Abstract
Traumatic peripheral nerve injury is not a common finding in the general population but is increasingly being recognized and diagnosed in trauma and emergency medicine settings. Peripheral nerve injury can cause temporary or long-term motor and sensory loss, as well as intense pain and disability, if left untreated. The cause of peripheral nerve injury is diverse and often involves soft tissue damage, fractures, and hemorrhage in cases of traumatic injury. Peripheral nerve injury does not always heal spontaneously and, depending on severity, may warrant surgical intervention and repair. Although electrodiagnosic testing and surgical exploration are considered gold standards for peripheral nerve injury, high-resolution sonographic examination supplemented by clinical findings may be of value. The use of sonography demonstrated utility in determining a strategic management timeline and the efficacy of a surgical intervention.
Peripheral nerves are prone to several disease processes including trauma, traumatic neuromas, infection, inflammation, malignant and benign tumors, and entrapment neuropathies. Sonographic evaluation of entrapment neuropathies is well documented in the literature but there is little sonographic representation of acute, traumatic peripheral nerve injury (PNI), especially in the sciatic nerve, the largest peripheral nerve in humans. Although uncommon in the general population, retrospective studies suggest a prevalence of major peripheral traumatic nerve injury somewhere between 1.8% and 2.8% in trauma populations.1,2 In addition to electrodiagnostic testing and magnetic resonance imaging (MRI), high-resolution sonography is quickly emerging as an important modality for diagnosis and management of traumatic peripheral neuropathies. Several studies have reported that sonographic findings of PNI modified management beyond electrodiagnostic findings in 43% to 58% of patients, especially in cases of acute PNI. 3 This is underscored where extensive soft tissue damage may complicate clinical and electrodiagnostic examination. 4 Although useful in determining specific nerve involvement, electrodiagnostic studies are often uncomfortable for the patient and limited in their ability to distinguish between degrees of PNI defined by Seddon and Sunderland. 5 Determining the degree of PNI is crucial in determining if surgical intervention is warranted. The following case study illustrated the limitations of MRI and electrodiagnostic testing in the setting of penetrating PNI. It also presents a rare sonographic evaluation of partial transection of the sciatic nerve.
Case Study
A 24-year-old man presented to the emergency department with a gunshot wound to the left thigh with arterial bleeding. Upon initial evaluation, the left foot was cold with a lack of pulse and motor function. There was an expanding hematoma near the sight of the wound in the thigh. Emergent left thigh surgical exploration was performed. The patient underwent hemorrhage control, left superficial femoral artery debridement and repair with reversed great saphenous vein interposition graft, left superficial femoral vein debridement and repair with non-reversed great saphenous vein interposition graft, and four compartment fasciotomies in the thigh. There was substantial soft tissue damage and although the sciatic nerve was not visualized during surgery based on surgical reports, there was documentation that it was likely in the trajectory of the bullet. Five days post surgery, the patient exhibited motor and sensory deficits in the left lower leg and foot. At this time, new concern for sciatic nerve injury warranted sonographic evaluation of the left lower extremity rather than MRI due to concerns for retained shrapnel.
Sonographic evaluation of the sciatic nerve was performed using a Philips iU-22 ultrasound system (Philips Medical, Bothell, WA) with a L12-5 MHz, L9-3 MHz, and C5-1 MHz transducer, depending on the depth at which the nerve traversed the posterior leg and thigh. Imaging revealed focal thickening and increased echogenicity of the sciatic nerve proximal and distal to the region of the gunshot wound discernable by an open wound and gauze packing. At the level of the packing, the nerve contour was irregular indicating disruption of the epineurium and an anechoic cleft was noted in longitudinal and transverse views, most likely representing partial thickness transection (see Figures 1 and 2). The hypoechoic defect could represent intraneural or perineural fluid, frequently seen with nerve compression, but more likely represented intraneural bleeding given the irregularity of the nerve contour and clinical history of penetrating trauma. Full thickness transection could not be excluded due in part to limited visibility at short segments of the nerve from acoustic shadowing and ringdown artifact posterior to small pockets of air in the gauze packing. An MRI evaluation was recommended to provide additional definitive information in regard to the extent of nerve injury. Diagnostic certainty outweighed the relatively low risk of MRI contraindications in this particular case. MRI findings were consistent with sonographic findings demonstrating circumferential, segmental thickening and indistinct margins of the sciatic nerve and possible low-grade partial laceration. Despite consistent ultrasound and MRI findings, conservative management was sanctioned, and surgical exploration was not yet warranted. The patient was scheduled for electromyography (EMG) testing in 4 weeks to assess nerve function. EMG evaluates electrical activity to a muscle. Abnormal EMG results are typically seen in PNIs that reduce the number of functional axons.
