Abstract
Objective:
Preeclampsia accounts for 15% of maternal deaths and may cause fetal morbidity and mortality. The aim of this research was to evaluate the efficacy of maternal uterine artery Doppler versus serum beta-human chorionic gonadotropin (β-hCG), during the first trimester, in predicting preeclampsia and intrauterine growth restriction (IUGR).
Materials and Methods:
In a convenient sample of 388 pregnant women, uterine artery resistive index (RI) and pulsatility index (PI) were assessed, and serum β-hCG level was measured at 11 to 13 weeks of gestation. The patients’ maternal blood pressure and fetal growth were monitored.
Results:
The patients with preeclampsia (n = 58) showed a significant uterine RI and PI increase with a significant β-hCG decrease compared with the normotensive patients (n = 330). The specificity of uterine PI and RI to predict preeclampsia and IUGR is higher than that of β-hCG. However, the sensitivity of combined diagnostic tools is higher than the singular use of these diagnostic tests.
Conclusion:
Uterine artery Doppler may be better than serum β-hCG in predicting preeclampsia and IUGR. However, combined diagnostic techniques may be better to screen at-risk patients.
Preeclampsia is known to be a syndrome of persistent hypertension in pregnancy. It is found in 3% to 7% of primigravida and 1% to 3% of multigravida women. 1 It is considered one of the main reasons for maternal and fetal morbidities and mortalities, accounting for approximately 15% of maternal mortality in developed countries. 2
Placental insufficiency plays a principal role in the pathogenesis of preeclampsia. The reduced placental perfusion, secondary to poor uterine vascular remodeling, gradually causes placental dysfunction. This results in the release, into the maternal circulation, of angiogenic factors leading to the activation, and perhaps injury, of the maternal endothelium, due to soluble fms-like tyrosine kinase 1 (sFlt-1) and endoglin. 3
These changes are associated with increased uterine artery resistance, which augments the sensitivity to vasoconstriction and leads to chronic placental ischemia and oxidative stress. This results in fetal complications, which may include intrauterine growth restriction (IUGR), intrauterine fetal death (IUFD), as well as low appearance, pulse, grimace, activity, and respiration (APGAR) score, in newborns. 4
The uteroplacental blood flow can be assessed by Doppler sonography that evaluates the capability and quality of blood flow, in the small branches of uterine arteries. During the antenatal period, the impedance of blood flow in the uterine arteries decreases with the increase of gestational age. However, in the case of inadequate trophoblastic invasion, there is an increase in blood flow impedance to the uterine arteries, as they become unable to reach low resistance. 5
A Doppler sonography study at 11 to 13 weeks demonstrated a correlation between increased vascular impedance in the uterine arteries and subsequent development of pregnancy-induced hypertension. 6 The increase in mean uterine artery pulsatility index (PI) and resistive index (RI) during the first trimester is accompanied by IUGR. 6 More than half of pregnant women who may develop preeclampsia and fetal IUGR can be detected during their first trimester using the uterine artery Doppler. 7
Placental hormones are considered early biochemical markers, as they indicate any disruption in the placental function. The level of maternal serum beta-human chorionic gonadotropin (β-hCG), released by the placenta, was found to decrease during the first trimester of pregnancy in women who may later develop preeclampsia or IUGR. 8 Whereas in the second trimester, preeclampsia and IUGR are associated with a rise in the level of β-hCG.9,10 Other biochemical markers, such as plasma cell-free fetal DNA level, could be considered a potential marker for preeclampsia during the third trimester. 11 Currently, studies show that the addition of maternal serum markers and uterine artery Doppler could improve the detection accuracy of adverse pregnancy outcomes. 12
Materials and Methods
Following the Medical Research Ethics committee’s approval, this cohort study was conducted at a University Hospital from September 2017 to February 2019. Written consent was obtained from all participants after they were informed of the study’s objective and design. All of the research participants were given the option to leave the study at any time.
The study population included 436 pregnant patients referred to a tertiary hospital during the study period, for routine antenatal care examination at 11 to 13+6 weeks of gestation. Patients were excluded from the study if they had any of the following conditions: multiple pregnancies, pyelonephritis, or thyrotoxicosis.
