Abstract
Pseudoaneurysm (PSA) formation, in an arteriovenous fistula (AVF) undergoing hemodialysis, can occur as a result of frequent venipunctures and can lead to a weakened vessel wall. Since many dialysis patients’ lives depend on their weekly hemodialysis sessions, complications that result from dialysis require treatment. A PSA can be a life-threatening complication, especially for these patients. Sonography has been demonstrated to be a useful modality of choice in the screening, diagnosis, and treatment of a PSA. The sonographic appearance of a PSA using gray-scale, color, and spectral Doppler can play a significant role in ruling out other diagnoses. A case study is presented of a dialysis-dependent patient who developed a PSA in his dialysis access site. Sonography played a key role in the diagnosis and follow-up for this specific case. Due to the risks involved with these patients, there is an essential need for sonographers to be competent in performing these examinations. Evaluation of the AVF to check for patency and a thorough assessment of the PSA are required.
Keywords
The formation of a pseudoaneurysm (PSA) in dialysis access sites can occur variably among the end-stage renal disease population. 1 A hemodialysis arteriovenous fistula (AVF) is a surgically created connection between an artery and a vein used most commonly for patients undergoing dialysis.2,3 A PSA can be a severe complication of an AVF, which can interfere with dialysis. There is disagreement in the literature on the definition and classification of PSA occurring in an AVF. 2 The natural history of aneurysms in vascular access (VA) is poorly defined, and although classifications exist, they are not uniformly applied in studies or clinical practice. 2 Treatment of pseudoaneurysms and true aneurysms of VA is not evidence based but relies on clinical experience and available facilities. As a result of reduced renal function, patients with chronic kidney disease (CKD) may have contraindications to imaging modalities that sometimes require contrast, such as computed tomography (CT) and magnetic resonance imaging (MRI). Contrast-induced nephrotoxicity (CIN) and nephrogenic systemic fibrosis (NSF) are both risks for CKD and dialysis patients who receive a CT or MRI. 4 Therefore, sonography can play a significant role in the screening, diagnosis, and treatment of pseudoaneurysms that occur in dialysis patients.2,3
Case Study
A male patient in his eighth decade of life presented to the emergency department for right arm pain, swelling, and a pulsatile mass proximal to the antecubital fossa near his dialysis access site (Figure 1). The patient’s history included end-stage renal disease (ESRD) and dialysis three times per week for the previous two years. The patient received dialysis through his brachiobasilic AVF (Figure 2). 5 The patient stated that he noticed the pulsatile mass one month earlier following one of his weekly dialysis sessions. When he brought it to the attention of his dialysis team, he was instructed to report to the emergency department to be evaluated for a suspected PSA in his VA. A sonogram of the right upper extremity was requested and performed to evaluate the pulsatile mass suspected as being a PSA. The sonographer performed the examination with a 12-MHz linear transducer on a Philips iU22 ultrasound machine (Philips Healthcare, Andover, MA). The patient was placed in the supine position with the right arm extended at 60 degrees from his side and placed on a pillow. The sonographer interrogated the right upper extremity, including the AVF, to check for patency; a thorough analysis of the basilic vein and the brachial artery was performed along with evaluation of the anastomosis, which connected the two vessels. Gray-scale, color Doppler, and pulsed-wave Doppler were used to aid in diagnosis. A sonolucent mass corresponding to the patient’s palpable mass was confirmed. The mass originated anterior to the basilic vein just inferior to the AVF anastomosis (Figure 3). The mass measured 3.2 × 2.5 × 2 cm (Figure 4A,B). The neck, which was 3 mm in width (Figure 5), extended from the basilic vein, which was the outflow site for the AVF (Figure 6). Color Doppler was applied to help identify flow direction and to demonstrate the “yin-yang sign,” which is a diagnostic finding in PSAs (Figure 7). 6 The “yin-yang sign” demonstrates a swirling pattern displayed as blue on one side and red on the other that indicates flow reversal and turbulence. 6 Pulsed-wave Doppler was then used to assess flow characteristics within the mass (Figure 7), within the suspected PSA neck (Figure 8), and within the fistula anastomosis (Figure 9). The flow characteristics of the mass and neck verified “to-and-fro” flow, which is visualized as flow above and below the baseline (Figures 7 and 8). 6 The AVF anastomosis had a normal high velocity (183 cm/s) and a low-resistant waveform, which is suggestive of a functioning AVF (Figure 9). 7 The radiologist concurred with the sonographer’s impression of a PSA originating from the AVF. A central venous catheter (CVC) was placed in the patient’s right internal jugular vein temporarily to receive dialysis until the PSA resolved into a hematoma or treatment was given. No treatment was given at that time for the PSA itself. The radiologist recommended close monitoring of the PSA with serial sonograms to assess its progression or resolution. Six weeks later, computed tomography angiography (CTA) and a follow-up sonogram were performed to reassess the PSA. The PSA resolved into a hematoma, and no further treatment was initiated. Use of the CVC was discontinued and the AVF was again used for dialysis.