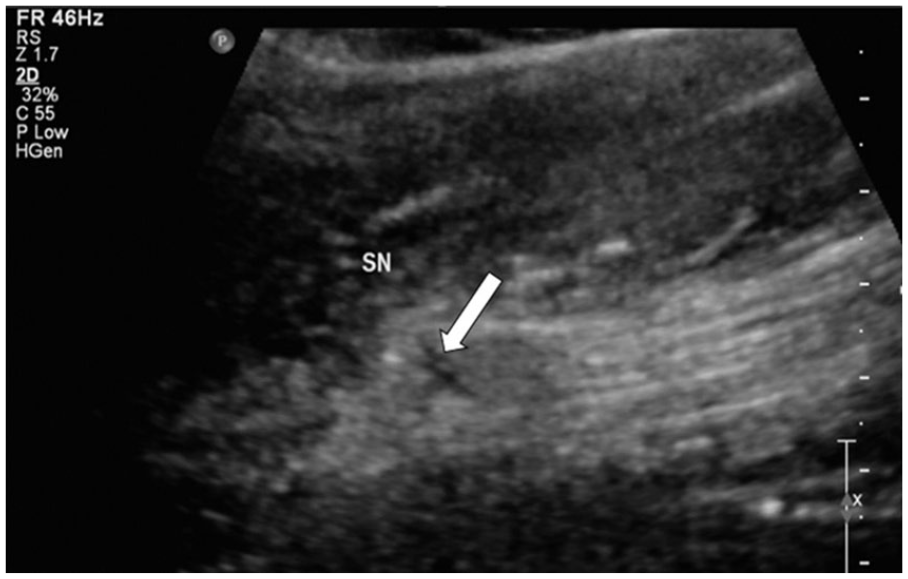
Longitudinal image of the sciatic nerve (SN) at the area of the gunshot wound in the posterior mid-thigh illustrating an anechoic cleft or defect (arrow). There is also focal thickening of the nerve contour and hypoechoic change in echogenicity at the retracted nerve ends on either side of the defect.
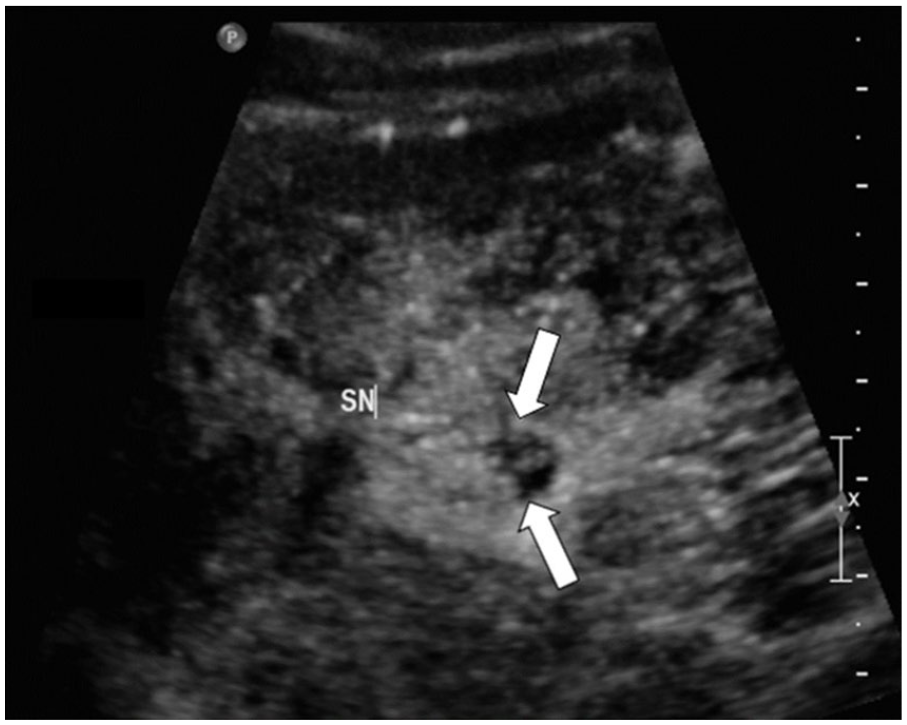
Short axis image of the sciatic nerve (SN) and anechoic cleft (arrows). Note the irregular contour of the encapsulating epineurium.
