Abstract
Objective:
Carotid atherosclerotic vascular disease (ASVD) represents an ongoing health problem and is responsible for a significant proportion of all cerebral ischemic events (CIEs).
Method:
A review of the literature was performed on the application of contrast-enhanced ultrasound (CEUS) to enhance the diagnosis of ASVD and further avoid CIEs.
Results:
Cerebral ischemic events are those resulting from reduction or cessation of perfusion to localized regions of the brain made manifest by neurological, typically stroke-like, symptoms. Traditional triplex ultrasound evaluation is a reliable and widely established method of identifying carotid ASVD lesions and grading the accompanying degree of focal stenoses and their hemodynamic impact. While this information plays an integral role in determining management of patients with significant carotid ASVD, it is less useful in classifying individual lesions as “vulnerable” or not. Vulnerable lesions are those that, based on their histological and morphological features, predispose a patient to an increased risk of a CIE due to plaque or thrombus embolization.
Conclusions:
The addition of CEUS to carotid artery diagnostic studies offers new potential in identifying vulnerable plaques and predicting which patients will progress to sequelae associated with a cerebral ischemic event.
Carotid atherosclerotic vascular disease (ASVD) represents a current health problem as it accounts for approximately 20% to 25% of all cerebral ischemic events in the United States. 1 The role of triplex ultrasound in the assessment of the extracranial carotid arteries is well established and has become, in most cases, the initial evaluatory modality for patients presenting with known or suspected ASVD.2–4 The mechanism giving rise to cerebral ischemic events (CIEs) is typically embolization of small blood clots (thromboemboli) or plaque fragments (atheroemboli) that originate from atherosclerotic lesions present within the lumen of the carotid bifurcation. While traditional triplex ultrasound has a clearly demonstrated efficacy in identifying flow-restricting ASVD lesions and quantifying the associated degree of stenosis, it has been admittedly less useful in determining whether a particular lesion can be classified as “vulnerable.” 5 Vulnerable atherosclerotic lesions are those that, because of their histological characteristics and morphological instability, are more likely to break up and give rise to subsequent ischemic embolic events. 6 While studies demonstrating the prevalence of vulnerable carotid plaques using ultrasound methods are scant, magnetic resonance imaging (MRI) studies have shown that they are present in approximately 8% of small lesions (1%–15%) and in 22% of larger carotid bifurcation lesions (16%–49%). 7 Emerging ultrasound technologies, particularly contrast-enhanced ultrasound (CEUS), offer exciting potential in identifying carotid atherosclerotic lesions that predispose an individual to an increased risk of a cerebral ischemic event. This article presents an overview of extant literature articles that focus on the role and efficacy of contrast-enhanced ultrasound in the characterization of vulnerable plaques.
Vulnerable Plaque—Definition
An atheroma, commonly referred to as an atherosclerotic plaque, is the accumulation of material on and beneath the intimal surface of an artery. Composition of these lesions varies, and each may contain an amalgamation of lipids, inflammatory cells, cellular debris, calcium, fibrous connective tissue, and thrombus. 8 The histological content ultimately determines the gross structural appearance, or morphology, of these lesions. There is ample evidence in the medical literature that particular structural types of plaque are predictive of an increased risk of both cerebral and cardiac ischemic sequelae.9,10 These plaques, termed vulnerable plaques, share distinct histological characteristics that include
Larger lipid core
Thinner fibrous cap
Plaque ulceration (a fissured fibrous cap)
Active inflammation within the lesion
The presence of vulnerable plaquing in the extracranial carotid circulation is clearly associated with an increased risk of a CIE and attendant symptomatic neurological sequelae.13,14 Of the various distinct histological characteristics listed above, neovascularization constitutes a major prognostic feature of vulnerability as these lesions are more prone to rapid bulk progression and rupture.15,16
Pathophysiology of Neovascularization in Atherosclerotic Plaques
Neovascularization is the process by which new blood vessels are generated and grow into a focal atheromatous lesion. In the absence of disease, the arterial vasa vasorum perfuses the outer (tunica adventitia) and middle (tunica media) arterial tissue layers. 17 The inner arterial layer (tunica intima) is normally perfused by oxygen diffusion from blood flowing within the vascular lumen. Arterial remodeling associated with ASVD results in a thickening of the intimal layer and impairs normal oxygen diffusion across the endothelial surface. Concomitant subintimal inflammatory processes also demand additional oxygen supply. 18 The subsequent intraplaque hypoxic state associated with these alterations signals the production of biochemical substances that stimulate the growth of microvessels into the evolving plaque.19,20 These neovessels arise from both the vasa vasorum extending through the tunica media into the base of the plaque and by epithelial lumen-derived microvessels penetrating the fibrous plaque cap 21 (Figure 1).