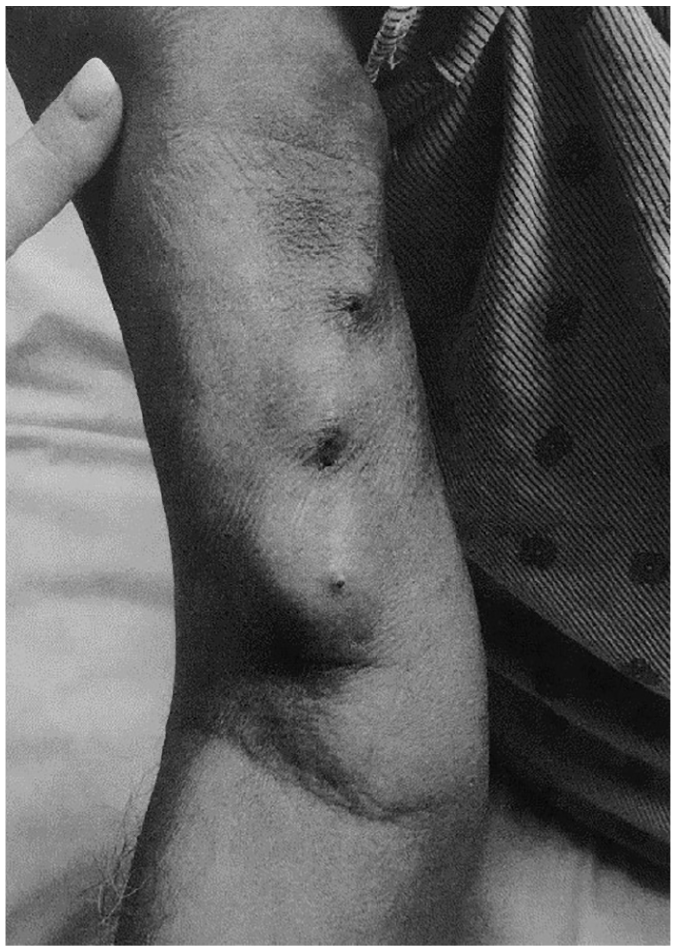
Pulsatile mass proximal to the antecubital fossa on the patient’s right arm.
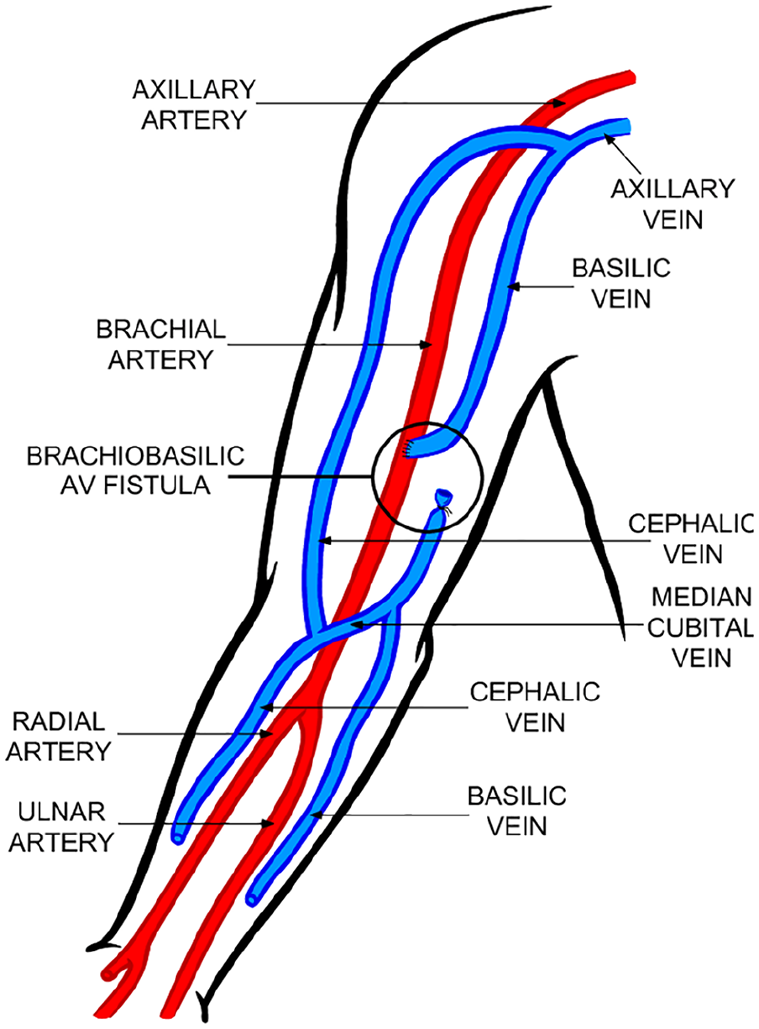
Arteriovenous fistula demonstrating an end-to-side connection between the right basilic vein and right brachial artery. 5
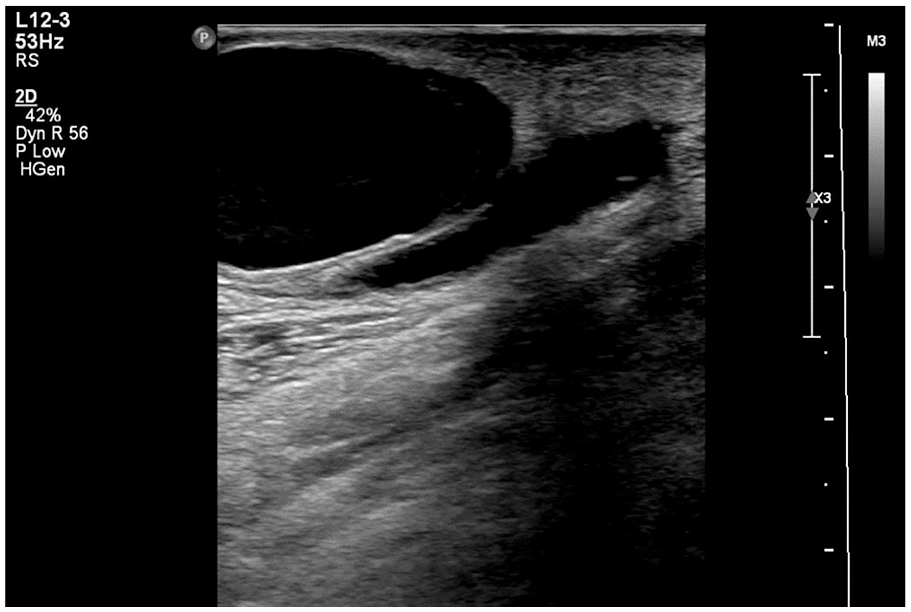
Gray-scale sonogram in the longitudinal scan plane. A sonolucent mass is seen originating from the basilic vein just inferior to arteriovenous fistula anastomosis.
