Abstract
The patient with a bronchopleural fistula and acute respiratory distress syndrome can present a therapeutic challenge for the treating clinician. In this case, the authors describe the use of bedside thoracic sonography to show real-time improvement in a pneumothorax after initiation of high-frequency oscillatory ventilation. Sonography may have a role in the evaluation of ventilator strategies in the future, although validation of this application is still needed.
Keywords
Background
The patient with a bronchopleural fistula (BPF) and acute respiratory distress syndrome (ARDS) can present a therapeutic challenge for the treating clinician. In this case, we describe the use of bedside thoracic sonography to show real-time improvement in a pneumothorax after initiation of high-frequency oscillatory ventilation (HFOV). Sonography may have a role in the evaluation of ventilator strategies in the future, although validation of this application is still needed.
Case Report
A 19-year-old man was admitted to the trauma intensive care unit (ICU) after a high-speed motor vehicle accident where he was ejected through the windshield. The patient was intubated in the field by paramedics for airway protection. His initial chest x-ray demonstrated bilateral hemopneumothoraxes, and bilateral tube thoracostomies were performed in the emergency department. Computed tomography (CT) of the abdomen demonstrated splenic and liver lacerations. Hypotension following his abdominal CT scan resulted in immediate transfer to the operating room, where splenectomy and liver packing were performed. Additional injuries included a left femoral neck fracture and a right clavicle fracture.
Over the course of the first three days of hospitalization, the patient had an additional three thoracostomy tubes placed in the right hemithorax for persistent pneumothorax and air leak. During this time, there was progressive worsening of his pulmonary contusions, leading to increasing oxygen requirements. On the afternoon of the third hospital day, the patient was placed on bilevel ventilation, with settings of pressure high = 30 mm Hg, time high = 1.5 seconds, pressure low = 10 mm Hg, time low = 1.5 seconds, and fractional inspiration of oxygen (FiO2) = 80%. On these settings, his arterial blood gas (ABG) was pH 7.40, PCO2 44 torr, PO2 122 torr (normal ABG values breathing room air: pH 7.35-7.45, PCO2 35-45 torr, PO2 80-100 torr), and bicarbonate 27 mEq/L, and his tidal volumes averaged 650 mL. On the morning of hospital day 4, with the same ventilator settings, his returned tidal volumes averaged 580 mL. An ABG at this time was PCO2 44 torr and PO2 56 torr. Later that afternoon, on FiO2 of 100%, his tidal volumes averaged 498 mL, and his ABG results were PCO2 53 torr and PO2 62 torr. A BPF was suspected but not identifiable on bronchoscopy. At 3
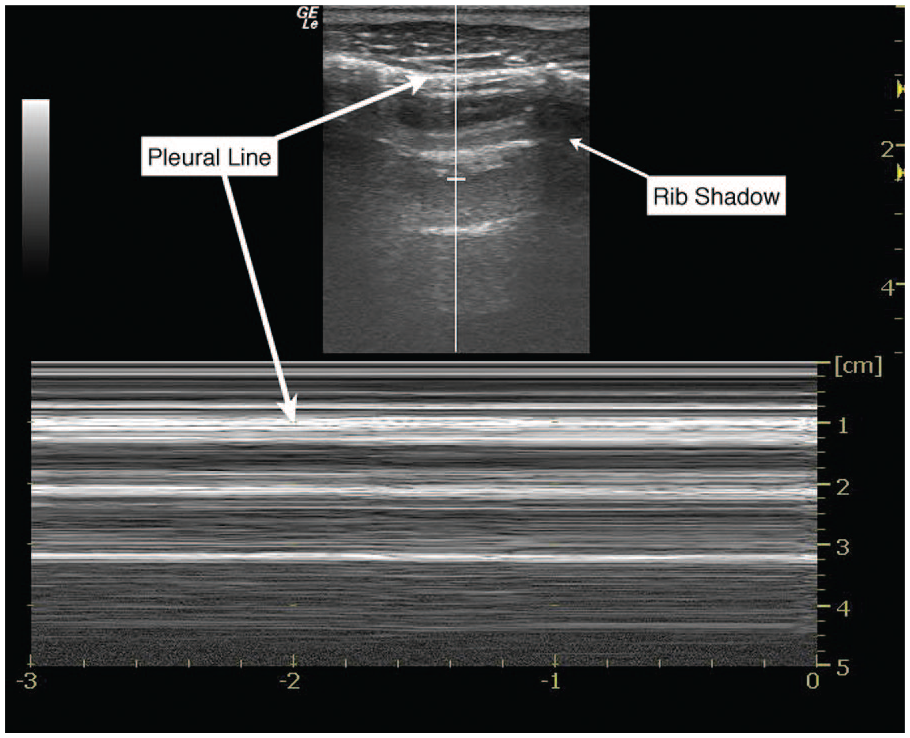
M-mode ultrasonographic appearance of pneumothorax.
