Abstract
Pelvic radiation therapy in patients with advanced rectal carcinoma improves oncological outcomes, but often raises infertility concerns in reproductive-age patients. Laparoscopic ovarian transposition is a surgical method which may offer preservation of ovarian function during and after cancer treatment. Sonographic evaluation of the ovaries post transposition gives insight into their condition and the efficacy of ovarian transposition in the continuation of ovarian function after radiation. This case describes how sonography was used to identify and evaluate surgically transpositioned ovaries in a patient with rectal adenocarcinoma.
Colorectal cancer is the third most common cancer worldwide and is the fourth most common cause of death from cancer. 1 Colorectal cancer survival is related to the stage at which it is diagnosed, with later stage diagnosis having poorer survival, and is usually staged with image modalities such as computed tomography (CT) and magnetic resonance imaging (MRI). Most colorectal cancers are colorectal adenocarcinomas, which originate from the glandular epithelium of the colon and rectum. Pelvic radiotherapy (PRT) is a common treatment for patients with advanced rectal cancer. The PRT improves the oncological outcomes of rectal cancer; however, it also raises concerns regarding infertility in patients of reproductive age. Preradiation laparoscopic ovarian transposition, aka ovarian suspension, oophoropexy, or ovariopexy, is an established method which addresses these infertility concerns. Ovarian transposition is a surgical procedure in which the ovaries are displaced from the pelvis away from the radiation field, with the goal of preserving ovarian function and fertility. Following transposition surgery, sonography can be used to assess the ovaries. For ovaries transpositioned far laterally or superiorly in the pelvis, a transabdominal sonographic approach is important to use as endovaginal imaging may not be able to adequately assess surgically transposed ovaries. The following case describes a patient with rectal adenocarcinoma for whom sonography located and evaluated transpositioned ovaries.
Case Report
A 37-year-old woman, G0P0, presented to the outpatient clinic with amenorrhea and a recent diagnosis of advanced rectal adenocarcinoma. Although previous endoscopic ultrasonography (EUS), CTs, and MRIs were completed at an outside hospital, the rectal adenocarcinoma was staged uT1N1M1. This abbreviation is used to indicate that the primary tumor was confined to the rectal mucosa and submucosa, but had lymphatic invasion and distant metastasis. The patient’s previous medical history included multiple rectal resections, a partial hepatectomy and cholecystectomy for hepatic metastases, bilateral salpingectomy, and ovarian transposition in conjunction with Mirena intrauterine device (IUD) placement. The patient’s bilateral salpingectomy and lateral ovarian transposition were performed laparoscopically. To describe the surgery in brief, the fallopian tubes were removed, and the ovaries were then transpositioned to the right and left lateral peritoneum. The ovaries were marked with metallic surgical clips to assist with identifying the ovaries for future imaging examinations. Prior to the patient’s presentation to the outpatient clinic, the outside imaging studies showed the high and superficial transposition of the ovaries from their normal location, deep in the pelvis (see Figure 1). The MRI examination demonstrated a well-defined cystic lesion measuring approximately 3.2 cm in length within the left ovary, suggestive of a benign hemorrhagic cyst (see Figure 2). In addition to previous imaging, the patient had ovarian hormone labs drawn, which consisted of follicle-stimulating hormone (FSH), luteinizing hormone (LH), estradiol (E2), and anti-Müllerian hormone (AMH). These labs were confirmed to be within the normal range of accepted values. At this point, the patient was referred to the outpatient clinic for a pelvic sonogram to locate and further evaluate the transpositioned ovaries in preparation for egg retrieval, as well as to assess and classify the cystic left ovarian lesion, as seen on MRI. A transabdominal pelvic sonogram was performed using a Philips EPIQ 7 ultrasound system (Philips Medical, Bothell, Washington) with a C5-1 MHz curvilinear transducer, a C9-2 MHz curvilinear transducer, and an L12-5 MHz linear transducer. A brief evaluation of the uterus showed an appropriately placed IUD. The right ovary was located just slightly medial to the iliac crest in the right pelvis and the left ovary was slightly lower, superolateral to the bladder and just medial to the iliac crest in the left pelvis (see Figure 3). The right ovary was deemed unremarkable given the transabdominal sonographic images and measured 3.8 × 3.6 × 1.6 cm. The left ovary measured 4.4 × 3 × 3.2 cm and showed a 3.6 × 3 × 2.8 cm complex cyst with low-level echogenic debris, without internal vascularity. This was dictated by the physician as an endometrioma versus hemorrhagic cyst. These sonographic findings correlated with the previous MRI results. Both the right and left ovaries demonstrated blood flow and appeared structurally normal. The results of the sonogram and the ovarian hormone labs indicated that the patient was likely a good candidate for ovarian stimulation. The patient’s treatment recommendations, following ovarian transposition, included chemotherapy and PRT, which were discussed in a colorectal multidisciplinary clinic conference, made up of a team of more than 20 specialists. Due to the patient’s desire to undergo ovarian stimulation, the team recommended deferment of PRT and close monitoring of the patient’s stage IV rectal adenocarcinoma while undergoing chemotherapy. In addition, the patient is being monitored by oncofertility through hormone labs and ovarian reserve testing for a year in preparation for ovarian stimulation for egg retrieval.
