Abstract
Objective:
To determine the capabilities of a sonographic examination to evaluate local and systemic hemodynamic disorders in patients with varicose disease.
Materials and Methods:
This study included 583 patients with varicose veins, among whom 348 had trophic disorders. All subjects underwent lower limb duplex sonography and superficial, perforator, and deep venous diameter measurements, as well as had a calculated velocity for antegrade and reflux flow. Peripheral resistive index (RI) in the arteries, accompanying perforator veins (PVs), was analyzed. In addition, echocardiography was performed on 46 of the participants.
Results:
A statistically significant difference in the diameter of the great saphenous vein was obtained between class C2 and C4 varicose veins (P < .05). Similar findings were obtained for the diameter of the PVs and the velocity reflux flow through PVs. Arteries in the perforator bundles demonstrated low RI, which may be attributed to the presence of arteriovenous shunts. Varicose veins were associated with an increased incidence of nonrestrictive type of blood flow through the tricuspid valve.
Conclusions:
Sonography demonstrated that additional factors rather than hemodynamics may be attributable to the development of venous ulcers. The most informative parameters of cardiac function, in a patient with varicose veins, are the indicators of diastolic function in both ventricles.
Duplex sonography (DS) of the lower extremities is a common and widely available diagnostic examination in subjects with varicose veins. DS of the superficial, deep, and perforator veins (PVs) has several advantages such as being minimally invasive. Multiple studies can be performed, with real-time evaluation of the anatomy of the venous system, assessment of valvular function, assessment of venous reflux, quantitative assessment of blood flow parameters such as blood velocities and volume, and venous reflux time. 1 However, in daily practice, DS is mainly used for qualitative assessment such as venous anatomy, quantitative parameters, and microcirculatory function. These are not routinely estimated but are necessary when assessing pathogenetic mechanisms in the development of trophic ulcers.1,2 The microcirculatory vascular bed is usually assessed using capillaroscopy, transcutaneous oxygen pressure measurement; thermography; and laser Doppler. 1 New-generation diagnostic ultrasound systems provide a possibility of imaging the microcirculatory vascular bed, such as superb microvascular ultrasound imaging or SMI mode, with the Aplio 500 ultrasound system (Toshiba Medical Systems Corporation, Tochigi, Japan). Although this equipment type has a high rate of accuracy, it still only provides an indirect assessment. 3 Regardless of the imaging methods used, it still requires additional equipment. Microcirculation may be assessed with the help of a standard DS ultrasound system.
Patients with chronic venous disease (CVD) routinely undergo lower limb DS, with special attention paid to the area in the distal parts on the medial surface of the lower leg. This is due to the frequent involvement and development of trophic disorders.1,4–7 According to classical anatomy, PVs are on the medial surface of the lower leg. The Cockett I, II, and III PVs are the main vessels involved in venous drainage in the area. 8 At the same time, PVs are accompanied by an arterial branch arising from the posterior tibial artery at the subfascial level.8,9 Above the level of the muscular fascia, these small arteries penetrate into the hypodermis, forming several minor “tree-like” branches. 10 These continue into the dermis, forming deep (subdermal) and superficial (between the dermis and epidermis) parallel arteriolar networks. The blood flows from the capillary loops into the venules, thus forming a superficial network immediately under the skin papillae. The second subcapillary network of venules, parallel to the first one, is located somewhat deeper. The third venous network is located in the next layer of the dermis. Venous outflow from epithelial tissues is carried out in the following manner: papillary capillaries, cutaneous plexus, superficial veins, perforator veins, and deep veins. 10 Thus, vessels at both macro- and microlevel are functionally interrelated. 7 Therefore, impaired hemodynamics at the microcirculatory level affects larger vessels, as well as changes in the venous portion of the microcirculation. 6 Consequently, calculation of hemodynamic parameters in the artery of the perforator bundle may provide useful information about the blood flow in the lower leg. However, in routine DS, venous lower limb perforator arteries, within the perforator bundles, are not assessed.11–14 Moreover, routine examination of subjects with CVD, on an outpatient basis, usually does not involve echocardiography. It may be difficult to not assume that the pathology of the venous system in the lower extremities would not affect cardiac function (e.g., inferior vena cava, which is directly involved in central hemodynamics).