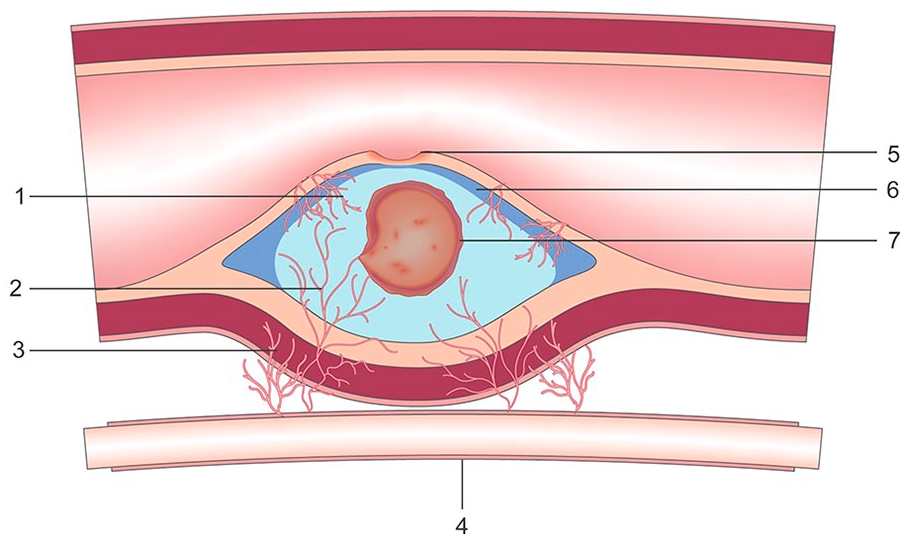
Vulnerable plaque—illustration. 1. Neovessels—epithelial origin. 2. Neovessels—vasa vasorum origin. 3. Vasa vasorum. 4. Capillary. 5. Fibrotic cap fissure (plaque ulceration). 6. Thin fibrous cap. 7. Intraplaque hemorrhage.
Because of the rapidity of their origin and growth, microvessels arising during the neovascularization process are, by nature, immature in histologic architecture and have a propensity to leak into and around a plaque. This vascular leakage consists of extravasated red blood cells (RBCs) and an accumulation of inflammatory fluids within the intima-media junction and at the edges of the plaque. Wider than normal gaps between cells in the walls of the microvessels also permit easy passage of lipids and macrophages into the actively evolving lesion. Intraplaque hemorrhage results from the erosion of the thin walls of these neovessels and/or terminal branches of vasa vasora and the spilling of blood into the atheroma. The net result of these processes is relatively rapid enlargement and an increase in plaque content and complexity. 22 A thinner fibrous cap, particularly in the presence of surface fissures (ulcerations), increases the likelihood of plaque rupture. When the lesion ruptures, a cascade of embolic debris is released into the carotid lumen and is ultimately transported to the anterior and middle portions of the brain via the internal carotid artery.
