Abstract
Hypoplastic left heart syndrome (HLHS) comprises a spectrum of left-sided heart anomalies resulting from left outflow or inflow obstruction. Obstruction most often occurs at the levels of the aortic valve and/or mitral valve due to stenosis or atresia. HLHS is a lethal cardiac anomaly if not treated within the first week of life. Knowledge of sonographic features could aid in early detection, which results in better planning and management of pregnancy. This report presents a case in which HLHS was discovered during a routine dating and viability sonogram, performed at 12 weeks’ gestation.
Congenital heart defects (CHDs) account for one-third of congenital anomalies and are the leading cause of infant mortality due to birth defects. 1 Hypoplastic left heart syndrome (HLHS) is a term that describes a spectrum of cardiac anomalies involving the underdevelopment of the left side of the heart. HLHS occurs in 1–2 per 10 000 live births. 1 These structures include the mitral valve, left ventricle (LV), aortic valve, ascending aorta, and aortic arch.2–7 Obstruction can occur at various levels of LV inflow or outflow tracks.3,6 Outflow obstructions arise through aortic valve atresia or stenosis and are the most common cause of underdevelopment of the LV. Inflow obstruction occurs through mitral valve atresia or stenosis or a restrictive foramen ovale. 6 The degree of left ventricular hypoplasia is directly related to the severity of obstruction except when a ventricular septal defect is present and allows for an alternate exit for ventricular blood flow. 8
Case Report
A 27-year-old woman, G3 T2 P0 A0 L2, presented to the maternal-fetal medicine clinic for a routine viability scan at 12 weeks 2 days based on her last menstrual period. She had a prepregnancy body mass index of 33, a history of gestational hypertension with G2, and a history of hypothyroidism. Sonographic images were acquired using Philips Epiq 7 with a 9-2-MHz curvilinear-array