Therefore, the aim of this study was to determine the possibility of using sonographic techniques in the assessment of local and systemic hemodynamic disorders in patients with varicose disease.
Materials and Methods
This study included 583 participants with varicose disease, of whom 348 had trophic disorders. A written consent was obtained from the local ethics committee of the Ryazan State Medical University. All participants gave their consent to join the study. Any participants diagnosed with arterial hypertension, diabetes mellitus, chronic lung diseases, structural heart disorders, or coronary artery disease were excluded from the study. Sonography was performed using Medison X8 and Sonoscape S20 Pro ultrasound equipment. Two types of transducers were used: a linear 7- to 10-MHz probe and a sector 1- to 4-MHz probe. The researchers evaluated superficial, deep, and perforator veins using functional tests such as standardized Valsalva and Siegel tests. The diameter and peak systolic velocity of the antegrade (PSV ant) were calculated in both superficial and deep veins, and retrograde blood flow characteristics were also assessed (peak reflux velocity [PRV]). When assessing PVs, the researchers calculated diameter, direction of blood flow, peak (PRV), and mean reflux velocities (MRVs), as well as the time of retrograde blood flow. For standardization, these parameters were measured three times, and the average value was used for further assessment. A retrograde flow of more than 0.5 seconds was regarded as pathologic. The study included participants with treatment for naive varicose disease. The DS study was performed during the morning. Great saphenous vein (GSV) diameter was measured in the proximal part of the thigh as recommended by Mendoza,15,16 in a standing position, with minimal probe pressure against the skin. Perforator diameter was measured at the level of entering the muscular fascia with a Doppler angle below 60°. Blood flow through the arteries and accompanying PV were also analyzed, including the calculation of the peripheral resistive index (RI). The RI in the posterior tibial arteries was also analyzed.
Echocardiography was performed with a Saote My Alpha Lab and Acuson Sequoia 512 ultrasound equipment. In addition to the standard echocardiographic protocol, the researchers precisely assessed right heart function, including the size of the right ventricle (RV), right atrial (RA) size, the thickness of the RV myocardium, RV ejection fraction, mobility of the lateral edge of the tricuspid ring, and the pressure gradient on the tricuspid valve. Diastolic function of the ventricles was assessed by analyzing blood flow patterns through the tricuspid and mitral ventricles, including early (E) and late (A) diastolic filling velocities and their ratio (E/A). There were three patterns of blood flow: normal, nonrestrictive, and restrictive.17,18 Echocardiography was performed in selected subjects (n = 46) because during the physical examination and while questioning the participants, some had chest discomfort, although there was no previous history of cardiac disorders or any changes revealed during the electrocardiogram (ECG). Diastolic dysfunction was evaluated using the E/A ratio. An E/A ratio of 0.75 to 1.5 was attributed to the normal flow, an E/A ratio lower than 0.75 to nonrestrictive flow, and an E/A ratio higher than or equal to 1.5 to the restrictive flow.19,20 Participants with pulmonary hypertension were not included in the study; another noninclusion criterion was smoking. 21
Statistical Analysis
Data analysis was performed using a Mann-Whitney U test as a nonparametric alternative test to the independent sample t test. The Wilcoxon test was used for paired samples, and the Spearman rank correlation was used to determine association between two ranked variables. With normally distributed data, the Student t test was used for estimating the mean of the sample population. A threshold probability value of P ≤ .05 was established a priori to indicate statistical significance.
