Abstract
Ultrasonography has shown that many T2 gallbladder carcinomas (GBCs) demonstrate a deep hypoechoic area, and many shallow T2 GBCs show a conically thickened, outermost hyperechoic layer. However, this report describes a case of a hyperplastic polyp, of the gallbladder, with similar sonographic findings. This case reviews and analyzes the ultrasonographic imaging, as well as the pathologic correlation. Endoscopic ultrasound (EUS) revealed a thickened, outermost hyperechoic layer beneath a protruding lesion in the gallbladder fundus. The lesion was coupled with a deep hypoechoic area and a pancreaticobiliary maljunction (PBM). Pre-contrast computed tomogram (CT) demonstrated a soft tissue density mass in the fundus. Magnetic resonance imaging (MRI) disclosed no decreased-diffusion lesion under the diffusion-weighted image. Without any finding suggestive of an obvious advanced GBC, a full-thickness laparoscopic cholecystectomy was performed. The histopathology report indicated a hyperplastic polyp, which consisted of hypertrophic muscle, increased fibrosis, and a conically thickened subserosal fatty layer underneath the polyp. A specific benign protruded lesion may, though rarely, demonstrate similar EUS findings to those of shallow T2 carcinoma.
A hyperplastic polyp of the gallbladder is a rare condition showing a protruded, pedunculated, or semi-pedunculated structure that can be accompanied by a pancreaticobiliary maljunction (PBM).1,2 A gallbladder polyp, with thickening of the mucosa, can frequently be associated with PBM. A PBM, without biliary dilation, is a high-risk condition inducing a gallbladder carcinoma (GBC). 3
Patients presenting with an early (pT1) GBC, confined to mucosa (pT1a) or muscle layer (pT1b), have a good prognosis, given the overall 10-year survival has been 87%. 4 Furthermore, although surgical outcomes for advanced GBC remain unsatisfactory, a radical resection provides a favorable prognosis for patients with a GBC, limited to a shallow subserosal invasion (subserosal invasion depth ≦ 2 mm: shallow pT2). 5 Hence, ultrasonographic findings of shallow T2 GBCs are gaining diagnostic attention. 6
Ultrasonography has shown that many T2 GBCs demonstrate a deep hypoechoic area, and many shallow T2 GBCs have a conically thickened outermost hyperechoic layer. 7 Hence, it is essential that the ultrasonographic diagnosis of gallbladder tumors need to determine whether a deep hypoechoic area can be demonstrated. It is also important to note whether an outermost hyperechoic layer is conically thickened. In contrast, this patient case illustrates a hyperplastic polyp of the gallbladder, with similar ultrasonographic findings that rely on image analysis and pathologic correlation of the lesion.
Case Report
A 40-year-old woman, with a history of appendectomy and resection of the left ovarian cyst, was discovered to have an asymptomatic gallbladder tumor on transabdominal sonogram, during her first health-check examination. She presented to the hospital for further investigation. She had no family history of malignancy, and no habit of smoking or drinking alcohol. The physical examination and laboratory values were within normal parameters except for mild anemia (hemoglobin = 10.8 g/dL). In particular, there was no elevation of carcinoembryonic antigen or carbohydrate antigen 19-9.
A transabdominal sonogram was performed with a Toshiba SSA-770A, a T5.0 MHz convex transducer, and a T7.2 MHz linear transducer (Toshiba, Tokyo, Japan). The images depicted a broad-based elevated lesion in the gallbladder fundus. It was noted that there was diffuse mural thickening, accompanied by a conically thickened outermost hyperechoic layer, without a distinct deep hypoechoic area. A color Doppler examination (advanced dynamic flow) did not show vascular signals in the lesion (Figure 1). However, the contrast-enhanced ultrasound (CEUS) revealed a dendritic enhancement of the lesion during the early vascular phase and no enhancement in the late phase. Endoscopic ultrasound (EUS) using an Olympus GF-UM2000 and a 6.0 MHz radial transducer (Olympus, Tokyo, Japan) revealed an elevated lesion in the fundus. The lesion had not only a conically thickened outermost hyperechoic layer but also a deep hypoechoic area and PBM (Figure 2). Pre-contrast computed tomogram (CT) revealed a soft tissue density mass in the fundus, and a dynamic CT demonstrated early and continuous enhancement of the lesion (Figure 3). Magnetic resonance imaging (MRI) disclosed an unclear mass intensity under the T1-weighted image, a low-intensity lesion under the T2-weighted image, and no decreased-diffusion lesion under the diffusion-weighted image. In addition, a magnetic resonance cholangiopancreatography (MRCP) showed PBM. Endoscopic retrograde cholangiopancreatography depicted an elevated lesion in the gallbladder fundus and PBM.
