Abstract
A 34-year-old patient developed an area of thrombus within her placenta. This resulted in severe fetal growth restriction which physicians felt was due to contracting and recovering from the coronavirus disease (COVID-19), during her pregnancy. Serial sonograms demonstrated a slowing of fetal growth and an area of thrombosis within the placenta. This was confirmed by pathologic examination of the placenta after delivery. Since COVID-19 and pregnancy are both known hypercoagulable states, it is vital for the ultrasonographer to recognize thrombosis in the placenta. It is also important to understand the possible pregnancy outcomes, based on the location and appearance of a nonvascular placental lesion.
Due to the newness of the coronavirus disease (COVID-19), its effects on pregnancy are not fully understood. The objective of this case study was to familiarize ultrasonographers with a potential COVID-19-related complication of developing thrombosis within the placenta.
Case Report
A 34-year-old gravida 4, para 3 patient presented to the sonography department for a fetal growth assessment sonogram, at approximately 30 weeks of pregnancy. The examination was ordered due to the patient’s fundal height measuring size less than the expected development date. She had recently recovered from the symptoms of COVID-19, which had been confirmed by a nucleic acid amplification test, which was done with a nasal swab, at 27 weeks gestation. The patient’s COVID-19 infection had been relatively mild, requiring no hospital admission, and her symptoms subsided before 30 weeks gestation. The patient had no other underlying bleeding disorders or coagulation risk factors.
At the 30-week follow-up sonogram, a newly developed structure was identified within the placenta. On this sonographic examination, the fetus measured at only the eighth percentile of growth. This was a significant change from 72% at the 20-week anatomy examination and suggested fetal growth restriction. The placenta still appeared homogenous at 30 weeks, apart from a complex nonvascular hypoechoic lesion with irregular, slightly hyperechoic borders (see Figure 1). The interpreting radiologist diagnosed this lesion as a probable area of intraplacental thrombus. This structure was not seen at 20 weeks gestation, prior to the patient’s contracting COVID-19.
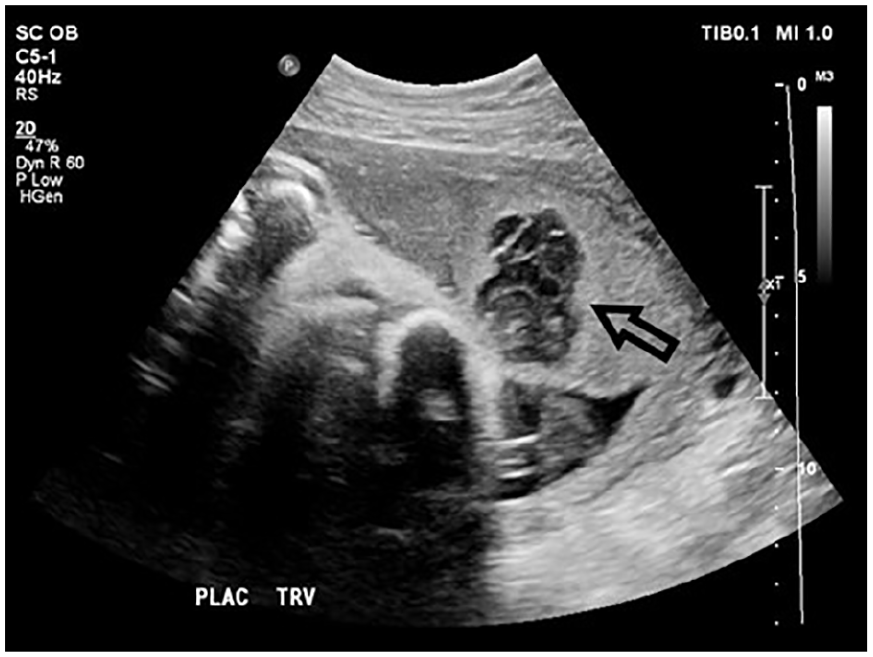
This sonographic image demonstrates an area within the placenta that contains a hypoechoic complex lesion (arrow).
A second follow-up sonogram at approximately 35 weeks gestation showed worsening fetal growth restriction, with the fetus measuring in only the fifth percentile. The intraplacental lesion appeared similar to the prior sonographic study.
The patient was referred to a maternal fetal medicine specialist, who performed another sonogram at 36 weeks gestation. The sonographic appearance of the intraplacental lesion remained unchanged. However, the fetal growth measurements dropped to the first percentile, with oligohydramnios, abnormal uterine artery Doppler, and nonreactive nonstress test. At this point, the patient was sent to the hospital for early induction. Subsequently, the patient delivered a healthy premature baby weighing 3 lb, 15 oz (1795 g), with reassuring Apgar scores of 9 at both 1 and 5 minutes of age. Postdelivery, the pathology report confirmed the presence of a 4-cm intervillous thrombohematoma near the fetal surface of the placenta. The pathology finding served as confirmatory evidence and correlated with the sonographic findings.