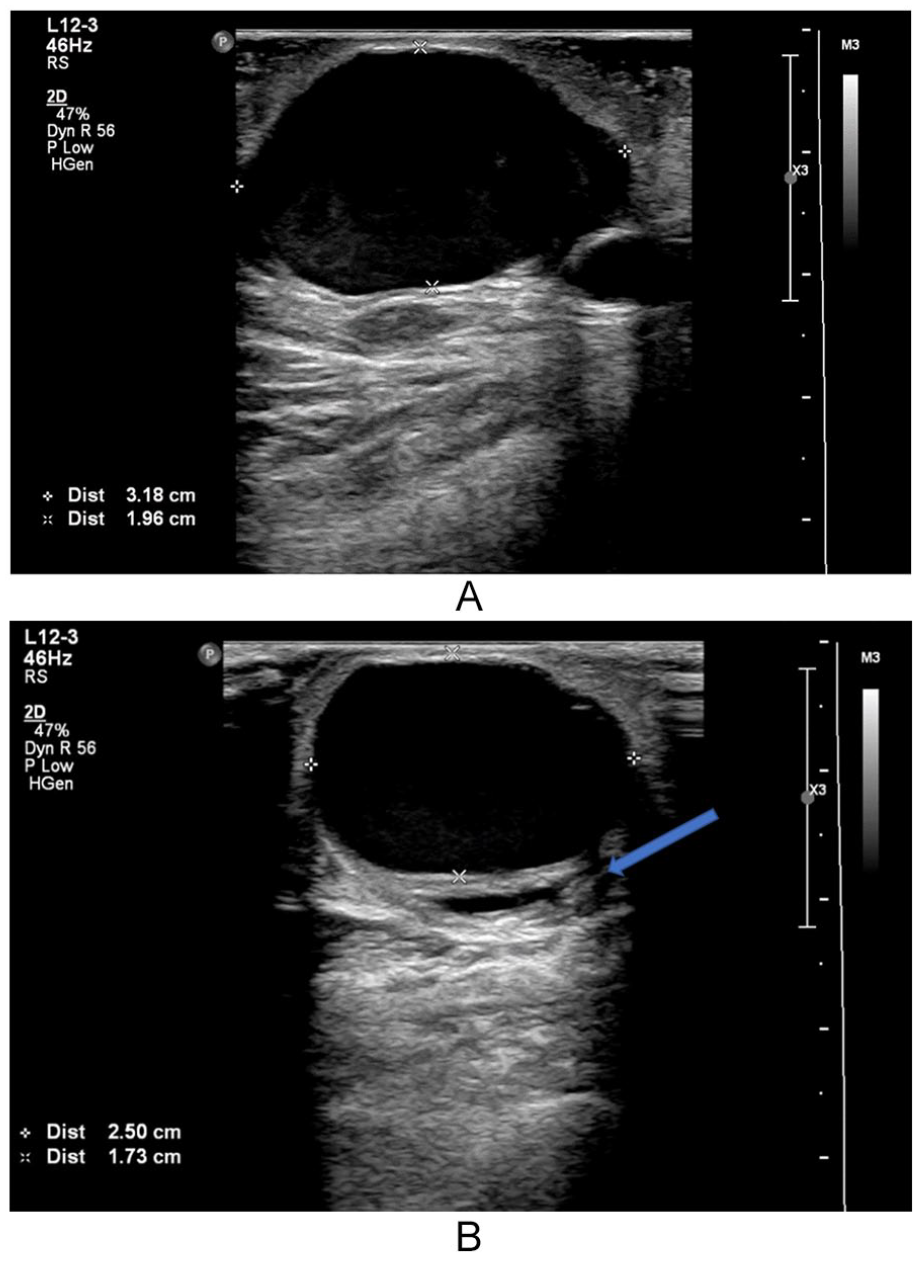
(A) Gray-scale sonogram in the longitudinal scan plane of the sonolucent mass. The mass measures 3.2 cm in length and 2 cm in anteroposterior diameter. (B) Gray-scale sonogram in transverse demonstrates a width measurement of 2.5 cm. The pseudoaneurysm appears to be compressing the proximal portion of the basilic vein (see arrow), which is the outflow site of this patient’s arteriovenous fistula.