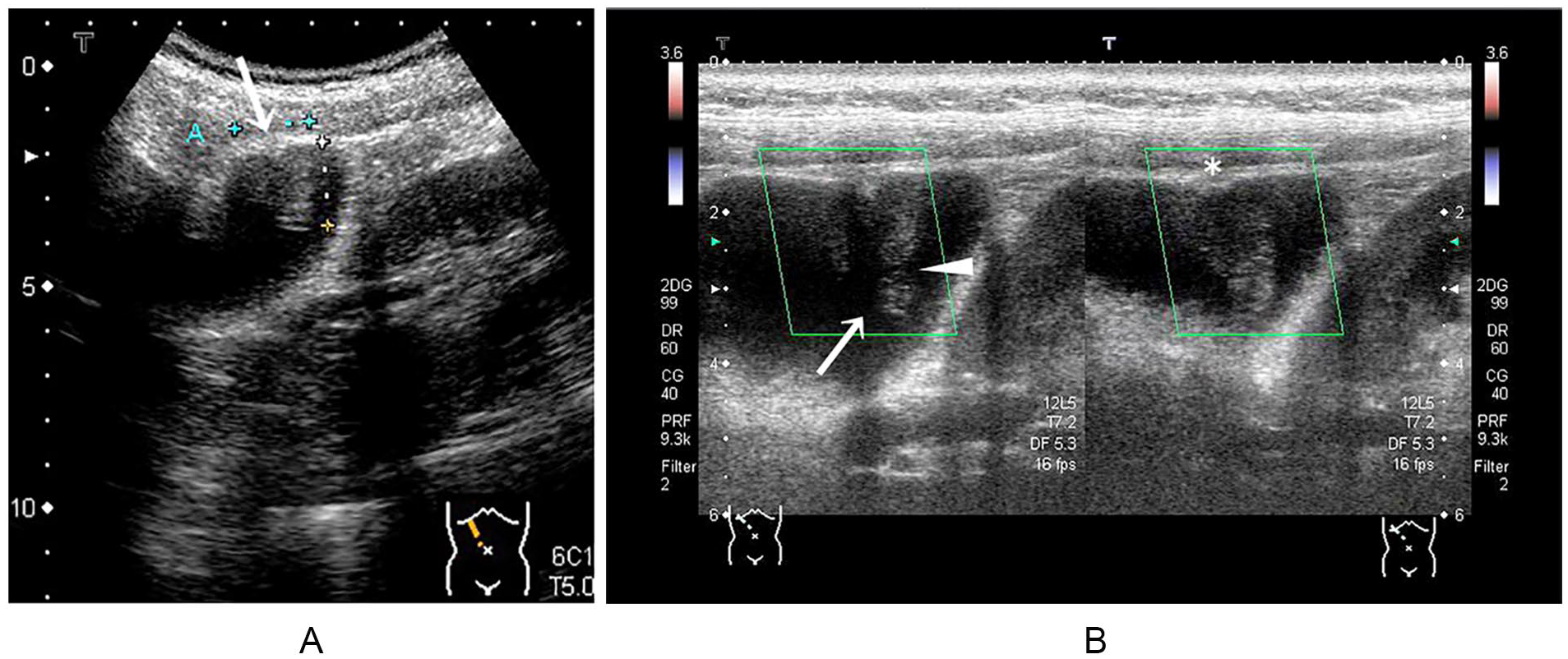
Transabdominal ultrasonogram. (A) Transabdominal conventional sonogram (T5.0 MHz) depicts a broad-based elevated lesion in the fundus, 20 mm in diameter, with a relatively smooth surface accompanied by a conically thickened outermost hyperechoic layer (white arrow) without a distinct deep hypoechoic area. Besides, there exists diffuse mural thickening of the gallbladder. (B) Color Doppler (advanced dynamic flow) does not show vascular signals in the lesion. High-frequency probe (T7.2 MHz) depicts a broad-based elevated lesion with constriction on the top (white arrowhead) accompanied by a conically thickened outermost hyperechoic layer with acoustic shadow (white arrow) and without a visible deep hypoechoic area. In contrast, there does not exist a prominent deep hypoechoic area also above a gently thickened outermost hyperechoic layer (*) without acoustic shadow.