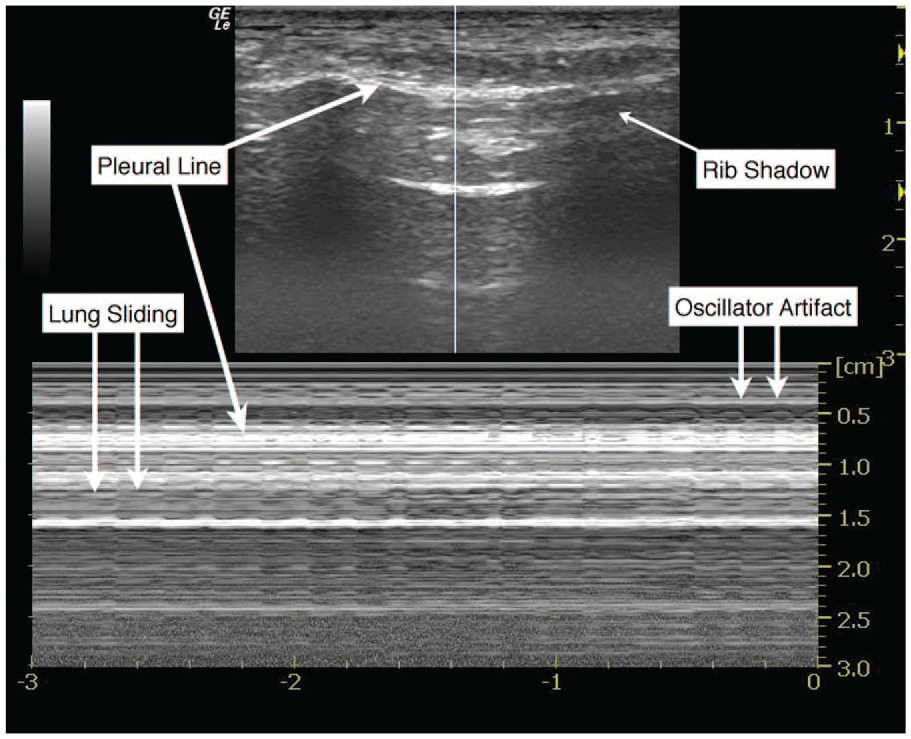
M-mode ultrasonographic appearance of normal pleural sliding on a high-frequency oscillatory ventilator.
Methods
Sonographic images were obtained using a General Electric LOGIQ e (GE Healthcare, Piscataway, New Jersey) with linear transducer 8L-RS (4-12 MHz). Using a systematic approach, four regions were scanned bilaterally on the supine patient: the upper and lower aspects of the anterior and lateral chest. The sonographic examination of the lung has been described as also including two posterior regions (upper and lower), but these were not attempted for this patient because of clinical instability.
The six total chest regions are delineated by the anterior and posterior axillary lines, with upper and lower aspects for each territory. The linear transducer is held along the cephalocaudal axis of the patient, and each intercostal space is evaluated. Using a rostral to caudal progression, moving from intercostal space to intercostal space, the sonologist can evaluate the majority of the superficial lung parenchyma. A complete sonographic examination will include a description of findings above (e.g., subcutaneous air), at (e.g., pneumothorax), and below the pleural line (e.g., consolidation). Depending on the clinical indication for obtaining thoracic imaging, brightness mode1–5 (B-mode), motion mode6,7 (M-mode), and power color Doppler 8 applications have been described.
