Abstract
We report an unusual case of autoimmune gastritis (AIG) complicated with a submucosal tumor (SMT) and two pedunculated polyps in a 60-year-old man. The patient was admitted for epigastric distention, heartburn, and anorexia. Endoscopy showed an SMT in the fundus, two pedunculated polyps in the body, and markedly atrophic mucosa of the body and fundus. The SMT, measuring 20 mm in diameter, was resected by endoscopic submucosal dissection and histologically diagnosed as a gastric hamartomatous inverted polyp (GHIP), which is characterized by submucosal glandular proliferation, cystic dilatation, and calcification. The gland structures consisted of foveolar cells and pseudopyloric or mucous-neck cell types. The two pedunculated polyps that were resected by endoscopic mucosal resection were histologically diagnosed as hyperplastic polyps, which are characterized by hyperplastic foveolar glands with pseudopyloric or mucous-neck glands in the inflamed stroma in the mucosa, which consisted of almost the same types of lining cells as the GHIP in the fundus. Findings may indicate the relationship between GHIP, hyperplastic polyp, and AIG. We highlight considering GHIP as a differential diagnosis for an SMT in patients with AIG.
Keywords
Introduction
Autoimmune gastritis (AIG), an immune-mediated disease, is considered to be closely related to gastric mucosal proliferative diseases such as hyperplastic polyps. 1 The gastric hamartomatous inverted polyp (GHIP) is a rare form of gastric polyp of unknown pathogenesis. 2 Because of their inverted growth pattern into the submucosal layer, GHIPs are difficult to diagnose accurately without endoscopic or surgical resection and subsequent histological examination. We report the case of a 60-year-old man diagnosed with AIG complicated with a GHIP in the fundus and two hyperplastic polyps in the body. Although the endoscopic GHIP type was determined to be a submucosal tumor (SMT), it was successfully resected by endoscopic submucosal dissection (ESD), indicating the utility of en bloc resection of an SMT-type GHIP with ESD. Based on the histopathological findings of the case, we discuss the relationship between GHIP, hyperplastic polyps, and AIG and explore the pathogenesis of GHIPs.
Case report
A 60-year-old man with no family history was admitted with chief complaints of intermittent epigastric distention, heartburn, and anorexia for more than half a year. The patient underwent an esophagogastroduodenoscopy (EGD) and endoscopic ultrasound (radial scan, 10 MHz). The EGD showed an SMT in the gastric fundus measuring 20 mm in diameter with a reddish area on the top (Figure 1a) and marked atrophy of the oxyntic mucosa with a considerable amount of mucus attached; however, no atrophic mucosa was observed in the antrum. An endoscopic ultrasound (radial scan, 10 MHz) revealed that the SMT was a heterogeneous tumor with multiple small hypoechoic or anechoic areas (Figure 1b) and hyperechoic calcified areas with acoustic shadow (Figure 1c). We suspected a gastrointestinal stromal tumor, neuroendocrine tumor, or GHIP. However, a definitive diagnosis was difficult without histological examination. Thus, an en bloc resection of the SMT was necessary. After obtaining written informed consent from the patient, we resected the SMT using ESD (Figure 1d). A hematoxylin and eosin stain of the resected SMT is shown in Figure 1e (low-power view), Figures 1f, 1g, and 1h (medium-power views), and immunohistochemical stains are shown in Figures 1i (mucin-5AC), 1j (mucin 6), 1k (pepsinogen I [Pep I]), and 1l (chromogranin A). Histologically, the hyperechoic solid portion (Figure 1b, orange circle) of the SMT had multiple small hypoechoic or anechoic cystic spots (Figure 1b, yellow arrows), which were the proliferation of foveolar glands and pseudopyloric or mucous-neck glands and had cystic dilatation and cytological atypia, fibroblast cells, and smooth muscle proliferation into the submucosal layer (Figures 1e–1g,1i,1j). The submucosal glands or cystic elements of the lesion were connected to the overlying gastric mucosa through a defect of the muscularis mucosa (Figure 1g, blue arrows). The hyperechoic solid portion with acoustic shadow (Figure 1c, red arrow) was shown to be the calcification lump (Figure 1m, red arrow) within a considerably dilated gland (Figure 1e). Histologically, the SMT was diagnosed as GHIP. Moreover, given the marked reduction of oxyntic glands, pseudopyloric and intestinal metaplasia in oxyntic mucosa (Figure 1h), positive serum parietal cell antibodies, a decreased level of Pep I, and increased serum gastrin level, the patient was diagnosed with AIG.