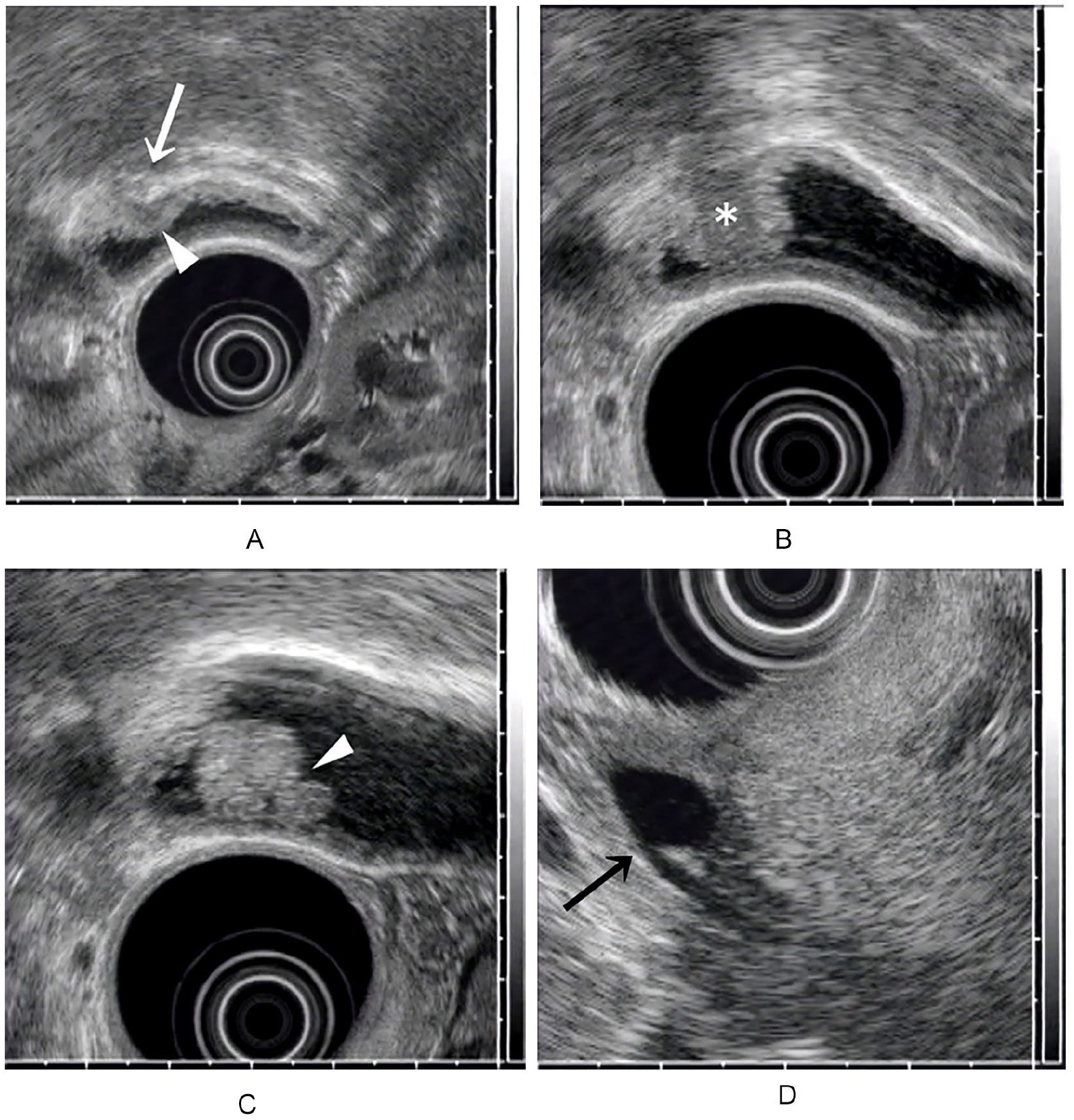
Endoscopic ultrasound (EUS). (A) The gallbladder, including an elevated lesion, is deformed by a balloon of the EUS unit. EUS shows an elevated lesion in the fundus with not only a conically thickened outermost hyperechoic layer (white arrow) but also a deep hypoechoic area (white arrowhead) accompanied by diffuse mural thickening. (B) The lower half of the lesion shows hyperechoic at the periphery and hypoechoic in the center (*). (C) The upper half of the lesion looks like a snowman with constriction on the top (white arrowhead). (D) Pancreaticobiliary maljunction (black arrow).
