Abstract
According to previously published papers, neurovascular injuries seem to be the most unfortunate complications after surgical procedures. In this report, we present our therapeutic approach to iatrogenic injury of the posterior tibial nerve that occurred during ankle arthroscopy in a 24-year-old patient. The outcome of the therapy was a full sensory return and partial motor return (S4 and M3 according to the Medical Research Council Grading System for Nerve Recovery). Our patient was able to resume her typical training. In comparison with available reports, our therapeutic approach enabled earlier functional recovery after nerve injury. While sensory return is beneficial, motor improvement is also important. However, we are conscious of the poor functional outcomes reported by other researchers.
Keywords
Background
According to Seddon’s classification, the most serious peripheral neural injury is neurotmesis, which means that the neural continuity is interrupted and its sheath is destroyed. 1 This results in symptoms associated with damage to the peripheral nervous system, such as motor palsy of the muscles innervated by the particular nerve, muscle atrophy, and loss of sensation in the dermatome supplied by the injured nerve.1,2 In such cases, spontaneous repair seems to be impossible and a proper combination treatment is necessary to enable satisfactory functional recovery. 1 In many cases, the nerve injury is a complication related to the primary injury; however, a subset of patients sustains an iatrogenic nerve injury attributable to treatment of other dysfunctions.3,4
With respect to sensory function, the cutaneous branches of the tibial nerve innervate the sole of the foot and toes and the posteromedial aspect of the shin. With respect to motor function, the tibial nerve and its distal branches innervate the posterior group of shin muscles, flexors of the foot and toes, abductor hallucis, and intrinsic muscles located on the plantar surface of the foot. This case report describes a patient with an iatrogenic injury to the posterior tibial nerve that occurred during ankle arthroscopy. Although such injury is one of the major complications of this surgical procedure, we believe that our case report is the first to combine two possible treatment options (both surgical and conservative) for such patients. We hope that this case will help other medical professionals to achieve more effective treatment planning with more significant improvements in their patients’ conditions.
Ethics and consent
This report describes a therapeutic procedure designed for our patient. We did not apply for bioethics board approval for this therapeutic procedure because we did not use an experimental or high-risk method of therapy. The patient participating in the study provided written informed consent for the therapeutic procedure and for publication of her case.
Case presentation
A 24-year-old female hurdle runner was eligible for ankle arthroscopy due to ankle pain exacerbated by physical activity. The pain was not accompanied by swelling, but the patient exhibited loss of ankle active range of motion for plantar flexion and dorsal extension. The sensation and blood supply were normal. The patient had a history of steroid injections into the affected ankle. The surgery was performed through two portals for ankle access; the posteromedial portal was located 1.5 cm dorsal to the posterior margin of the medial malleolus, and the posterolateral portal was located 2.5 cm dorsal to the posterior margin of the lateral malleolus. Immediately after the surgery, the patient was unable to perform plantar flexion of either the toes or foot, and she felt neither touch nor pain in the plantar aspect of the foot. The patient was admitted to our medical center for evaluation of these symptoms. An ultrasonographic examination performed 3 weeks after the surgery revealed a pathological condition of the nerve. An electromyographic (EMG) examination confirmed an injury of the posterior tibial nerve characterized by axonal demyelination changes in both the sensory and motor fibers (Table 1).
Results of the preoperative electromyographic examination.

MCV, motor conduction velocity; SCV, sensory conduction velocity; Lat., latency; Ampl., amplitude; Stim., stimulation
Interventions
Surgical procedure
Open surgical revision of the posterior tibial nerve was performed 40 days after the ankle arthroscopy. The previous posteromedial arthroscopic portal was elongated into an incision. After the skin incision, massive scar tissue entrapping a tendon of the flexor digitorum longus was discovered. During removal of the scar tissue, the tendon of the flexor digitorum longus was liberated, inspected, and sewed in the area of the tendon fissure. The posterior branch of the tibial nerve and its branch innervating the heel were then identified. No injury was present at the junction, but the surgeons confirmed an incision of the posterior tibial nerve located proximal to the junction, reaching three-fourths of the neural width (Figure 1). The injured nerve was repaired by a perineural suture, and the continuity of the tibial nerve was restored (Figure 2). Immediately after the surgery, the patient exhibited visible improvement in her ankle and toe movements. After the surgical procedure, the patient was prescribed cyanocobalamin, pyridoxine, thiamine, and galantamine hydrobromide.

