Abstract
Gastric outlet obstruction secondary to pyloric thickening in adolescents is an extremely rare finding. The condition is caused by a number of benign or malignant etiologies that cause an impediment to gastric emptying. Common and uncommon etiologies as well as the pathogenesis of gastric outlet obstruction are discussed. A case study of gastric outlet obstruction in a 14-year-old male is presented, and the crucial role of sonography in the diagnostic process of his condition is considered.
Gastric outlet obstruction, also referred to as pyloric obstruction, is not a single medical condition but rather the clinical and pathophysiological consequence of any disease process that causes an impediment to gastric emptying. 1 In an adolescent, gastric outlet obstruction is a rare finding and can prove to be challenging to diagnose. Although sonography is not the modality of choice used to diagnose the condition, it is useful in the preliminary stages of the diagnostic process. It is especially favorable for the pediatric and adolescent patient because it is noninvasive, it does not rely on radiation, and sedation is not often necessary.2,3 Sonographic evaluation of the gastric outlet allows the radiologist to observe the pylorus functioning in real time. This dynamic evaluation is crucial to the diagnosis of gastric outlet obstruction resulting from a hypertrophied pylorus. 3 Indications for gastric outlet evaluation include postprandial vomiting, early satiety, and abdominal distention. 4
Case Report
A 14-year-old male presented to the radiology department with postprandial, nonbilious vomiting with a duration of 6 weeks that had become continuous. Early satiety was present after consuming minimal amounts of food or liquid. Laboratory workup revealed mild leukocytosis and elevated C-reactive protein. He had a low-grade fever that fluctuated between 99.0°F and 100.5°F. He was lethargic and had physical signs of dehydration.
During the preliminary workup, the patient had a series of abdominal and thoracic radiographs (Figure 1) that were reported as unremarkable. There were no obvious foreign bodies, abnormal gas patterns, or free air under the hemidiaphragm noted.
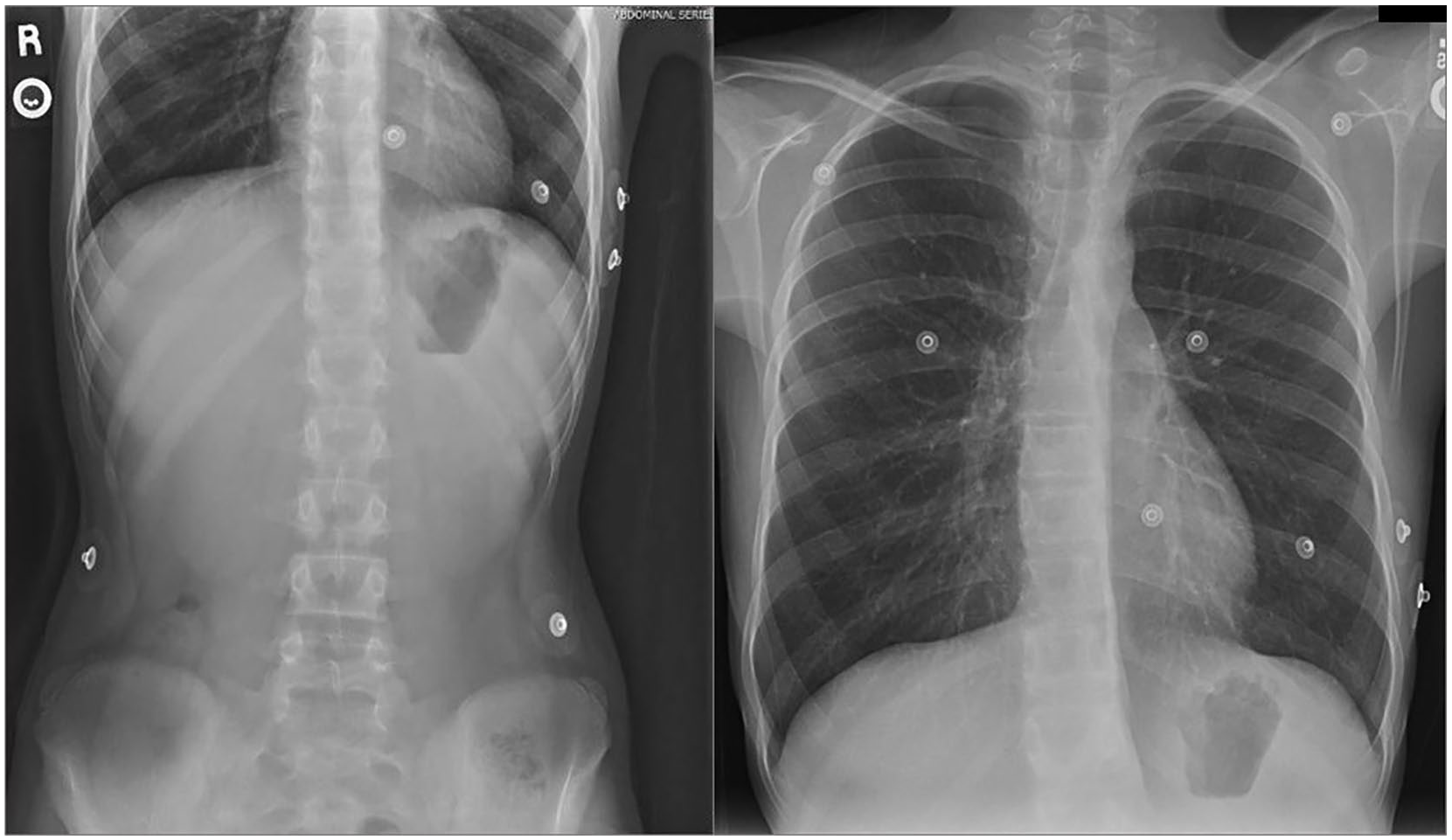
Abdominal and thoracic radiographs with no obvious foreign bodies, abnormal bowel gas patterns, or free air under hemidiaphragm.