Results
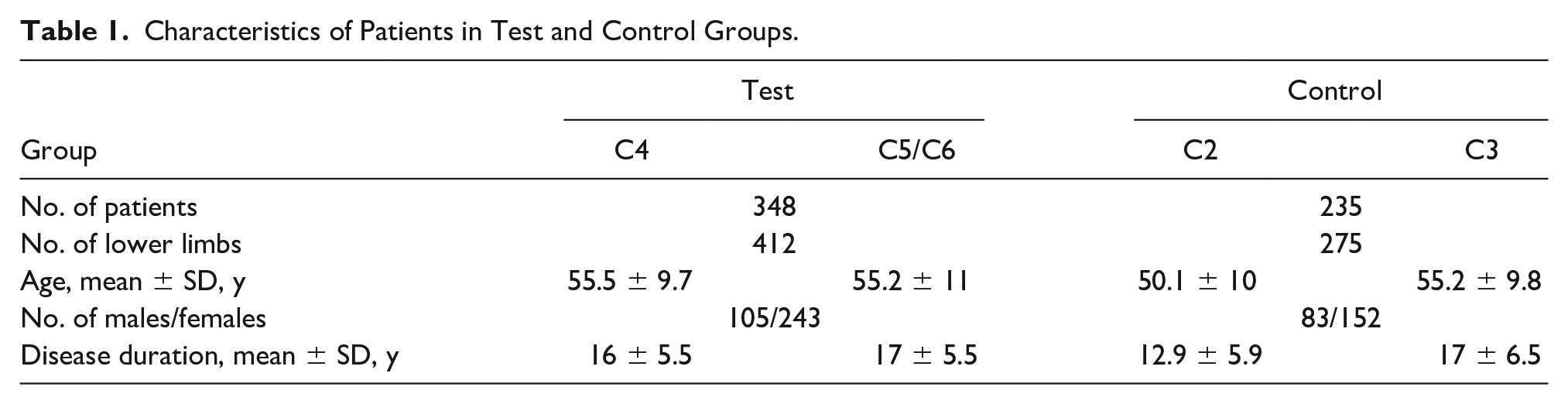
The participants were divided into a test group that included 348 patients with trophic disorders. Among that group, 214 participants were determined to have C4 clinical class varicose veins, according to the Clinical-Etiological-Anatomical-Pathophysiological (CEAP) classification. In addition, 134 participants were found to have C5 to C6 clinical class varicose veins. In the control group, there were 235 participants without trophic disorders. Among that group, 137 subjects had CEAP class C2 varicose veins, and 98 subjects were found to have CEAP class 3 varicose veins. The descriptive statistics of the participants in both the test and control groups are presented in Table 1.
Characteristics of Patients in Test and Control Groups.
The CEAP classification of participants who underwent echocardiography was as follows:
CEAP class С2: 22 patients (13 males and 9 females); median age 40.3 ± 12.2 years; duration of disease 12.3 ± 4.8 years
CEAP class С3: 11 patients (8 males and 3 females); median age 43.2 ± 12.6 years; duration of disease 15 ± 6.3 years
CEAP class С4: 10 patients (8 males and 2 females); median age 51.1 ± 11.7 years; duration of disease 13.9 ± 4.5 years
CEAP classes С5 and C6: 3 patients (all males); median age 58.3 ± 2.9 years; duration of disease 18.3 ± 7.6 years
The median age of participants with CEAP classes С2 and С3 was lower than compared to the main group (P < .05). There was no statistically significant difference between the median age in CEAP class C4 and class C5/C6 patients. There was also no statistically significant difference between the duration of disease among the groups.
The average age of participants in the test group (55.35 ± 10 years) was higher than in the control group (52.65 ± 9 years); disease duration in the test group was also slightly higher as compared to the control group: 16.5 ± 5.5 and 15 ± 6.2 years, respectively (P < .05).