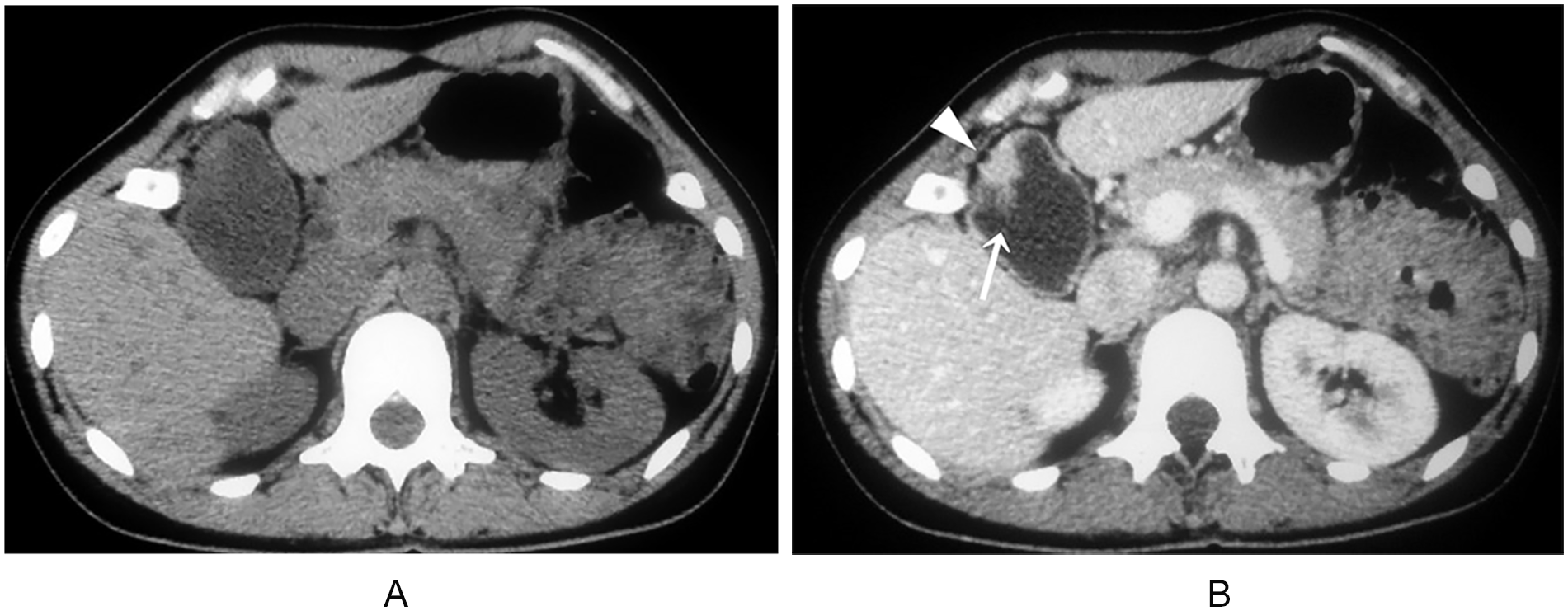
Computed tomogram (CT). (A) Pre-contrast CT demonstrates a soft tissue density mass except for apex in the fundus accompanied by diffuse mural thickening of the gallbladder. (B) Dynamic study shows early and continuous enhancement of the lesion, including diffuse mural thickening of the gallbladder. The apex (white arrow) of the elevated lesion is not depicted until the intravascular injection of contrast agent, and the outline of the gallbladder is slightly irregular (white arrowhead) (portal phase).
