired structures were consistent with seminal megavesicles (Figure 2A), as seen on a prior CT (Figure 2B). The patient was treated for a cystic infection confirmed by an abnormal urinalyses test. The emergency department’s attending physician suggested follow-up with a primary care physician for further patient care. The patient was subsequently discharged from the hospital.
Discussion
Renal tissue affected by ADPKD may have various sonographic appearances. In the advanced stages of the disease, the bilateral renal tissue will appear cystic and enlarged. Due to the number and size of the cysts, normal renal parenchyma may be difficult to identify. Typically, patients present clinically in the third and fourth decades of life. 2 In younger patients, multiple cysts may have not yet formed, making the disease more difficult to diagnose. Simple renal cysts, noted on sonography, appear round or oval, anechoic, thin walled, and as clearly delineated structures, with acoustic through transmission. Complex cysts may exhibit internal layering, debris, septations, or calcifications. Complex diagnostic findings can be seen in cases of cyst infection, rupture, or post hemorrhage. 3 One limited biomarker used to assess the progression of ADPKD is eGFR, particularly in younger patients. 2
There are many complications and comorbidities associated with ADPKD. Findings in these patients may include intracranial aneurysms, dilation of the aortic root, and aortic dissection, 1 all of which can be life threatening. The disease can lead to further organ pathologies such as cyst formation in the kidneys, liver, pancreas, uterus, testicles, prostate, and seminal vesicles. 4 Two subtypes of ADPKD exist, specific to gene mutation: PKD1 and PKD 2. These genetic mutations result in the development of renal cysts and the continued loss of renal function. 2 The most common type of ADPKD is caused by the PKD1 gene mutation, which affects 85% to 90% of these patients. The PKD2 gene affects the remaining 10% to 15% of ADPKD patients. Literature has shown that ADPKD patients with a PKD1 mutation are ten times more likely to have megavesicles than patients with a PKD2 mutation. 5 Gene mutation analysis shows that PKD1 patients with megavesicles have non-truncating mutations, which likely reflects the less severe presentation of renal involvement in the phenotype. Among PKD1 patients, those with megavesicles have a higher eGFR, lower height-adjusted total kidney volume (HtTKV), and less severe renal disease, than those without. Furthermore, the presence of megavesicles may help to differentiate PKD1 and PKD2 mutations. 5 This finding may be used clinically as one predictor of the severity of progression of disease.
Seminal vesicles are paired glands located posterior and inferior to the bladder, and lateral to the vas deferens. 6 Development of the seminal vesicles is closely related to the embryological development of the vas deferens, ureters, and kidneys. Seminal vesicles, vas deferens, and ureteric buds are all derived from the Wolffian (mesonephric) ducts. The normal development of the ureteric buds is necessary for the proper formation of the metanephric blastema into the kidneys. Because of this embryologic association, there is a close connection with developmental anomalies of the seminal vesicles, vas deferens, ureters, and kidneys. 7
Dilated seminal vesicles may cause infertility, testicular pain, pain with ejaculation, hematospermia, or perineal pain. Less common symptoms may include back pain, thigh pain, urinary obstruction, dysuria, or pain with defecation. 8
Seminal vesicles are paired, oval shaped tortuous tubular structures of the male reproductive tract that lie in the retroperitoneum. 7 Located at the base of the prostate, seminal vesicles are bordered posteriorly by the rectum and vas deferens, and anteriorly by the bladder. Limited literature exists describing the sonographic appearance of seminal megavesicles and they are not routinely seen by transabdominal sonography. The small size of normal seminal vesicles in addition to the depth of their anatomical location makes them unlikely to be visualized by this approach. With transrectal ultrasonography (TRUS), the average width of normal seminal vesicles is 0.97 cm on the right side and 0.93 cm on the left. 9 When enlarged, seminal vesicles can appear cystic or dilated. Seen together in the transverse view, megavesicles will likely have a “bowtie” shaped appearance by sonographic examination. 10 In normal cases, seminal vesicle size can vary, and volume will slightly decrease with age. This may not be true in cases of dilated seminal vesicles associated with ADPKD, as their size is unlikely to change. 9
Seminal vesicle development and health is imperative to fertility. Disorders specific to the seminal vesicles are rare. Common diseases to affect seminal vesicle are caused by trauma, obstruction, neoplasm, infections, and genetic anomalies and can lead to secondary findings of normospermia, immotile sperm, seminal vesicle cysts, megavesicles, and ejaculatory duct cysts. Providing 80% to 90% of male ejaculate fluid, these organs are crucial to male reproductive health. The alkaline substance provides nutrient-rich energy, including vitamin C, enzymes, and fructose, to source the spermatozoa. 7 Male infertility issues related to ADPKD are not widely published in reference books and thus are lesser known.