Contrast-Enhanced Ultrasound in Assessment of Plaque Morphology
Traditional Imaging
Gray-scale sonography has long been used in identifying the presence of plaque in the extracranial carotid arteries, and many attempts have been made to correlate sonographic appearance with clinical and prognostic significance. 23 While various schema have been devised to characterize plaques using subjective descriptors such as simple, soft, fibrotic, calcific, and complex, none have borne the weight of an evidence-based association with prognostic certainty or clinical outcomes associated with a CIE.23–25 Ulceration, as an indicator of plaque vulnerability, may be useful predicting neurological sequelae. However, using gray-scale sonography alone, the sensitivity in detecting ulceration is 77% in lesions less than 50% but only 41% in lesions greater than 50%. 26 This is similar to the data obtained using catheter-based angiography (77% and 48%, respectively). 27 The addition of color Doppler imaging does little to improve gray-scale accuracy, whereas CEUS as a stand-alone sonographic modality in identifying plaque ulceration does with a sensitivity of 88% and a specificity of 59%. 28
Contrast-Enhanced Imaging
Emerging ultrasound technologies have added expanded capabilities in characterizing the morphological features of carotid plaques.29,30 Chief among these is CEUS, which improves the detection and characterization of pathologies based on perfusional dynamics of the tissue being studied. 31 Intravenously injected ultrasound contrast agents are considered “blood pool” agents as they remain intravascular at all times and do not permeate into adjacent parenchymal tissues. As such, they act effectively as red blood cell tracers and are ideally suited in imaging the vascular perfusional patterns feeding and flowing through a tissue bed. 32
The use of high-frequency broadband transducers, harmonic received frequencies, and advanced digital signal processing methods yield ultrasound imaging techniques that can accurately display and track CEUS microbubbles as they course through tissue vasculature. Enhanced spatial, contrast, and temporal resolving capabilities permit real-time display of blood flow through larger feeding vessels and spilling into arterioles and capillary beds with good sensitivity and specificity. 33 This is particularly efficacious in plaque evaluation for detecting tissue microperfusion, a hallmark of vulnerable neovascularized plaques.34,35 CEUS has been shown to be an efficacious method of differentiating vulnerable from “nonvulnerable” carotid plaques. 34
In a study done by Xiong et al., 36 CEUS demonstrated 80% sensitivity and 70% specificity in correlating CIE symptoms with sonographic findings consistent with the presence of plaque neovascularization. In addition, when quantitative CEUS assessment was performed, they concluded that positive findings were “significantly greater” in symptomatic versus asymptomatic patients. As such, contrast-enhanced imaging provides the potential for improving plaque risk stratification and identifying those patients with greater potential for experiencing a CIE, offering a new standard for managing patients with carotid ASVD.37,38
Quantitative Contrast-Enhanced Imaging
In addition to 2D real-time display of CEUS images, the interpretation of which relies on the skill and experience of the interpreter, analytical methods have been devised that can quantify microbubble concentration within a given region of interest (ROI). The ability to visualize and quantify tissue perfusion is a useful imaging adjunct in the clinical assessment of a wide range of pathological conditions involving changes to local blood flow into tissue. 39 There are a number of methods available to do this; however, the most widely used are values obtained from a time-intensity curve (TIC).