As the pre-contrast CT demonstrated a soft tissue density mass in association with PBM, it made it easier to rule out a cholesterol polyp, fibrous polyp, but not a malignancy. 8 However, a malignancy could be mostly ruled out, given that the lesion did not show decreased diffusion, based on the diffusion-weighted images. These two conditions conflicted with each other. Hence, without findings suggesting an obvious advanced GBC, a full-thickness laparoscopic cholecystectomy was performed. The amylase activity of the gallbladder bile collected during the operation was 160 000 U/L.
Macroscopically, a broad-based elevated lesion, 15 mm in diameter, was evident in the fundus of the gallbladder on a background of coarsely granular mucosa and mural thickening (Figure 4). Histopathology reports indicated that the lesion was a hyperplastic polyp with diffuse papillary hyperplasia of the mucosa. Neither mucosal dysplasia nor adenoma was discovered. A broad-based elevated lesion, consisting of Rokitansky-Aschoff sinuses, with prominent pseudo-pyloric gland metaplasia and massive fibrosis, beneath the muscle layer, was evident in the fundus. Subserosal adipose tissue was a bit drawn into this enormous fibrosis beneath which a conically thickened subserosal adipose tissue layer existed (Figure 5).
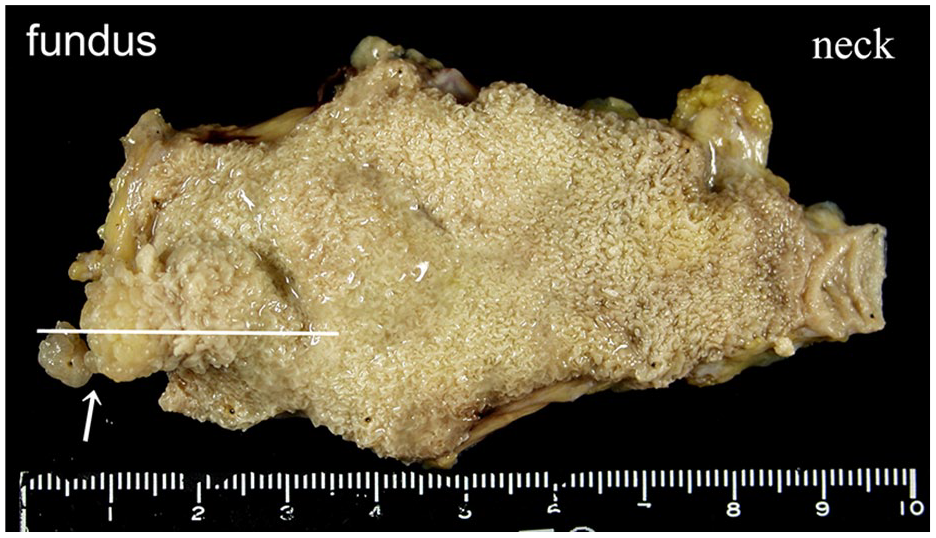
Fixed specimen.