Thorough imaging of the seminal vesicles is imperative in the diagnosis and treatment of male infertility, especially in the presence of ADPKD. Seminal vesicle cysts are reported to be present in 40% to 60% of the ADPKD population. 11 However, many studies report a high prevalence of seminal vesicle cysts. A large portion of these findings may actually be megavesicles, which resemble cysts, with sonography. 6 Studies that use percutaneous vesiculography, in ADPKD patients, to examine presumed seminal vesicle cysts on a sonogram can prove to be dilated seminal vesicles, without evidence of obstruction. This defines the presence of megavesicles. 11
When seminal vesicles become dilated or engorged, with a lumen measurement greater than 1.5 cm, they are considered megavesicles. If only one seminal vesicle is dilated to greater than 1.5 cm, it is not considered a megavesicle and needs to be evaluated for other pathologic conditions. Megavesicles typically appear cystic and anechoic by sonography due to the engorged tubules. The term “megavesicles” does not apply to vesicles dilated by cysts. Depending on the method of imaging used, up to 60% of men with ADPKD in fact have megavesicles. It should be noted that no volume change will be noted in megavesicles after ejaculation, because of a structural inability to contract. Distention of the seminal vesicles typically indicates obstruction. 1 However, in the setting of ADPKD, megavesicles are not caused by obstruction and patency of the vesicles can be demonstrated by contrast enhanced CT. In the case of megavesicles, the free flow of dye can be identified in the ejaculatory duct, by percutaneous vesiculography. 6
It is unclear why seminal vesicles become distended in men with ADPKD. The presence of megavesicles can be linked to a functional failure of contraction by an atonic or missing muscular wall, likely contributing to infertility. Studies show thinned or absent muscular walls in vesicles and ejaculatory ducts of corpses with megavesicles. This finding validates that dyskinesia and vesicle atonicity may be the cause of dilation. 11
Healthy seminal vesicles will appear tortuous with sonography. Megavesicles will appear as simple, dilated, tapering collagenous tubes that narrow from their proximal to distal ends. 12 Although megavesicles are patent, the inability to propulse the contents lead to stagnation and result in sperm death. 13 ADPKD is a disease that affects the extracellular matrix, which is the reason for its association with mitral valve prolapse and intracranial aneurysms. It is likely that this aspect of the disease plays a role in the abnormal propulsion of seminal vesicle contents. 12
Patients with ADPKD typically receive routine magnetic resonance imaging (MRI) to evaluate total kidney volume, which serves as a predictor of acute renal failure. As ADPKD progresses and kidney volume becomes greater, it is recommended to include the pelvis as a part of imaging studies. This allows to adequately measure renal volume, as the kidneys can extend deep into the pelvis. 5 If the pelvis is not included in the MRI study, there is a risk of incomplete assessment of the extent of disease. By routinely adding pelvic imaging (including the testes), a more thorough assessment of the kidneys will be acquired as well as identifying possible urogenital manifestations of megavesicles, cysts in the seminal vesicles, testes, prostate, or dilation of the vas deferens in association with ADPKD. 5
Understanding differential diagnoses of the male genitourinary system is important for the accurate diagnosis of megavesicles. Congenital seminal vesicle cysts are most often caused by the internal accumulation of fluid due to obstruction by atresia of the ejaculatory duct. Sonographically, they will appear as well-defined, anechoic structures, similar to the appearance of cysts found elsewhere in the body. Acquired seminal vesicle cysts develop due to inflammation, infection, or obstruction. In cases of infection or inflammation, sonographic findings may include the appearance of a thickened or enhanced seminal vesicle wall, internal debris, cystic dilation during the acute phase, and atrophy in the chronic phase. 7 It is possible that prostate cysts in ADPKD are associated with dilation of the seminal vesicles. Prostate cysts in ADPKD patients have been found midline near the verumontanum, where the ejaculatory ducts join to enter the urethra. Dilation of the ampulla of the vas deferens may also be noted where it enters the seminal vesicles in ADPKD subjects, where it is unlikely to be found in individuals without megavesicles. 7
The number of reported cases of seminal vesicle cysts varies widely. Approximately 60% of cases are reported by TRUS, while approximately 6% are reported by CT. 4 Dilated seminal vesicles in men with ADPKD are more commonly found incidentally during fertility evaluations than in renal examinations. Due to the technical limitations of medical imaging systems, it can be very difficult to differentiate dilated seminal vesicles from seminal vesicle cysts. MRI with contrast provides the highest resolution imaging. However, due to the high cost of MRI, TRUS is an important diagnostic tool to assess obstructions of the seminal vesicles and ejaculatory ducts. TRUS is the primary imaging modality used to evaluate the extent and location of dilation present in megavesicles and can specifically indicate their relationship to the prostate. 9
Prior to modern imaging techniques, disorders of the seminal vesicles or ejaculatory ducts were often underdiagnosed and undertreated, particularly in cases of infertility. The quality image improvement of sonography, MRI, and CT has led to more accurate diagnoses and improved treatment outcomes. Following preliminary imaging and a diagnosis of megavesicles, a symptomatic patient may undergo further imaging, such as MRI with an endorectal coil and a vasogram, which can provide greater diagnostic specificity. Patients suffering from pain, azoospermia, or oligospermia may undergo sperm analysis and treatment by way of a transurethral resection of the ejaculatory duct (TURED). 8 In some cases, this allows for a reduction in pain, and improvement of sperm quality, and motility. The presence of megavesicles is benign and may not impair the patient’s quality of life. Due to the minimal impact on the health, for asymptomatic patients with megavesicles, there may be less need for treatment. 8
Conclusion
Sonography plays a critical role in identifying seminal megavesicles. During transabdominal renal and bladder sonography, megavesicles may be incidentally detected. Among adult men, visualizing paired elliptical structures, posterior to the bladder, are likely megavesicles and ADPKD may be a contributing factor. Megavesicles can be visualized bilaterally, at the base of the prostate, with the lumen measuring more than 1.5 cm. A sonographer should be aware of the different types of polycystic kidney disease, the age of clinical onset, the associated organ sonographic abnormalities, and the linked comorbidities. Men with ADPKD may suffer from reproductive and extra-renal disease. At the time of renal sonogram, this imaging modality can be the initial diagnostic tool to detect the presence of megavesicles.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
