Abstract
In the female, the canal of Nuck is a tubular fold of parietal peritoneum that travels along with the round ligament through the inguinal canal to its attachment on the labia majora. The canal of Nuck is analogous to the processus vaginalis in the male. In embryonic development, this pathway typically obliterates early in life; however, in some cases, it may partially or completely fail to close. This failure to obliterate can lead to complications ranging from a cyst of the canal of Nuck (also known as a female hydrocele) to herniation and incarceration of abdominal and pelvic organs. There is little information available in the medical literature concerning this rare condition; therefore, it is important for sonographers, physicians, and surgeons to be familiar with this developmental abnormality. This report represents the first prenatal case of a cyst of the canal of Nuck, along with the unique combination of a large left fetal ovarian cyst and acute polyhydramnios. The embryology, incidence, differential diagnosis, management, and treatment of these female conditions are discussed.
Case Report
A 31-year-old woman was referred at 33 weeks of gestation for a fetal abdominal cyst that had been discovered during a fetal growth ultrasound. Records from the referring office indicated that a routine anatomy scan had been performed at 19 weeks of gestation, which was reported as normal. A detailed anatomic survey of the fetus at our institution identified significant polyhydramnios with the four-quadrant amniotic fluid index at 38.7 cm and a 9.5 × 10.1 × 7.5 cm round anechoic cyst occupying the fetal abdomen and pelvis. Compression of the fetal bowel and abdominal viscera was appreciated without evidence for renal obstruction. Because the differential diagnoses included ovarian cyst, determination of the fetal gender was necessary. Upon evaluating the external genitalia, a 2.5 × 1.6 × 1.6 cm ovoid cystic mass was noted in the right labia majora of a female fetus (Figure 1). Three-dimensional surface rendering was used to further delineate the abnormal findings, and the reconstructed images of the fetus emphasized the magnitude of the abdominal distention, as well as the right labial swelling (Figure 2). The working diagnosis was a suspected fetal left ovarian cyst, as well as a concomitant cyst in the right canal of Nuck.
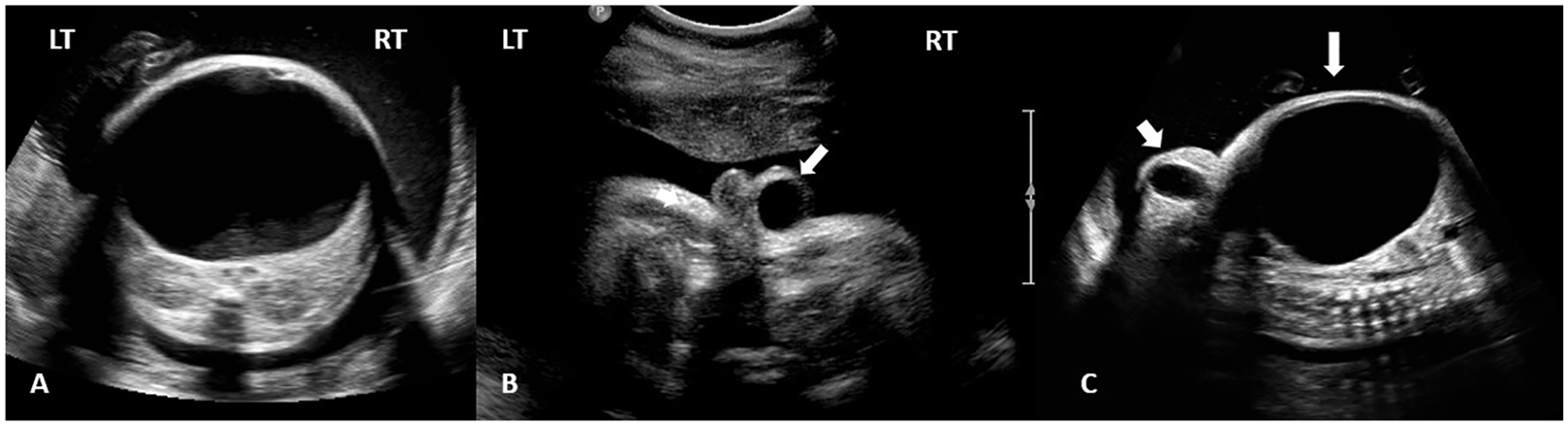
Transverse and oblique sonographic images of the fetal abdomen and pelvis at 33 weeks of gestation. (A) Transverse section of the mid fetal abdomen at the level of the kidneys demonstrates the left ovarian cyst occupying the entire abdomen and pelvis without evidence of renal obstruction. (B) Transverse image of the fetal pelvis shows a well-defined cystic mass within the subcutaneous tissue of the right labia majora, which is the equivalent to a scrotal hydrocele in a male fetus (arrow). (C) Oblique right parasagittal image of the abdomen and pelvis demonstrates the huge abdominopelvic cyst (arrow) compressing the bowel and elevating the diaphragm. The ovoid cyst of the canal of Nuck is observed extending from the inguinal canal into the right labia majora (arrowhead).