Since it is assumed that the returning acoustic signal intensity is proportional to the volume of microbubbles present within the ROI, digital signal processing techniques can be used to quantify blood flow into that region. As contrast microbubbles remain strictly intravascular, variations in signal intensity over time offer data on the degree of vascularization within the analyzed tissue sample. Intensity curves can be plotted that can be correlated with various perfusional patterns and, ultimately, various tissue types. 40
CEUS Sonographic Findings
Several sonographic findings have been reported that demonstrate the utility of CEUS in enhancing carotid artery diagnosis. Microbubble enhancement of the carotid lumen can reveal previously undetected plaque irregularities and morphology, particularly plaque surface ulcerations. 41 It also provides improved resolution of the common carotid artery intima-media thickness (c-IMT), particularly on the near wall, which can serve as a surrogate marker for ASVD 42 (Figure 2). And, within the context of this discussion, neovascular changes feeding into and within focal plaques can be clearly imaged. 43
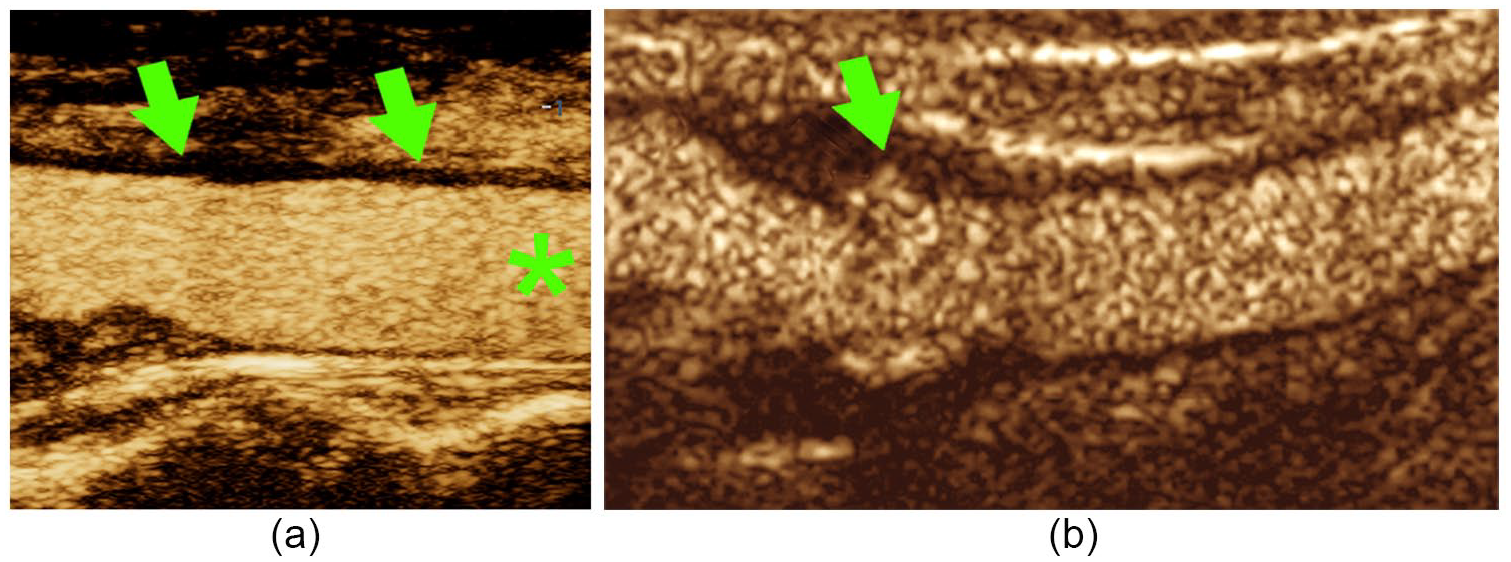
Contrast-enhanced ultrasound carotid imaging enhancements. (a) Microbubble enhancement of carotid lumen (*) and improved resolution of carotid artery intima-media thickness, particularly on the near wall (arrows). (b) Enhanced visualization of plaque surface ulceration (arrow).
Neovascularization—CEUS
Sonographic findings associated with plaque neovascularization are fairly straightforward. Overall contrast enhancement (increased echogenicity) of the lesion itself has been correlated with both histological evidence of neovascularization and clinical symptomatology associated with the presence of a vulnerable carotid artery plaque. 44 These findings have been subjectively semiquantified in some studies ranging from 0, indicating the absence of neovessels (Figure 3), to 3, where real-time imaging reveals the presence of pulsating arterial flow into the base and body of the plaque 45 (Figure 4).