During routine antenatal care examination at 11 to 13+6 weeks of gestation, two blood pressure recordings were obtained. Laboratory investigations, such as complete blood chemistry panel, urine analysis, blood glucose, and quantitative β-hCG, were done. All participants underwent a sonogram and uterine artery Doppler, using a Voluson ultrasound equipment system (Voluson 730 Pro, GE Healthcare, Chicago, Illinois, USA) and a curvilinear transabdominal multifrequency (3–7 MHz) transducer. The number of fetuses, fetal heart activity, fetal biometry, congenital anomalies, amount of amniotic fluid, placental site, and the maternal uterine artery Doppler (RI and PI), in the first and/or second trimesters, were assessed. All the measurements recorded from 30 symptomatic patients were taken twice by the same examiner and then by another examiner. This measurement procedure occurred on the same day and was done to evaluate both intraobserver variability and interobserver variability.
The uterine artery was easily identified by its characteristic flow velocity waveform. The patient was sitting in a semi-recumbent position with her head and chest supported at approximately 45°, from the supine position. To obtain recordings from both right and left uterine arteries, the transducer was placed longitudinally in the right and left lower quadrant of the abdomen (iliac fossa just above the inguinal ligament). Color Doppler was employed to identify the uterine artery at the point where it crossed the external iliac artery. An angle of insonation of 50° or less was used to obtain waveforms acceptable for analysis. The maximum systolic and minimal diastolic velocities were computed by electronic caliber and produced the systolic-diastolic ratio, the RI, and the PI of the two vessels (see Figure 1). The RI, PI, and the presence or absence of a diastolic notch (unilateral or bilateral) were noted. Waveform profiles were obtained without the patients experiencing a uterine contraction (lax maternal abdominal wall).
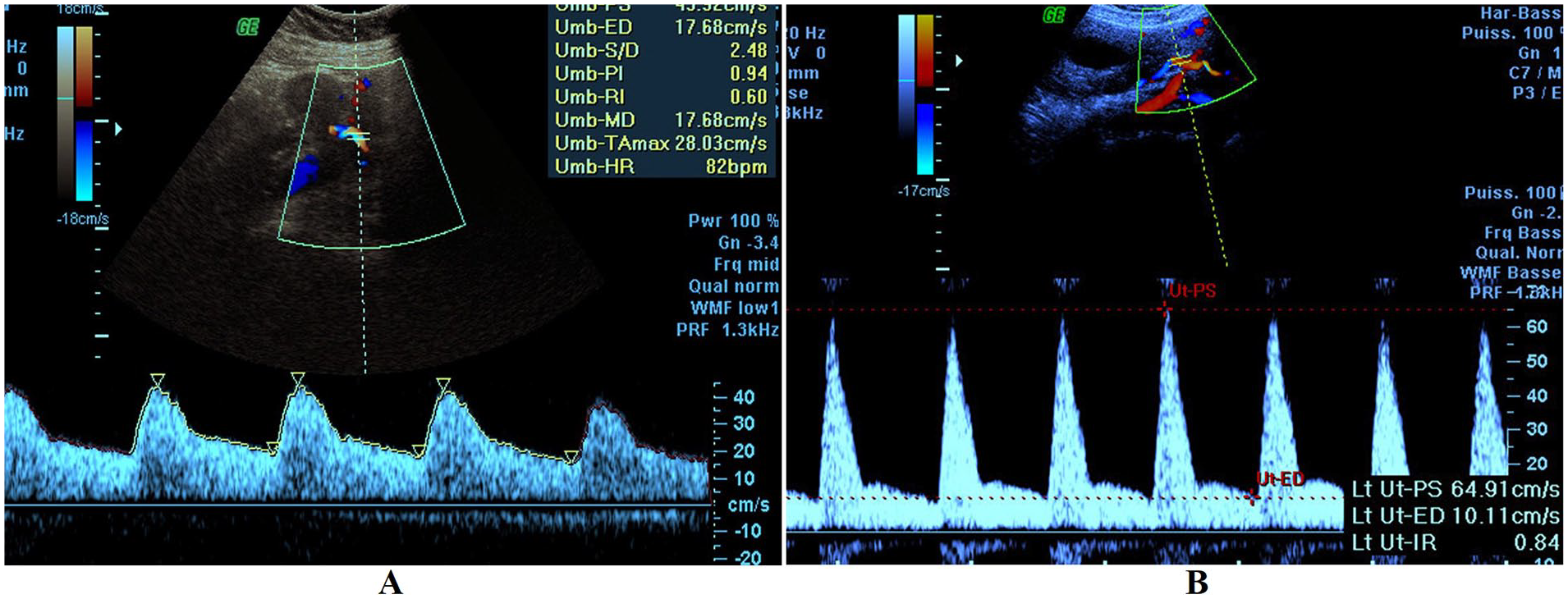
An example of a normal uterine artery Doppler (A) compared with a uterine artery Doppler taken on a first-trimester preeclampsia patient (B). PI, pulsatility index; RI, resistive index.
