Abstract
Objective:
To determine whether an evaluation of a wireless ultrasound transducer could be completed remotely.
Materials and Methods:
Video conferencing was used for communication between a sonographer and a minimally trained operator to allow evaluation of a flat linear array transducer that communicated via Wi-Fi with a smartphone and the probe vendor’s software. A tissue-mimicking phantom was used for quantitative testing, while scanning human models allowed qualitative assessments. Video conferencing using a tablet camera directed at the smartphone screen or transducer allowed the sonographer to view images, and guide the operator on probe positioning, and image optimization techniques.
Results:
All tests were completed during three 60-minute video conferences. Assessments of models were more challenging than tests using the phantom. The glare from ambient lighting sometimes made it difficult to view the smartphone screen. A connectivity limitation, which has since been resolved, prevented simultaneous use of video conferencing and the transducer software on the smartphone.
Conclusion:
This study confirmed that an evaluation of a wireless ultrasound transducer can be successfully performed remotely.
Key Takeaways
Video conferencing can be employed to facilitate communication between remote sites for technical collaborations.
Video conferencing can be used to enhance product evaluations by facilitating collaboration between subject matter experts and staff who may not have expertise in the technology.
Planning and preparation can reduce the amount of video conferencing required and allow participants to focus on performing time-efficient tests and tasks.
Challenges may be encountered when integrating video conferencing services and software apps that use the display of the connected device.
The testing institution is an independent, member-supported, nonprofit medical device testing, and patient safety organization. It has been performing evaluations of medical products, including capital equipment and consumables for more than 50 years.1,2 Vendors’ marketing devices that meet the prerequisites of an evaluation (i.e., are determined to be clinically and technically equivalent) are invited to voluntarily participate in evaluations. Vendors are not charged to participate in the evaluations and do not receive any compensation for their participation.
Typically, capital devices are loaned to the institution for testing, and consumables are provided at no charge. Testing is performed by subject matter experts (SMEs) in the testing laboratories located near Philadelphia, PA and in Selangor, Malaysia. When in-house testing is not feasible, for example, to evaluate magnetic resonance imaging equipment, testing may be performed at a vendor’s educational, or research and development facility, or at a health care facility that has the device.
The institution’s evaluation process includes creating a test protocol that describes test criteria and test methods, testing using industry accepted methods and test objects, as well as review of test results and drafts prior to publication. All aspects of the evaluations include input from equipment vendors, as well as internal and external clinical and technical experts. The results of these evaluations are published online and available to our institution member facilities and organizations. Evaluations performed in the United States include products that marketed in the United States.
To evaluate devices not marketed in the United States, the institution established a testing laboratory in 2019 at its facility in Selangor, Malaysia, which is staffed by two biomedical engineers. In the first year, evaluations performed in Malaysia included infusion pumps, continuous positive airway pressure devices, surgical lights, wireless handheld ultrasound transducers, and point-of-care blood gas and pH analyzers. Subject matter experts from headquarters provided on-site assistance to the Malaysia staff for all evaluations except the wireless ultrasound transducer evaluation. The wireless ultrasound transducer evaluation was performed using a combination of individual assessments by the Malaysia staff, and remote assessments using Zoom (Zoom, Zoom Video Communications, San Jose, CA) video conferencing.
This pilot project was performed to determine whether an evaluation of a diagnostic ultrasound device could be successfully completed remotely using video conferencing between a qualified sonographer instructing a minimally trained operator in the performance of the required tests.
Materials and Methods
Video conferencing was used to establish a real-time Internet communication between a sonographer located in the United States and an operator located approximately 9500 miles away in Selangor, Malaysia. A Samsung Galaxy Tab S5e (Samsung, San Jose, CA) tablet was used for video conferencing at the Malaysia site, and a Dell Latitude 7300 (Dell, Round Rock, TX) laptop was used in the United States.