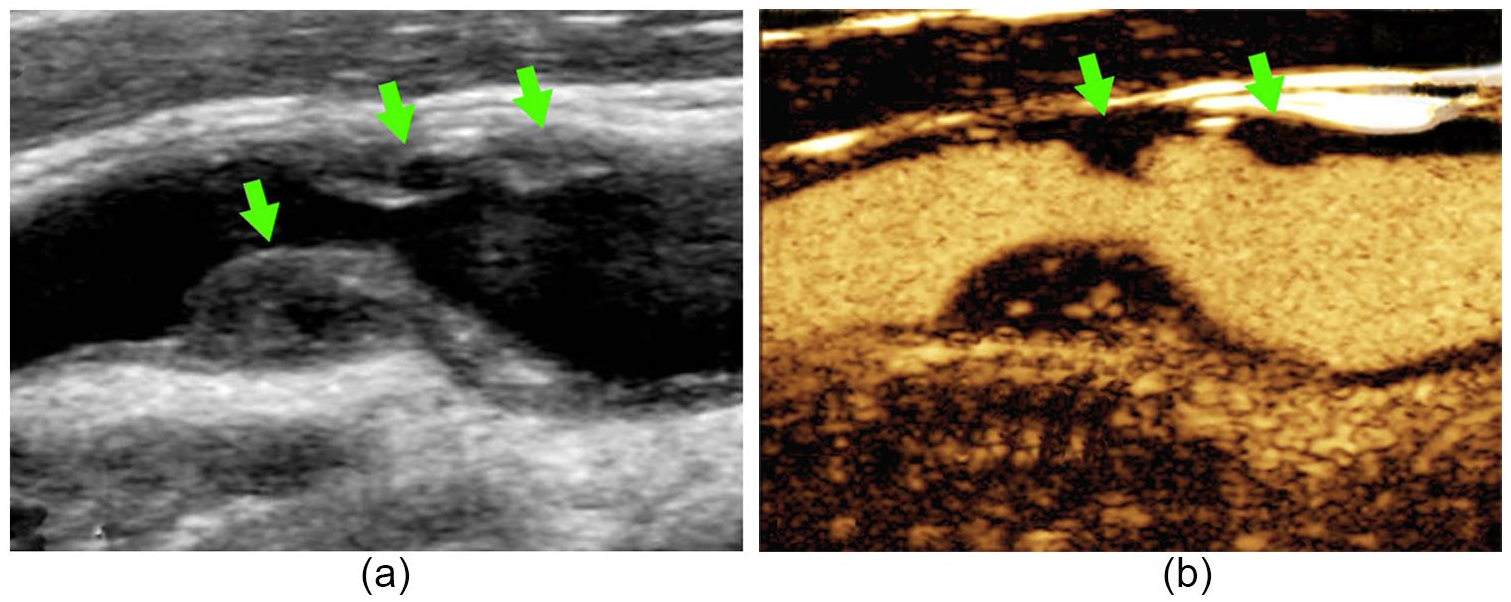
Contrast-enhanced ultrasound (CEUS) findings. (a) Gray-scale imaging demonstrates the presence of several complex plaques in the carotid bulb and proximal internal carotid artery (arrows). (b) CEUS demonstrates absence of overall enhancement in body of several nonvascularized plaques (arrows).
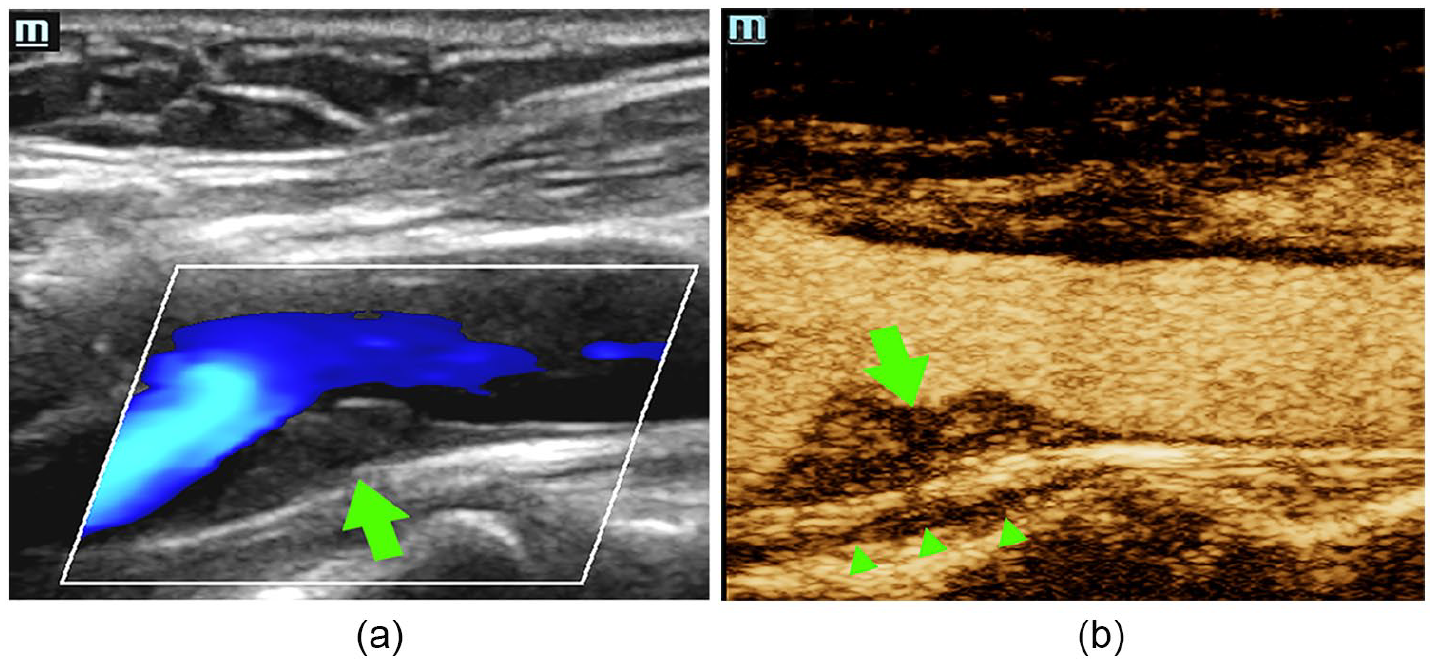
Contrast-enhanced ultrasound findings. (a) Gray-scale and color Doppler imaging demonstrating the presence of a focal complex plaque in the proximal internal carotid artery that does not significantly restrict flow (arrow). (b) Same lesion demonstrating increased enhancement in microvascularity within the plaque (arrow) and microbubble flow through the vasa vasorum (arrowheads).
Neovascularization—Quantitative CEUS (Q-CEUS)
Quantitative CEUS (Q-CEUS) provides data on microbubble intensity within a plaque by comparing values obtained from three related ROIs:
Within the atherosclerotic plaque
Normal arterial wall (a virtually nonperfused reference region)
Carotid lumen (region of maximum contrast enhancement) 46 (Figure 5a)
Intensity values in a neovascularized plaque fall between those obtained from the carotid lumen and those obtained from the normal arterial wall (Figure 5b). Increased microbubble intensity within the plaque, as compared to normal carotid wall, has been correlated histologically with plaque neovascularity and confirms the gray-scale sonography findings.34,47 While TICs provide measurable intensity values useful for relative comparisons, there are no widely accepted, specific numerical values that are pathognomonic for plaque neovascularity reported to date.