Intraoperative view of the injured posterior tibial nerve.

Intraoperative view of the sewed posterior tibial nerve.
Physiotherapy
After revision of the injured nerve and removal of sutures, physiotherapy was applied. At the very beginning of the rehabilitation process, we assessed the patient’s range of feeling. The sensory examination was performed with the patient’s eyes closed and was based on area localization. 5 We assessed the patient’s feeling of touch and compared the operated and uninjured sides. The patient was asked to evaluate the feeling on a percentile scale where 100% represented the feeling in the uninjured foot.
Our physiotherapy program began with manual mobilization of the scars and surrounding tissues, stimulation by soft tissue massage, and work at the neural interface for nerve mobilization, We used techniques of manual therapy for mobilization of the longitudinal and transverse foot arches, gentle grade I and II mobilization of the talocrural and subtalar joints, and mobilization of the tarsus in the restricted translatoric direction [accessory movement]). We also performed slider neural mobilization 6 for the tibial nerve according to the instructions provided by the Neuro Orthopaedic Institute. We added some anti-edematous actions to increase the axonal transport speed, including lymphatic kinesiology taping applications, lymphatic drainage, and simple non-weight-bearing lower limb exercises to arouse the muscles’ pumping activity. The patient was instructed to keep her foot and shin warm.
Three weeks after the surgery, new procedures were added to the protocol. We began grade III manual mobilization techniques for the neural interface and physical exercises involving both lower limbs using unstable surfaces and multiple types of structures to enhance exteroceptive stimuli. The patient was encouraged to perform the exercises with plantar flexion of the foot. The protocol was supplemented with infrared light exposure (red filter, 15 minutes daily; the intensity was chosen according to the patient’s feeling) and low-level laser therapy (LLLT) (820-nm laser wave and three areas of stimulation: the space around the nerve injury, the motor points of the tibial nerve located proximal to the point of injury, and the projection of the tibial nerve located distal to the point of injury2,7).
The second assessment of the range of feeling was performed 4 weeks after the nerve revision. Scar tissue mobilization was performed until full mobility of the soft tissues around the surgical wounds was achieved. We increased the level of physical activity during the strengthening exercises of the lower extremities and used the elements of proprioceptive neuromuscular facilitation for work in functional conditions. The protocol was supplemented with electrical stimulation involving low-frequency stimulation with a triangular impulse and the following procedural sequence: 3 minutes of stimulation and 3 minutes of break repeated three times. With respect to the electrode arrangement, the anode was located above the medial malleolus, and the cathode was placed at the plantar surface of the foot around the head of the first metatarsal bone. The intensity was chosen according to the minimal visible muscle reaction. Hydrotherapy was also applied in the form of hydro massages of the lower extremities (water temperature of 38°C, duration of 15 minutes). The patient had physiotherapy appointments three times a week, during which she was instructed in neural mobilization strategies so that she could perform them on her own. The electrical and laser stimulation and hydro massage were performed five times a week.
Ten weeks after the neural repair, another sensory and motor examination was done. During physiotherapy, the patient’s exercises progressed until she was performing unilateral tasks to strengthen the lower extremity kinematic chains. She also started dynamic tasks beginning with mini jumps and plyometric exercises, first on a stable surface and later on an unstable surface. Manual therapy included tensile loading techniques in different positions of the tibial nerve.
Twelve weeks after the surgery, the patient began performing jogging and sports-specific tasks including dynamic jumps and activities with a dynamic change in the movement direction. The patient was encouraged to obtain an at-home electrical stimulation set, and she performed this procedure twice a day. We found the motor point for the abductor hallucis muscle, and one daily procedure was performed with an intensity strong enough for visible abduction motion. The second procedure was dedicated to the toe flexors.
Six months after the neural repair, the patient was allowed to resume her typical training and ended the physiotherapy at our medical center.
At the 9-month follow-up, the patient was able to perform six trainings a week. She complained of a weaker feeling in her operated foot than uninjured foot, and she sometimes still experienced pain in the transverse arch of the foot and in the space between the fourth and fifth metatarsal bones. She also exhibited symptoms of patellofemoral pain syndrome in her operated leg.
Outcomes
We continuously assessed the patient’s condition during the treatment. The first physiotherapeutic examination was performed 2 weeks after the surgery at the beginning of physiotherapy. The subsequent examinations were performed 4, 10, 16, 24, and 36 weeks after the neural repair. The patient was tested for sensory and motor function of the tibial nerve.