transducer. Initial sonographic evaluation revealed a single, live fetus with a crown-rump length of 51.6 mm (11 weeks 6 days). Upon further investigation, a single right ventricle was noted on the four-chamber view (4CV), and only the main pulmonary artery (PA) could be identified from the three-vessel view (3VV) (see Figures 1 and 2). The aorta was difficult to visualize, causing suspicion for aortic atresia or hypoplasia. Higher resolution imaging was attempted, transvaginally, to better visualize the suspected CHD. A C10-3V endovaginal transducer was used to obtain these images. The right ventricle appeared significantly larger than the LV. Minimal color Doppler could be visualized by entering the LV through the mitral valve (see Figure 3)
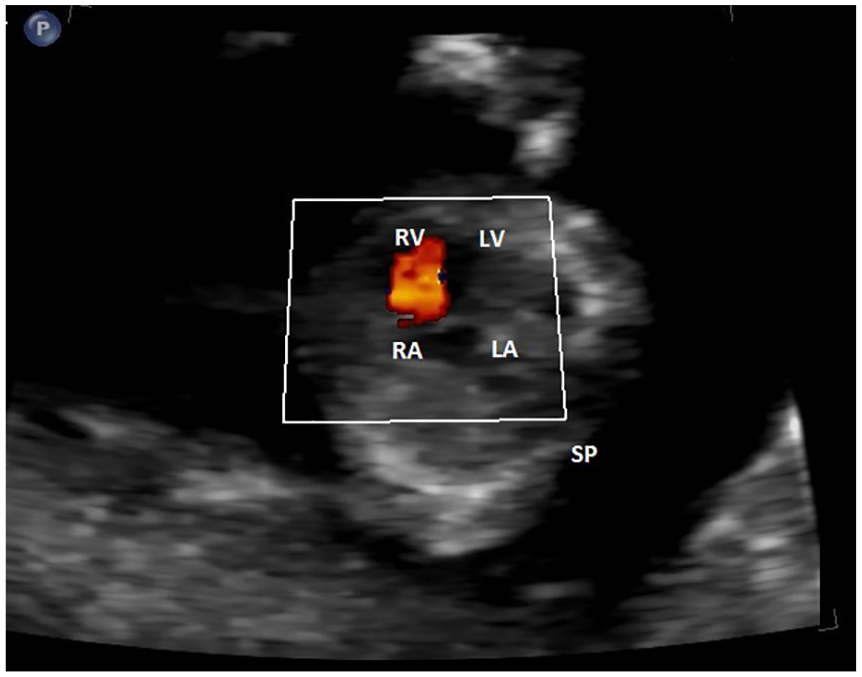
A transabdominal sonographic image of the four-chamber view taken at 12 weeks’ gestation. Color Doppler flow is only visualized through the RV. SP, spine; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
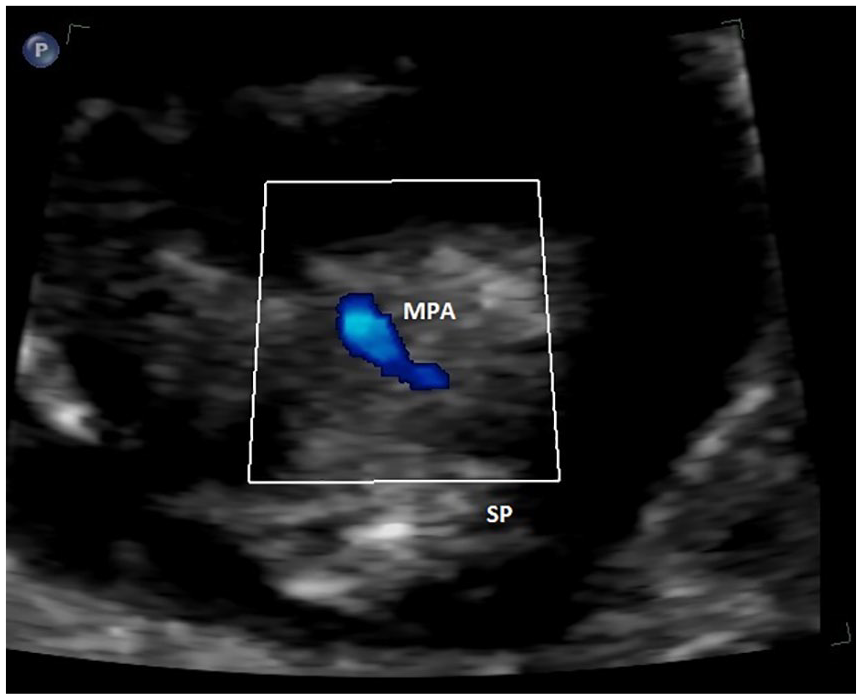
A transabdominal sonographic image taken in the region of the three-vessel view showing a prominent main pulmonary artery at 12 weeks’ gestation. The ascending aorta could not be identified in this image due to diminished blood flow. SP, spine; MPA, main pulmonary artery.
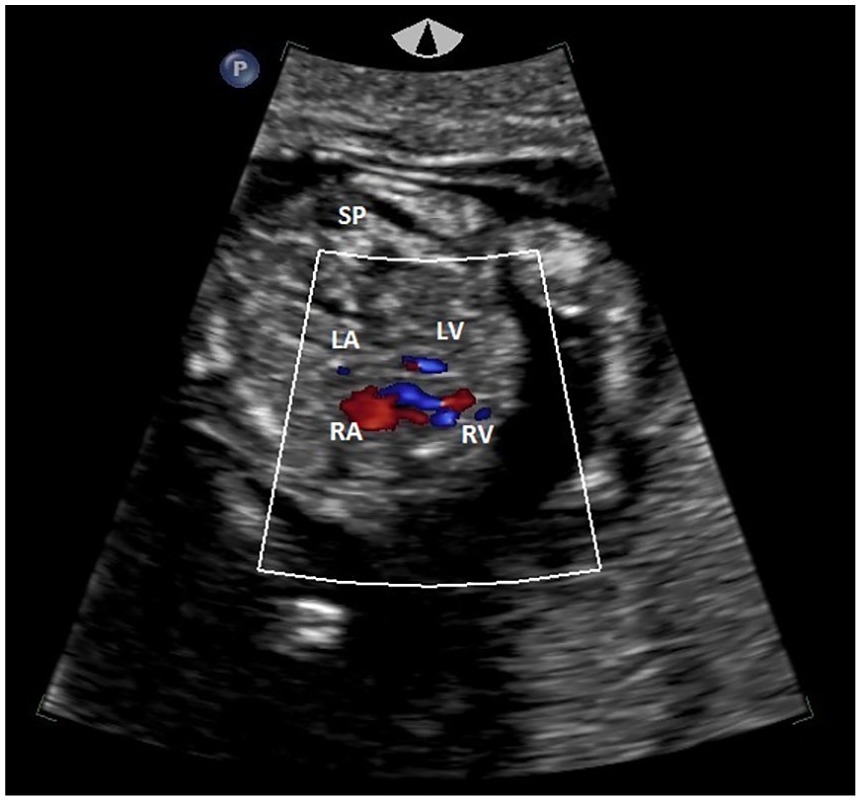
A transvaginal sonographic image of the four-chamber view taken at 12 weeks’ gestation. Diminished color Doppler flow is seen through the LV with a prominent RV and RA. SP, spine; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
The patient decided to pursue a first-trimester fetal echocardiogram at an outside facility at 14 weeks 3 days. It was reported that there was a single-ventricular disease with a severely hypoplastic LV, severe aortic valve hypoplasia, aortic hypoplasia, retrograde flow within the ascending aorta, and an inability to identify a ductus venosus (DV), suggesting its absence.