The structure of the lesions in the superficial, perforator, and deep veins is as follows:
Test group: superficial veins (3.2%), superficial veins and PVs (92.8%), and superficial veins, PVs, and deep veins (6.9%)
Control group: superficial veins (23.8%), superficial veins and PVs (72.8%), and superficial veins, PVs, and deep veins (3.4%)
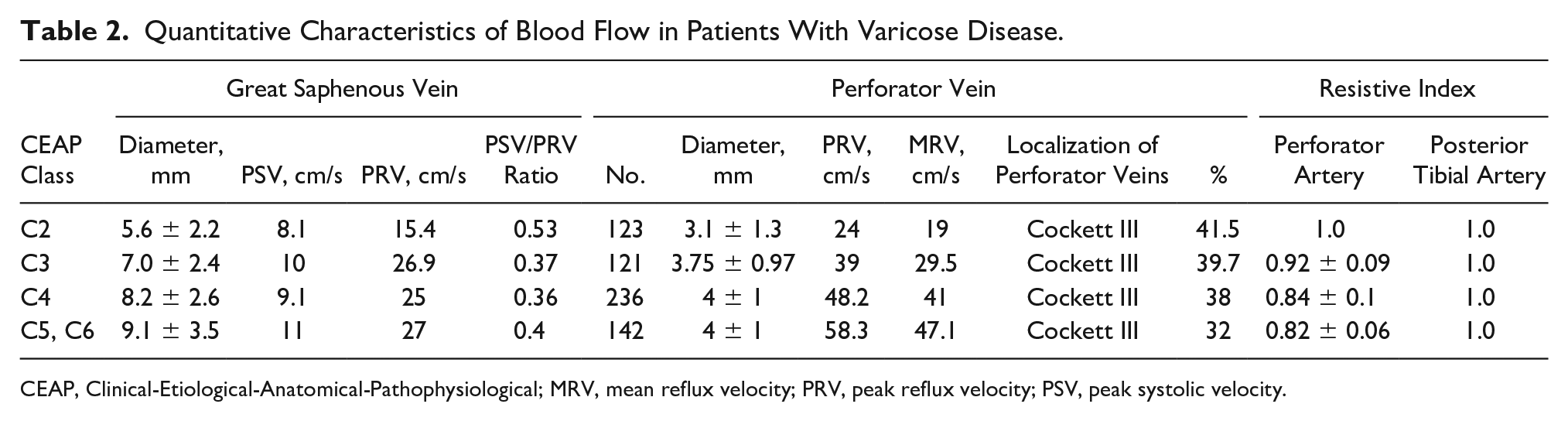
The GSV was the most affected. The diameter of the GSV was 8.4 ± 2.9 mm in the test group and 6.2 ± 2.4 mm in the control group. In addition, in patients in the test group, the mean diameter was 4 ± 1 mm for PVs, 51 ± 33.8 cm/s for PRVs, and 43 ± 24.2 cm/s for MRVs. In the control group, the mean diameter was 3.5 ± 1 mm for PVs, 32.7 ± 20 cm/s for PRV, and 25 ± 13.8 cm/s for MRVs. The Cockett II and III PVs were the most affected. The key parameters for this study are provided in Table 2.
Quantitative Characteristics of Blood Flow in Patients With Varicose Disease.
CEAP, Clinical-Etiological-Anatomical-Pathophysiological; MRV, mean reflux velocity; PRV, peak reflux velocity; PSV, peak systolic velocity.
A statistically significant difference in the diameter of the GSV was obtained between classes C2 and C3, as well as C3 and C4 (P < .05). Despite the fact that the diameter of the GSV in patients with CEAP classes C5 and C6 was larger than in patients with class C4, a statistically significant difference was not noted. Similar findings were obtained for the diameter of the PVs and reflux velocities. Higher clinical class according to the CEAP classification was associated with a gradual increase in venous diameter and reflux velocities. However, a statistically significant difference was obtained only between classes C2 and C3 in terms of the diameter of the PVs and PRV; as for MRV, a statistically significant difference was noted between classes C2 and C3, C3 and C4 (p < 0.05).