Discussion
BPFs are relatively rare but feared complications in pulmonary medicine.9,10 A BPF is a communication between the bronchial tree and the pleural space. The condition is most commonly a perioperative complication, although it can occur in other settings (e.g., infection, malignancy, trauma, postradiation, postchemotherapy, persistent pneumothorax, ARDS, etc.). ARDS, on the other hand, is a common condition in the ICU, with descriptions dating back to the 1960s. A 1994 consensus conference defined the syndrome as acute-onset severe hypoxemia, with bilateral infiltrates on chest radiograph, not caused by heart failure. 11 There are both ventilator- and non-ventilator-based strategies for treating ARDS; conventional ventilator management includes maintaining titrated amounts of positive pressure in the ventilator circuit at the end of a patient’s respiratory cycle.12,13 Increasing the amount of positive pressure improves oxygenation for patients with less severe disease. Management of ARDS in the setting of BPF, however, presents a particular challenge, as the same positive pressure employed to improve oxygenation may worsen the defect between the bronchial tree and pleural space. This has the potential consequence of perpetuating a pneumothorax as well as preventing adequate ventilation of the patient.
In the face of increasing positive pressure requirements to maintain acceptable saturations, HFOV was employed here in the hope that oxygenation could be improved through an increased mean airway pressure and at the same time minimize fistula flow by increasing airway impedance and reducing the importance of lung compliance on air flow distribution. In addition, HFOV maintains the mean airway pressure with smaller cyclical changes and lower peak airway pressures. 14 To this end, HFOV has previously been described in adults with BPF and ARDS but not rigorously studied. 9 Furthermore, although several studies have investigated the role of HFOV in ARDS,15–17 controversy remains over its role in patients with BPF and concomitant parenchymal diseases such as ARDS. 9
In this case, on initiation of HFOV, bedside transthoracic sonography was performed by the treating clinician. The examination demonstrated immediate improvement in the patient’s pneumothorax, accompanied by resolution of the thorocostomy circuit air leak (a finding consistent with closure of the BPF). Image acquisition took less than five minutes to perform and did not require the patient to be transported out of the ICU.
The evaluation of pneumothorax by bedside sonography has been well described. 2 The presence of air between the visceral and parietal pleura creates distinctive artifact patterns that are easily identifiable. Classic findings of pneumothorax using B-mode sonography include the absence of a “comet-tail” artifact (see Figure 3) and the absence of pleural sliding. 18 In M-mode, the stereotypical pattern of pneumothorax has linear streaks across the near field of the sonographic image, abutted by a bright pleural line, and then again linear streaks across the bottom of the image (also known as the “stratosphere sign”; Figure 1). 6 Above the pleural line, sonographic images of normal lung have a similar ultrasonographic appearance to the pneumothorax pattern; however, below the pleural line, there is a granular pattern that represents artifact from normal lung movement during respiration (also known as the “seashore sign”; Figure 4). 6 In cases where B-mode evaluation for pneumothorax is equivocal, M-mode may provide additional support for the presence or absence of lung sliding. Figure 2 shows a 5-Hz repeating pattern starting at the pleural interface, with interposed areas of lung sliding that correspond to the respiratory cycle. In patients without a pneumothorax who are receiving conventional ventilator support or who are spontaneously breathing, the M-mode sonographic appearance of lung sliding is similar. Figure 5 (online at http://jdms.sagepub.com/content/by/supplemental-data) is a B-mode cine-clip that demonstrates lung sliding and the comet-tail artifact while on the HFOV. It is worth noting that the features of pneumothorax described above are not limited to patients with BPFs; rather, these pathological findings are found with any etiology of pneumothorax.