The sonographer is credentialed by the American Registry for Diagnostic Medical Sonography (ARDMS) and has more than 35 years of experience in academic ultrasound research and clinical sonography. Prior to the video conferences the operator in Malaysia, a biomedical engineer had reviewed the SonoStar Uprobe-C (SonoStar Technologies, Guangzhou, China) wireless transducer operator’s manual, approximately 30 minutes of video on the use of SonoStar wireless probes, approximately 40 minutes of in-house created video training modules on ultrasound equipment performance testing, and the user manual for the tissue-mimicking phantom (ATS Model 549, ATS Laboratories, Bridgeport, CT) that would be used for performance testing.
The Uprobe-C wireless transducer (see Figure 1) was evaluated using an established wireless ultrasound transducer test protocol that had been reviewed by wireless transducer vendor representatives and external clinical experts. The transducer is a flat linear array that has a 25-mm footprint and transmits 10 MHz or 14 MHz ultrasound signals. It supports B-mode, M-mode, and combined B-mode/M-mode. The transducer communicated via Wi-Fi with an Apple iPhone XR (Apple, Cupertino, CA) smartphone that had the probe vendor’s proprietary “Wireless USG” software application, which is available as a free download from the Google Play Store and Apple App Store. The sonographer had previous experience evaluating a different model SonoStar wireless transducer that used the Wireless USG app.

The Uprobe-C wireless ultrasound transducer with needle guide attached.
The institution’s medical device evaluations include seven test categories: Performance, Safety, Workflow, Cybersecurity, Interoperability, Maintenance, and Total Cost of Ownership. The wireless transducer evaluation had a total of 66 criteria, of which 13 were not applicable to this transducer (e.g., criteria related to pulsed Doppler and color Doppler imaging, which the probe does not support). Of the remaining 53 criteria, 22 (42%) were completed, or confirmed by reassessment of the device during video conferences. The Zoom conferences were focused on test criteria that required operation of the probe and software app. Scanning gel was applied to the scanning surface of the tissue-mimicking phantom, and the sonographer verbally guided the operator through the required B-mode quality assurance tests of axial resolution, lateral resolution, dynamic range, near field (dead zone), anechoic target, penetration (sensitivity), and measurement accuracy.