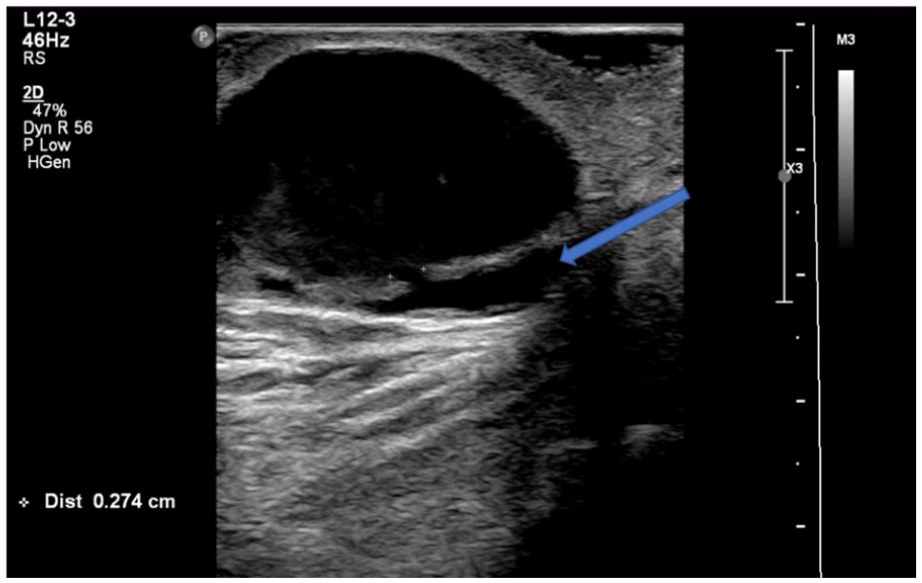
Gray-scale sonogram of the pseudoaneurysm’s neck measuring 3 mm in width. It is evident that the neck originates off the basilic vein (see arrow). Identification of a neck rules out a true aneurysm.
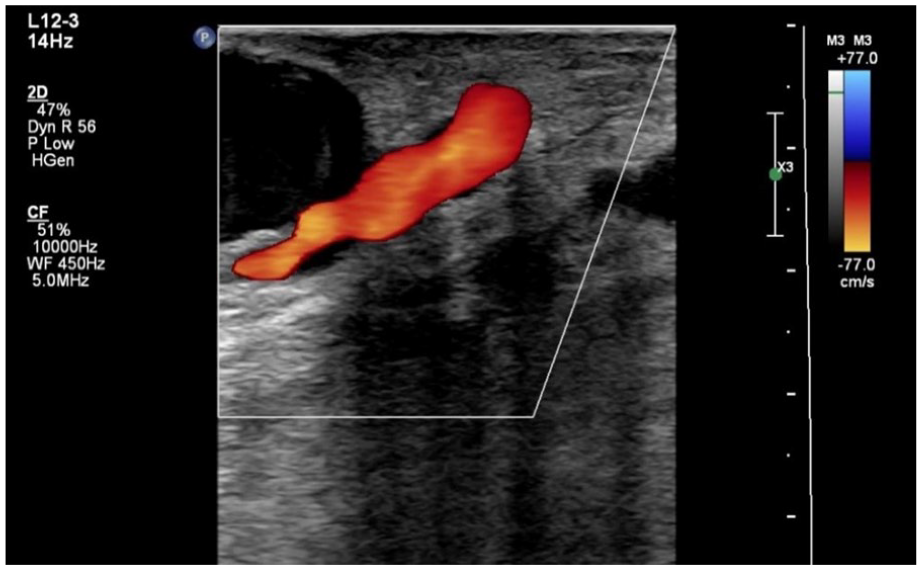
Color Doppler was used to demonstrate flow within the basilic vein.