Upon return for EMG testing 4 weeks post injury, the patient endorsed stabbing pain in the left foot and continued motor function deficits in the ankle and toes. EMG results were abnormal, revealing the absence of left sural sensory and peroneal motor responses. Unfortunately, the patient refused testing of the left tibial motor nerve likely due to discomfort. These findings suggested severe left sciatic nerve injury; however, due to limited and incomplete testing, the degree of nerve continuity could not be fully assessed. Classifications of nerve injuries were discussed with the patient, as well as their potential progressions and outcomes. The patient and the plastic surgery team opted once again for conservative management considering lack of clear evidence for nerve transection. Conservative management gives the nerve an opportunity to regenerate on its own.
EMG was performed again 3 months later revealing abnormal and more definitive results. Findings included severe left sciatic neuropathy above the branch innervating the short head of the biceps femoris muscle which is consistent with the location of the gunshot wound. There was no definite electrophysiological evidence of sciatic nerve continuity to the left lower leg or foot. Left superficial fibular and sural sensory responses, as well as left fibular and tibial motor responses were absent. All of these nerve branches stem from the sciatic nerve below the level of the gunshot wound (Figure 3). Six months following the initial injury, surgical exploration and debridement of the left posterior thigh revealed significant nerve injury, scar tissue, and a neuroma of the sciatic nerve at the area of interest. There was a sciatic nerve tibial bundle gap of 4.5 cm and a sciatic nerve common peroneal bundle gap of 5.5 cm, supporting clinical, sonographic, MRI and EMG findings. A cable grafting with the autologous left sural nerve was used to bridge these gaps.
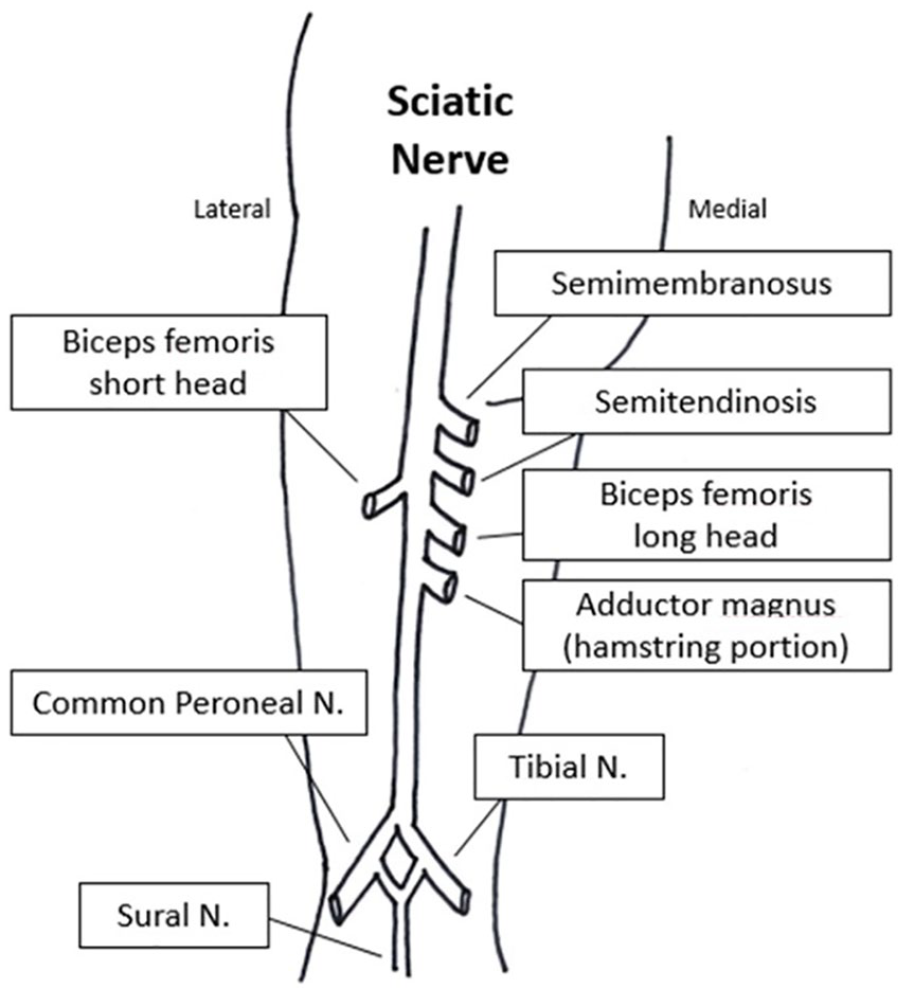
Sciatic nerve anatomy and innervation of lower leg muscles.