Two weeks after the surgery, the motor function of the muscles innervated by the affected nerve was assessed as 2/5 for plantar flexion of the foot and 3/5 for flexing the toes. The reaction of the skin of the plantar surface of the foot suggested active hyperesthesia.
The result of the sensory examination at the 4-week follow-up is depicted in Figure 3. The motor function of the muscles innervated by the affected nerve was assessed as 2/5 for plantar flexion of the foot and 3/5 for flexing the toes. We still observed the presence of dysesthetic pain, assessed by the patient as 9 on the numeric pain scale.

Result of the sensory examination 4 weeks after the neural revision. The lined area is the area of extant feeling.
The control EMG examination performed 12 weeks after the neural revision confirmed improvement in the sensory fibers of the posterior tibial nerve (Table 2). A complete assessment of motor function was impossible due to the location of the surgical scar. During the follow-up examination, the patient presented 5/5 muscle strength for flexing the foot and toes and 1/5 for abducting the hallux.
Results of 12-week follow-up electromyographic examination.

MCV, motor conduction velocity; SCV, sensory conduction velocity; Lat., latency; Ampl., amplitude; Stim., stimulation
At 16 weeks postoperatively, manual assessment of muscle strength revealed 5/5 for flexing the foot and toes and 1/5 for abducting the hallux. The result of the sensory examination is depicted in Figure 4. The dysesthetic pain assessment result was 4/10.
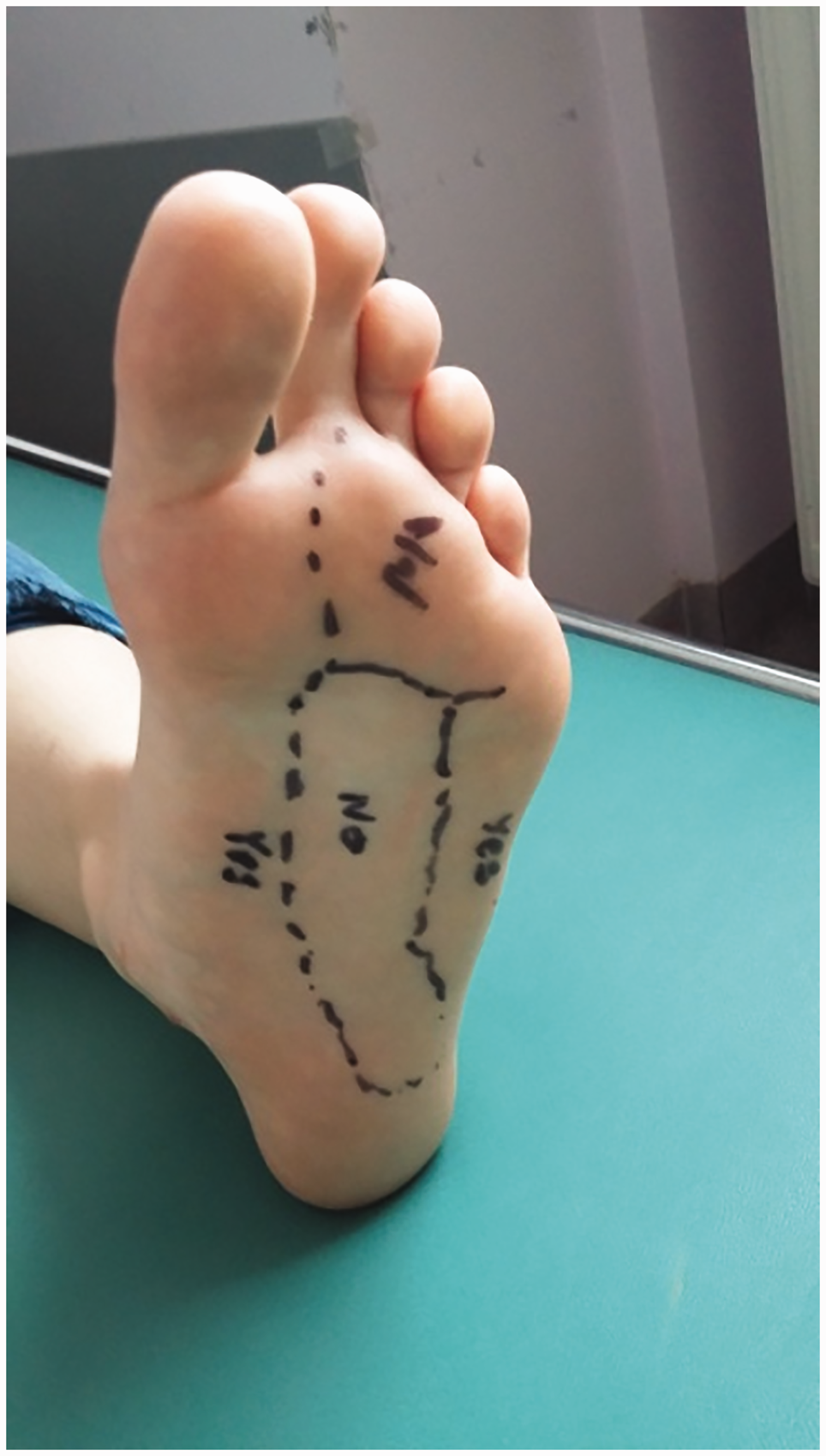
Result of the sensory examination 16 weeks after the neural repair. The area marked with “yes” is an area of feeling the touch assessed as equal to the uninjured side.