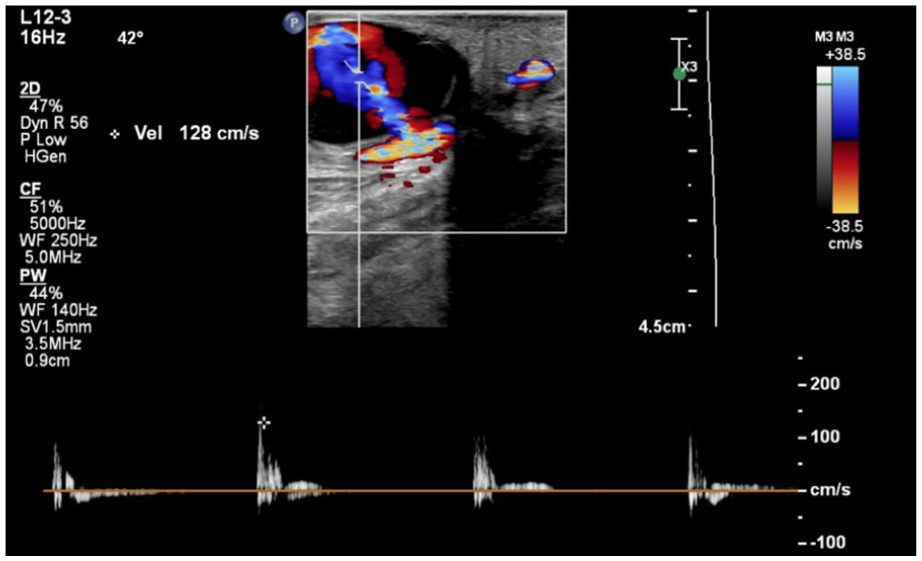
Duplex Doppler imaging displays the turbulent flow within the pseudoaneurysm. The “yin-yang sign” is appreciated within the mass.
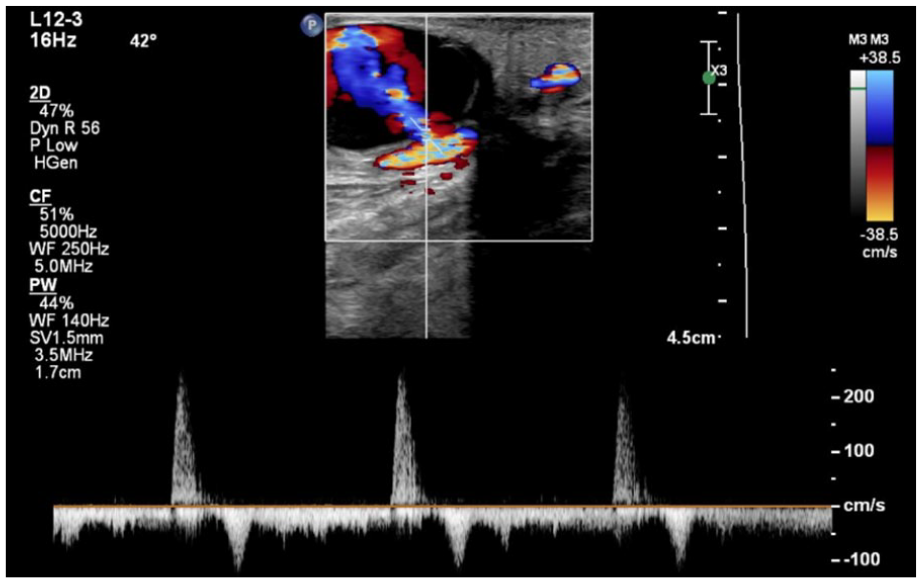
A spectral Doppler sample was taken within the pseudoaneurysm’s neck. “To-and-fro” flow is seen within the tracing, which is a classic characteristic of a pseudoaneurysm.
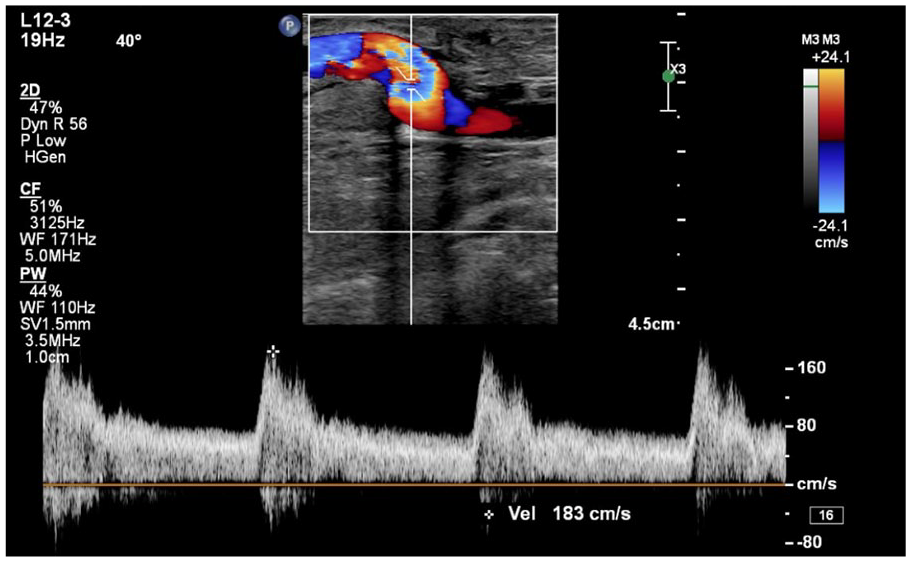
A spectral Doppler sample at the anastomosis of the right basilic vein and right brachial artery. The high-velocity, low-resistant waveform is indicative of a functioning arteriovenous fistula. The velocity is approximately 183 cm/s, which is within normal limits.