All the consented patients were followed until delivery during routine antenatal care with particular concern for blood pressure measurement, urine analysis for proteinuria, and sonographic monitoring for fetal growth. Preeclampsia was diagnosed by the presence of high blood pressure after the 20th week of gestation (140/90 mm Hg or above on two separate occasions at least 4 hours apart) and proteinuria (urinary protein ≥300 mg/24 hours or persistent 30 mg/dL protein in random urine samples). IUGR was diagnosed if the estimated fetal weight, by sonography, was below the 10th percentile for gestational age. After delivery, the APGAR score at minute 5 was recorded to assess the neonate’s condition.
Statistical Analysis
Data were coded and analyzed using the Statistical Package for the Social Sciences (SPSS, Inc., Chicago, IL, USA, v.25 for Microsoft Windows). Statistical analysis was done to obtain the mean ± standard deviation (SD), frequencies, and percentages. A Student t test was used for numerical data, while a chi-square test was used for categorical data. Multivariate regression analysis was used to calculate the relative weight of several predictors and to assess the best prediction model. The receiver operating characteristic curve analysis was done to estimate the sensitivity and specificity of the obtained cutoff values. A P value <.05 was set a priori and considered statistically significant.
Results
In this study, 436 pregnant patients who met the inclusion criteria were recruited and consented to the study. Only 388 patients were enrolled in the study as 29 women were missing during follow-up data and 19 women had a miscarriage. All patients underwent an initial sonogram, uterine artery Doppler, and measurement of the serum level of β-hCG, between 11 and 13+6 weeks of gestation. During the diagnostic follow-up, 330 women remained normotensive, while only 58 women developed preeclampsia.
There was no significant difference in age between patients who developed preeclampsia and those who remained normotensive. On the contrary, the mean body mass index (BMI) was significantly higher in patients with preeclampsia (P < .001).