Six months after the surgery, manual assessment of muscle strength revealed 5/5 for flexing the foot and toes and 2/5 for abducting the hallux. The result of the sensory examination is depicted in Figure 5.

Result of the sensory examination 24 weeks after the neural revision. The area marked with the star is the area in which feeling was blocked.
At 36 weeks after the neural revision, the patient had achieved full recovery of sensory function in the injured nerve. Manual assessment of muscle strength revealed 3/5 for abducting the hallux. The result of the sensory examination is depicted in Figure 6. The EMG examination did not show significant differences between the new results and those of the earlier test (Table 3). The dysesthetic pain assessment result was 2/10.
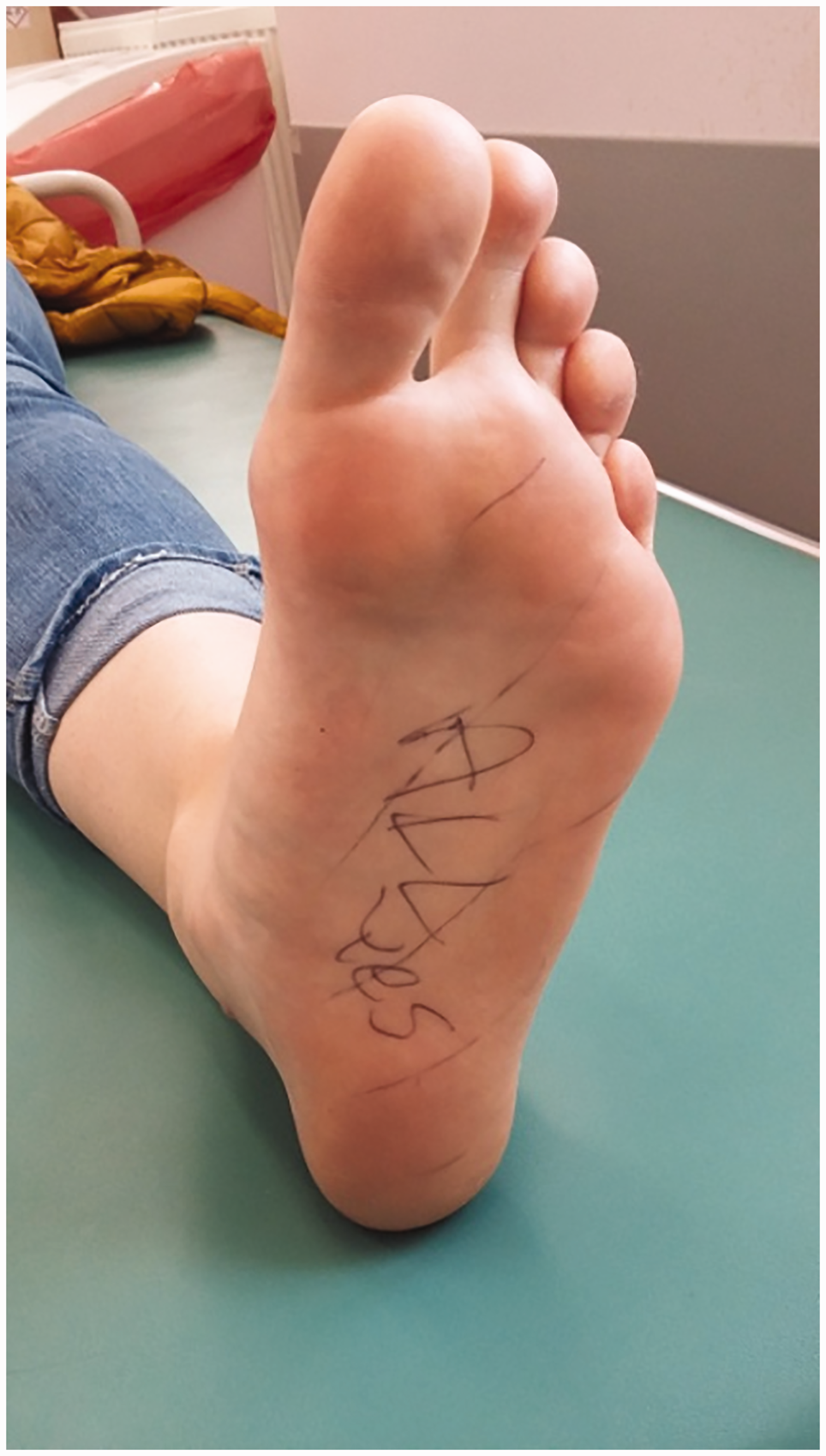
Result of the sensory examination 36 weeks after the neural repair.
Results of 36-week follow-up electromyographic examination.
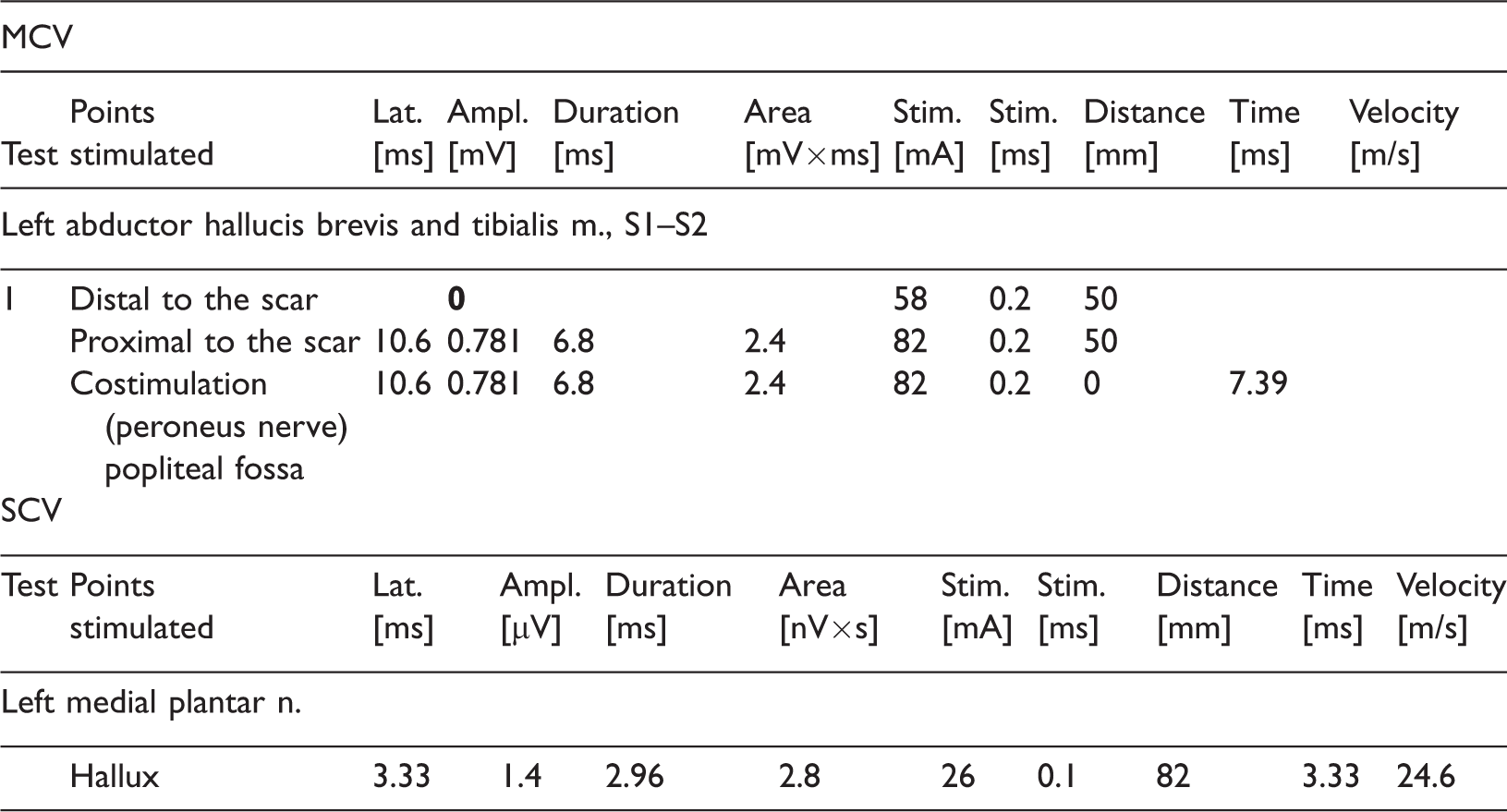
MCV, motor conduction velocity; SCV, sensory conduction velocity; Lat., latency; Ampl., amplitude; Stim., stimulation
Two years after the neural revision, the patient had achieved full recovery of sensory function for the injured nerve. Manual assessment of muscle strength revealed 3/5 for abducting the hallux. The results of the EMG examination are presented in Table 4. The patient was able to practice her typical training, beat her personal best, and win the national championships.