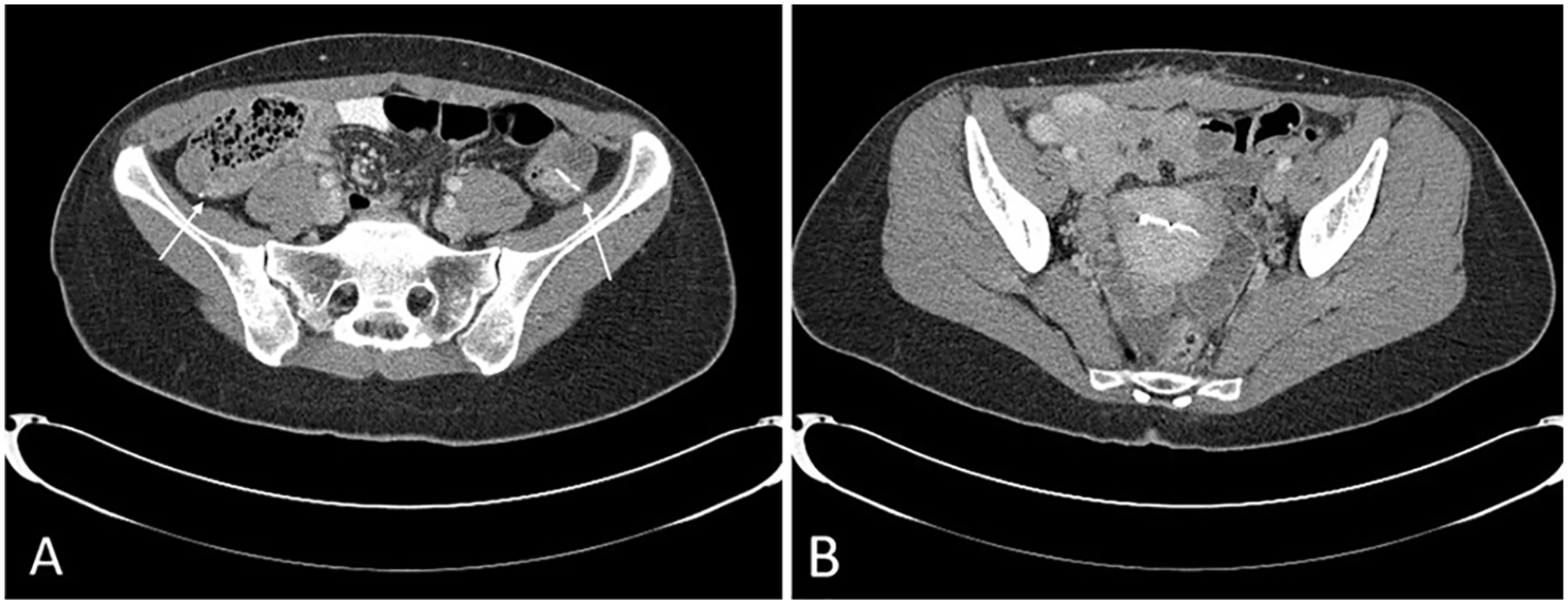
(A) Axial computed tomography image showing the high and superficial location of the ovaries marked with surgical clips after transposition (arrows). (B) Axial computed tomography image showing typical location of ovaries adjacent to the uterus with intrauterine device.