Those patients with preeclampsia showed a significant increase in the mean uterine RI and PI compared with the normotensive women (P < .001). The level of β-hCG at 11 to 13+6 weeks of gestation also was found significantly reduced in women with preeclampsia (P < .001). All the patients’ follow-up data revealed that those who developed preeclampsia had a significant risk for IUGR, preterm delivery, low birthweight, and low APGAR score at minute 5 for their neonates (P < .001). Among those patients who developed preeclampsia, three women developed IUFD (see Table 1). In the first trimester, 31 patients (8% of participants) presented bilateral notches, which disappeared in the second trimester of gestation.
A Descriptive Data Comparison Between Those Study Patients Who Did and Did Not Develop Preeclampsia.
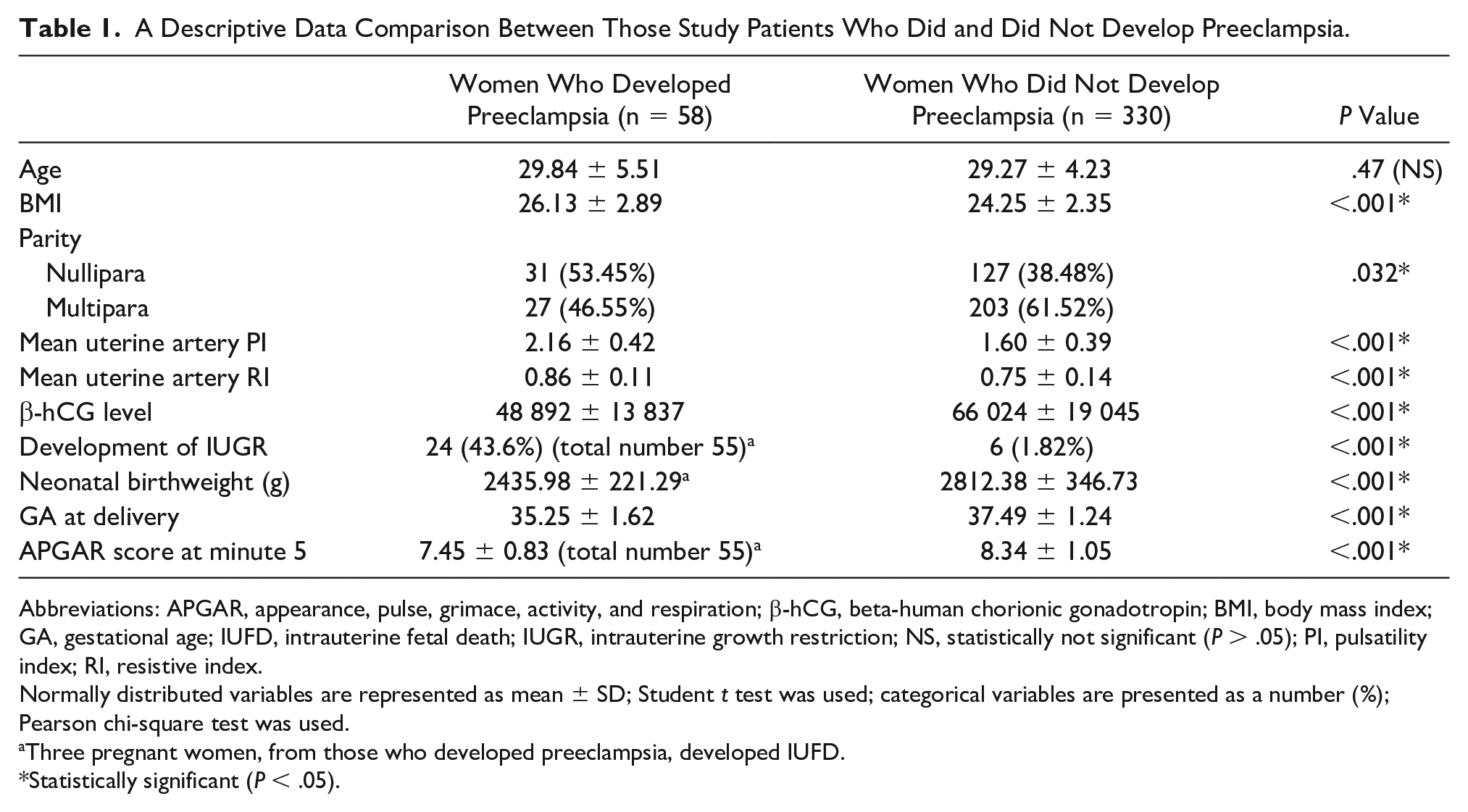
Abbreviations: APGAR, appearance, pulse, grimace, activity, and respiration; β-hCG, beta-human chorionic gonadotropin; BMI, body mass index; GA, gestational age; IUFD, intrauterine fetal death; IUGR, intrauterine growth restriction; NS, statistically not significant (P > .05); PI, pulsatility index; RI, resistive index.
Normally distributed variables are represented as mean ± SD; Student t test was used; categorical variables are presented as a number (%); Pearson chi-square test was used.
Three pregnant women, from those who developed preeclampsia, developed IUFD.
Statistically significant (P < .05).
The specificity of mean uterine artery PI (76.4%) and RI (70.3%) to predict preeclampsia was higher than that of serum β-hCG level (66.1%) and almost near that of combined diagnostic tools (74.5%). However, the sensitivity of combined diagnostic assessment tools (81%) was higher than using each diagnostic test alone (see Table 2 and Figure 2). Regarding IUGR, the specificity of mean uterine artery PI (75%) and RI (79.6%) to predict IUGR was higher than that of serum β-hCG level (74.5%). However, the sensitivity of combined tools (90%) was higher than using each diagnostic test alone (see Table 3 and Figure 3).
The Sensitivity and Specificity of Mean Uterine PI and RI, Serum β-hCG, and Combined Uterine Artery Doppler Parameters With Serum β-hCG, for Detection of Preeclampsia.
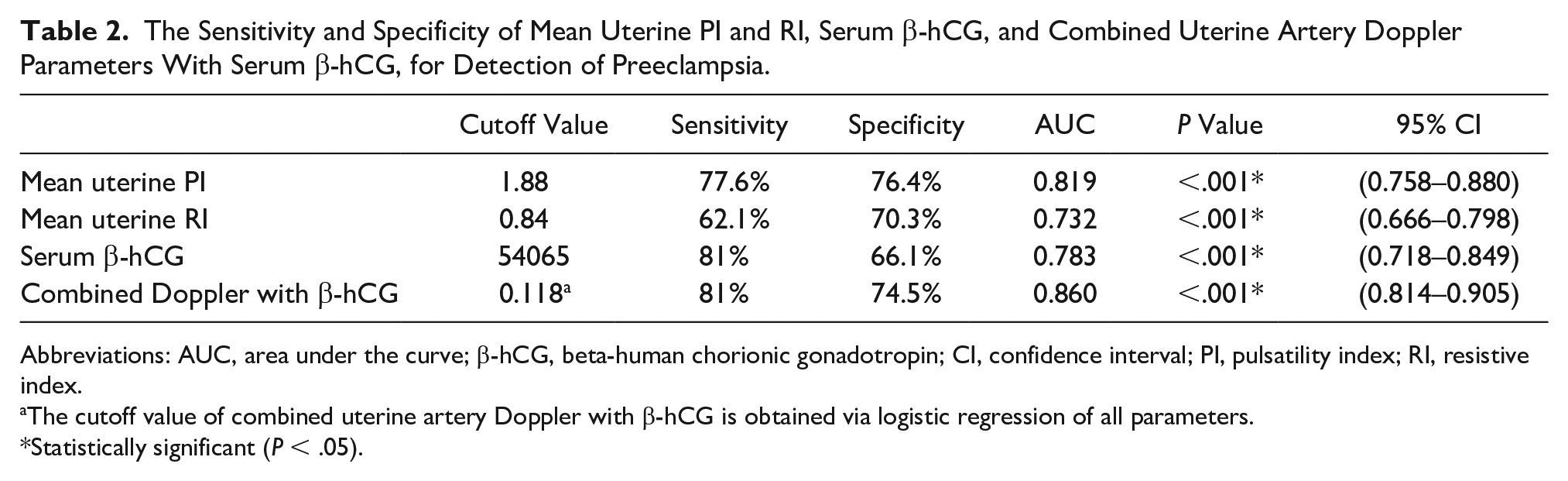
Abbreviations: AUC, area under the curve; β-hCG, beta-human chorionic gonadotropin; CI, confidence interval; PI, pulsatility index; RI, resistive index.