Results of 36-week follow-up electromyographic examination.
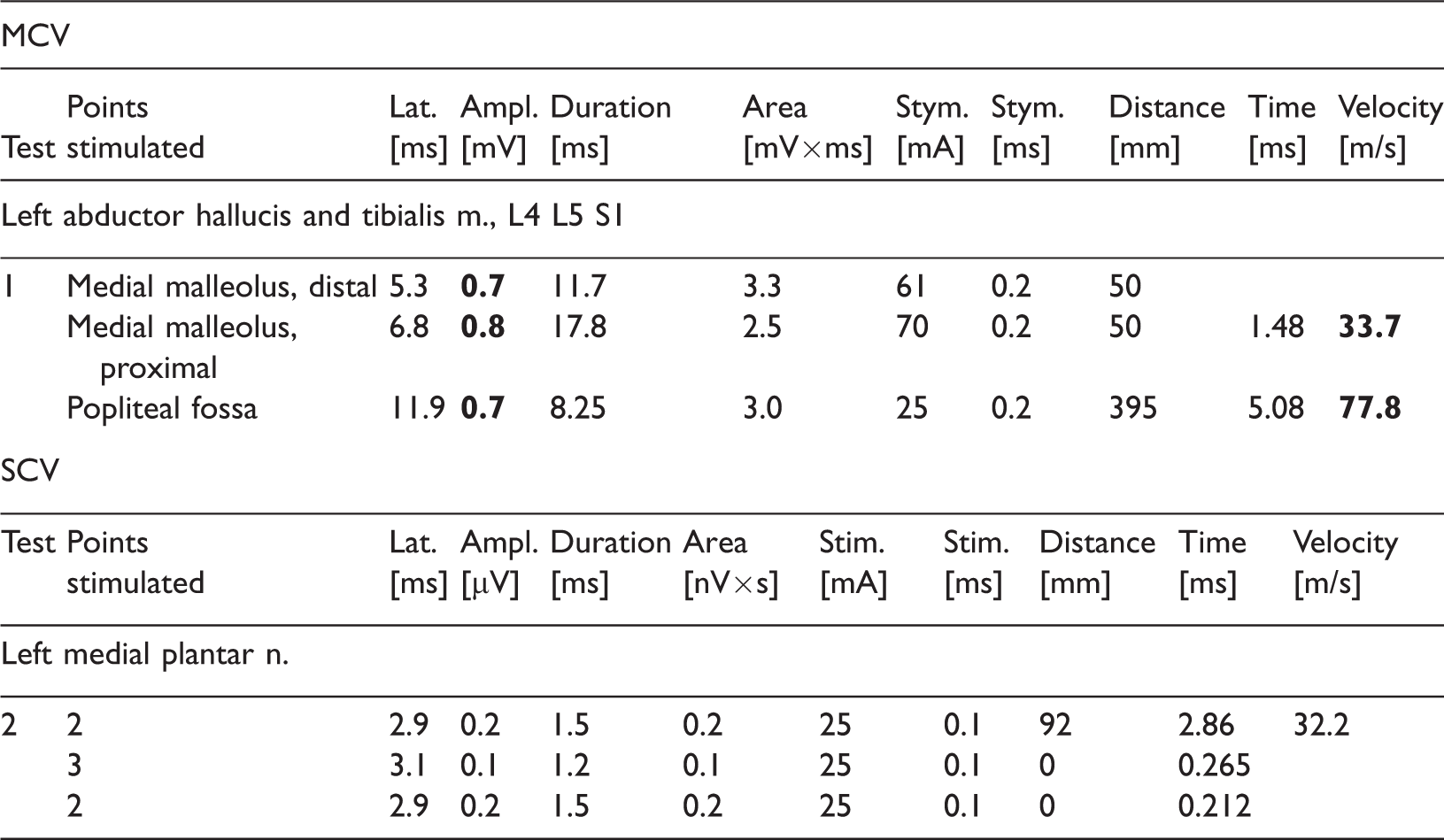
MCV, motor conduction velocity; SCV, sensory conduction velocity; Lat., latency; Ampl., amplitude; Stim., stimulation
Final conclusion of the last electromyographic examination: Neurography confirmed a mixed lesion (axonopathy and demyelination) of the left tibial nerve in the region of the medial malleolus (data of examination: 17 March 2016). The consecutive test (performed 30 June 2016) revealed progression and thereafter slight improvement in the motor fibers (response from the abductor hallucis brevis) and sensory fibers (assessment of the medial branch of the left plantar nerve).
Discussion
Neurovascular injuries seem to be the most common and unfortunate complications of ankle arthroscopy. Their prevalence may reach 80% of all complications after ankle arthroscopy.3,8
Cugat et al. 9 reported excellent results in a patient with a smaller lesion of the posterior tibial nerve after an 18-month follow-up. According to the Medical Research Council Grading System for Nerve Recovery, 1 we achieved scores of S4 and M3 at 9 months after surgery in the present case. Our patient managed to return to her training routine and fully participate in the trainings despite the massive lesion of her posterior tibial nerve. This outcome confirms that the rehabilitation program was suitable for our patient.