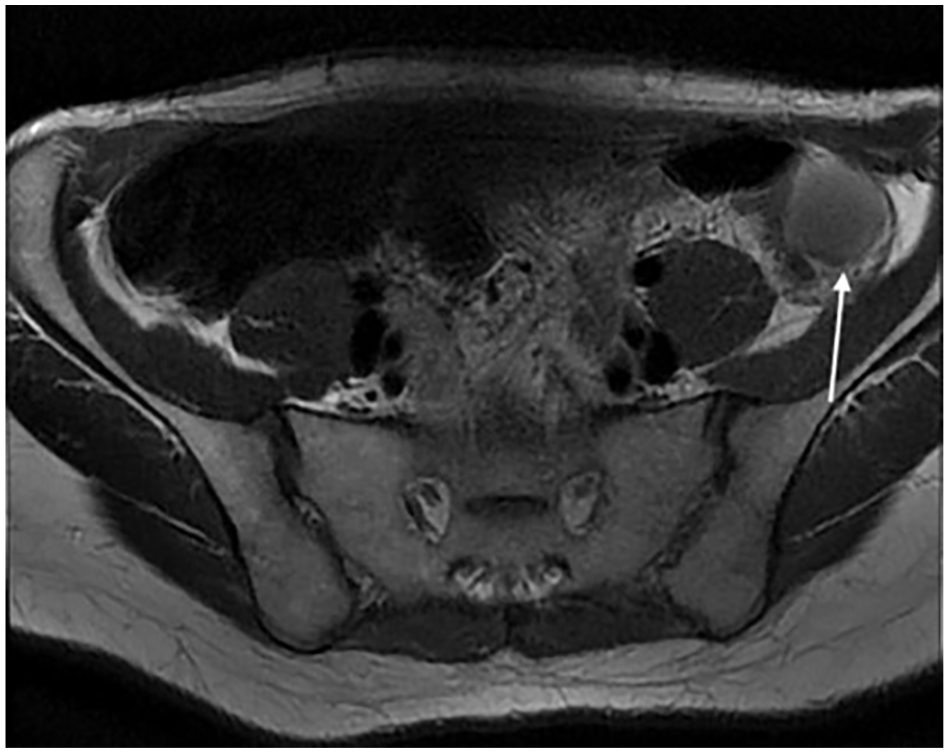
Axial magnetic resonance image showing the complex hemorrhagic cyst (arrow) on the left transpositioned ovary.
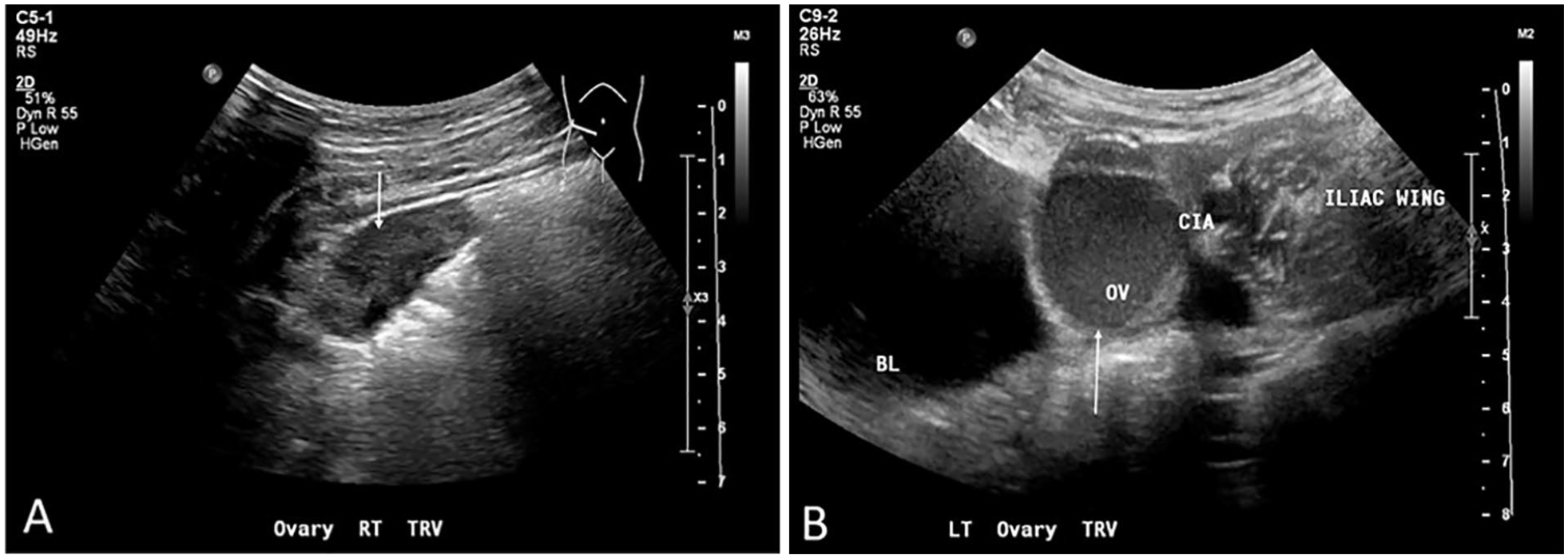
(A) Transabdominal sonogram of the right ovary (arrow) just medial of the iliac crest. (B) Transabdominal sonogram of the left ovary (arrow) superolateral to bladder and just medial to the common iliac artery and iliac crest.