The patient returned to the original maternal fetal medicine department to continue her care. A detailed fetal anatomic survey was performed at 19 weeks 4 days. Normal fetal growth was noted during this sonogram, with an estimated fetal weight in the 44th percentile. In addition, a dominant right ventricle was visualized with left atrial and ventricular hypoplasia. The aorta also appeared hypoplastic from the 3VV (see Figure 4). No extracardiac anomalies were identified during this examination.
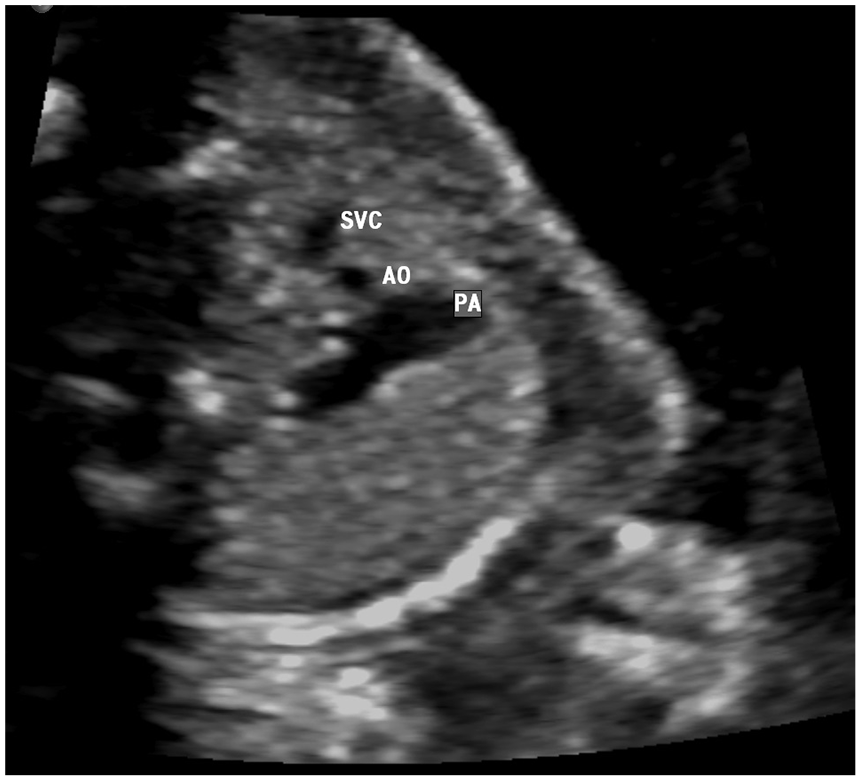
A sonographic image taken at the level of the three-vessel view at 19 weeks’ gestation. The ascending aorta appears hypoplastic in size compared to the PA and SVC. SVC, superior vena cava; AO, aorta; PA, pulmonary artery.