Electrical stimulation also proved to be an effective way to accelerate reinnervation after the neural injury in our patient. Few reliable guidelines are available regarding the most effective evidence-based dosage of electrical stimulation. We used a method that we considered to be the safest and most effective for our patient. We did not exceed a daily 1-hour period of electrical stimulation as suggested by Gordon. 10 Although Pieber et al. 11 suggested a slightly different dosage and electrode arrangement than presented here, they examined other nerves and muscles, and it seems that their findings cannot be precisely applied to our case. Nevertheless, electrical stimulation is thought to be a beneficial way to enhance the axonal regeneration process, even in cases of delayed neural repair. 12 Moreover, with respect to the weaker biokinematic chain in the affected lower extremity, at-home electrical stimulation seems to be the most effective way to alleviate muscle soreness after physical effort associated with athletic training. 13
Still, no strict consensus regarding the dosage of LLLT has been established. Many different types of LLLT appliances are available, each with a different wavelength and diameter of the laser beam. Nevertheless, LLLT seems to be the perfect method to contribute to the neural restoration process. In vitro tests have shown that the major therapeutic mechanisms of LLLT include enhancement of neurite sprouting, Schwann cell proliferation, and stimulation of the cascade processes involving mitochondrial and cellular activity. 7
A worrisome point regarding patients’ outcomes is the incomplete recovery of neural function. Adiguzel et al. 4 reported that most of their patients presented no change in the EMG test results and no change in the strength of motor function. Moreover, another study showed that even in the cases regarded as successful, patients with similar tibial nerve injuries were not able to perform dynamic or strenuous actions such as running or long standing. 14 In contrast to these findings, we observed distinct progress in our patient’s functional muscle strength outcomes and a slight improvement in the EMG examination results. Our patient achieved full functional recovery and was eventually able to perform at the top level of competition and win the national championships even after such devastating complications of her primary surgical procedure.
Footnotes
Declaration of conflicting interest
The author(s) declared that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors. The publication of the research was paid by the Medical University of Lodz (500/7-048-01/500-41).