Discussion
Surgical techniques for laparoscopic ovarian transposition using a lateral approach have been described in literature. While there are some variations in technique between patients, the basics of the procedure remain the same. The procedure is completed under general anesthesia and the patient is placed in the Trendelenburg position. A midline incision is made near or at the umbilicus, and the utero-ovarian ligament is transected using a vessel-sealing and cutting device (e.g., LigaSure) for mobilization of the ovaries. 2 At this point, the fallopian tubes can be removed or left intact depending on the various patient treatment plans. Each ovary is then carefully mobilized with its corresponding ovarian vessels so as to not compromise blood supply and pulled superolaterally. 2 Finally, the ovaries are permanently sutured to the peritoneum, marked with surgical clips, and the laparoscopic incision is closed. 2 While CT is the diagnostic gold standard for postsurgical pelvic imaging and to visualize surgical clips, sonography may be beneficial for further evaluation of the transpositioned ovaries and the true pelvis, from which they were removed. While there is little literature on complications within the true pelvis after ovarian transposition, the ovaries can be sonographically assessed for postoperative fluid collections, hematomas, metastatic disease, cyst formation, and vascular complications.
Ovarian insufficiency and infertility due to pelvic radiation for advanced rectal adenocarcinoma is a major concern for female patients of reproductive age. The oocytes contained within the ovaries are particularly susceptible to damage from PRT. The lethal dose of radiation for an oocyte is approximately 2 gray (Gy). The unit of measure, Gy, is the absorption of 1 joule of radiation energy per kilogram of matter. The dose of radiation, which results in complete ovarian failure, is approximately 20 Gy. 3 The average or usual dose of radiation for most pelvic, colon, and rectal cancers is 30 to 70 Gy, well above the threshold for oocyte injury. 4 Transposition of the ovaries out of the radiation field, in combination with the use of lead protective shields, placed centrally on the abdomen, can minimize the ovaries’ exposure and decrease the chance of ovarian insufficiency. 3
Careful monitoring of patient hormones in conjunction with sonographic screening of transpositioned ovaries can help to evaluate ovarian structure and function following a transposition surgery. Surgically transposed ovaries should maintain similar morphological characteristics. Sonographically, normal ovaries appear as oval-shaped hypoechoic structures that contain small anechoic follicles. 5 Following transposition, ovaries may be identified by surgical clips marking their location, although it may be difficult to visualize these sonographically. Laterally transpositioned ovaries may be identified by tracking ovarian blood vessels, which will run laterally near the iliac fossa, rather than their normal inferior course within the pelvis. 6 When evaluating the ovaries post transposition, it is vital to recognize sonographic features and abnormalities which may be associated with ovarian transposition to avoid mistaking the ovaries for other abdominopelvic lesions. Surgically transpositioned ovaries should maintain typical cyclical variations in blood flow as seen in normal premenopausal ovaries. Such cyclical variations can be noted on an arterial spectral Doppler waveform, taken within the dominant (ovulating) ovary and the nondominant ovary. The dominant ovary should demonstrate a low-resistance arterial waveform, indicating an increase in blood flow for ovulation and support of the corpus luteum cyst after ovulation. In comparison, the nondominant ovary may demonstrate higher arterial resistance waveform. 5 Bilateral ovaries should demonstrate continuous venous return with Doppler. Typical ovarian color and spectral Doppler documentation is an important predictor of functionality of transpositioned ovaries, as reduced blood supply to the ovaries inhibits function.