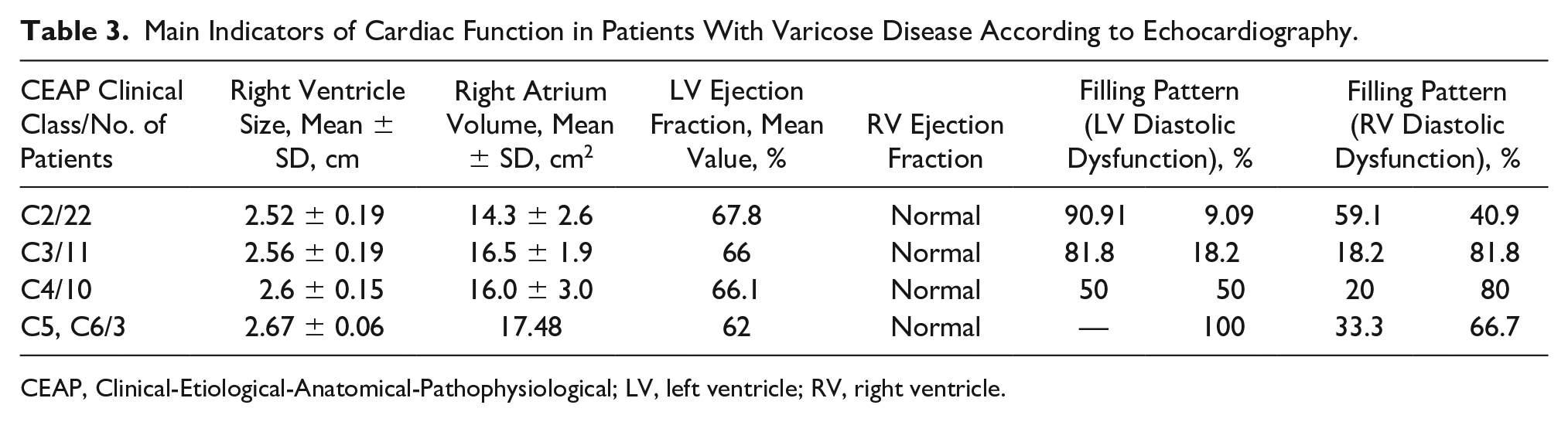
The perforator arteries, as a component of the PV bundles, were visualized in 100% of the participants. Perforator arteries demonstrated lower resistive indices; also, pseudo-pulsating blood flow was detected in 50% of PVs, which may indicate the presence of arteriovenous shunting. 12 Statistical analysis demonstrated statistically significant differences between CEAP classes C2 and C3, C3, and C4 (P < .05), while there was no statistical significance between CEAP classes C4 and C5/C6. The results of echocardiography for participants with varicose disease are presented in Table 3.
Main Indicators of Cardiac Function in Patients With Varicose Disease According to Echocardiography.
CEAP, Clinical-Etiological-Anatomical-Pathophysiological; LV, left ventricle; RV, right ventricle.
Despite the fact that most of the indicators of cardiac function were within the normal diagnostic range, there was a tendency to detect an increased size of the right heart chambers associated with a higher clinical CEAP class. In addition, there was an increase in the thickness of the interventricular septum, from 0.8 cm to 1.1 cm, and the thickness of the anterior wall of the RV, from 0.3 to 0.5 cm. Ejection fraction of both RV and left ventricle (LV) was also within the normal range but with a tendency to decrease with a higher clinical class according to the CEAP classification.
Changes in the diastolic function of the ventricles, mostly RV, were the most demonstrative. Higher clinical classes of varicose disease were associated with an increased incidence of nonrestrictive blood flow through the tricuspid valve, which might be associated with a decreased inflow into the right heart in subjects with advanced chronic venous insufficiency (CVI). No restrictive filling patterns were identified in patients with varicose disease.