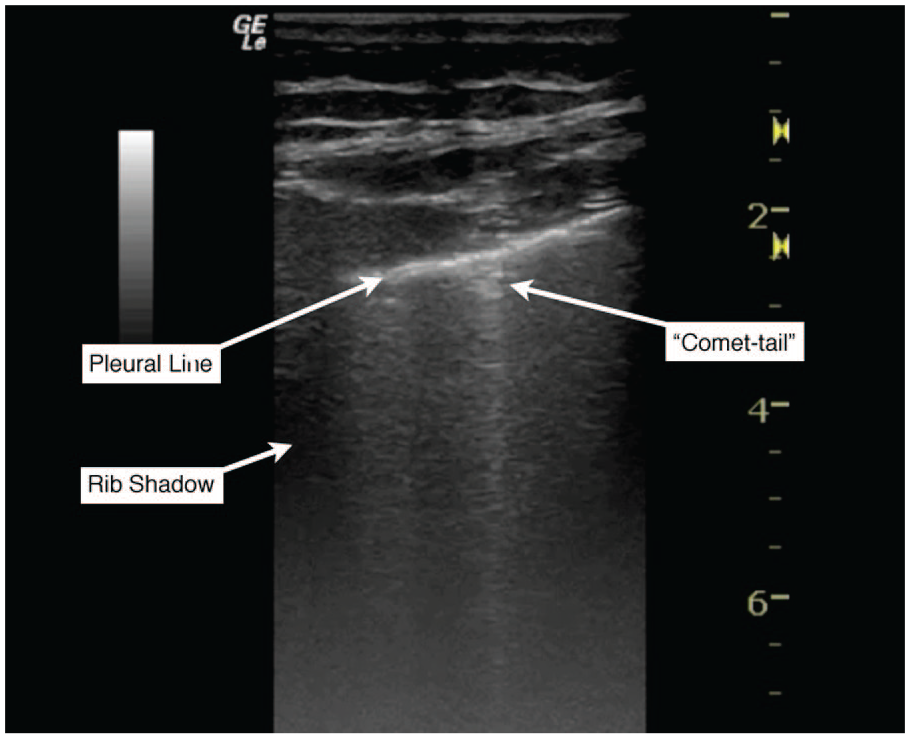
B-mode ultrasonographic appearance of normal pleural sliding and comet-tail artifact.
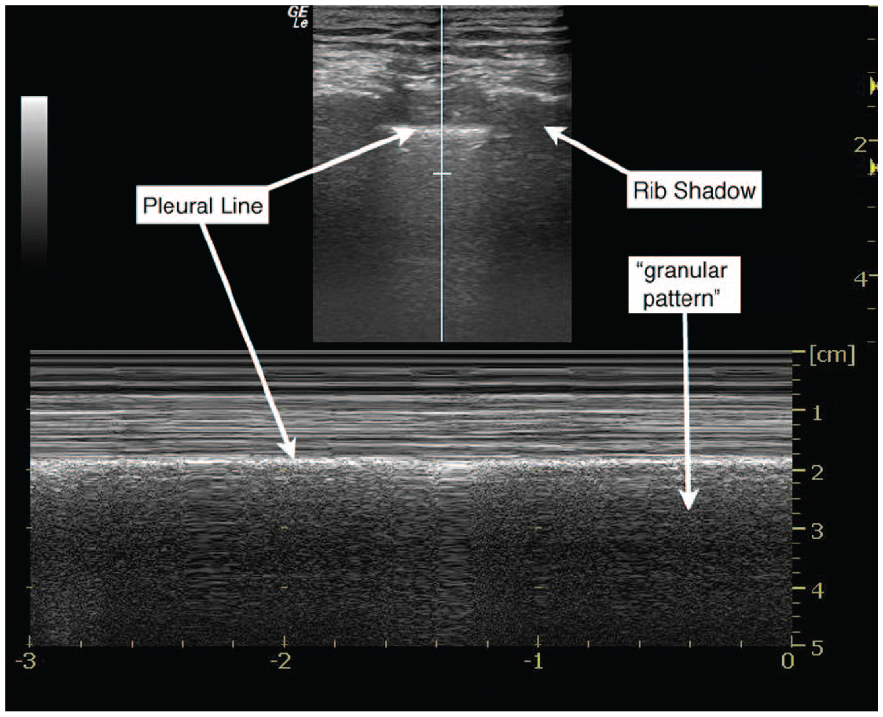
M-mode ultrasonographic appearance of normal pleural sliding.
Conclusion
The management of patients with BPF and ARDS is challenging. HFOV is a modality with reported benefits in selected patients; however, factors predicting success for individual patients have not been identified. Bedside sonography may offer a real-time opportunity to evaluate the effectiveness of this and other mechanical ventilation strategies. In this case, the sonographic findings corresponded with improvements in the pneumothorax, as well as subsequent pulmonary function recovery. As an easily attained noninvasive study that does not require the patient to leave the intensive care unit and has performance characteristics that complement the portable chest x-ray, thoracic sonography will increasingly have a role in the evaluation and care of the critically ill.
Footnotes
Acknowledgements
The authors thank respiratory therapists Nicole Woods and Kyle Crago, whose expertise with high-frequency oscillator ventilation was invaluable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
Funding
The authors received no financial support for the research and/or authorship of this article.