During the video conferences, the Samsung video camera was directed at the iPhone screen to allow the sonographer to view the sonographic images, parameter settings, and user interface, or at the transducer to allow the sonographer to guide the operator in transducer positioning and manipulation (see Figure 2).
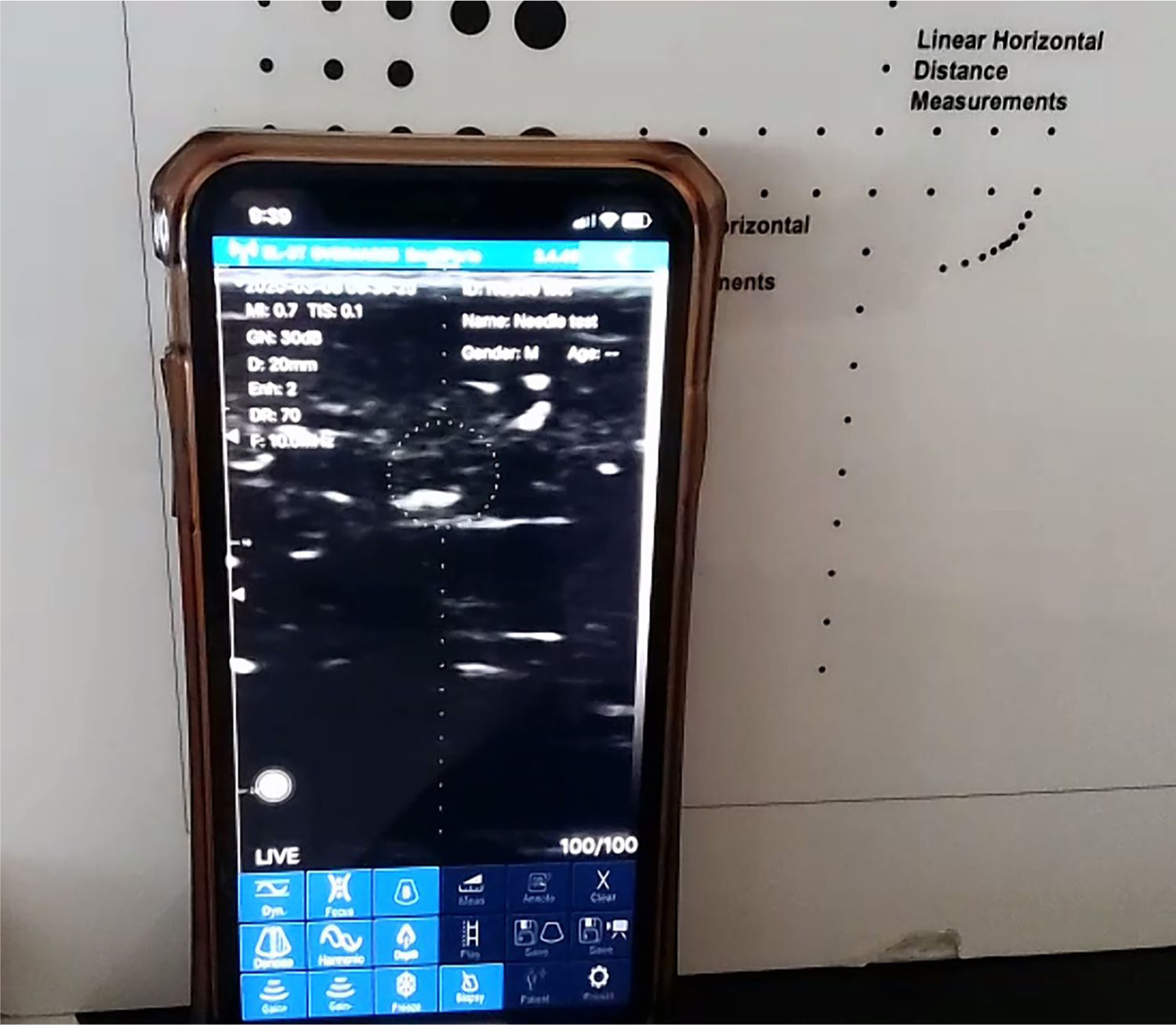
A screen capture from the sonographer’s laptop shows the operator’s iPhone and the sonographic display during tests of interventional guidance features. The iPhone is leaning against the tissue-mimicking phantom.
The interventional guidance features of the transducer, including an enhanced needle visualization mode, and graphics on the sonographic display to indicate needle location were assessed using a 20-gauge (g) spinal needle inserted into a soft tissue phantom (i.e., a boneless chicken breast) (see Figure 3).

A photo of the operator performing tests of the interventional guidance features of the wireless transducer, using a soft tissue phantom.
The performance of the transducer for imaging humans was qualitatively assessed by having the operator scan a volunteer model and herself. Prior to the scanning sessions, the model was provided with a description of the scanning procedures that would be performed, reasons for the scans, and they provided verbal consent. No protected health information (PHI) was obtained from the model or recorded during testing. Scanning gel was applied to the transducer face as an acoustic coupling. The sonographer guided the operator to perform simulated imaging examinations that included assessment of the volunteers’ peripheral blood vessels and musculature in the wrist and forearm, thyroid gland, internal jugular veins, and carotid arteries.
Findings from all assessments were recorded in a logbook and documented using still images and video clips. Data were temporarily stored on the iPhone and later uploaded to a private file sharing platform for permanent documentation of evaluation results.
The video conferences were recorded for documentation and later review as needed. After each video conference the sonographer reviewed the archived images and video clips on the laptop to compare the findings (e.g., axial resolution and the depth of penetration) with those obtained during the video conferences.
Results
Three video conferences, each lasting approximately 60 minutes, were conducted to complete the required assessments. Video conferencing provided an effective means of real-time communication between sites. There was minimal lag and temporary disruptions to the internet connections, which did not adversely impact testing.