Following the radiographs, an upper gastrointestinal study with oral contrast was performed under fluoroscopy that also yielded normal findings (Figure 2). The duodenal bulb was unremarkable, and although the filling time was slightly delayed, no obvious filling defects or masses were seen within the esophagus or the gastric mucosa. The patient was placed on NPO (nothing by mouth) status and a pylorus sonogram was ordered for the next day to evaluate the gastric outlet.
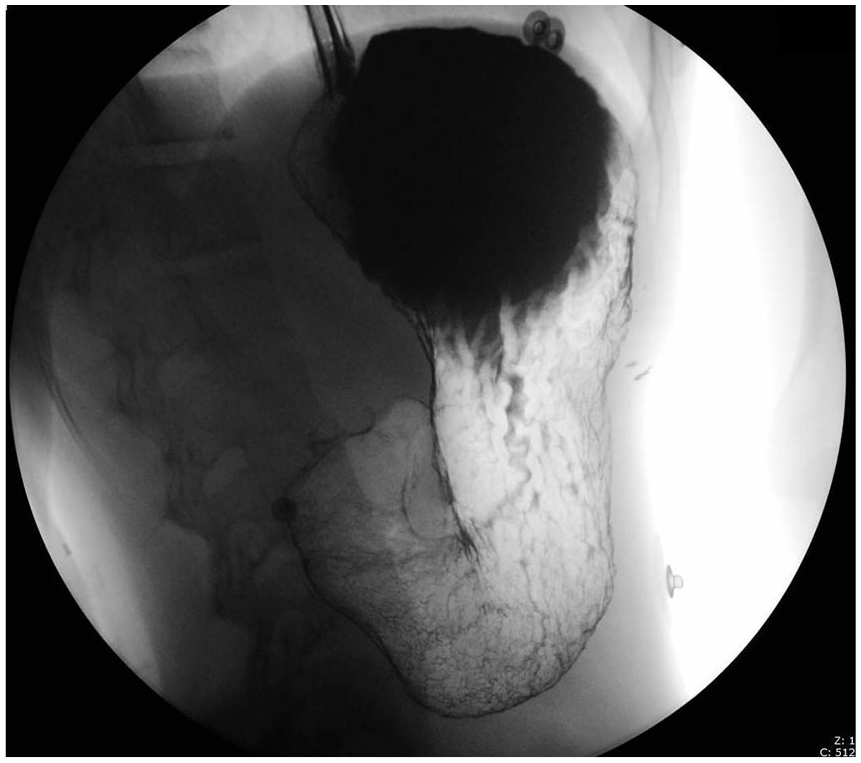
Upper gastrointestinal with contrast.
A linear transducer at 15 MHz on a GE Logiq E9 (GE Healthcare, Milwaukee, WI) was used to perform the examination. Typically, the normal pylorus is not easily appreciated sonographically unless there is pathology present (Figure 3). The initial impression of the patient’s pylorus upon visualization was that it was, at the very least, prominent (Figure 4).
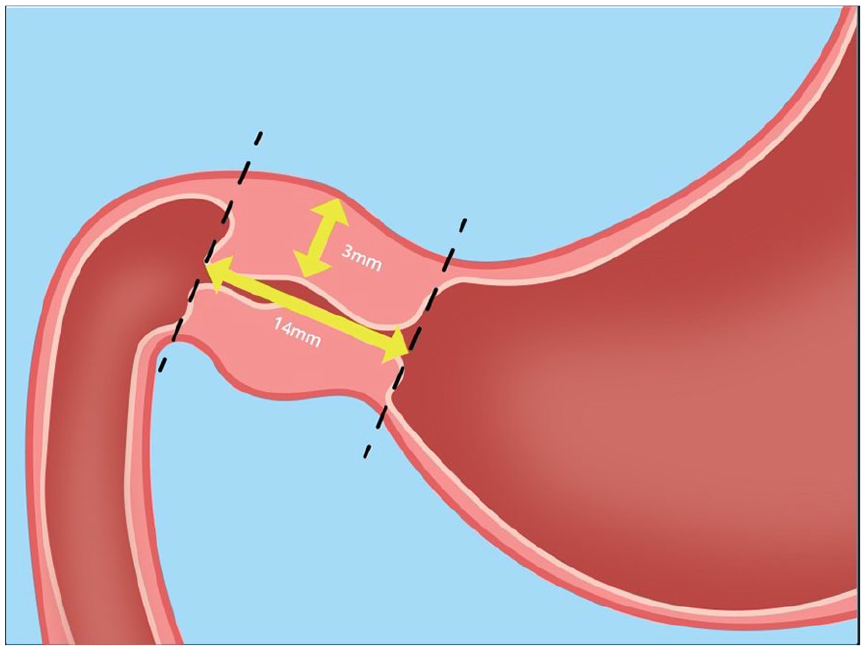
Normal pylorus measurement parameters and location. Anterior wall measurement should be less than or equal to 3 mm with a pyloric canal length less than or equal to 14 mm.