The cutoff value of combined uterine artery Doppler with β-hCG is obtained via logistic regression of all parameters.
Statistically significant (P < .05).
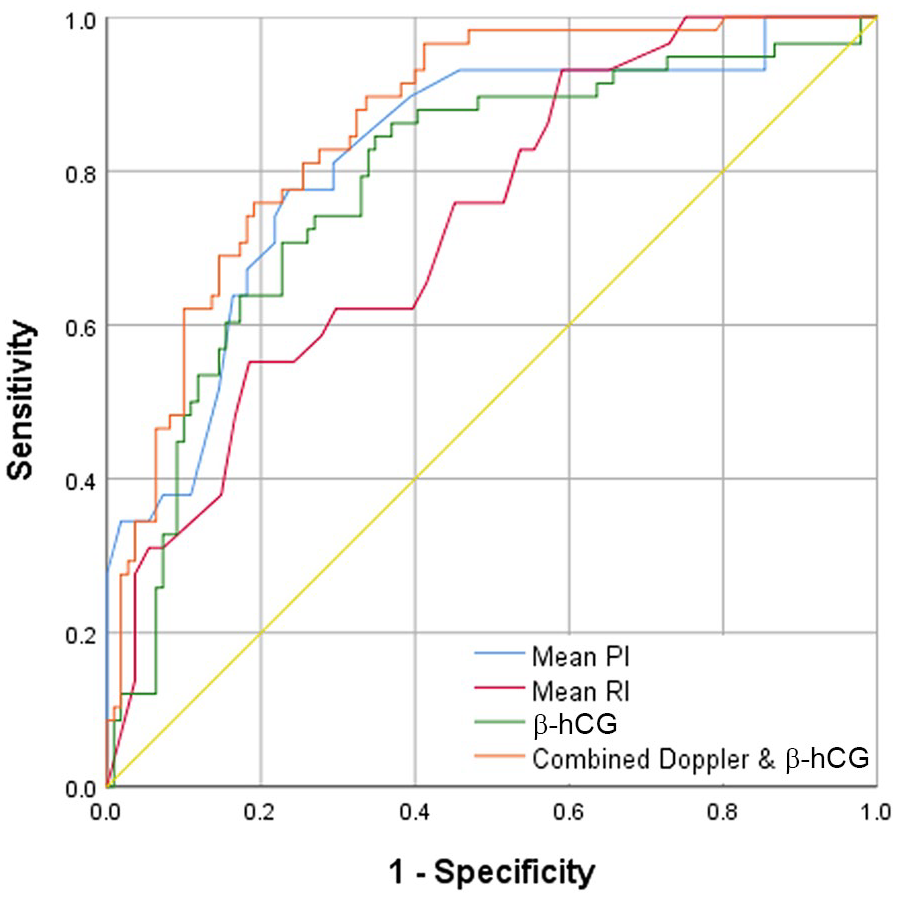
The receiver operating characteristic curve for mean uterine PI and RI, serum β-hCG, and combined uterine artery Doppler parameters with serum β-hCG, for early detection of preeclampsia. β-hCG, beta-human chorionic gonadotropin; PI, pulsatility index; RI, resistive index.
The Sensitivity and Specificity of Mean Uterine PI and RI, Serum β-hCG, and Combined Uterine Artery Doppler Parameters With Serum β-hCG, for Detection of IUGR.