Discussion
Originating from the anterior and posterior divisions of the lumbosacral spine and extending down the posterior compartment of the thigh into the leg and foot, the sciatic nerve is the largest peripheral nerve in humans. The sciatic nerve plays a major role in mobility providing motor innervation to the muscles of the posterior compartment of the thigh responsible for knee flexion and hip adduction. Proximal to the popliteal crease, the sciatic nerve splits to become the tibial nerve and the common peroneal nerve, responsible for plantarflexion and dorsiflexion of the foot, respectively.
The course of the sciatic nerve can be reliably identified with sonographic evaluation in the posterior compartment of the thigh as it courses proximal to distal beneath the piriformis near the subgluteal fold all the way to the popliteal crease. The sciatic nerve can be differentiated from surrounding tendons and soft tissue as it runs parallel and superficial to the popliteal artery and vein in the popliteal fossa (Figure 4). In the transverse plane of the popliteal fossa, the sciatic nerve has a round, “honeycomb” appearance with small hypoechoic areas representing fascicles each encased in perineurium, and the hyperechoic background of the surrounding connective tissue (see Figure 4). Everything is bundled together in the encapsulating epineurium. In longitudinal, the sciatic nerve has a cable-like appearance and maintains its cross-sectional area unlike surrounding tendons which transition to hypoechoic muscle at their myotendinous junctions (see Figure 5). Another way to differentiate the sciatic nerve from surrounding tendons in cross-section is by toggling the transducer. Tendons will produce anisotropy to a greater degree than nerves.6,7 Ideally, a linear transducer with a high frequency should be utilized to visualize peripheral nerves. It is recommended that a 15 to 18 MHz transducer is used for more superficial nerves and 8 to 12 MHz for deeper nerves such as the sciatic.6,8 However, a lower frequency curved transducer may be necessary to accommodate for patient habitus and nerve depth. In this particular case study, a Philips C5-1 transducer was utilized to visualize the sciatic nerve through the posterior compartment of the thigh due to depth of the nerve and surrounding soft tissue swelling and edema.
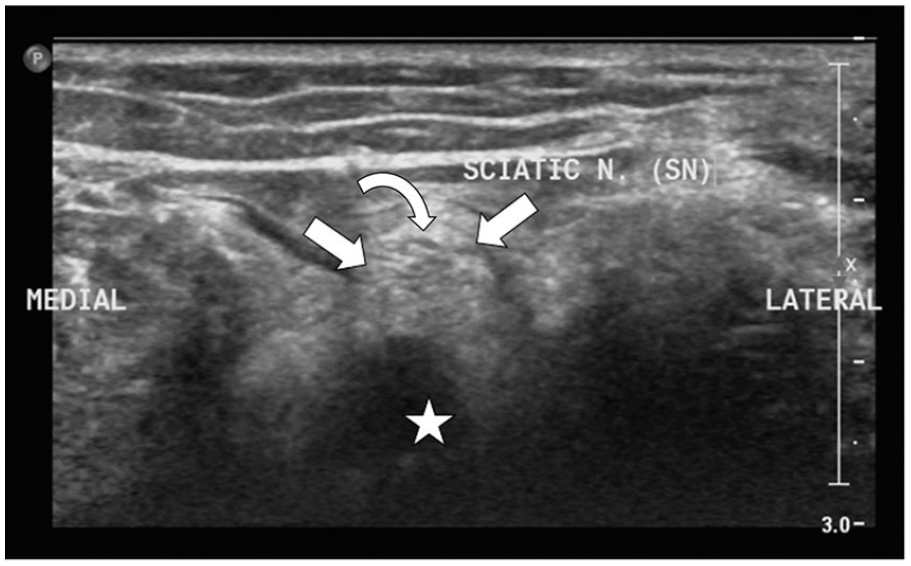
Short axis image of the popliteal fossa illustrating the superficial position of the sciatic nerve and encasing epineurium (open arrows) compared with the popliteal vein (star). Notice the “honeycomb” appearance of the sciatic nerve (SN) with more hypoechoic areas representing individual fascicles (curved arrow).