Discussion
End-stage renal disease has become prevalent in the United States due to the increase of diabetic and hypertensive patients. 1 Hemodialysis is essential for those with end-stage renal disease and is the method of treatment for a large percentage of this patient population. 1 To effectively receive this treatment, a reliable VA site is necessary. Complications associated with VA sites occur frequently, and those affected account for 36% to 39% of dialysis access-related admissions. 2 There are three main types of dialysis access that are in current use in the United States: the AVF, the arteriovenous graft (AVG), and the tunneled catheter. The preferred choice for dialysis is the AVF. This is the safest form of dialysis access with the least complications.2,3 A native vein and artery are surgically connected usually in the forearm or upper arm for this type of VA.2,3 The vein, which is the outflow site, matures up to threefold in diameter and takes on arterial characteristics due to its abnormal connection to the artery, the inflow site. 2 The vein becomes more durable and likely to sustain repetitive venipuncture. The AVF helps provide substantial blood flow to the dialysis machine that is filtered and quickly returned to the patient’s bloodstream. 2 Dialysis grafts and tunneled catheters are at a higher risk for various complications. Grafts use a synthetic tube that is more prone to infection, inflammation, and other complications. 2 Tunneled catheters, or CVCs, are usually placed in the internal jugular vein for temporary use while an AVF is maturing. Infection, central venous stenosis, thrombosis, and restricted blood flow are all complications of tunneled catheters. 8 Although an AVF is the preferred choice for dialysis access, it is still susceptible to numerous complications. Complications may include AVF immaturity and failure, intimal hyperplasia, thrombosis, ischemic steal syndrome, aneurysm, and PSA.
A PSA occurs due to vessel wall injury that is caused by inflammation or trauma. With dialysis patients, the most common cause is repetitive venipuncture.1,2 Due to the high pressure and flow hemodynamics of the AVF, the blood begins to flow into the surrounding tissue and is confined between the two outermost layers of the vessel wall (Figure 10). The perfused sac connects to the supplying vessel by a communicating channel or neck.6,9 PSA formation in dialysis access sites occurs more frequently in dialysis grafts than dialysis fistulas. They commonly occur at the anastomosis or the venipuncture site and arise more often in a proximal AVF than a distal AVF. 2 There is thought to be a 2% to 10% incidence in the dialysis population according to several published articles.1,10 –12 On average, patients receive dialysis up to three times per week, which puts them at a higher risk of vessel wall injury leading to PSA formation. 1 A normal vein is low in pressure and quickly thromboses when injury, such as a needle stick, occurs. As the AVF matures, the outflow vein contains elevated pressure and velocity resembling the arterial system, which limits vessel wall injury as seen in normal venous anatomy. Consequently, the new flow hemodynamics may lead to PSA formation in the outflow vein resulting from frequent venipunctures. Clinical signs and symptoms include a pulsatile mass, palpable thrill, audible bruit, swelling, pain, erythema, and occasionally systemic issues. The surrounding tissue can become ischemic resulting from the hemodynamics. Compression of nerves can cause neurological symptoms, and venous compression may cause thrombosis or edema. 6