The initial second-trimester fetal echocardiogram was completed at 23 weeks 4 days, and the pediatric cardiologist’s report provided a detailed assessment. He noted a modest hypoplasia of the LV, a hypoplastic mitral valve, and an aortic valve with prograde flow through both. A hypoplastic ascending aorta, a widely patent ductus arteriosus, and no endocardial fibroelastosis were seen. In addition, he described reasonable systolic activity of the LV, dominant right ventricle with good systolic function, and widely patent foramen ovale (PFO), with possible severe preductal coarctation. As the pregnancy progressed, other complications of HLHS were found, and this included a hypertrophied right ventricle with preserved systolic function, dilated pulmonary valve annulus, moderately severe aortic valve hypoplasia, and possible hypoplastic or atretic DV. In addition, mitral valve hypoplasia and stenosis with scant prograde valvular flow developed (see Figures 5 and 6).
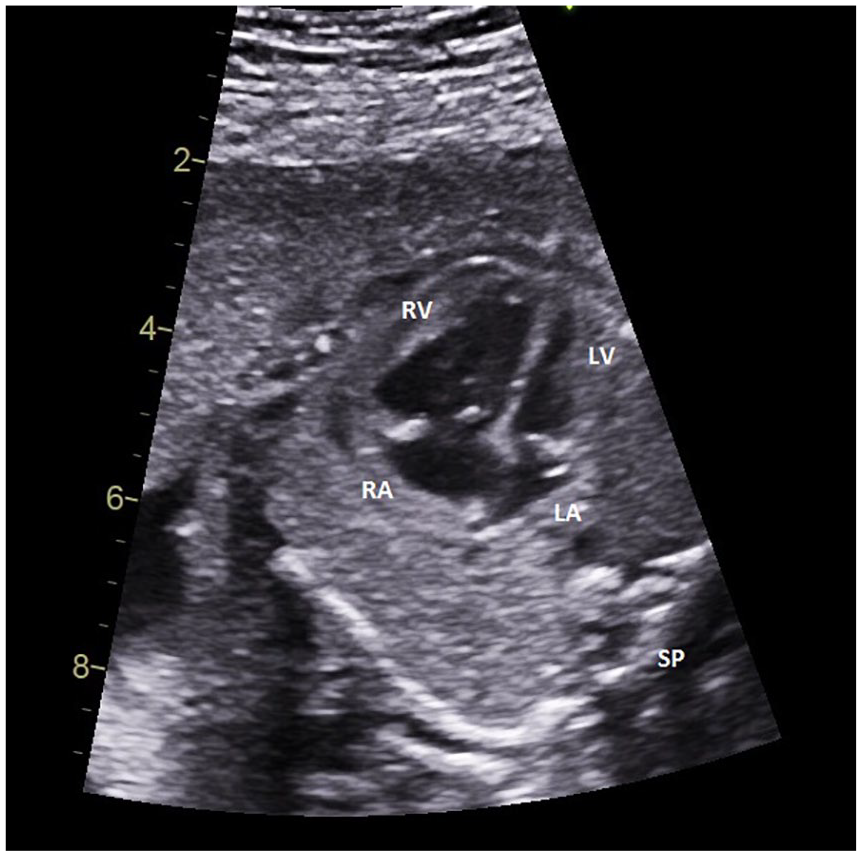
A sonographic image at the level of the four-chamber heart showing prominence of the RV and RA and a hypoplastic LV and LA at 26 weeks’ gestation. SP, spine; LA, left atrium; LV, left ventricle; RA, right atrium; RV, right ventricle.