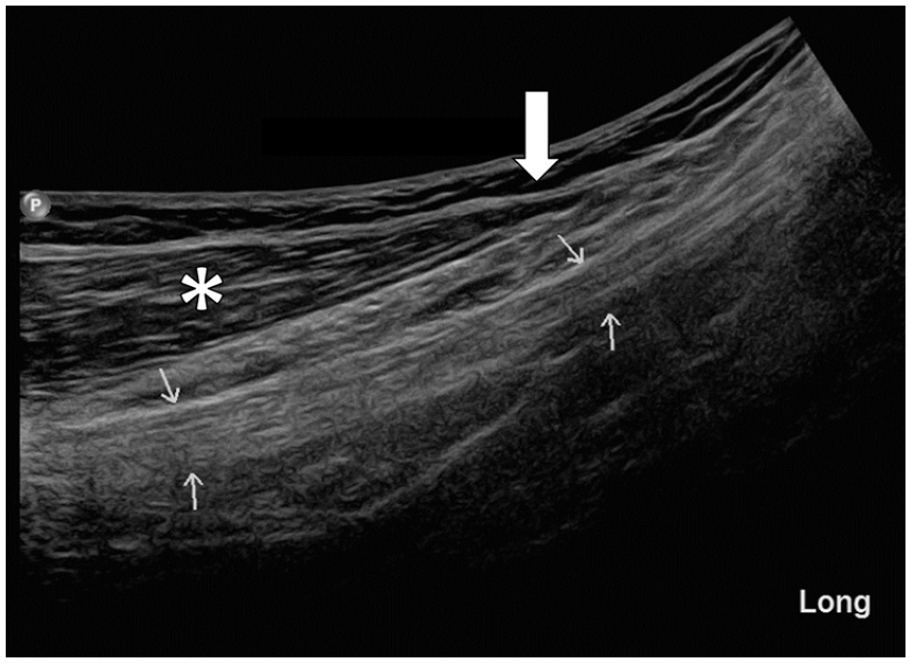
Long axis image accessed from the posterior lateral thigh of normal sciatic nerve (arrows) distal to the gun shot wound. Note the cable-like appearance and consistent in cross-sectional area of the nerve (arrows) compared with the tapering appearance (open arrow) of biceps femoris muscle (asterisk) at the musculotendinous junction.
Like other peripheral nerves, the sciatic nerve is susceptible to trauma, traumatic neuromas, infection, inflammation, malignant and benign tumors, and entrapment neuropathies. Characterizing PNIs and determining physiologic changes secondary to injury is important for optimal treatment and recovery. Some types of PNIs may heal on their own while others require surgical intervention and repair. The Seddon and Sunderland classifications are the most commonly used classifications for PNI.9,10 Seddon classified nerve injuries into three categories according to the destructive forces involved. These categories include neuropraxia, axonotmesis, and neurotmesis. Neuropraxia is the first degree of PNI involving mild and temporary symptoms and no loss in nerve continuity. Axonotmesis is considered second degree PNI, typically involving crush injuries and disruption of the axon and myelin sheathing. The perineurium and epineurium remain intact, thus maintaining anatomic nerve shape and continuity. Neurotmesis is the most severe form of PNI often involving penetrating and pulling mechanisms and is associated with nerve transection. There is permanent functional loss, and surgical intervention is warranted. Sunderland classified nerve injuries into five, more detailed, categories according to physiologic changes and tissue damage. These classifications range from first-degree injury, equivalent to neuropraxia and partial disruption in nerve conduction, to fifth-degree or complete nerve transection requiring surgical repair. Together, these classifications can be helpful in determining management. Sonography is limited in its ability to visualize individual axons, myelin, and endoneurium.11,12 However, advances in resolution have allowed for visualization of individual fascicles, perineurium, and epineurium (see Figure 4). Changes in echogenicity of nerve connective tissue, disruption of normal shape and cross-section areas of the nerve, interruption, and discontinuity of fascicles and/or the epineurium, swelling of retracted ends, and presence of intraneural bleeding can all indicate PNI and help to determine injury classification.