Tests using the tissue-mimicking phantom were performed in a systematic fashion, which followed the steps and imaging techniques demonstrated in the performance testing training videos. This facilitated the testing process and allowed time-efficient data acquisition. Real-time imaging was used to optimize image quality and allow assessments of the various targets in the phantom (Figure 4). Still images documented the findings. In addition to instructing the operator through the series of phantom tests, the sonographer also explained the concepts and clinical significance of the performance tests to enhance the operator’s knowledge of the clinical utility of the modality and the ultrasound equipment testing processes.
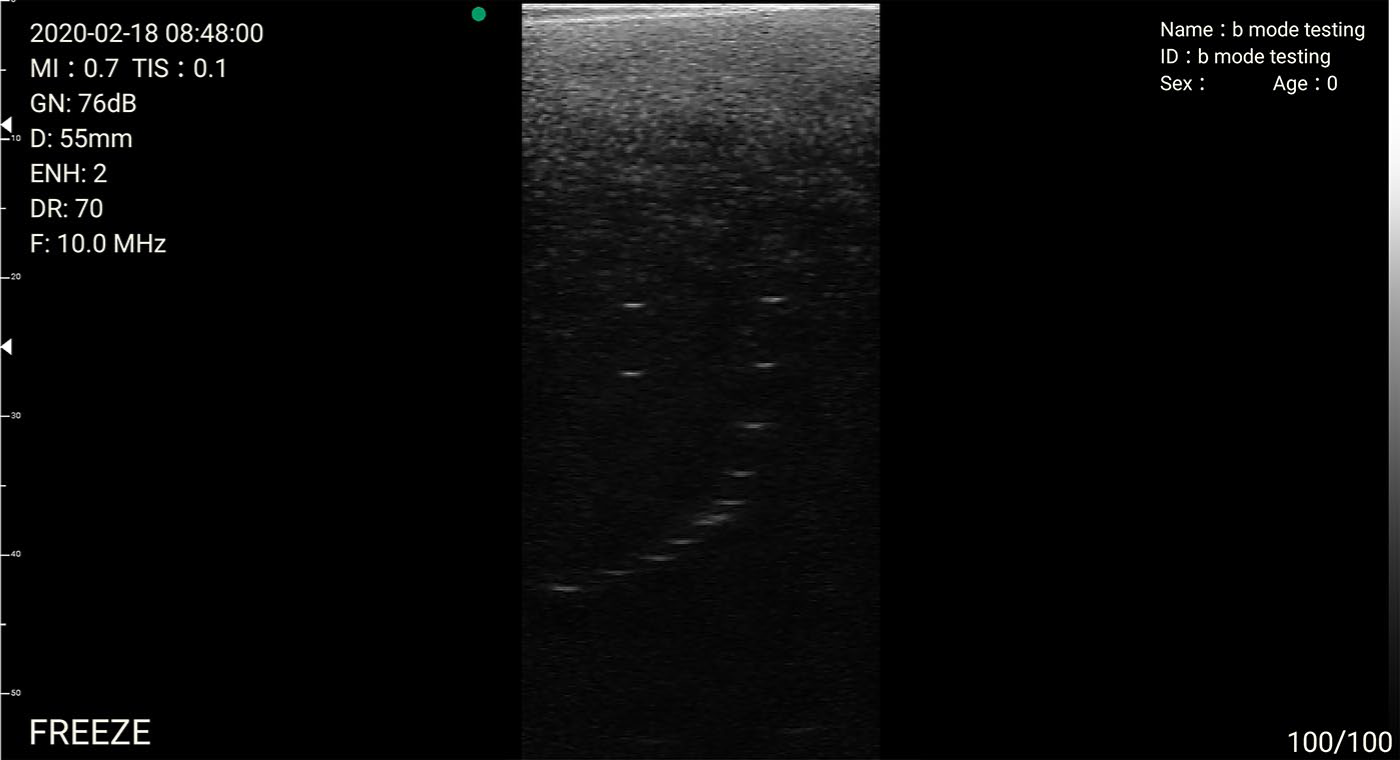
A 10-MHz image of the spatial resolution targets in the tissue-mimicking phantom.