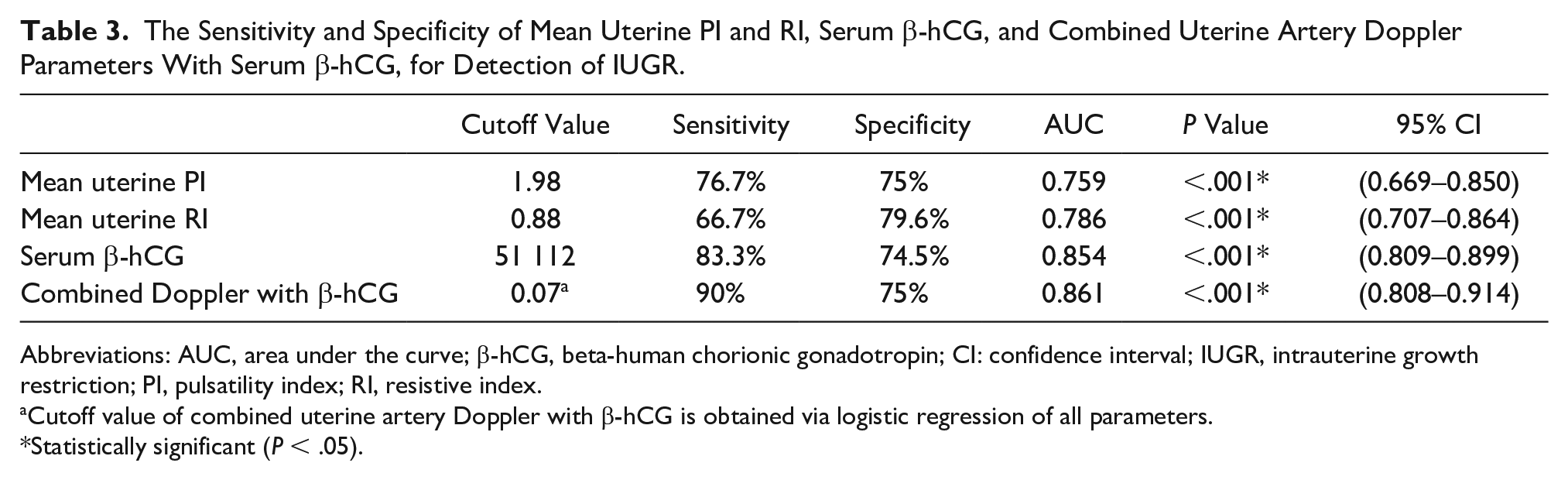
Abbreviations: AUC, area under the curve; β-hCG, beta-human chorionic gonadotropin; CI: confidence interval; IUGR, intrauterine growth restriction; PI, pulsatility index; RI, resistive index.
Cutoff value of combined uterine artery Doppler with β-hCG is obtained via logistic regression of all parameters.
Statistically significant (P < .05).
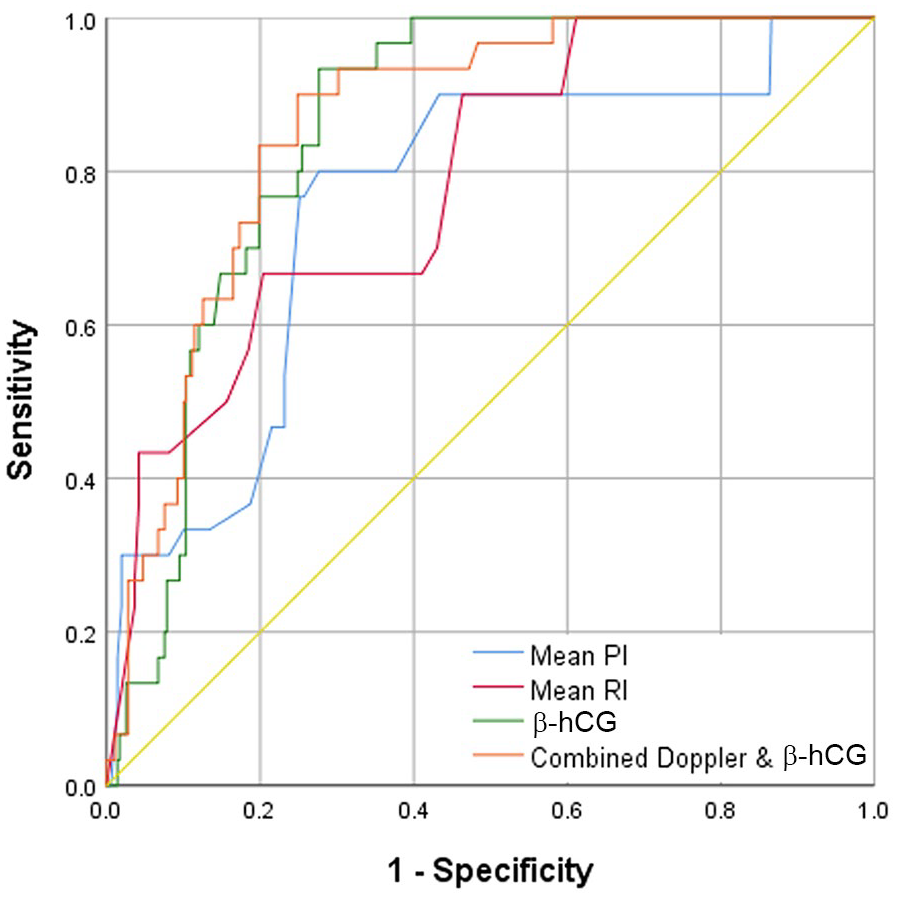
The receiver operating characteristic curve for mean uterine PI and RI, serum β-hCG, and combined uterine artery Doppler parameters with serum β-hCG, for early detection of IUGR. β-hCG, beta-human chorionic gonadotropin; IUGR, intrauterine growth restriction; PI, pulsatility index; RI, resistive index.
Discussion
Preeclampsia complicates 5% to 8% of all pregnancies worldwide that can lead to serious complications for both mother and baby. 13 Apart from preeclampsia being a primary placental disorder, it is a multiorgan disorder and is labeled as a disease of theories.14,15 As a result of developing preeclampsia, maternal morbidity usually affects coagulation as well as possible renal and hepatic failure. 16 IUGR and other fetal complications are caused by diminished uteroplacental blood flow as a sequence of improper trophoblastic infiltration of the placental vascular bed. In addition, women with preeclampsia deliver infants with lower APGAR scores than those of healthy women.17,18
An overall better understanding of this pathophysiology would advance the prediction and clinical management of these patients. It is increasingly recognized that early- and late-onset preeclampsia result from different pathophysiologic pathways. Preeclampsia is currently being addressed as a broader concept than pregnancy-induced hypertension and proteinuria, which these findings support the hypothesis that preeclampsia is more than a single disorder. 19 Given the irreversibility of the pathophysiologic mechanisms underlying preeclampsia and IUGR, delivery remains the only definitive treatment to prevent the development of maternal or fetal complications, from disease progression. 20 Currently, the implementation of low-dose aspirin, before 16 weeks of gestation, has reduced the incidence of early-onset preeclampsia, by more than 60%. 21 Therefore, screening for preeclampsia and preventive treatment are likely to be effective for the mother and infant.
In fact, many screening tests have been used to predict pregnancy complications. Prediction of preeclampsia has been a subject of various studies. The traditional approach to screening, proposed by the National Institute for Health and Care Excellence (NICE) or the American College of Obstetricians and Gynecologists’ (ACOG) guidelines, is based on a maternal risk factor checklist, which achieves detection rates of 41% and 34%, respectively. 22 These prediction rates can no longer be considered sufficient to effectively predict preeclampsia. Therefore, a more effective screening model for preeclampsia is the Bayes theorem based on maternal characteristics and medical history combined with the results of biomarkers and has the advantage of being individualized and dynamic. 23