Electrodiagnostic studies are also an essential part of any clinical workup for neuropathology; however, there are major limitations including accessibility in emergency and tertiary settings and delays in PNI treatment in cases of traumatic, acute injury. In cases of extensive soft tissue damage, a patient’s treatment plan is often complicated by the critical need to stabilize hemodynamics, manage bone injury, and prevent infection. These complications can greatly hinder clinical assessment. A study by Noble et al concluded that the mean time from hospital admission to PNI diagnosis was 6.7 days. 2 This case study illustrates the all-too-common delay in PNI evaluation and diagnosis in emergency settings. Another disadvantage of electrodiagnostic studies is that, although accurate in their assessment of specific nerve involvement, they are often unable to distinguish axonotmesis from neurotmesis and they are unable to precisely localize a lesion which can be especially problematic in cases of multiple injury sites, entry points, ballistics, and shrapnel.5,13 Furthermore, relying on electrodiagnostic testing can make for a conservative and lengthy management plan. Conservative management is not uncommon in PNI as injured nerves undergo a complex physiologic process known as Wallerian degeneration before regrowth begins. 14 There is typically an initial inflammatory and regenerative response 4 to 7 days post injury and a second, more sustained response around 4 weeks. 15 However, if and when regrowth begins, studies have shown that axon regeneration only proceeds at a rate of about 1 mm per day in humans over long periods of time and poor functional outcomes are often associated with delayed surgical repair.16,17 Advances in initial imaging could save a patient months of hopeful, and often painful, conservative management.
Imaging is used to localize peripheral nerve lesions and their relationship to surrounding soft tissue. Sonography and MRI are the most commonly used methods for visualizing peripheral nerves. Retrospective studies have shown that high-resolution sonography may actually be more sensitive than MRI as an initial imaging modality in traumatic PNI.7,8,18 When combined with electrodiagnostic testing, high-resolution sonography is quickly emerging as an important tool for management and surgical planning in cases of penetrating PNI. Several studies have reported that sonographic findings of neuropathology modified management beyond electrodiagnostic findings in 43% to 58% of patients, especially in cases of acute PNI.3,4 Research has demonstrated the advantages of high-resolution sonography in evaluating acute gunshot and combat-related PNI and its role in treatment and surgical planning outcomes.5,19,20 Ultrasound is cost-effective, efficient, portable, and allows for real-time evaluation. Sonographic findings provide valuable information for refining the potential for surgical intervention by assessing specific localization of nerve injury, degree of nerve transection or discontinuity, and the presence of retained foreign bodies and neuromas.5,11 It should be noted, however, that although sonographic findings show high positive predictive values in characterizing peripheral nerve trauma, surgical inspection is still considered the gold standard. 20 This case study illustrates the evolving role of high-resolution sonography in musculoskeletal ultrasound and peripheral nerve evaluation.
Conclusion
In the presence of clinical signs and symptoms of neuropathology following traumatic or penetrating injury, PNI should be considered. High-resolution sonography can provide valuable information in the characterization of PNI, therefore guiding management, treatment, and surgical planning. Following clinical assessment and identification of expected peripheral nerve involvement, sonographic evaluation requires knowledge of peripheral nerve anatomy and surrounding soft tissue and bony anatomy. The peripheral nerve in question should be identified and evaluated in longitudinal and short axis views with careful consideration of changes in echogenicity, cross-sectional area, and continuity of individual fascicles and the epineurium. It is important to note that PNI may be subtle and less conspicuous than the depiction in this case study. Higher resolution linear transducers are ideal for evaluating individual fascicles and more minute physiologic changes; however, a lower frequency curvilinear transducer may be necessary to penetrate deeper peripheral nerve anatomy such as the sciatic nerve. Although rare in the general population, sonographers should be aware of the signs, symptoms, and sonographic features of PNI as the scope of musculoskeletal sonography continues to grow. Sonographers should expect to see increasing orders for evaluation of PNI not only in trauma and emergency settings but also in outpatient clinical settings following up traumatic injury where nerve assessment was complicated or postponed in initial emergency assessments.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