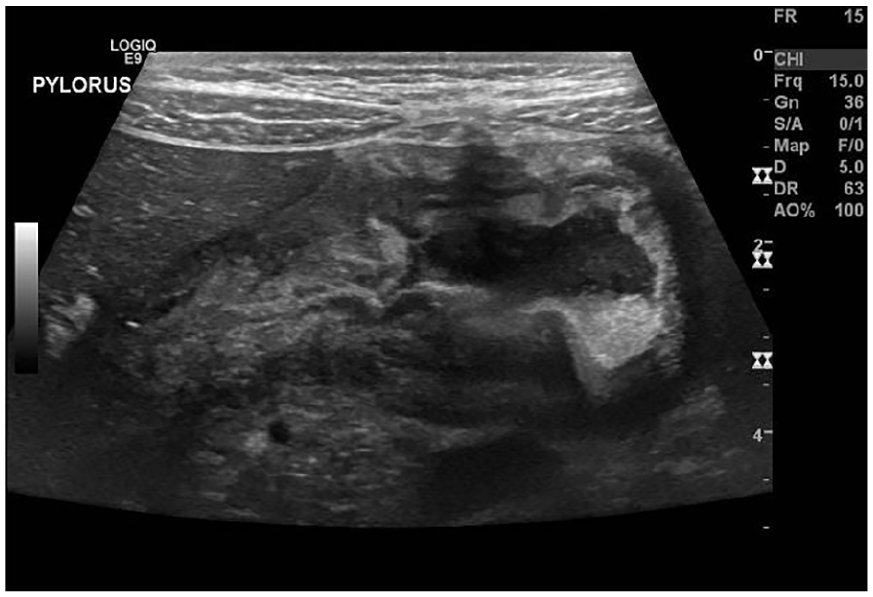
Sonographic image of a longitudinal view of the pylorus on initial visualization.
The anterior wall of the pylorus measured 9.3 mm and the posterior wall 9.6 mm (Figure 5). The pyloric canal in a long-axis view measured 31 mm (Figure 6). In a short-axis view, the pyloric canal appeared closed and displayed a target sign that is indicative of an abnormal pylorus (Figure 7).
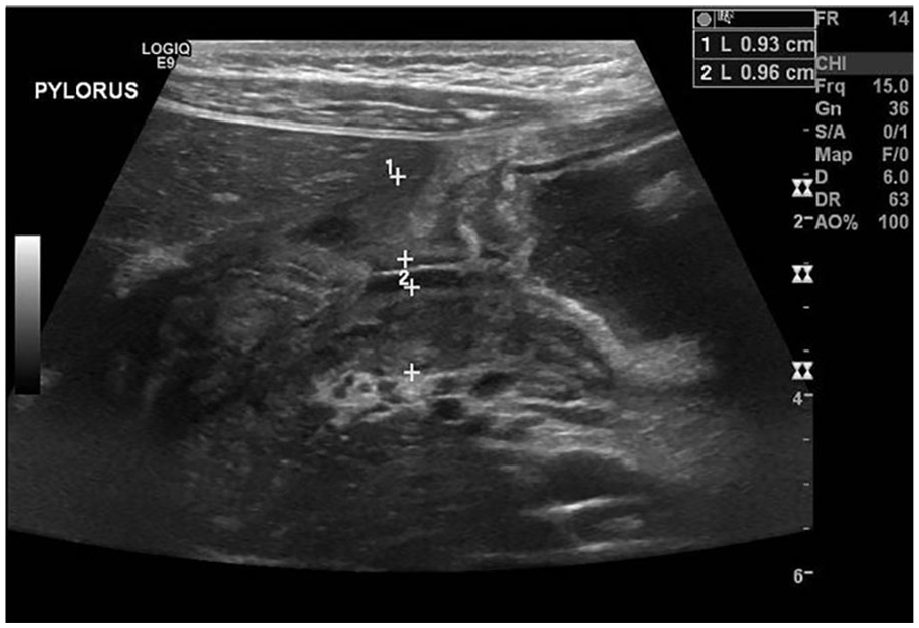
Sonographic image of a longitudinal view of the pylorus. Anterior wall measures 9.3 mm and posterior wall measures 9.6 mm.
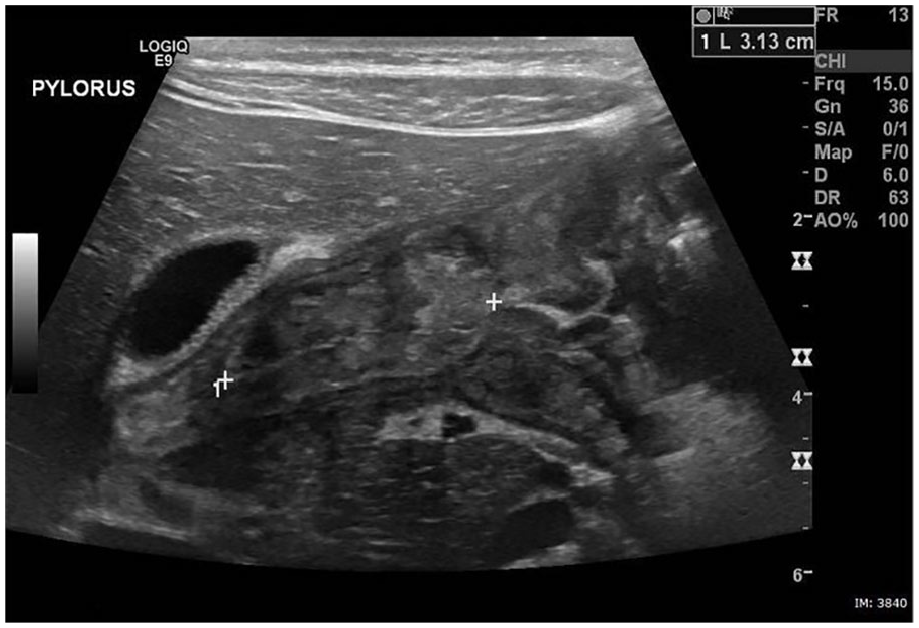
Sonographic image of a longitudinal view of the pyloric canal measuring 31 mm.