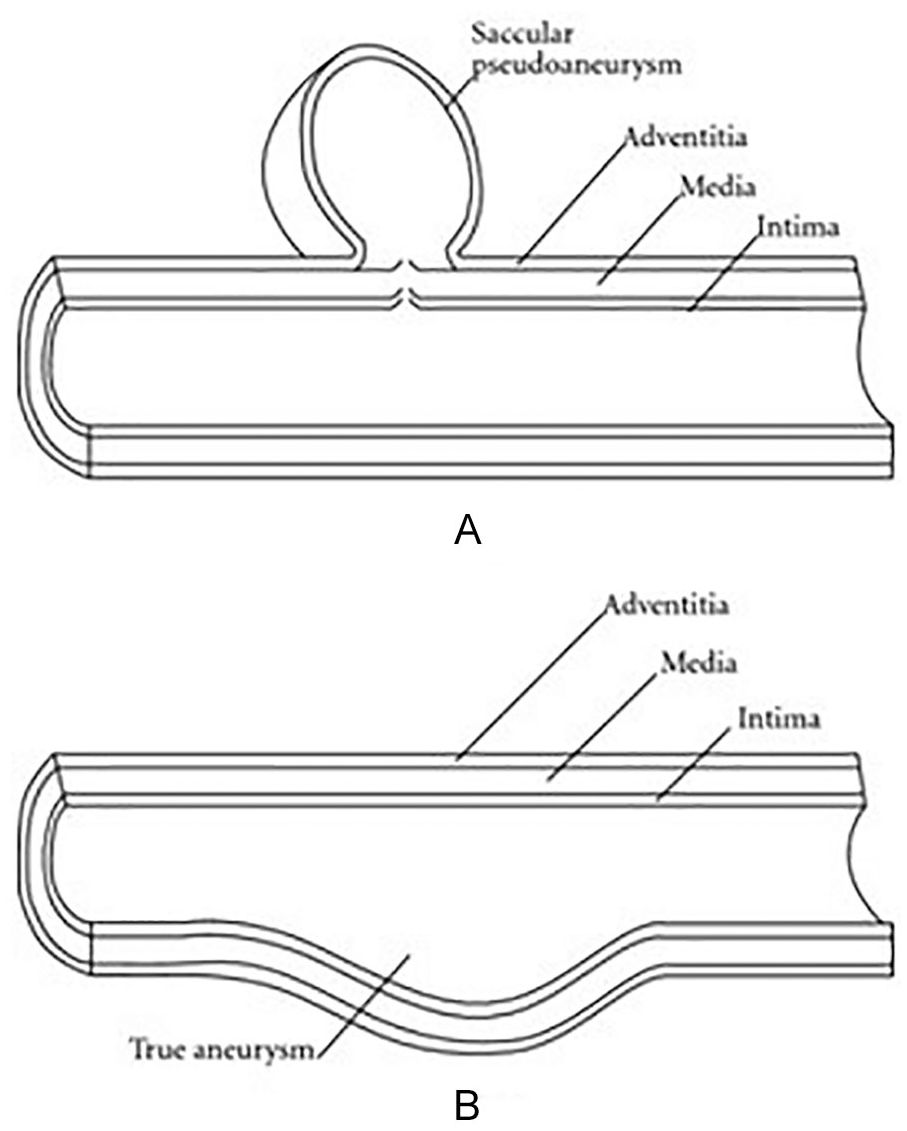
(A) Pseudoaneurysm demonstrates an abnormal dilation between the adventitia and media layer of the vessel. (B) A true aneurysm includes all three vessel wall layers.
For correlative imaging, angiography remains the gold standard in diagnosing a PSA; however, it is invasive in nature. In addition, angiographic hazards include the use of ionizing radiation and an increased risk of related procedural complications due to renal toxicity. Complications that arise from angiography occur at a rate of approximately 0.02% to 9%. 6 CTA is another imaging modality used for evaluation of pseudoaneurysms. A study demonstrated a high sensitivity and specificity rate of approximately 95% and 99% with CTA. 6 Decreased spatial resolution and presence of imaging artifacts are the disadvantages of CTA. 6 Magnetic resonance angiography (MRA) is ordered less frequently due to contraindications, expense, and its time-consuming nature. 6 Sonography is an excellent modality choice for the screening and diagnosis of pseudoaneurysms. It is comparatively inexpensive, is portable, can be performed quickly while providing valuable diagnostic information, is noninvasive, has no contraindications, and does not use ionizing radiation. Real-time visualization using spectral Doppler of the flow patterns within a PSA is useful to help confirm a diagnosis. 6 Studies have shown that it has a 94% sensitivity and a 97% specificity rate.6,13
Sonography helps determine specific features of a PSA that other modalities cannot. The sonographic appearance of a PSA is mostly uniform throughout. Detailed characteristics will consistently be demonstrated regardless of the pseudoaneurysm’s origin. 14 To help clarify the main difference in aneurysms, a true aneurysm has been defined as the dilation of all three layers of the vessel while a PSA, or false aneurysm, normally involves the two outermost layers of the vessel wall (Figure 10). With sonography, in gray-scale, a PSA usually appears as an anechoic to a hypoechoic cystic structure close to a feeding vessel (Figure 3). Gray-scale sonography is helpful in assessing the size and the origin. Gray-scale alone is not enough to diagnose a PSA due to its similar appearance with other diagnoses such as a hematoma or abscess.13,14 Color and spectral Doppler is the most definitive tool for confirming diagnosis of a PSA. Color Doppler will demonstrate the “yin-yang sign.” This may also be demonstrated with true aneurysms. To differentiate from a true aneurysm, the sonographer must identify the channel that connects the PSA to the supplying vessel. This channel is most commonly referred to as the neck of a PSA.13,15 Spectral Doppler will demonstrate a “to-and-fro” flow pattern. This will be demonstrated as sharp peaks above and below the baseline (Figure 8).13,15
A proper sonographic protocol is essential for screening and confirming diagnosis of pseudoaneurysms. Depending on body habitus, a curved or linear-array high-frequency transducer should be used. Sonographers must take special care with examining dialysis patients with arteriovenous fistulas displaying symptoms of a PSA. The sonographer should evaluate the AVF and the PSA (see Table 1 for protocol and diagnostic criteria).15 –17 Additional diagnostic criteria may exist and should be reviewed.