Images of endoscopy, endoscopic ultrasound, and histology of the submucosal tumor in the fundus. (a) Esophagogastroduodenoscopy showed a gastric submucosal tumor with a reddish area on the top in the fundus. (b) Endoscopic ultrasound (radial scan, 10 MHz) revealed the submucosal tumor as a heterogeneous tumor with multiple small hypoechoic or anechoic areas (yellow arrows). (c) Endoscopic ultrasound (radial scan, 10 MHz) revealed a hyperechoic calcified area with acoustic shadow (red arrow). (d) Operative specimen. The tumor (20 mm in diameter) was completely resected. (e) Low-power view illustrated the inverted growth lesion, which was almost covered by normal gastric mucosa, resulting in a submucosal tumor. The lesion consisted of dilated glands in various sizes and shapes and fibroblast cells in the submucosa. (f) Medium-power magnification demonstrated foveolar and pseudopyloric or mucous-neck glands without cytological atypia and partial cystic dilation and the infiltration of inflammatory cells that were scattered in the stroma. (g) Medium-power magnification demonstrated that the submucosal glands or cystic elements were connected with the overlying gastric mucosa through a defect of the muscularis mucosa (blue arrows). (h) Medium-power magnification demonstrated a marked reduction of oxyntic glands and Continued.pseudopyloric and intestinal metaplasia in the oxyntic mucosa. (i) The foveolar epithelium of normal mucosa on the surface and foveolar type glands in the submucosal lesion were positive for the mucin-5AC immunohistochemical stain. (j) Immunohistochemical stain for mucin 6 showed positive glands in both the normal mucosa and the submucosal lesion. (k) A few propria gland cells were positive in the normal mucosa and no positive cells were observed in the submucosal lesion with Pep-I immunohistochemical stain. (l) CgA immunohistochemical stain highlights linear and nodular enterochromaffin-like cell hyperplasia and (m) Three calcification lumps within considerably dilated glands in the specimen.