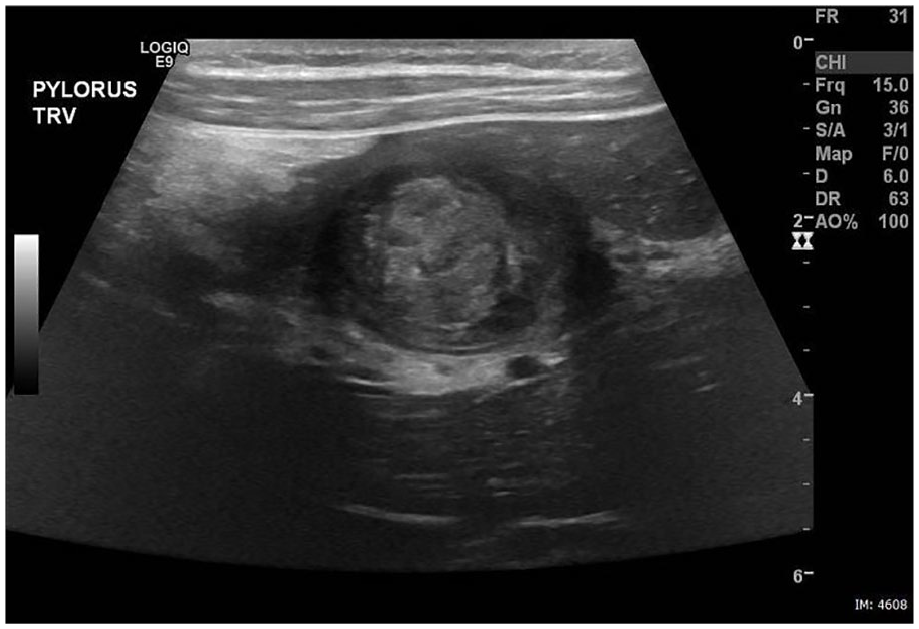
Transverse sonographic image of the pylorus showing a closed pyloric canal with target sign.
A prominent chain of lymph nodes was visualized lateral to midline in the left upper quadrant (LUQ), and another lymph node was seen in the right upper quadrant (RUQ). The sonographer modified the protocol and interrogated the lower abdominal quadrants. Free fluid was seen in the right lower quadrant along with another prominent chain of lymph nodes (Figures 8 and 9). Because appendicitis often presents with lymphadenopathy, the appendix was interrogated. It was not hyperemic and measured within normal limits (Figure 10). The patient was given water to drink and images were taken at 2, 5, 8, and 10 minutes after water ingestion. In both the supine position with the bed tilted at 20 degrees (Figure 11) and in a right lateral decubitus position (Figure 12), the pyloric canal remained closed and no passage of fluid within a 10-minute period was reported.
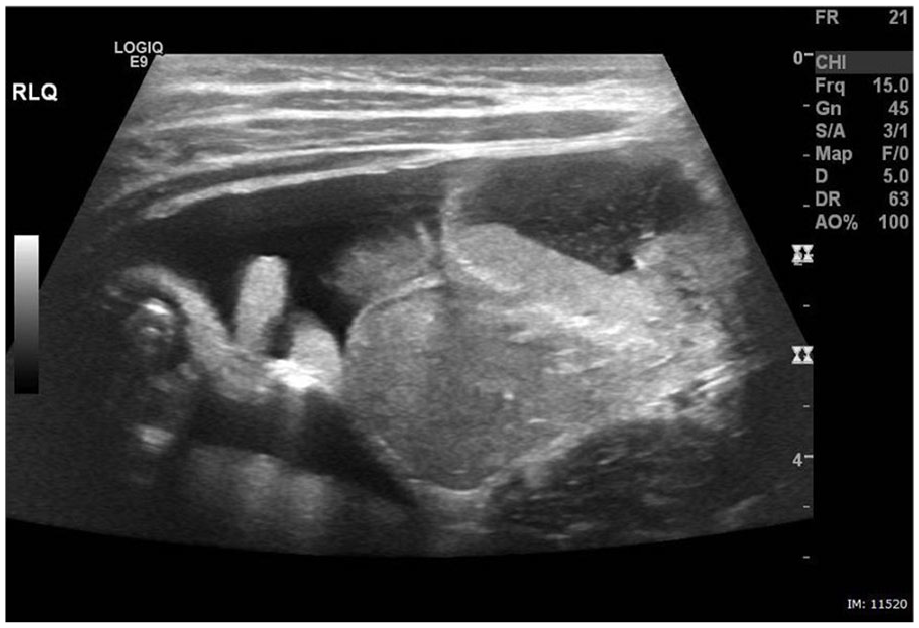
Transverse sonographic image showing free fluid in the right lower quadrant.