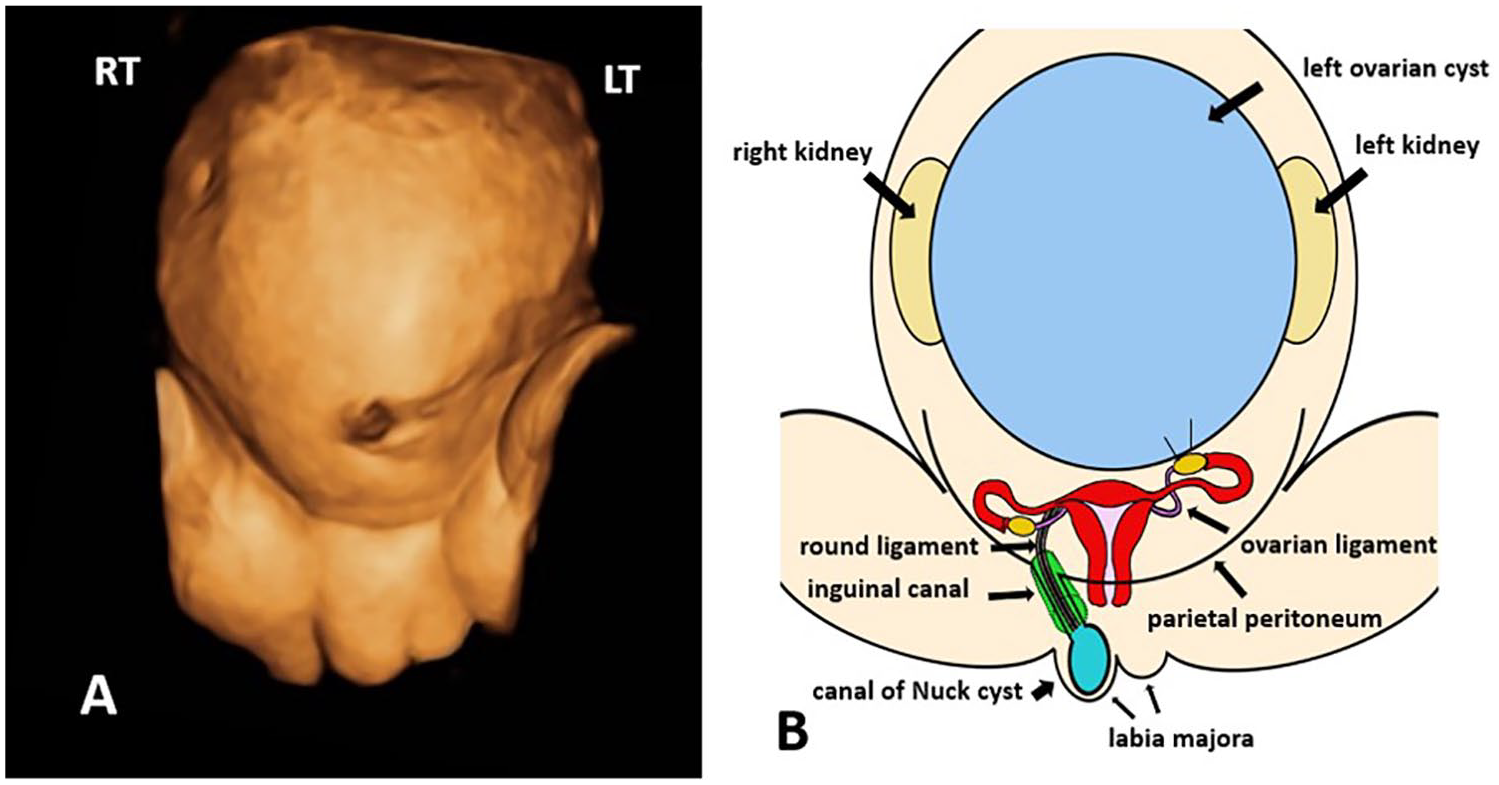
(A) Three-dimensional surface rendering of the fetal abdomen and pelvis at 33 weeks of gestation. Significant abdominal distention is appreciated, resulting in laxity of the abdominal musculature, and inguinolabial soft tissue swelling is apparent. (B) Corresponding medical illustration demonstrates the abdominal and pelvic anatomy, as well as the pathology on the right side that was reported in this case. A patent canal of Nuck allows for fluid accumulation within the labia majora. The proportional distention of the fetal abdomen is a consequence of the magnitude of the left ovarian cyst.
A multiplanar fetal MRI was performed at 34 weeks of gestation, which reported a 10.5 × 8.0 x 9.4 cm cystic intensity lesion causing a mass effect in the fetal abdomen, intermediate signal on