Continued.
The EGD further showed two markedly reddish pedunculated polyps in the posterior and anterior walls of the upper-middle body (Figure 2a, 2b). We resected the two polyps by endoscopic mucosal resection. The polyps were histologically diagnosed as hyperplastic polyps (Figure 2c), which are characterized by irregularly shaped, haphazardly dilated hyperplastic foveola with pseudopyloric or mucous-neck glands lying in the edematous, inflamed stroma and small smooth muscle bundles in the mucosal layer.
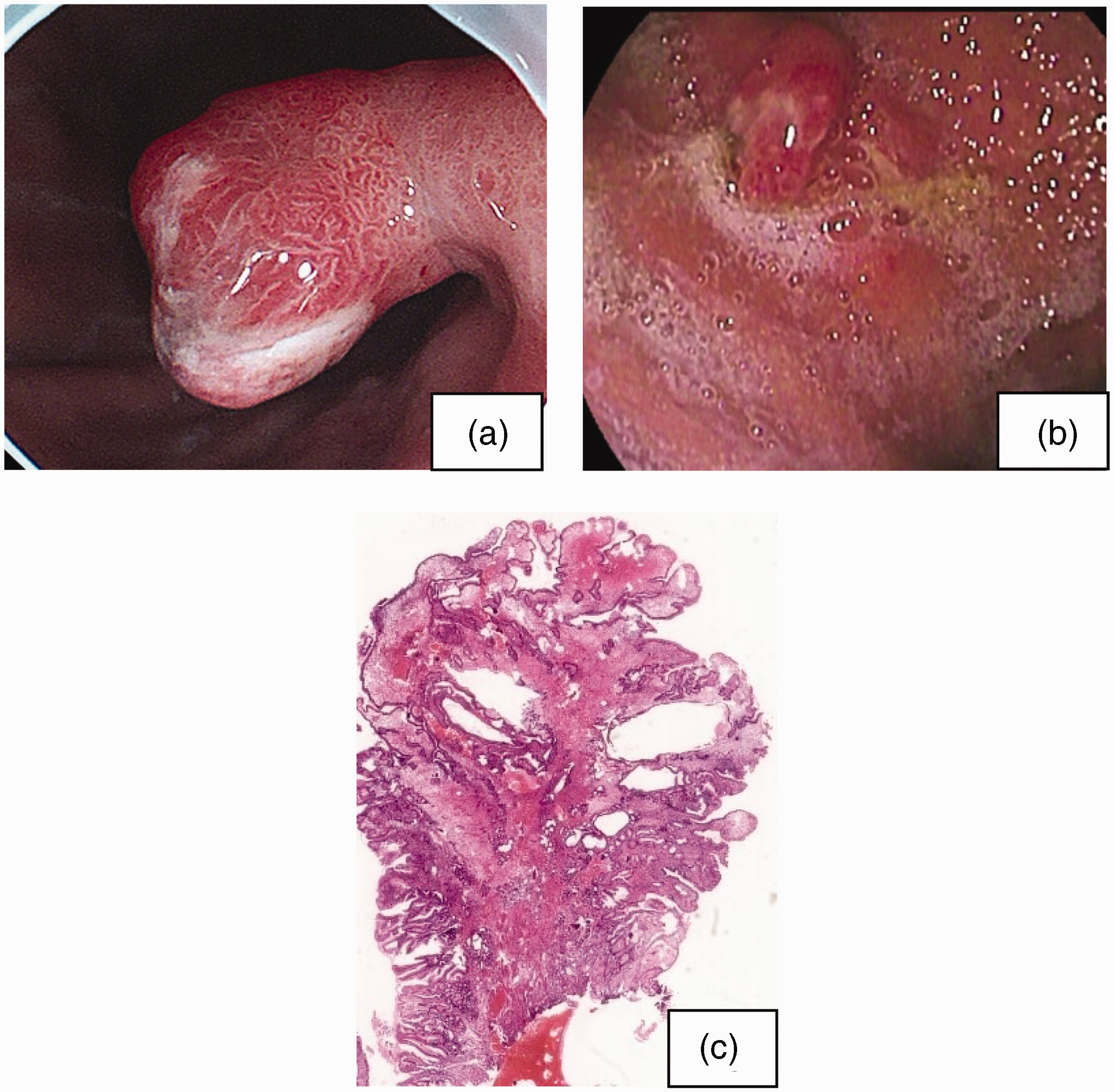
Endoscopic and histological images of pedunculated polyps in the body. (a, b) The esophagogastroduodenoscopy showed two markedly reddish pedunculated polyps in the posterior and anterior walls of the upper-middle body, with markedly atrophic manifestation in the oxyntic mucosa and a considerable amount of mucus attached and (c) Low-power view illustrated that the lesion consisted of irregularly shaped, haphazardly dilated hyperplastic foveola with pseudopyloric or mucous-neck glands in a background of edematous, inflamed stroma, and small, haphazardly distributed smooth muscle bundles in the mucosal layer.
No abnormal findings were obtained following routine blood tests, biochemical blood tests, or coagulation function tests. Major laboratory test results are listed in Table 1. A 99mTc-octreotide scan showed negative results.
Major laboratory findings.

IIFA = indirect immunofluorescence assay; TIBC = total iron binding capacity; ELISA = enzyme-linked immunosorbent assay.