AVF and PSA Protocol With Diagnostic Criteria. a
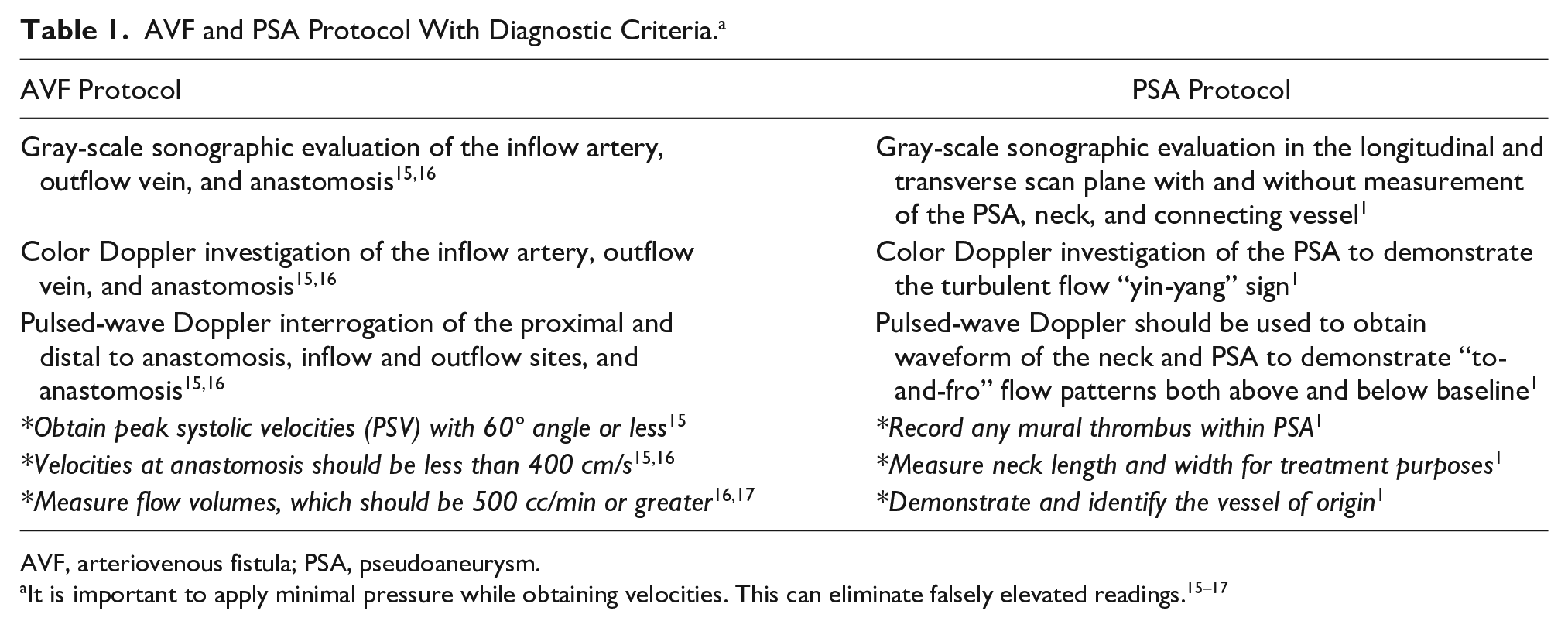
AVF, arteriovenous fistula; PSA, pseudoaneurysm.
In general, the development of a PSA carries a high risk of AVF failure, thrombosis, infection, rupture, and hemorrhage.1,2,6 The treatment and prognosis of a PSA in an AVF depend on variable factors. Surgery is the gold standard and most optimal treatment option for a PSA that is greater than 2 cm in diameter and displaying severe symptoms.6,13,17,18 Depending on the case, surgical resection or ligation may be the treatment. Although it is the traditional treatment of choice, open surgery has its disadvantages. Infection, rupture, anesthesia-related risks, and even death can occur from surgery. 6 Dialysis patients may have contraindications, and surgery is not always the best treatment option. Ultrasound-guided thrombin injections have been recognized as an excellent choice for treatment. Thrombin injections are considered safe, regardless of the neck measurement, for non-groin-related pseudoaneurysms. 19 Thrombin is injected into the PSA sac, which results in thrombosis of the swirling blood. Sonography is used to visualize the PSA as thrombin is injected. Another treatment option, ultrasound-guided compression therapy, is used to compress the neck of a PSA. It is not the ideal choice due to operator and patient discomfort and the risk of occluding the vessel or fistula.1,6 With angiography, covered stents may be placed across the neck of a PSA to prevent it from receiving blood flow from the adjoining vessel. The use of contrast, infection, and stent migration are the main drawbacks for this form of treatment. 20 For patients with end-stage renal disease who are experiencing minimal or no symptoms, monitoring with sonography may be the ideal conservative treatment of choice.
Conclusion
Hundreds of thousands of patients undergo hemodialysis in the United States per year due to the increased prevalence of diabetes and hypertension. 21 A statistic from 2015 stated that over 661,000 patients had CKD, and 468,000 of those received dialysis. 21 For patients with CKD and ESRD, survival is dependent on hemodialysis until they receive a renal transplant. A PSA can be a severe complication for these patients and life-threatening. Sonography is an excellent method for the screening and diagnosing of a PSA in dialysis patients with arteriovenous fistulas and may even play a role in treatment. Sonography has a high sensitivity and specificity rate, is inexpensive and portable, uses no radiation, and has no contraindications. A major advantage of sonography is its real-time visualization of the flow patterns of a PSA with spectral Doppler analysis. The main pitfall of sonography is that it is highly operator dependent. Knowledge of normal and abnormal vasculature, technical skill, and proper sonographic protocol performance are crucial for a diagnostic examination.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