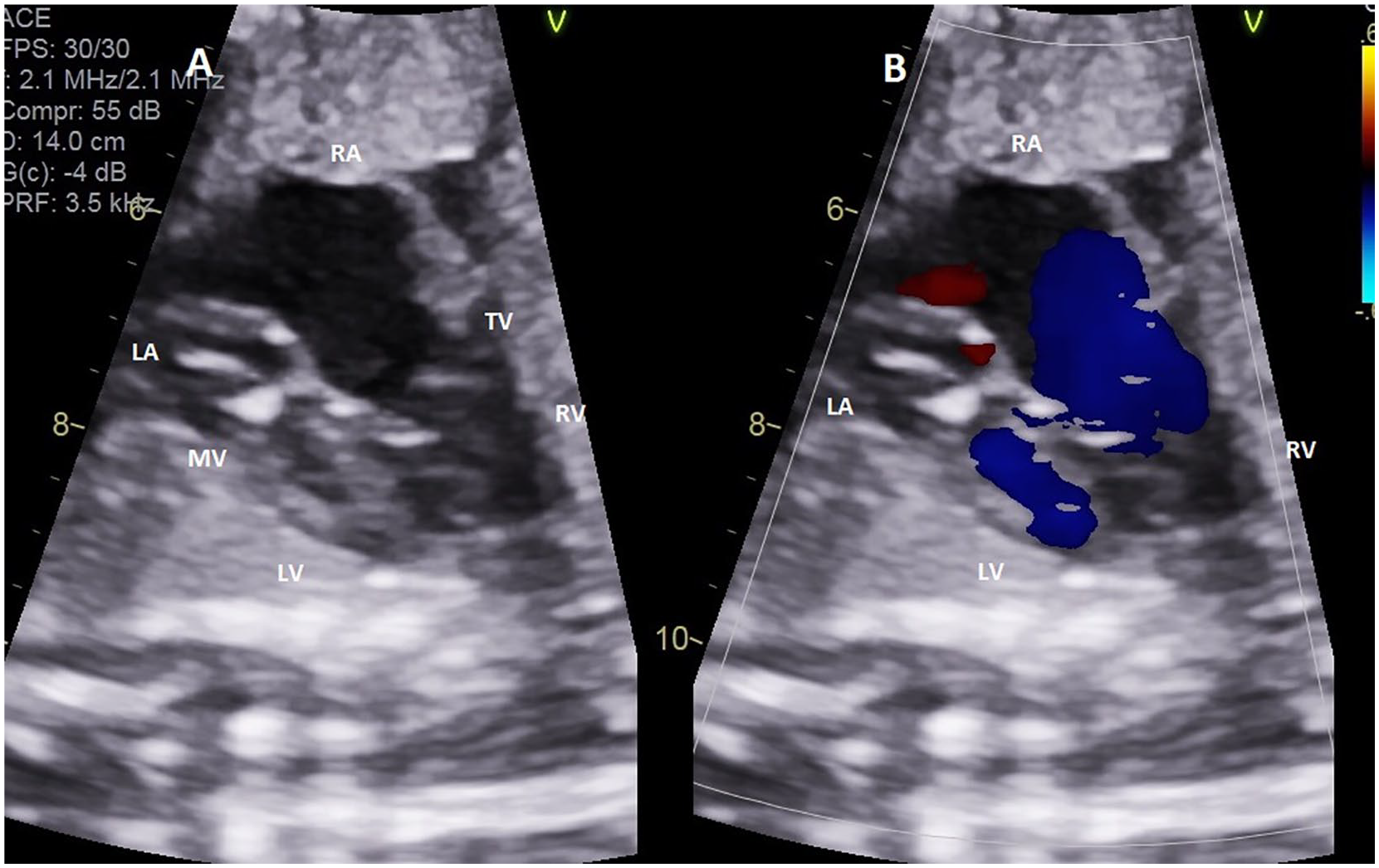
A sonographic image through the apical four-chamber view taken at 31 weeks’ gestation. (A) Moderately hypoplastic LV and LA and prominent right-sided heart structures; (B) prograde flow through both the mitral valve and tricuspid valve. LA, left atrium; RA, right atrium; TV, tricuspid valve; RV, right ventricle; LV, left ventricle; MV, mitral valve.
During a serial growth sonogram that was performed at 34 weeks 3 days, the fetus was found to have fetal growth restriction based on an abdominal circumference in the fifth percentile. The patient was diagnosed with gestational hypertension at 37 weeks 2 days. Therefore, the fetus was delivered the following day with a birthweight of 2650 g and Apgar scores of 8 and 9 at the 1- and 5-minute marks, respectively. The neonate was found to have glanular hypospadias and a congenital sacral dimple. Both renal and spinal canal sonograms were performed and had normal diagnostic findings.