T1 and hyperintense on T2. In addition, a 1.8 × 2.2 × 3.0 cm fluid intensity of the right aspect of the labia majora was described (Figure 3). At 35 weeks of gestation, a decompression amniocentesis was performed due to persistent symptomatic polyhydramnios, and 1.8 liters of straw-colored fluid was removed from the amniotic space. A sample of the amniotic fluid was sent for karyotype and fluorescence in situ hybridization (FISH) analysis, which subsequently resulted in a normal female karyotype. The maternal patient was referred for prenatal consultations with pediatric surgery and neonatology to discuss the postnatal treatment and management of the fetus. At 38 weeks of gestation, the abdominopelvic cyst remained stable and the right labial cyst had increased slightly in size, measuring 3.6 × 2.4 × 2.7 cm. In addition, the amniotic fluid index had increased to 42.1 cm. Although in utero aspiration of the cyst was considered, based on advanced gestational age, clinical progression of the polyhydramnios and significant maternal discomfort, the decision to move forward with delivery was made. The patient delivered a 3890 g female by scheduled repeat Cesarean section.
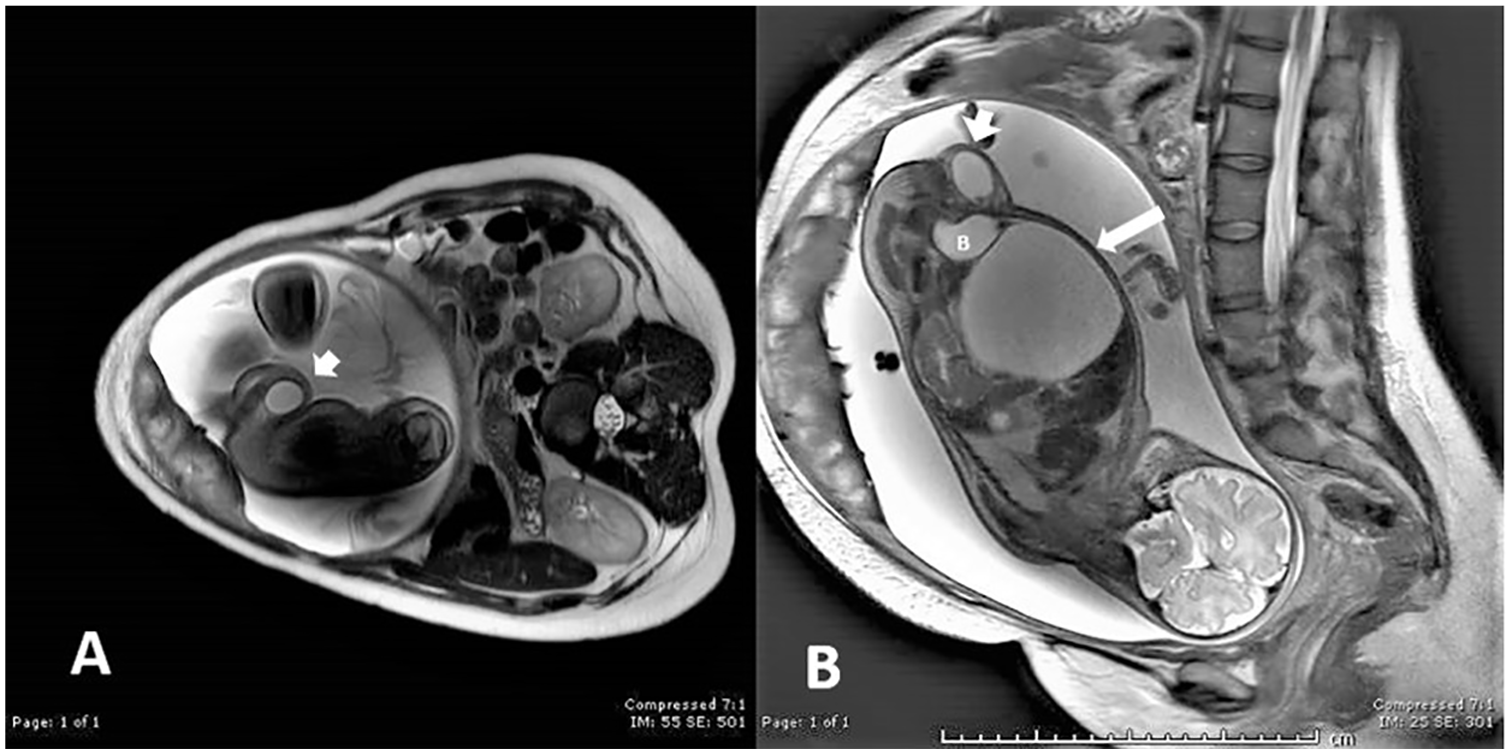
Fetal T2-weighted MRI at 34 weeks of gestation. (A) Oblique image of the fetal pelvis reveals the hyperintense fluid collection in the right labia majora corresponding to the cyst of the canal of Nuck, or female hydrocele (arrowhead). (B) Right parasagittal image of the fetus demonstrates the relationship among the left ovarian cyst (arrow), urinary bladder (B), and the ovoid fluid accumulation in the right labia majora (arrowhead).