Testing the interventional guidance features of the transducer was adequately performed by the operator. The 20-g needle could be identified on the sonographic image by the sonographer and the operator. Assessment of the transducer on humans proved to be more challenging than scanning the phantoms. The operator was not familiar with conventional radiologic image display presentations (e.g., longitudinal and transverse image planes, left and right orientations) and had never scanned a person before. Therefore, the sonographer provided a brief review of the concepts used when performing clinical exams, such as the importance of transducer pressure and positioning, adequate gel, proper ergonomics related to positioning the iPhone display in a comfortable location for viewing, and other practical aspects of scanning. While the operator performed the scanning, and held the iPhone, an assistant directed the Samsung video camera at the probe on the skin surface or at the sonographic image on the iPhone screen. It was not possible to adequately show both simultaneously. Furthermore, the transducer’s limited tissue penetration of approximately 30 mm, and the small, 25-mm-wide field of view made it more difficult for the sonographer to interpret the image based on anatomical structures. Nonetheless, adequate images were obtained to permit assessment of the transducer’s potential clinical capabilities for selected applications (Figure 5).
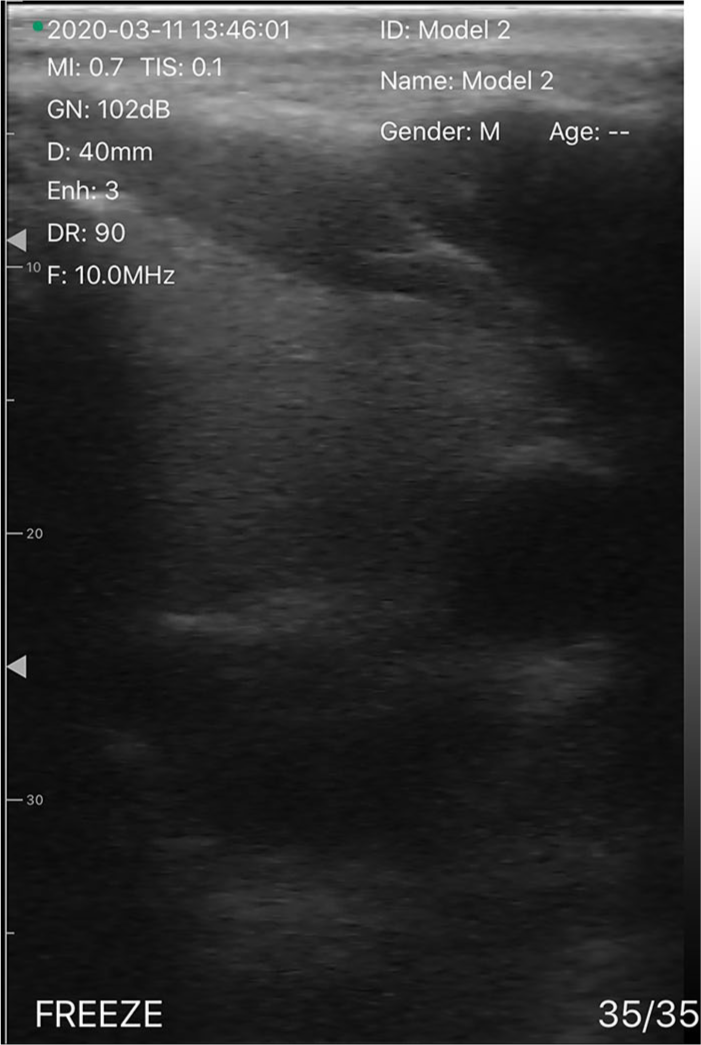
A 10-MHz transverse sonographic image of the left thyroid gland and carotid artery in a volunteer.
The Malaysia facility was not equipped to conduct tests that are typically performed in low-level lighting, such as diagnostic sonographic imaging. The lab was limited to having all of the lights on, which caused glare on the iPhone screen that adversely impacted the sonographer’s ability to view the sonographic image, or all lights off, which created difficulties for the staff performing the tests. Repositioning the iPhone and using a desk lamp to provide low-level ambient light reduced the glare and allowed the sonographer to adequately view the sonographic data on the iPhone.
Retrospective review of the still sonographic images and video clips obtained during the video conferences did not result in any changes to the test results.