A postnatal transesophageal echo (TEE) was performed and had a variety of diagnostic findings. The TEE confirmed a large PFO with mildly restrictive left-to-right shunting, trace tricuspid valve regurgitation, and trivial flow acceleration across the mitral valve. It also confirmed a moderately hypoplastic LV and unobstructed left outflow tract. A mildly hypoplastic aortic root, ascending aorta with laminar flow, and low-normal LV systolic function were seen. The examination also recorded a good right ventricular systolic function. Six days after the neonate’s birth, a stage I Norwood procedure was completed using the Sano modification technique.
The postsurgical TEE reported a functionally common biatrial communication via a widely patent septectomy with left-to-right shunting. Mild tricuspid regurgitation with no mitral regurgitation was noted. A dominant right ventricle with neoaortic outflow unimpeded through the neoaortic valve with full systolic excursion was seen. There was trace neoaortic insufficiency and the proximal end of the Sano shunt was seen traversing the right ventricular free wall with diffusely turbulent flow through the shunt. The examination documented a left PA, right PA, and hypoplastic LV with mitral valve and aortic valve hypoplasia with prograde flow through both.
At 3 months of age, the infant underwent stage II palliation consisting of the placement of a right bidirectional Glenn shunt and bilateral central pulmonary arterioplasty. A postsurgical TEE was performed, and it documented continued diagnostic issues. The TEE demonstrated normal veno-atrial situs, right inferior vena cava (IVC), and coronary sinus to right atrium. It was also documented that the right superior vena cava was anastomosed to the right PA, and a balanced atrial size was seen. This examination also found unrestrictive left-to-right shunting through central atrial communication, a large tricuspid valve annulus with mild tricuspid regurgitation, and combined right ventricular enlargement and hypertrophy. There was also a fair right ventricular systolic function, intermittent neoaortic insufficiency, and moderate hypoplasia of the mitral valve and aortic valve (see Table 1).
Sonographic Findings Throughout Pregnancy and Postnatally.

At 3 months of age, the infant is currently doing well postsurgically, with a mild complication of gastroesophageal reflux. The infant has demonstrated appropriate weight gain since birth and average O2 saturation in 80 seconds. The third surgical procedure will likely occur when the infant is 2–3 years of age.
Discussion
HLHS accounts for up to 9% of CHDs and will result in inadequate systemic circulation after birth.2,4,5 In normal fetal circulation, both ventricles contribute to a supply of blood to the developing fetus. This is due to important fetal circulation connections such as PFO, patent ductus arteriosus (PDA), and DV. 9 The purpose of both the PFO and the PDA is to bypass the fetal lungs. The PFO shunts blood from the right atrium to the left atrium, and the PDA connects the main PA to the aorta. The DV connects the umbilical vein to the IVC to bypass the liver. 10 Due to these connections, a single healthy ventricle can compensate for a hypoplastic contralateral ventricle in utero while sustaining fetal circulation and allowing for normal growth and organ development.2,9
In HLHS, pulmonary venous blood flows into the left atrium where some of it may still move through the mitral valve into the LV. Most of the blood flow is diverted through the PFO in retrograde fashion, from left to right, into the right atrium. Both oxygenated and deoxygenated blood is pumped through the right ventricle into the PA and reaches systemic circulation through the PDA. Often, blood is supplied to the brain and upper extremities by reverse flow from the PDA to the aortic arch.2,9 HLHS is a ductal-dependent lesion after birth, meaning closure of the PDA will result in a decrease in pulmonary vascular resistance, which could progress to hemodynamic shock. 2 Therefore, initial management of newborns consists of maintenance of ductal patency with the administration of prostaglandin E1 (PGE1).2,6
Approximately 4%–5% of cases with HLHS have an associated chromosomal anomaly and 10%–25% have an extracardiac defect. 4 Some of the associated chromosomal anomalies include Monosomy X, Trisomy 12, Trisomy 18, Trisomy 21, Partial Trisomy 9, Holt-Oram Syndrome, Smith-Lemli-Opitz Syndrome, Jacobsen Syndrome, and DiGeorge Syndrome.6,9 Associated extracardiac anomalies may include agenesis of the corpus callosum, congenital diaphragmatic hernia, omphalocele, holoprosencephaly, and many others. A higher morbidity and mortality rate and longer postsurgical hospital stay have been associated with the presence of a chromosomal anomaly in conjunction with HLHS. 6 One-fourth of neonates diagnosed with HLHS will have an associated early central nervous system (CNS) abnormality, but many will have impaired late CNS outcomes as well, such as learning disabilities, attention deficit hyperactivity disorder (ADHD), fine and gross motor abnormalities, and speech and behavioral problems. 11 Due to these associations, a detailed fetal anatomic sonogram is recommended at 18–20 weeks. 4