Discussion
Since pregnancy and COVID-19 both cause hypercoagulable states, it is important for ultrasonographers to be familiar with the risk of increased clotting that can occur in a typical pregnancy. Equally important is to understand the potential risk for different types of nonvascular placental lesions and how they might relate to hypercoagulability.
Sonography plays a vital role in differentiating between intraplacental lesions, such as thrombus or venous lakes, and retroplacental lesions. Retroplacental lesions have the potential to cause placental abruption, which is the separation of the placenta from the uterine wall. Careful attention should be paid to any placental lesion to accurately document its position, within or behind the placenta. Intraplacental lesions and retroplacental lesions are independent of each other and are associated with different risks to the pregnancy (see Figure 2). A retroplacental hematoma may appear earlier in pregnancy and result in an increased risk of premature rupture of membranes, possibly due to amniotic irritation from the adjacent hematoma. Intraplacental hematoma is defined as a nonvascular hypoechoic mass surrounded by placental tissue. It tends to appear later in pregnancy and carry a greater risk of placental insufficiency, which can result in fetal growth restriction. 1
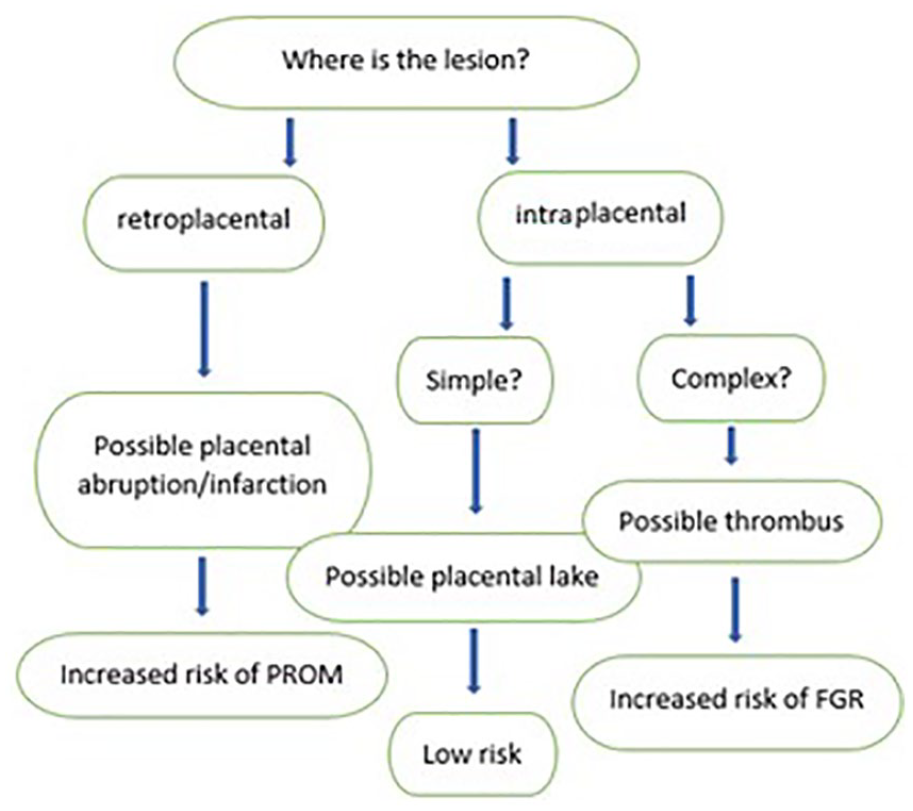
This diagram is used to assist in predicting various outcomes, due to the location and appearance of a nonvascular placental lesion.
Venous lakes are common sonographic findings in the second and third trimester placenta. A British research study reported that venous lakes are seen in approximately 18% of pregnancies and demonstrated no associated increased fetal risk. 2 Venous lakes typically appear as large anechoic nonvascular spaces within the placenta (see Figure 3). In comparison, this COVID-19 patient’s intraplacental lesion appeared complex, with fibrous-appearing strands throughout and was suggestive of thrombosis (see Figure 4).
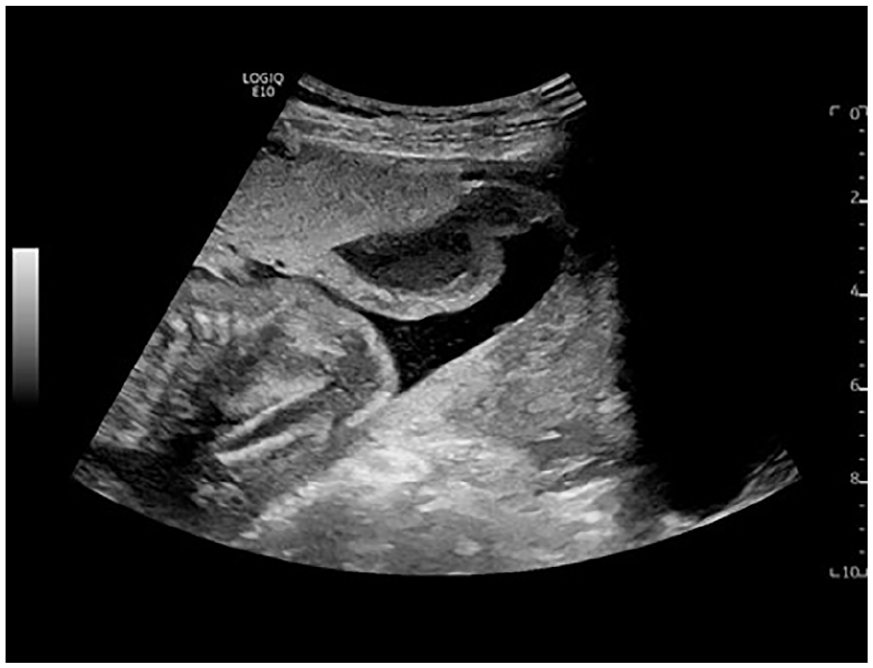
An anechoic venous lake is noted within the placenta.