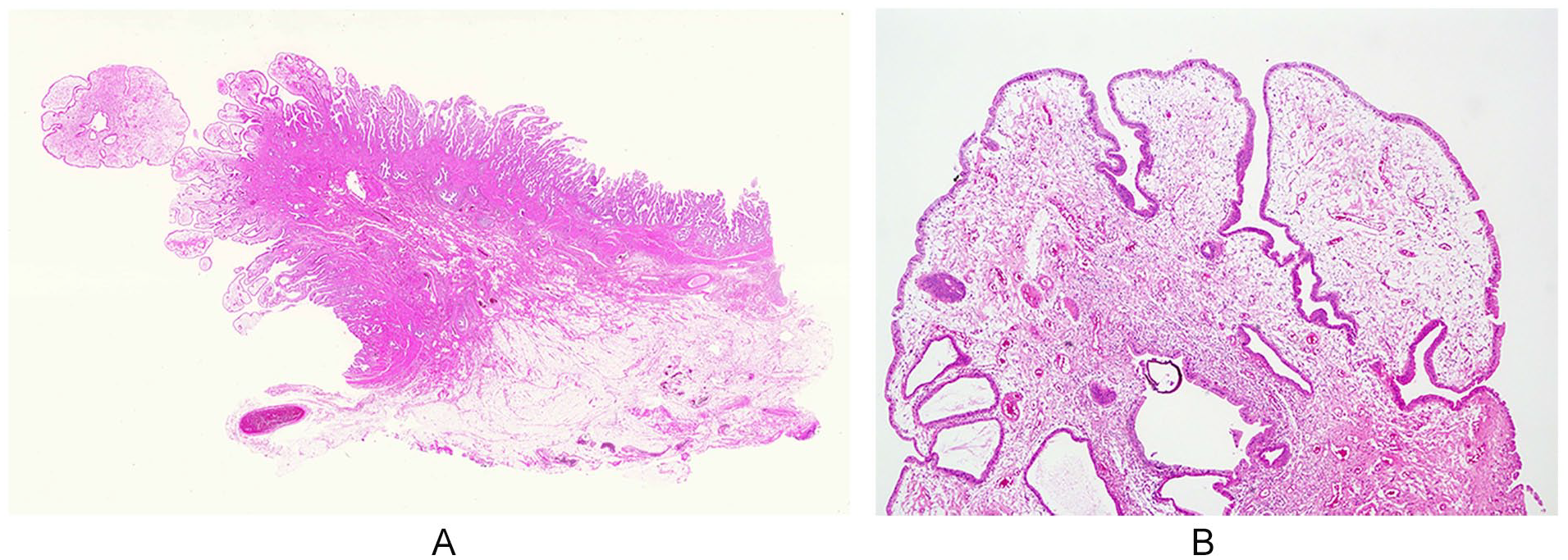
Loupe and low power view (H&E). (A) Loupe view of the cross-section of Figure 4: the specimen shows papillary hyperplasia of the mucosa. Massive fibrosis is evident beneath the muscle coat of protruded lesion in the fundus. Subserosal fatty tissue is drawn into extensive fibrosis beneath which is a conically thickened subserosal fat tissue layer. (B) Moderate power view of apex of (A) (original; 200×): mild papillary hyperplasia and edematous interstitial tissue.
Discussion
A hyperplastic polyp of the gallbladder is a rare benign condition. There have been no published reports of a case with a deep hypoechoic area and a conically thickened outermost hyperechoic layer. This is based on a literature search using PubMed (1990–present), with the keywords gallbladder, polyp, or ultrasound.
The discussion associated with this case’s imaging diagnosis, is centered on the following points: (1) reaching a definitive diagnosis of PBM, after obtaining ultrasonographic findings that indicate a diffuse mural thickening of the gallbladder; (2) differential diagnosis of a broad-based elevated lesion in the fundus, to determine a benign or malignant lesion; and (3) diagnosing an invasive depth carcinoma when malignancy is suspected.