Discussion
As a general rule, the absolute values of velocity and time for retrograde blood flow are used to characterize venous reflux. In this particular study, reflux time was not taken into account since the retrograde blood flow lasted throughout the entire duration of the Valsava test. Indicators of venous reflux vary, which may be explained by the fact that cardiac and respiratory function significantly affected the blood flow in the femoral popliteal segments in the deep and superficial veins. 12 Therefore, the authors of the article used a PSV/PRV ratio in GSV diameter to characterize reflux. Interestingly, this indicator differed only between classes C2 and C3. This is compared to participants with classes C3, C4, and C5/C6, and the values did not vary.
There are several theories to explain the development of trophic ulcers, including the consequences of chronic phlebostasis, phlebohypertension, and hydrodynamic stress in subjects with incompetent PVs. There also assumptions about the microcirculation, with increased resistance to venous outflow, that lead to impaired drainage function and arteriovenous shunting.
Some scientists have provided data that point to blood pressure in varicose veins being increased as compared to normal vessels. 12 In the initial stages of vein enlargement, with no signs of valvular insufficiency, the venous pressure taken with the patient in the upright position will be in the normally expected range (e.g., 75–120 mm Hg). Advanced stages of venous insufficiency are associated with higher venous pressures of up to greater than 500 mm Hg. 13 An increase in venous pressure in the superficial veins leads to the opening of arteriovenous precapillary anastomoses. This then promotes arterial blood to flow into the veins, thereby further increasing venous pressure. This leads to difficulties in transferring blood from the capillaries of the skin and subcutaneous tissue into the venules and can promote stasis and the formation of trophic ulcers. However, some scientists have reported that venous pressure in the superficial and deep veins, taken in the upright position among CVI patients, corresponds to the distance from lower leg to the heart and can act as a constant value.7,22 At the same time, dynamic venous hypertension cannot be excluded given the effects of high pressure from incompetent lower leg perforators (Cockett) and decreased muscle pump function. Pressure values of 180 to 200 mm Hg are destructive to the microcirculation and the surrounding soft tissues.7,23,24 High pressure at the microcirculatory level leads to an increased postcapillary resistance and increased capillary pressure. With an increase in postcapillary resistance, arteriovenous shunting can be activated and mobilize the blood deposited in lower leg veins.7,14 The blood flow bypasses the capillaries, which leads to an impaired transcapillary metabolism, microcirculatory dysfunction, and the subsequent development of trophic ulcers. 25
The results of this study showed that there was no statistically significant difference between C4 and C5/C6 CEAP classes in terms of superficial and perforator venous reflux parameters. This study would suggest that none of the developmental theories of trophic ulcers find their full hemodynamic confirmation. Trophic ulcers may only be a possible complication of the varicose disease and are not an inevitable symptom. This conclusion has been confirmed by morphological studies of venous trophic ulcers. 7
Some authors state that the duration and higher velocity of retrograde flow (over 20–30 cm/s) may be regarded as predictors for the development of trophic ulcers.26–28 The present study data do not support such a statement. During the present study, the researchers did not consider the timing of the reflux, due to the retrograde flow being detected, throughout the whole period of the Valsalva maneuver. Therefore, these results agree with Pittaluga et al., 29 which indicates that the “time of the retrograde flow” should be considered only for the detection of reflux. Data indicate that the time and velocity of the retrograde flow, through the GSV, decrease after diameter reduction following the removal of the varicose tributaries. The same effect is seen in subjects who wear elastic stockings. 30 Such an effect is explained by the increased peripheral resistance due to the decreased diameter of the vessel. At the same time, increased arterial inflow leads to the decreased reflux time; some authors have reported that the above-mentioned phenomenon may occur during physical activity. 31 Other authors have reported that females with idiopathic arterial hypotension and varicose diseases present with increased reflux time.32,33 The effects of the diaphragm on blood flow in the abdominal part of the inferior vena cava (IVC) are explained by its compression at the vena caval foramen during inhalation. During exhalation, the blood enters the intrathoracic part of the IVC and, during inhalation, enters the right atrium. The pressure gradient between the intrathoracic and intraabdominal veins has an effect on the volume and velocity of the venous inflow to the heart. Such modulations explain high variability of venous patterns of blood flow. 12 Therefore, the researchers for this study chose the ratio between the antegrade and retrograde velocities to characterize venous reflux.