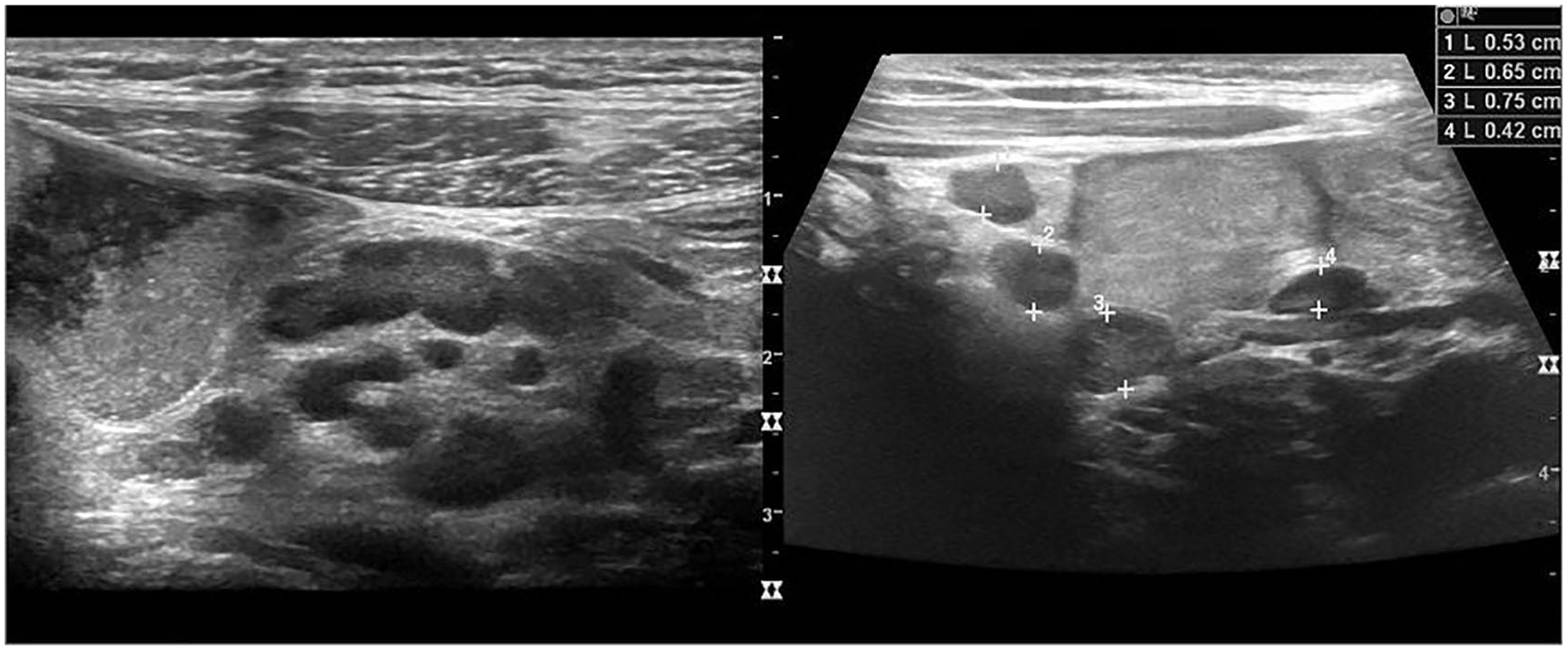
Sonographic image showing a chain of lymph nodes in the right lower quadrant.
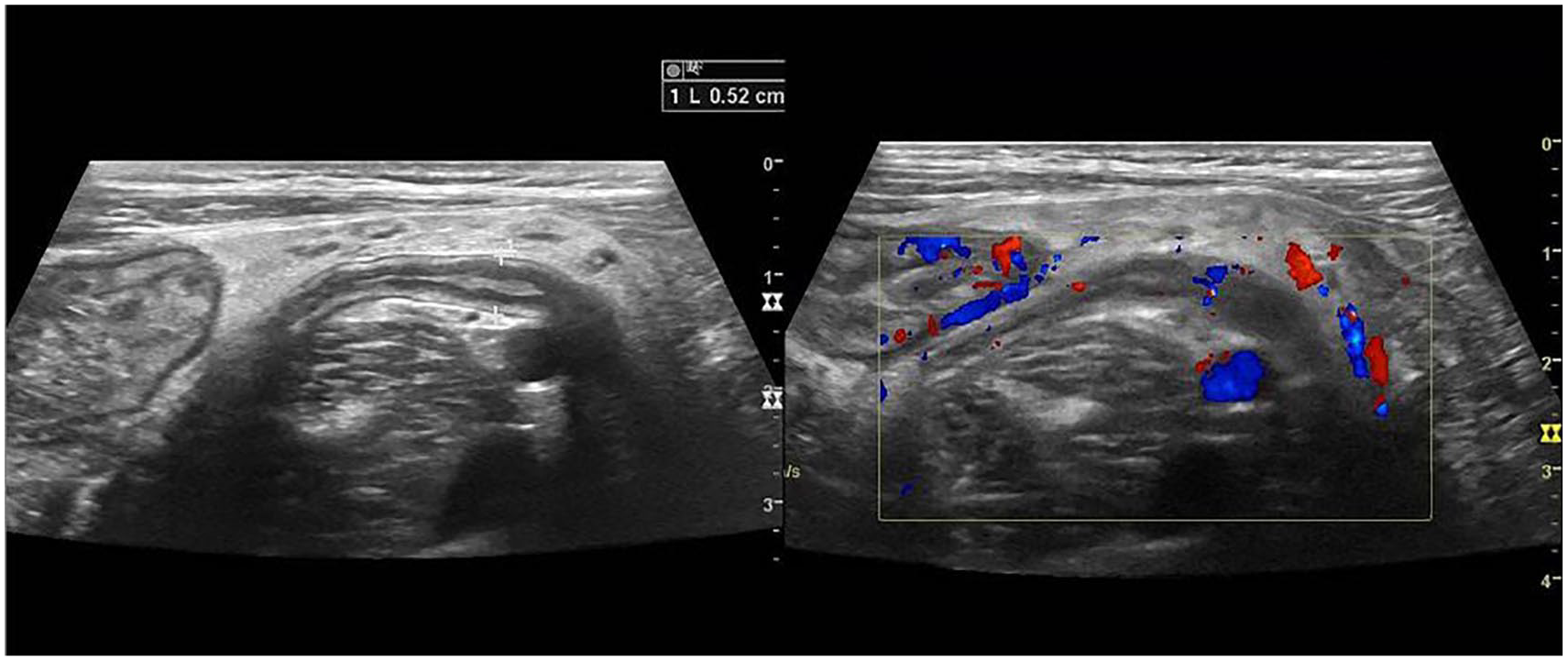
Longitudinal view of appendix with anteroposterior measurement of 5.2 mm. Appendix displays normal vascularity.