Prenatal guidelines recommend systematic risk assessment of preeclampsia in early pregnancy and low-dose prophylaxis of aspirin for high-risk women. 24 However, the early prediction of preeclampsia poses several problems. First, the relatively low frequency of preeclampsia, in a low-risk population (2.1%), makes prediction difficult. Second, there is no biomarker or radiologic examination that provides adequate predictive performance to be used in isolation for clinical practice. 25 Finally, most studies on preventive strategies to reduce preeclampsia incidence have reported low to moderate predictive performance. 26
Previous studies investigating the predictive accuracy of maternal uterine artery Doppler indices have revealed considerably varied results. Thus, it is questionable whether uterine artery Doppler should be used as a predictive test. 27 In the Pongrojpaw et al 28 study, they showed that 16 patients presented with IUGR, due to preeclampsia (4.84%). Their patients with preeclampsia had elevated impedance to blood flow, in the uterine arteries, in the second trimester. In that study, those women with mild preeclampsia were able to deliver safely, at term, compared with those with severe preeclampsia, who required early delivery as well as had small for gestational age newborns. 28 Among low- and high-risk patients, an increased PI with notching in the second trimester can precisely predict preeclampsia. Accordingly, increased PI or bilateral notching could be the best indicator for severe preeclampsia. Increased PI alone or together with notching was used to estimate IUGR in low-risk patients. In addition, RI recorded with high-risk patients can be considered the best predictor for complicating changes. 28
Recently, many authors found a uterine artery Doppler examination to be an effective noninvasive screening tool, for preeclampsia, in high-risk pregnancies and particularly in health systems with limited means to assess other biomarkers.29–31 According to Elwakel et al, 31 uterine Doppler provides a noninvasive assessment of uteroplacental circulation, by comparing systolic and diastolic waveforms, as a direct assessment of trophoblast invasion, which is not normally discernable. Murtoniemi et al 32 found that the best prediction of early preeclampsia was obtained using the area under the curve (AUC) of a receiver operating characteristic curve. Conversely, this could indicate that uterine artery PI is much less useful for predicting preeclampsia and its subtypes, in the first trimester of a high-risk patient population compared with low-risk population. 32 Moreover, Prasad et al 33 demonstrated that first-trimester uterine Doppler alone had a good predictive value in predicting preeclampsia in healthy pregnant patients with risk factors. In comparison, other authors have found that uterine artery Doppler is a moderate to poor screening tool for preeclampsia, with low sensitivity (34%–76%). 34 In the current study, abnormal uterine artery Doppler waveforms appeared when uteroplacental blood flow was impaired. Therefore, in the present study, uterine artery Doppler was enough to exclude pregnancy complications.