The standard of care for surgical repair of HLHS consists of a three-stage treatment approach. Stage I consists of the Norwood procedure and is performed within the first week of life.2,9 The goal of this initial surgery is to use the right ventricle as the main pumping ventricle for systemic circulation by creating a connection between the right ventricle and the aorta. This develops a new source of pulmonary blood flow and creates an atrial septectomy to maintain atrial communication. The goal of stage II is to unload the right ventricle to decrease the risk of a coronary steal and limit exposure of pulmonary vasculature to systemic pressures. This is accomplished by placing a bidirectional Glenn shunt or performing a hemi-Fontan procedure at 4–6 months of age. The final stage of surgical treatment is the Fontan operation performed between 2 and 3 years of age. In this procedure, blood in the IVC is diverted directly into the PA, bypassing the right ventricle, allowing deoxygenated blood to flow through the lungs.6,9,12 Heart transplantation is another treatment option but is only reserved for neonates who are at too high risk to undergo a staged repair, fail to progress through the staged repair, or develop complications of the Fontan circulation.2,6 Fetal intervention, such as in utero balloon atrial septostomy and stenting or balloon dilation of the aortic valve, may also be necessary in severe cases of HLHS.9,13
HLHS is responsible for 23% of all cardiac deaths occurring in the first week of life. 6 Without treatment, it has a 100% mortality rate because the LV is unable to support systemic circulation after birth. With staged palliation, survival figures range from 40% to 61%. However, some advanced care centers report a survival rate of 90%, although other facilities have had trouble duplicating these results. 3 In addition, lower survival figures of 17%–33% have been reported in fetal HLHS studies compared to pediatric studies.3,6 Approximately two-thirds of children with HLHS survive to the age of 5. 6
HLHS is most commonly diagnosed in the first half of pregnancy during a routine ultrasound through an abnormal 4CV.2–4 Normally, the ventricles should appear similar in size in this view, with the LV forming the apex, but in the case of HLHS, the LV will appear much smaller than the right ventricle. In some cases, the right ventricle may even be apex-forming. Because HLHS is evolutionary during fetal life, the size of the LV tends to become smaller as gestation progresses, while the right ventricle and right atrium tend to become dilated due to increased volume load. This volume overload in the right side of the heart may result in impaired systolic function of the right ventricle. Decreased flow through an atrioventricular valve, semilunar valve, or PFO restricts the growth of the structure it feeds. For example, if the mitral valve is atretic and flow cannot reach the LV properly, the LV will not develop normally. Other sonographic findings of HLHS that may be identified from the 4CV are endocardial fibroelastosis in the left ventricular wall, cardiomegaly, mitral valve hypoplasia or atresia, presence of a parachute or arcade mitral valve, poor mitral valve motion, diminished color Doppler flow into the LV, tricuspid valve regurgitation, and a restrictive or reversed interatrial shunt.6,7,9,14
Sonographic views of the left ventricular outflow tract (LVOT) and aortic arch will often appear abnormal in HLHS. A normal LVOT will show uniform patency with good aortic valve motion. In HLHS, the aortic valve often has poor valve motion, resulting in retrograde flow through the LVOT.6,7 Furthermore, the ascending aorta visualized in this view will appear hypoplastic or atretic. 7 Aortic atresia is the most common manifestation of the HLHS, and the aortic valve is also often bicuspid or unicuspid.8,9 Like the LVOT, in normal circulation the aortic arch will appear widely patent and uniform in size with three head and neck vessels branching from it. In HLHS, the aortic arch will appear hypoplastic, and the aortic root will measure less than 5 mm. 7 (see Table 2)
Comparison of Sonographic Findings in Normal Hearts and HLHS.

Abbreviations: HLHS, hypoplastic left heart syndrome; PFO, patent foramen ovale; PDA, patent ductus arteriosus.