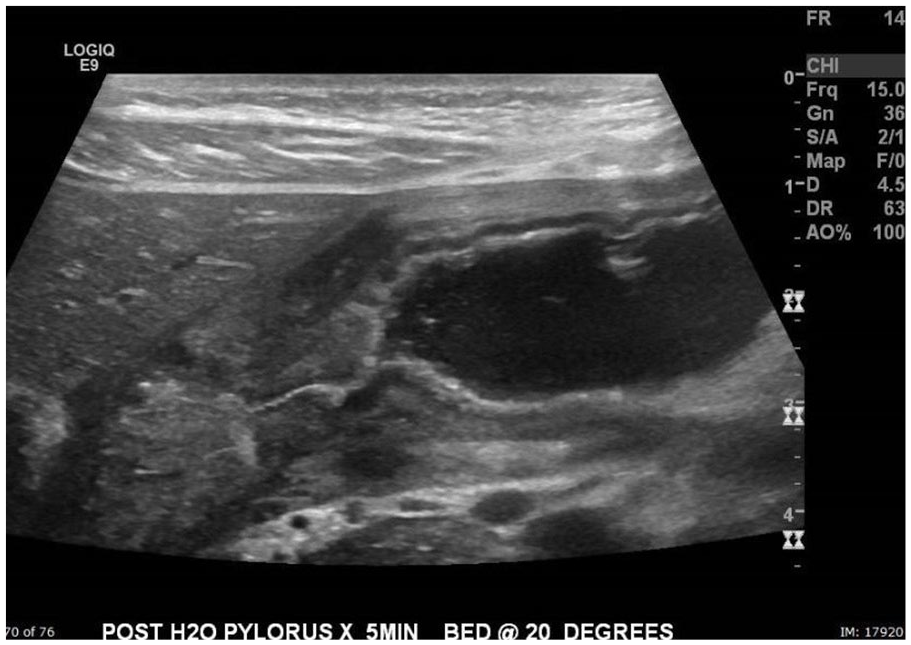
Sonographic image of the pylorus after water ingestion. Patient was placed in a supine position with bed tilted at 20 degrees.
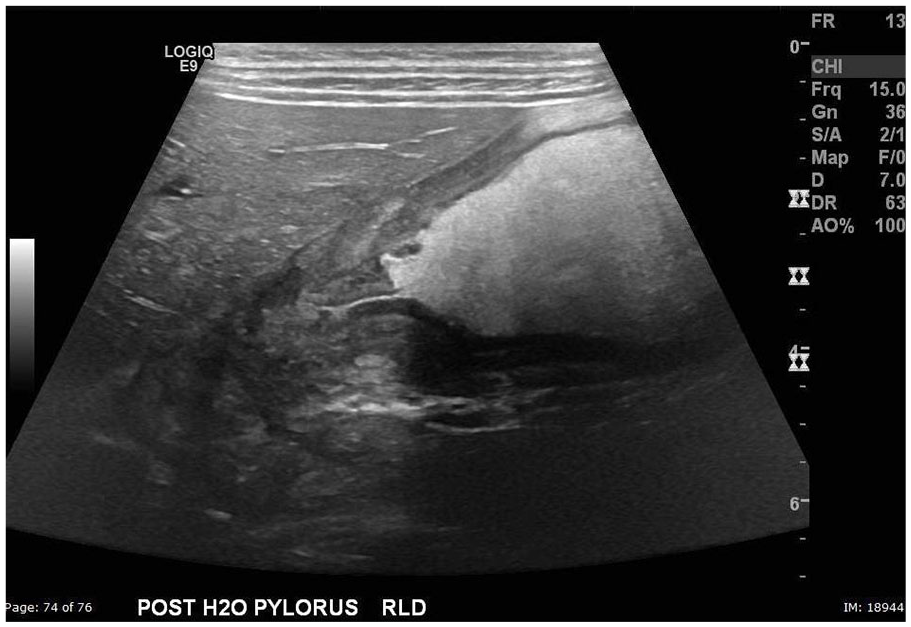
Longitudinal sonographic image of the pylorus after water ingestion in the right lateral decubitus position.
The final study was an abdominal magnetic resonance imaging (MRI) examination with oral contrast (Figure 13). Due to the patient’s early satiety, only one of the two bottles of contrast necessary for the examination was ingested. A markedly thickened pyloric muscle was reported as described during the sonographic examination, and no large masses causing obstruction were seen. The stomach was severely distended, and delayed passage of contrast was noted, although a small amount was reported to pass through to the jejunum. Retroperitoneal and mesenteric lymph nodes seen during the sonographic evaluation were confirmed and reported by the radiologist as likely reactive, consistent with mesenteric lymphadenitis secondary to a gastrointestinal infection.
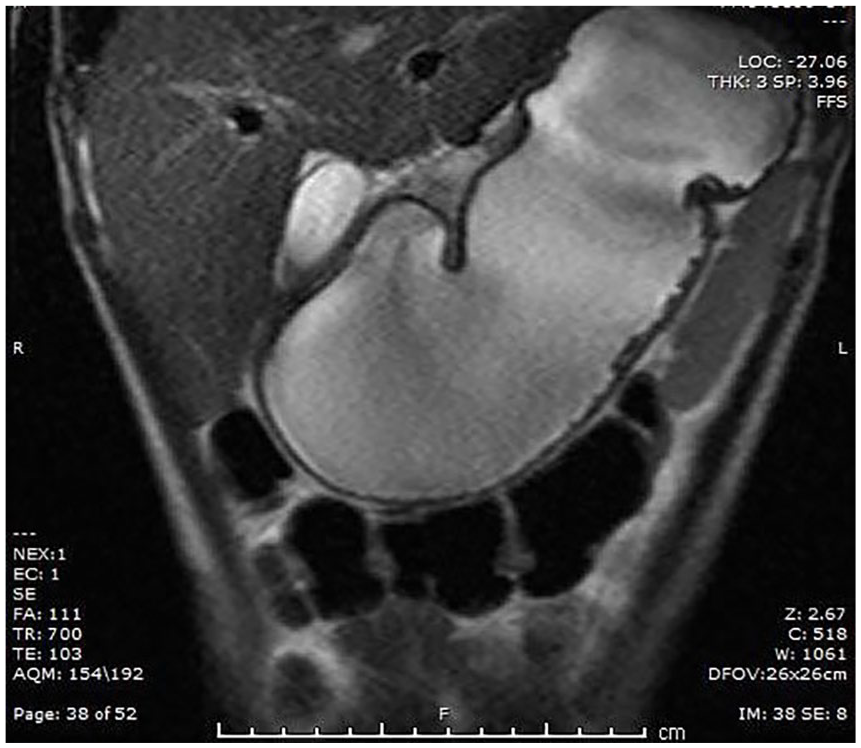
Abdominal magnetic resonance imaging with contrast showing a distended stomach with a markedly thickened pylorus.