Regarding biochemical markers, another group of researchers measured maternal serum β-hCG at 11 to 13+6 weeks in 4859 candidates. Out of these pregnant women, 462 women developed preeclampsia and 350 women developed IUGR. In their study, those had a lower value of β-hCG than the control group. 35 However, other studies that assessed the relationship between maternal serum β-hCG in the first trimester and subsequent pregnancy complications due to poor placentation provided conflicting results. 36 Contradictory to the present research results, Barjaktarovic et al 37 showed a relation between increased levels of β-hCG in early pregnancy and successive pregnancy-related hypertensive complications. The increased β-hCG levels may cause alterations on the balance between pro-angiogenic and anti-angiogenic factors and may explain the pathophysiology of preeclampsia.
In another study, Di Lorenzo et al studied the role of uterine artery Doppler and serum biomarkers in the prediction of preeclampsia. However, they found no improvement in the detection rate among their patients. 38 On the contrary, this study results would indicate that utilizing uterine artery Doppler together with measuring maternal serum β-hCG, during the first trimester, is better to predict preeclampsia and IUGR, than using each parameter singularly. However, the present use of capturing only uterine artery Doppler, during the first trimester, was enough to exclude women at risk, of developing preeclampsia and IUGR.
Given the number of patients who had a bilateral uterine notch in the first trimester, which then disappeared in the second trimester, would make uterine artery Doppler a more specific diagnostic tool than β-hCG. In addition, uterine artery Doppler can be used as a prognostic parameter, making it a better predictive and prognostic screening tool than only using β-hCG. It should be noted that the specificity of PI for predicting preeclampsia and IUGR is nearly the same. In comparison, the specificity of RI is better for predicting IUGR (79.6%) than (70.3%). In both, the sensitivity of the combined tools is higher than using a single diagnostic tool for screening. However, the sensitivity of the combined tools is higher for predicting IUGR (90%) than for predicting preeclampsia (81%).
Study Limitations
The main study limitation is due to the threats to internal and external validity, associated with the research design. The cohort of patients, who developed preeclampsia, was limited to only 58. Therefore, these results do not allow for a general assessment of the predictive accuracy of the serum β-hCG level and uterine artery Doppler, in early- and late-onset preeclampsia.
Conclusion
Uterine artery Doppler velocimetry, at 11 to 13 weeks of gestation, could be a specific diagnostic screening tool to exclude subsequent development of preeclampsia and IUGR. Combining serum β-hCG level and uterine artery Doppler may be a better screening set of tools for predicting preeclampsia and IUGR, than the singular use of these tests. Further research is required on a wider scale to evaluate these diagnostic tests’ predictive value, to detect early- and late-onset preeclampsia.
Footnotes
Acknowledgements
We would like to thank our colleagues from Kasr Al-Aini Hospital, who provided insight and expertise that greatly assisted our research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Medical Research Ethics committee of the National Research Center, Cairo, Egypt, under Registration Number 18-171.
Informed Consent
Written consent was obtained from all participants after they were informed of the objective and design of the study, and they were free to leave the study at any time if they wished.