In this case study, there was question as to whether the DV was absent. An absent ductus venosus (ADV) has been reported to be associated with HLHS as well as with ventricular septal defects, atrial septal defects, interrupted IVC, and heterotaxia syndrome. 15 There is a paucity of data regarding ADV and its clinical implications. 16 In normal fetal circulation, the DV connects the umbilical vein with the left portal system and the left hepatic vein.15,16 Oxygenated blood enters the umbilical vein, flows into the DV, bypasses 20%–30% of liver sinusoids, and flows into the IVC so that more oxygenated blood can flow into systemic circulation. 15 There are three main patterns of abnormal connection of the intra-abdominal umbilical vein in ADV: (1) the umbilical vein bypasses the liver and drains directly into the right atrium (46%), (2) the umbilical vein bypasses the liver and drains directly into an iliac or renal vein (25%), or (3) the umbilical vein connects directly to portal circulation without giving rise to DV (21%).15,16 The third combination is the most favorable, while the other two are associated with fetal cardiac failure. 15
Differential diagnoses for HLHS often include other left-sided obstructive lesions that are also considered ductal-dependent after birth, such as aortic coarctation, critical aortic stenosis, and interrupted aortic arch. Aortic coarctation is the most common differential and is seen in conjunction with HLHS in 7% of cases. It is considered a differential diagnosis because it does not always result in severely hypoplastic left heart structures. In addition, aortic coarctation may not be present until after birth when the ductus arteriosus closes. After this closure, the neonate often presents with the same signs and symptoms that would be seen with HLHS.4,8
Syngelaki et al performed a study, the largest of its kind, involving 44 859 pregnancies using first-trimester ultrasound as part of routine screening for aneuploidies. It was found that HLHS was able to be identified at 11–13 weeks in more than 90% of cases. 17 However, recognizing HLHS in the first trimester can be difficult due to poor image resolution and limited clarity due to fetal position, movement, and size. Increased maternal body habitus can also inhibit ultrasound beam penetration. In a study performed by Khalil and Nicolaides, HLHS had a 45% identification rate at 11 weeks versus 90% at 13 weeks. Furthermore, it was reported that a 4CV success rate was only 20% at 11 weeks and 92% at 13 weeks. 1 Other studies have suggested that prenatal diagnosis of HLHS has no impact on surgical mortality rates in a neonate. However, it is important to note that only neonates who made it to surgery were included in those data sets. Neonates who had a postnatal diagnosis of HLHS and passed away before reaching a tertiary care center were excluded. 13
Various technological advancements have facilitated imaging and identification of complex cardiac anomalies such as HLHS. 18 This allows for fetal karyotyping and genetic counseling to be completed earlier, allowing parents and physicians to partake in complex decision-making prior to birth.13,18–20 Termination of pregnancy could be decided upon earlier, resulting in lower rates of post-traumatic stress. 21 Earlier termination also presents less of a medical risk to the mother. 22 A first-trimester diagnosis of HLHS allows time for neonatal management planning and to assemble a multidisciplinary team at a tertiary care center.13,18–20 Moreover, early identification will prompt a detailed fetal anatomic survey to potentially diagnose any extracardiac defects that may be present.13,19,20 In severe cases, there is an opportunity for fetal intervention if necessary.9,13 There is also the prospect of fetal listing for heart transplantation if anatomy is unfavorable for staged palliation. 9 One study showed that women who have a prenatal diagnosis of HLHS who deliver within or near a cardiac center have neonates with lower mortality rates than those with a postnatal diagnosis in a geographically remote location. 13
Conclusion
HLHS is a severe cardiac anomaly that will result in neonatal death, if untreated. Familiarity with typical sonographic findings could allow for earlier detection in pregnancy. The first-trimester diagnosis aids in management planning from an earlier gestation. A prompt referral to a tertiary care center may ultimately improve maternal-fetal outcomes.
Footnotes
Acknowledgements
I would like to thank Michele P. Monaco, MD for insight and guidance on the topic of hypoplastic left heart syndrome and for providing useful articles for reference. I would also like to thank Oliver T. Chen, MD for his advice and assistance with revisions, as well as continuing to push me to venture outside of my comfort zone.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