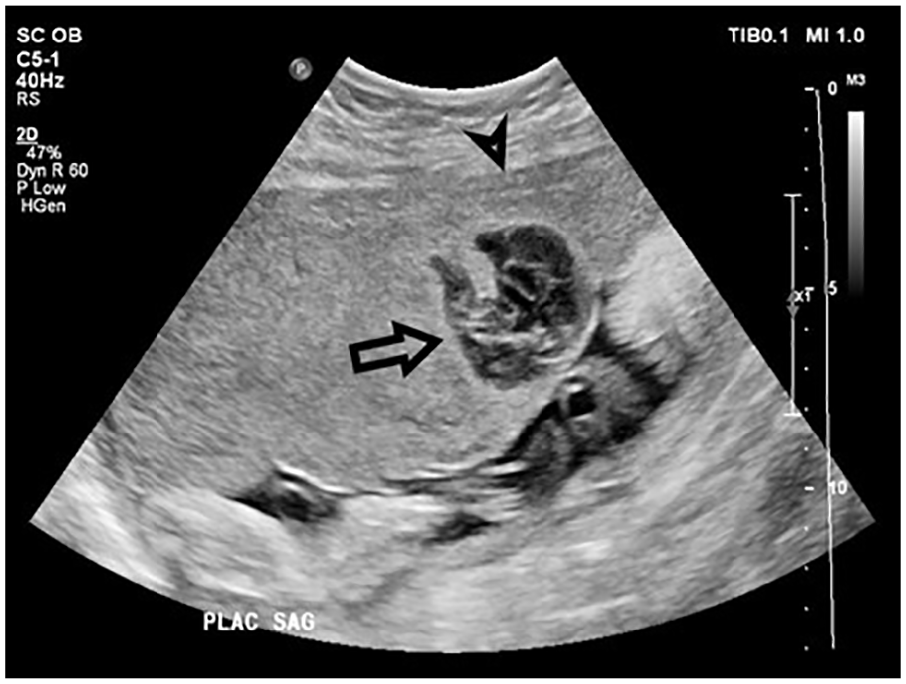
Compared with a normal placental venous lake, this nonvascular lesion (arrow) appears complex, suggesting thrombosis. It lies along the fetal surface of the placenta, rather than the maternal surface (arrowhead) which differentiates it from a retroplacental hemorrhage.
One of the accepted clinical manifestations of COVID-19 is increased coagulability. On a cellular basis, hypercoagulability from COVID-19 shares some features with other known categories of coagulopathy, but does not match any known pattern. This would suggest that COVID-19 may cause a brand-new type of coagulopathy. 3 Evidence suggests that COVID-19 causes hypercoagulation through a unique process of thromboinflammation, starting in the pulmonary vessels, triggered by the virus. 4
Increased coagulability during pregnancy is considered a normal state. Growth of the pregnant uterus puts pressure on intra-abdominal vessels, increasing the risk of venous stasis. Pregnancy hormones increase several clotting factors, which may play a role in protecting early pregnancies from spontaneous abortion and may help protect patients from antepartum hemorrhage. 5
Higher, and more concerning, degrees of thrombophilia can develop in patients with inherited coagulation disorders such as a factor V Leiden mutation or other acquired disorders, such as antiphospholipid antibody syndrome. Pregnancy complications associated with these conditions include fetal growth restriction, placental abruption, and preeclampsia. 5
Maternal COVID-19 infection, during pregnancy, has been reported to cause vascular malperfusion in the placenta and resulted in higher rates of fetal growth restriction, as well as fetal loss. 6 However, current studies tend to focus on pregnant patients who are infected with COVID-19 at the time of their delivery. Due to the recent onset of COVID-19, few pregnancy outcomes have been assessed for those patients who suffered from the virus, during pregnancy and recovered prior to delivery.
Conclusion
Until the effects of COVID-19 during pregnancy are better understood, there are some key diagnostic takeaways for ultrasonographers and sonologists. It is important to understand the hypercoagulable state of pregnancy and the associated potential risk of placental malperfusion, due to COVID-19 coagulopathy. In addition, careful diagnostic attention should be applied to the position and appearance of placental lesions, as these findings can elicit varied risks for the fetus.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