Discussion
This pilot project has shown that an evaluation of a wireless diagnostic ultrasound transducer could be successfully performed remotely, between an experienced sonographer SME instructing an operator who had minimal experience with the modality. This project was not intended to determine whether a minimally trained operator could perform clinical sonography assessments.
Previous reports have described the use of tele-sonography for a range of clinical applications and in various settings including the International Space Station.3–11 Early reports described the use of off-the-shelf hardware and Voice Over Internet Protocol (VOIP) software. 12 The more recent introduction and availability of video conferencing services has now made video conferencing a commonly available and utilized technology for both personal and professional activities.
It is believed that this is the first report describing the use of video conferencing and tele-sonography to successfully evaluate a diagnostic ultrasound device. Key components that contributed to the success of this project were planning, and creation and use of training videos that demonstrated how the various phantom tests are performed, and having the sonographer participate in the video conferences. Prior to the video conferences, the operator reviewed the training videos as well as the user manual for the phantom and ultrasound transducer so that they were familiar with the concepts, terminology, and techniques that would be used during the remote tests. The operator also completed tests that did not require ultrasound expertise, without the assistance of the sonographer such as determining the battery life of the transducer, battery recharge rate, and maintenance requirements. This allowed the participants to conduct time-efficient tests that required ultrasound scanning and data interpretation expertise.
It was also beneficial that the sonographer had prior experience performing ultrasound equipment evaluations using the same phantom and had evaluated a similar wireless SonoStar probe that used the same app, as the transducer tested remotely. Familiarity with the user interface of the device facilitated such processes as optimizing image quality, performing measurements, and storing data on the iPhone.
Assessment of the imaging capabilities of the transducer using volunteer models proved to be more challenging than tests using phantoms. The operator was not familiar with conventional medical imaging display formats, which required additional instruction to allow effective communication regarding probe positioning and maneuvers. The additional training provided to the operator to address these deficiencies will be useful during future imaging device evaluations.
Because of the 12-hour difference in time, the video conferences were held in the evening for the sonographer located in the Eastern US time zone, which was the next morning in Malaysia. This sequence allowed the operator to upload the images obtained from each session to the data-sharing platform for review by the sonographer the next day.
At the time of the video conferences, it was not possible to participate in the Zoom video conference and simultaneously use the Wireless USG app on the same smart device. This required use of a third device to serve as a video camera to stream data during the conferences. This connectivity problem, combined with the limitations of the lighting in the Malaysia facility, described above lead to problems with glare on the iPhone screen that hampered the sonographer’s ability to view the ultrasound image. The connectivity problem has now been addressed; it was noted that the operator must be the host for the Zoom meeting and must be using the same device as that used as the ultrasound display. Once the Zoom meeting has begun, the operator as the host can share the iPhone screen and then launch the SonoStar ultrasound app. The ultrasound equipment display will then be directly viewable by all Zoom participants. Thus, in the future, it will be possible to share the display as seen on location, with the remote sonographer, and glare will not be a problem.
The results of this project may serve as a model for future product evaluations performed in this Malaysia facility. The laboratory there has limited staff who are generalist biomedical engineers responsible for evaluating a variety of devices, whereas the Device Evaluation staff in the United States are SMEs for specific technologies. The use of video conferencing has the potential to improve utilization of expertise and reduce the need for SMEs from the United States to travel to Malaysia to assist in product evaluations. This, in turn, will reduce costs associated with travel and may contribute to more effective evaluations that include more staff members with greater skills and expertise. Remote collaborations are also likely to become more necessary as a result of global issues such as pandemics, which restrict travel and require social distancing.
Conclusion
This pilot study confirmed that an evaluation of a wireless diagnostic ultrasound transducer can be successfully performed remotely. This can be done by having an experienced sonographer instruct a minimally trained operator in the performance of tests to assess the imaging capabilities and the user interface of the device. This project may serve as a model for future product evaluations.
Footnotes
Acknowledgements
Health Best International, a global medical product distributor, provided the SonoStar wireless probe to ECRI for evaluation.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