Transpositioned ovaries have a noted increase in cyst formation, which is thought to be due to the surgery itself. The transposition surgery often requires extensive mobilization of the vascular pedicles of the ovaries, temporarily compromising their vascular supply, causing cyst formation. 7 The cysts may be symptomatic, and require intervention, or may be asymptomatic. The incidence of symptomatic cyst formation in transpositioned ovaries has been reported to be up to 24% more than normally positioned ovaries. 7 These cysts may be follicular, corpus luteum, or hemorrhagic. Cysts with complex components may be mistaken for malignant abdominopelvic lesions or masses. 7 Transpositioned ovaries with complex cysts could resemble reactive lymphadenopathy, although lymph nodes typically contain internal flow and are extraperitoneally located near or around musculature. In one case of ovarian transposition, a large complex cyst in the ovary was mistaken for a mucocele of the appendix due to its close proximity to the cecum, while in another case, a large ovarian cyst was thought to be metastatic disease or a lymphocele. 8 The intraperitoneal location of most transpositioned ovaries can help to differentiate them from extraperitoneal and retroperitoneal masses. The use of color and spectral Doppler can help to evaluate internal vascularity of lesions and determine whether they are ovarian in origin or some other abdominopelvic lesions.
Peritoneal inclusion cysts may also be a complication of ovarian transposition surgery and can mimic other abdominopelvic lesions or masses. Associated with surgery, trauma, endometriosis, and pelvic inflammatory disease, these cysts are a result of thick postoperative scarring or adhesions due to decreased peritoneal absorption of the fluid secreted by the ovaries. The normal peritoneum absorbs the fluid from the ovaries, but when damaged by surgery or infection, its absorption of fluid is slower and can cause adhesions to form on structures within the peritoneum, such as ovaries. 9 Peritoneal inclusion cysts may cause pain and appear sonographically as fluid-filled cystic masses that can be unilocular or septated and surround one or both ovaries. 10 Although these are typically a benign finding, the septated and complex appearance of these cysts may mimic malignant lesions also found within the abdominopelvic cavity. 10
As ovarian transposition is gaining traction as a surgical method to preserve ovarian function while treating advanced cancers, the recognition of sonographic characteristics of transposed ovaries and their associated abnormalities is essential. Furthermore, it is important to have an accurate patient surgical history and understand ovarian transposition technique and location changes to avoid mistaking ovaries for other differential diagnoses that could raise suspicion for benign or malignant lesions. An understanding of surgical history can guide future treatment plans and patient care during recovery. In addition, accurate surgical history and sonographic assessment is vital for evaluating the success of ovarian transposition in patients with fertility concerns.
The success rate of ovarian transposition in literature has been variable. It has been successfully completed in many patients with advanced cervical cancer, Hodgkin lymphoma, and other malignant pelvic tumors. Described in a 2019 study, 39 reproductive-age patients with various malignancies underwent laparoscopic ovarian transposition prior to PRT. Forty percent of patients with cervical cancer, 71.4% of patients with Hodgkin lymphoma, 90% of patients with soft tissue tumors, and 100% of patients with rectal and anal cancer retained ovarian function after transposition. Overall, 67.6% of these patients with varying malignancies had ovarian function preserved. 11 The success of ovarian transposition, specifically in patients with colorectal/anal cancer, remains fairly consistent over the last few decades. In a 2017 study of 18 patients with colorectal/anal cancer, ovarian function was preserved through ovarian transposition in 67% of the patients, with a higher percentage of function retention (90%) in the patients younger than 40 years and a lower percentage of function retention (38%) in the patients older than 40 years, thus suggesting that preservation is age-dependent. 12 In addition to the age of the patient population, the success rates of ovarian transposition in literature may be accounted for by the differences in surgical technique and ovarian placement, the use of chemotherapy, the dose and delivery path of pelvic radiation, and the shielding of the ovaries from radiation. 10 These aforementioned studies suggest that laparoscopic ovarian transposition is both feasible and effective in preservation of ovarian function before and after PRT.
Conclusion
Laparoscopic ovarian transposition before PRT in patients with advanced colorectal/pelvic cancer is a surgical technique that is becoming more prevalent as a method for the preservation of ovarian function in reproductive-age patients. Sonography, in conjunction with ovarian hormone labs, can give important insights into the status of the ovaries after transposition. Thus, a keen recognition of the normal sonographic appearance of transpositioned ovaries and the other various associated abnormalities plays a crucial role in their accurate diagnostic evaluation.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