To relieve the patient’s symptoms, a nasogastric postpyloric feeding tube was placed into the proximal jejunum under fluoroscopy (Figure 14). This allowed the patient to receive enteral nutrition while receiving antibiotic and steroidal therapy. This medically conservative treatment was attempted first to see if treating the infection would reduce the inflammation at the pylorus and relieve the obstruction before resorting to a more invasive intervention. The patient was transferred to a pediatric facility for further treatment.
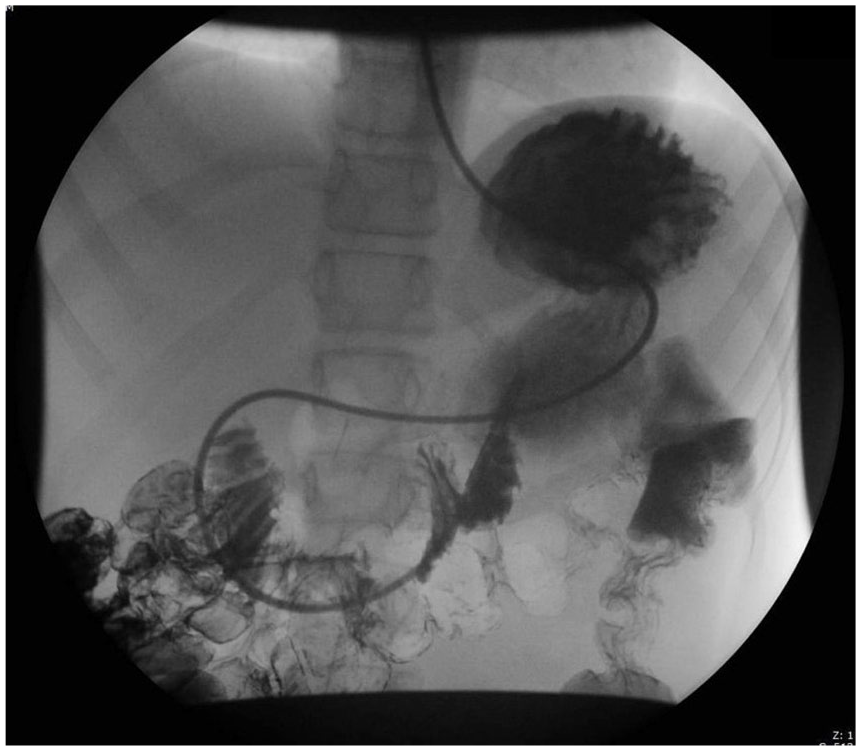
Nasogastric postpyloric gastric feeding tube placed under fluoroscopy.
Discussion
A benign form of gastric outlet obstruction, infantile hypertrophic pyloric stenosis (HPS), occurs in 2 to 4 of every 1000 infants. 5 Occurrence of adult cases of gastric outlet obstruction has been well established, especially in the over 50 population. The incidence of gastric outlet obstruction in adolescents, however, has not been established as it is extremely rare. 6
The usual pathophysiology of gastric outlet obstruction is an intrinsic or extrinsic obstruction of the pyloric channel or duodenum. 7 The mechanism that leads to obstruction depends on the underlying etiology. Intrinsic obstruction may result from acute inflammation, scarring, or chronic fibrosis of the pyloric channel or duodenal bulb, as can occur with chronic pancreatitis. Extrinsic obstruction can occur secondary to compression of the pyloric canal or duodenal bulb by a mass external to the gastric region or by metastatic disease at the gastroduodenal junction. The etiology that leads to the condition is classified as benign or malignant, and this classification facilitates the management and treatment of the obstruction.
Benign Etiologies
Most cases of gastric outlet obstruction in adolescents are of benign etiologies, including inflammatory bowel disease, acute or chronic pancreatitis, Crohn disease, and eosinophilic gastroenteritis. Infectious diseases, whether viral or bacterial, can cause gastric outlet obstruction in an immunocompromised individual. If the infection is severe, obstruction can occur in an otherwise healthy patient. Peptic ulcer disease used to account for over 70% of benign cases of gastric outlet obstruction, but with the use of proton pump inhibitors and other acid-suppressing medications, it now accounts for less than 10% of cases.5,7 Bouveret syndrome, a rare condition that is characterized by a proximal migration of an impacted gallstone into the duodenal bulb or pylorus,8,9 has been reported as a cause of gastric outlet obstruction. Bouveret syndrome typically affects elderly patients, which increases the risk of a misdiagnosis in younger patients. 9 Strictures caused by the use of nonsteroidal anti-inflammatory drugs (NSAIDs) may lead to obstruction with overuse and, in some patients, can develop with infrequent use. 10 Idiopathic or acquired HPS rarely occurs beyond infancy. 11 In adolescents and adults, the etiology of HPS is unknown and is thought to be a manifestation of the infantile form that is triggered by a specific cause such as inflammation, infection, or edema.7,12 Other etiologies include benign tumors such as adenomas or lipomas. Bezoars, which are solid masses of indigestible material that accumulate at the gastric outlet, may cause obstruction. 13 They can be composed of particles of food or even substances such as hair and often require surgical intervention to relieve obstruction at the gastric outlet.
Malignant Etiologies
Malignant causes are typically neoplastic in nature, with pancreatic masses being the most common etiology.7,14 Research shows that 15% to 20% 14 of patients with pancreatic cancer present with gastric outlet obstruction. Other malignancies that may cause a mechanical impediment of gastric emptying are gastric carcinoma, gastric lymphoma, and gastrointestinal stromal tumors.