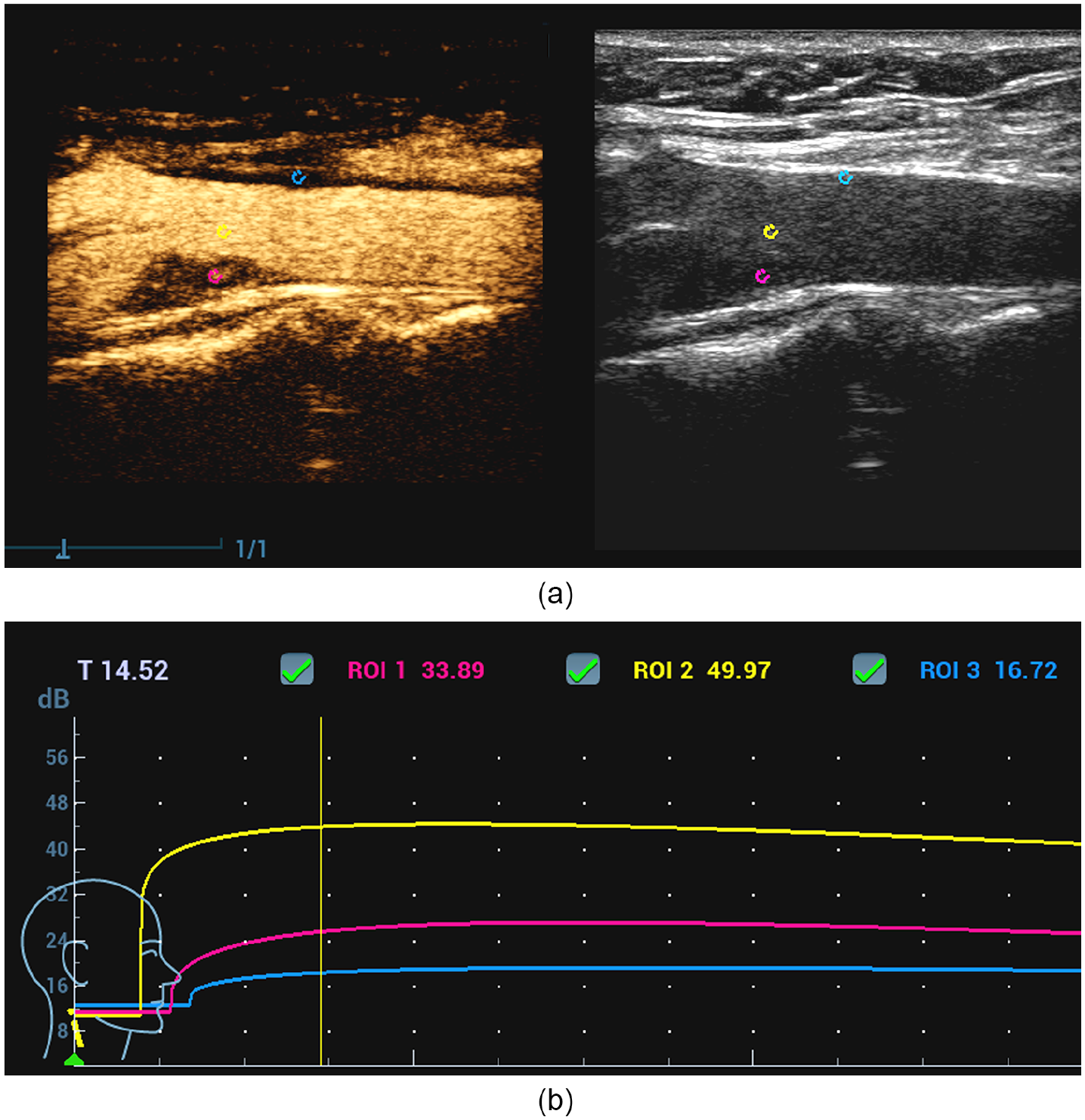
Quantitative contrast-enhanced ultrasound (CEUS). (a) CEUS and gray-scale reference image demonstrating placement of contrast intensity samples within the same vessel seen in Figure 4. Samples are placed within the plaque (magenta), normal arterial wall (cyan), or carotid lumen (yellow). (b) Time-intensity curve demonstrates increased microbubble intensity in the plaque (magenta) compared to the normal arterial wall (cyan) but decreased compared to lumenal flow (yellow).
Conclusion
Triplex sonography has a long history of providing an accurate assessment of carotid artery pathology. Estimation of plaque bulk, subjective description of morphology, and hemodynamic impact of focal lesions all provide valuable information in managing patients with ASVD, both symptomatic and asymptomatic.
The introduction of contrast-enhanced ultrasound to the sonographic armamentarium permits differentiation of lower-risk atherosclerotic lesions from those that are higher risk. In addition, these lesions are known to place the patient at higher risk for a significant cerebral ischemic event.
Footnotes
Acknowledgements
The author thanks Kendall Dea for creation of the original illustration demonstrating vulnerable plaque morphology and Dr. Zhao Lu, Mindray Innovation Center, for providing the CEUS images.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