Addressing the first point, Igarashi 3 discussed the diagnosis of PBM and demonstrated that a sonographic finding of a thickened gallbladder wall (≧3mm), is a clue for PBM. This is due to papillary hyperplasia or thickening of the gallbladder mucosa, which is frequently associated with PBM. 3 The current case corresponded to a PBM without biliary dilatation and a high-risk condition inducing a gallbladder carcinoma, based on Japanese clinical practice guidelines. 9
Second, the differential of a benign or malignant condition was confirmed with the pre-contrast CT findings, which did not rule out a malignancy. However, a malignancy could be mostly denied by the findings of the diffusion-weighted MRI. Thus, these two pathologic conditions were in opposition, which ruled out an obvious advanced GBC.
As for the diagnosis of invasive depth carcinoma, interpretation of the layer-structure of the gallbladder wall with ultrasonography or EUS was critical. Ultrasonography has demonstrated that the gallbladder wall, during fasting, could increase to 3.0 mm, in thickness. It is usually composed of two layers, an innermost hypoechoic layer and an outermost hyperechoic layer. Occasionally, it can be noted to have three layers, an innermost hyperechoic layer, a middle hypoechoic layer, and an outermost hyperechoic layer. There has been much discussion regarding the interpretation of the middle hypoechoic layer. However, a commonly accepted theory is that the middle hypoechoic layer consists of not only the muscularis propria but also the subserosal fibrous layer. Besides, the outermost hyperechoic layer represents the subserosal adipose layer plus serosa.7,10 According to the ultrasonographic interpretation of the layer-structure of the gallbladder wall, a gallbladder tumor with a thickness-unchanged outermost hyperechoic layer does not exclude a subserosal carcinoma, confined to the subserosal fibrous layer.
However, regarding a polypoid gallbladder tumor with a deep hypoechoic area, the ultrasound and pathological correlation study shows that a superficial hyperechoic part coincides with a papillary adenocarcinoma and a deep hypoechoic area represents an adenocarcinoma invading subserosa, accompanied by abundant fibrosis and lymphocytic infiltration, even if a thickness-unchanged outermost hyperechoic layer is noted.7,11 Meanwhile, a deep hypoechoic area represents not only a medullary tumor but also hyperplastic lymphoid follicles or noncancerous fibrosis. For this reason, a pT1 GBC (limited to the muscular coat) with a deep homogeneous lesion, or a benign protruded lesion with hyperplastic lymphoid follicles or massive fibrosis beneath the lesion, might be overestimated as invading the subserosa. 7 So far, these types of clinical examples have not been reported. This case is unique and represents the latter description. This case confirmed that the EUS findings were based on similar histological findings of shallow T2 GBC, that is, massive fibrosis beneath which a conically thickened subserosal adipose tissue layer existed.
Regarding the differences in results between a transabdominal sonogram and EUS, the following mechanism is a likely explanation. The transabdominal sonogram failed to depict a distinct deep hypoechoic area due to an acoustic shadow derived from the conically thickened outermost hyperechoic layer. In contrast, the EUS detected a deep hypoechoic area because the ultrasound beam was aimed from top to bottom of the lesion, opposed to the direction of the transabdominal sonogram. Therefore, a deep hypoechoic area beneath a protruded lesion can be detected even with a transabdominal sonogram, if the lesion exists in the dorsal gallbladder wall. As an aside, the discrepancy between the transabdominal sonogram and the EUS, in the shape of the polypoid lesion, may have been caused by the absence and presence of compression, respectively.
Conclusions
The EUS findings described above, that is, a protruded lesion with a deep hypoechoic area and a conically thickened outermost hyperechoic layer, are essential and should be spread to depict a shallow T2 GBC. However, a specific benign protruded lesion may, though rarely, demonstrate similar EUS findings to those of shallow T2 carcinoma as in the present case. For the moment, meticulous full-thickness laparoscopic cholecystectomy and histopathologic examination of the resected gallbladder should be performed for the suspected “shallow T2 GBC” lesions to avoid unnecessary surgery.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Compliance With Ethical Standards
All procedures followed were by the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. Written informed consent was obtained from the patient for being included in the study.