Clinical Presentation
Gastric outlet obstruction typically presents with nausea and nonbilious vomiting, with vomiting being the hallmark symptom of the condition. 15 These symptoms occur intermittently in the early stage and more continuously in the later stage until there is ultimately a full obstruction. When the obstruction becomes chronic, the continuous vomiting can cause dehydration, electrolyte imbalance, and malnutrition. 16 Initially, the patient may demonstrate better tolerance to liquids rather than solid food. In the later stages, due to the inability to retain food and liquid, the patient may exhibit severe weight loss and clinical signs of malnutrition. With persistence of obstruction, there will be a progressive gastric dilation, and the stomach eventually loses contractility, allowing for undigested gastric contents to accumulate, putting the patient at risk for aspiration pneumonia or other complications. Occasionally, there will be bleeding in the upper gastrointestinal tract detected during diagnostic imaging or laboratory workup.
Treatment and Prognosis
The treatment and prognosis of gastric outlet obstruc-tion vary depending on the etiology and the severity of obstruction. The initial treatment plan consists of relieving the patient’s pain, decompressing the stomach, and restoring a nutritional pathway. 14 The patient’s condition can then be further assessed to determine which type of intervention will be most effective. For mild gastric outlet obstruction of benign etiology, endoscopic balloon dilation (EBD) has proven to be an effective and less invasive option to open pyloric surgery. 17 Cases of gastric outlet obstruction due to inflammatory conditions such as mesenteric gastroenteritis or Crohn disease may require multiple EBD procedures and increase the risk of obstruction recurrence unless properly treated. Malignant or more severe cases of obstruction may require a more invasive surgical approach. 17 Traditionally, open gastrojejunostomy was the surgical method of choice. In recent decades, prevalence has been given to laparoscopic gastrojejunostomy, which is a minimally invasive procedure that results in less postoperative pain and a shorter recovery time for patients. 18 If the patient has been given a poor prognosis and a limited life expectancy, surgical repair may not be an option. Administering nutrition through a nasogastric feeding tube or the placement of a stent through the stricture is an option to keep the patient comfortable in these cases.
Sonographic Evaluation of the Pylorus
To evaluate the pylorus, a 6- to 10-MHz linear transducer is most often used with the patient in the supine position.3,6,19 A curvilinear or sector transducer is encouraged if the structure is difficult to locate or if the patient has a larger body habitus. The pylorus is typically located medial and posterior to the gallbladder.3,19 To locate it, begin by placing the transducer in a transverse position beneath the xiphoid process and use the liver as an acoustic window. If the stomach is empty, the pylorus is likely to be visualized midline in this area.3,19 If the stomach is full and the pylorus is not seen midline, it may be displaced by gastric contents, so sliding over toward the area of the gallbladder may help locate it. An important component of the protocol is to determine whether gastric contents pass through the pyloric canal into the duodenum as the pyloric sphincter contracts and releases. 20 The patient can be given water to drink to evaluate whether the fluid passes.
Once the pylorus is located, the appearance of the structure is evaluated, and a measurement of the anterior wall and length of the pyloric canal is taken. A hypertrophied pyloric muscle will appear hypoechoic and will display a hyperechoic central mucosa. Normal measurements for the pylorus are typically an anterior wall thickness less than 3 mm and a pyloric canal length less than or equal to 14 mm (Figure 3).2,6,19,20 It is important to note that while these measurements are typical for determining HPS in infants, there are no established pylorus measurement parameters for adolescents. Analysis of current ultrasound measurement guidelines was conducted in a study by Said et al. 21 in 2012. In this study, retrospective analysis posed the question of whether pyloric measurements among infants diagnosed with HPS correlated with varying weights and ages. The study consisted of 189 patients over a five-year period who had undergone laparoscopic or open pyloromyotomy confirmation of HPS. Although sonographic findings suggesting HPS such as gastric emptying failure, pylorus contraction and release failure, and thickened pyloric muscle are useful to the diagnostic process, these criteria are not objective. Numerical criterion has become the gold standard for diagnosis, but the authors hypothesized that the measurements obtained during ultrasound evaluation could be misleading. The study found that 5% to 8% of patients with confirmed HPS had a numerically abnormal study. An attempt was made by the authors to develop a measurement criterion that would correlate the patient’s age and weight with pyloric muscle thickness or pyloric canal length. The results of their data showed that there was no correlation with pyloric length and the weight or age of the patient. The authors theorized that the reason for the variability in pyloric length was due to the spasm of the antral muscle proximal to the pylorus. While in spasm, the antral muscle can resemble the pylorus, causing the sonographer to include the antrum in the pyloric canal measurement. A correlation was found, however, between the pyloric muscle thickness and the patient’s age and weight, although a method to avoid a misdiagnosis in younger, smaller patients was not found. The authors concluded that while they support the sonographic diagnostic criterion of a pyloric muscle thickness of 3 mm or greater, a pyloric canal length of 12 mm or greater can be misleading.
Conclusion
Gastric outlet obstruction is a rare finding in adolescents and can be challenging to diagnose. Any delay with a diagnosis can result in severe malnutrition and electrolyte imbalance, which can have detrimental effects on the patient. Although an upper gastrointestinal study is the preferred diagnostic imaging modality, in adolescents, the role of sonography can prove to be instrumental to appreciate thickened pyloric walls, free fluid in the abdominal quadrants, and any lymphadenopathy present. The sonographer’s role in documenting the appearance and function of the pylorus as well as investigating findings in a methodical manner is crucial. This will help provide a definitive diagnosis and a clearer path to treat the patient.
Footnotes
Acknowledgements
I thank Lynn Schluns, BS, RDMS, RVT, and Maria Johnson, RDMS, RVT, for their guidance and support. A special thank you to Stephen and Olivia Liddick who helped make all of this possible.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