Specific patient information was completely de-identified in the manuscript. The patient provided informed consent for treatment and for the publication of this report. The reporting of this case report conforms to the CARE guidelines. 3
Discussion
AIG is an increasingly prevalent disease characterized by loss of oxyntic mucosa and the presence of metaplastic epithelium and enterochromaffin-like cell hyperplasia. AIG is a CD4+ T cell-mediated organ-specific chronic inflammatory disease that occurs most commonly in older women. Our case was an elderly man and was therefore easy to misdiagnose. Diagnosing AIG in the early stage is difficult because of its non-specific clinical and subtle endoscopic manifestations. As the disease progresses, the mucosal atrophy in the gastric body and fundus is more obvious than that in the antrum, and flattened mucosal folds and visible submucosal vessels are observable on endoscopy. Histological diagnosis has been considered the gold standard for AIG. Furthermore, histological manifestations change with disease progression. 4 Typical histological characteristics are marked reduction of oxyntic glands, pseudopyloric and intestinal metaplasia, and the proliferation of mucous-neck and foveolar cells. Moreover, positive serum parietal cell antibodies, a decreased level of Pep I, and increased serum gastrin level are helpful in the diagnosis of AIG. Several patients do not experience anemia. The histological and laboratory findings of this case are consistent with the characteristics above. The damaged parietal cells in patients with AIG directly result in decreased gastric acid secretion. A low acid state leads to a compensatory increased number of G cells on the antrum, resulting in hypergastrinemia. 5 Several in vivo and in vitro experiments have confirmed that gastrin is the most important nutrient hormone in the stomach and can play an important role as a growth factor in the proliferation and differentiation of gastric mucosa, mucosal tissue remodeling, and vascular regeneration.6–9 Thus, AIG is closely related to gastric mucosal proliferative diseases such as hyperplastic polyps, adenomas, type I gastric neuroendocrine tumors, and gastric cancer; 1 long-term hypergastrinemia is speculated to be the most important inducing factor.10,11 Consequently, the early recognition and diagnosis of AIG in the clinic are crucial.
GHIPs account for fewer than 1% of all clinical gastric polyps. 2 GHIPs are mostly located in the upper body or the fundus of the stomach and are mostly asymptomatic.2,12 GHIPs with a stalk are classified as the polyp type, whereas those without a stalk are classified as the SMT type, which must be differentiated from neuroendocrine tumors, gastrointestinal stromal tumors, and the ectopic pancreas. 12 GHIPs are diagnosed using the histological characteristics of the tumor, which include glandular hyperplasia, cystic gland dilatation, the presence of fibroblast cells, smooth muscle proliferation, and nerve components in the submucosa.2,12–15 Endoscopic ultrasonography is helpful for preoperative diagnosis. GHIPs mostly originate from the second or third layer of the gastric wall, are heterogeneous, and have anechoic or hypoechoic cystic areas and hyperechoic calcified areas with acoustic shadow.12,16 In this case, the endoscopic ultrasonography findings are consistent with these previous reports. The pathogenesis of GHIPs remains unclear. Some researchers13,15,17 believe that the rupture and thinning of the muscularis mucosa are caused by repeated erosion and the conversion of gastric glands into the submucosa. The submucosal glands or cystic components are connected with the covered gastric mucosa through the defect of the muscularis mucosa. Rather than being considered hamartomas or being congenital in nature, GHIPs are believed to be formed by the ectopic inversion into the submucosa of proliferated mucosal glands. Therefore, GHIPs should be classified as inverted hyperplastic polyps. Other researchers 12 speculate that hamartomatous changes occur in the submucosa and that some of these components expand to the mucosa layer, resulting in the formation of hypertrophic lesions.
This case of AIG is complicated with a GHIP and two pedunculated hyperplastic polyps in the markedly atrophic oxyntic mucosa with intestinal metaplasia. To the best of our knowledge, such a complicated case has not been reported previously. The two types of polyps derived from the same background mucosa consisted of almost the same types of gland lining cells. In this case, the submucosal gland or cystic elements of the GHIP were connected with the overlying gastric mucosa through a defect of the muscularis mucosa, suggesting that the polyp may have been formed by the heterotopic inverted downgrowth of mucosal glands into the submucosa. These pathological findings may elucidate the relationships between GHIPs, hyperplastic polyps, and AIG and the underlying pathogenesis of GHIPs.
Conclusion
Like hyperplastic polyps, GHIPs may be a type of gastric mucosal proliferative disease. However, GHIPs are heterotopic in the submucosa through the defect of the muscularis mucosa. We highlight the importance of considering GHIPs as a differential diagnosis for SMTs in patients with AIG.
Footnotes
Authors’ contributions
JH conceived the report and participated in data acquisition and the drafting of the manuscript. ND critically revised the manuscript. BY was responsible for the pathological analysis. FM participated in the conception and design of the report. YW performed the endoscopic operations. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
The patient provided written informed consent for treatment and for the publication of this report. All details have been de-identified to ensure that the identity of the patient cannot be ascertained. The requirement for review board approval was waived because of the nature of this study (case report).
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