In this study, the participants’ ejection fraction of the RV and LV was within the normal range. However, the study has identified diastolic dysfunction of both RV and LV in participants with varicose disease. This may be a precursor for the presence of systolic dysfunction. 18 At the same time, all study participants had a lack of concomitant coronary artery disease. An experimental study has shown that an increased preload to the right chambers of the heart worsened the symptoms of myocardial ischemia. 34 Subjects with varicose disease and chest discomfort presented with a nonrestrictive flow pattern through the tricuspid valve. Such a phenomenon is associated with decreased relaxation of the right ventricle and a slower pressure drop. The myocardium in patients with varicose disease faces intermittent isotopic overload while supine and underload in the upright position. Frequent change in myocardial function may serve as an additional factor that leads to decreased contractility and the Frank-Starling curve shifting along the axis of myofibril stretching. 3 This study revealed that the ejection fraction of both right and left ventricles in subjects with varicose disease was within normal values, but both ventricles demonstrated signs of diastolic dysfunction. It has been noted that decreased myocardial relaxation is often a precursor of systolic dysfunction.35–38
Diastolic dysfunction is usually attributed to primary myocardial hypertrophy, infiltration cardiomyopathy, ischemia, and arterial hypertension. All of the factors mentioned above are associated with two components of ventricular filling disorder 12 :
Impaired active relaxation of myocardium
Decreased ventricular wall extensibility
At the same time, one of these factors associated with ventricular diastolic dysfunction may have impaired inflow to the right heart. As mentioned above, varicose disease is associated with excessive accumulation of blood in the lower extremities. Physiologically, preload is the maximum extensibility of the myocardial sarcomeres and is under the influence of diastolic filling of the ventricles. According to the Frank-Starling law, the higher the preload and myocyte stretching, the more forceful is systolic contraction. Chronic venous insufficiency is associated with the absence of complete myocardial relaxation in orthostasis as well as impaired inflow to the right heart. Moreover, in the supine position, the amount of blood accumulated in the lower extremities will be redirected to the right heart. Such overload with a pathological amount of blood may lead to the increased thickening of the interventricular wall and ventricular walls (mainly of the right ventricles). This would support the current research data being reported. Similar findings have also been reported by Leukhnenko. 39
In patients with CVI, similar conditions are observed in a supine position when increased blood volumes are deposited in the venous bed and are easily returned to the right heart. This study reports an indirect confirmation of this phenomenon, as participants with advanced varicose disease had a tendency toward an enlargement of the right heart. Therefore, even uncomplicated cases of varicose disease should not be considered solely a cosmetic defect.
Conclusions
Routine DS provides useful information on local hemodynamics and microcirculatory function in patients who have trophic changes due to varicose veins and need an assessment of the flow in their perforating vessels. An echocardiographic evaluation of systemic hemodynamics, with special attention to diastolic dysfunction, should be considered. This may be especially important when managing patients with varicose disease. Changes in cardiac structure and function are an integral part of chronic venous insufficiency, which is why “varicose veins” should be considered a “varicose disease.”
Footnotes
Authors’ Note
The results of the work were presented at the 20th European Venous Forum (Zurich, Switzerland, June 27–29, 2019) and 35th International Conference, “Introduction of High Technologies in Vascular Surgery and Phlebology” (Saint Petersburg, Russia, June 21–23, 2019).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