Physical examination of the neonate found a large for gestational age newborn with significant abdominal distention, laxity of the abdominal wall musculature, and swelling of the right labia majora. There was clear and equal breath sounds bilaterally with fair aeration and limited expansion due to the abdominal mass, which required intubation. In the right labia majora, there was a palpable cystic mass with soft tissue edema. A postnatal sonogram of the neonatal pelvis reported a large cystic abdominal mass in the lower abdomen and pelvis situated slightly to the left of midline, measuring 10.1 × 11.7 × 7.3 cm. The uterus was noted in the midline pelvis and measured approximately 3.6 × 1.7 × 0.8 cm. Measuring 1.1 × 1.5 × 0.8 cm, the right ovary was identified in the right adnexa. The left ovary could not be identified with confidence as a separate structure from the abdominopelvic cyst. Imaging over the right inguinolabial swelling confirmed a cyst of the canal of Nuck. The sonographic features included an avascular, tubular, thick-walled anechoic cystic structure measuring 2.9 × 1.8 × 0.8 cm (Figure 4). Evaluation of the contralateral inguinal canal found no abnormality. On day three of life, the neonate was taken to the operating room for an exploratory laparotomy and cystectomy. Upon identifying the cyst under the peritoneum, a fine needle aspiration was performed, yielding 180 milliliters of clear gold fluid. Approximately 40 milliliters of the fluid was sent to cytopathology, which was negative for malignancy. Following the aspiration, the surgeon was able to mobilize the remaining cyst out of the abdomen and identify that it originated from the left ovary. The cyst was then carefully dissected away from the fallopian tube and uterus using a combination of cautery and blunt dissection and passed off the surgical field intact (Figure 5). No intervention was performed for the cyst of the canal of Nuck to avoid further surgical trauma. It was believed that the Nuck cyst would resolve spontaneously once the increased abdominal pressure had been eliminated. Pathology confirmed the diagnosis of an atypically large left ovarian follicular cyst. The newborn’s postsurgical course was unremarkable and discharge to home occurred on day nine of life. Due to the potential for future complications, such as recurrence of the Nuck cyst or the development of an inguinal hernia, the patient will be closely monitored by the pediatrician. Follow-up evaluation at one year of age reported complete resolution of the cyst of the canal of Nuck.
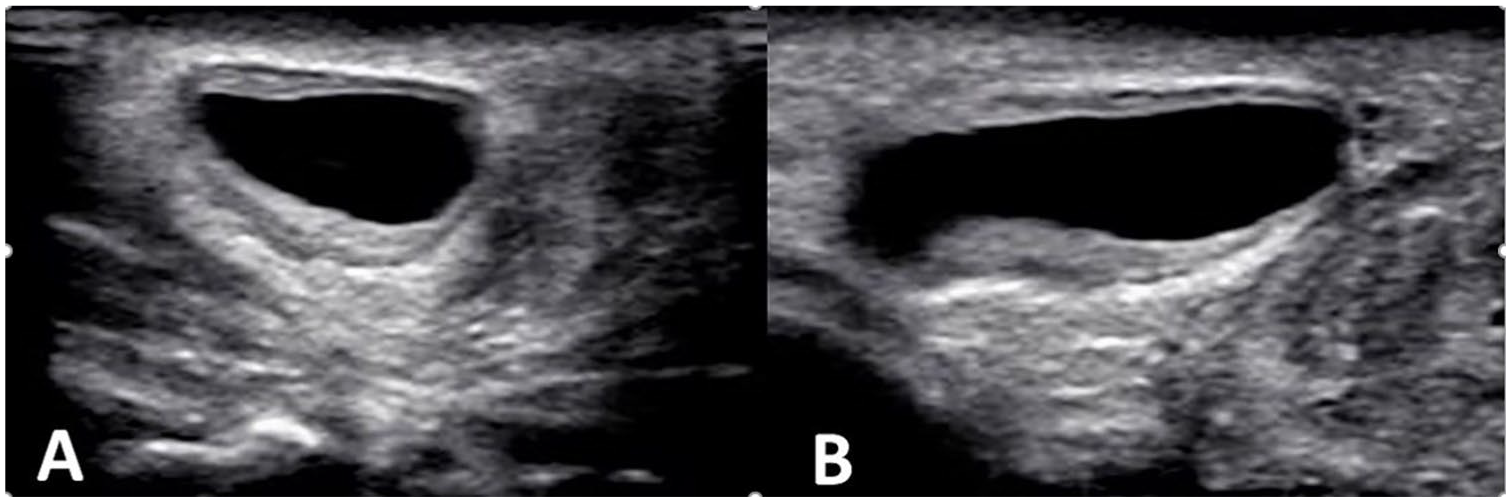
Postnatal sonographic imaging over the right inguinolabial swelling. (A) Transverse and (B) longitudinal images of the cyst of the canal of Nuck. The typical sonographic features are present, including a superficial, subcutaneous, tubular cystic structure in the area of the inguinolabial swelling. The cyst is well-circumscribed with a thick wall, and it contains anechoic fluid without evidence of debris, septations, or abdominal/pelvic organs.
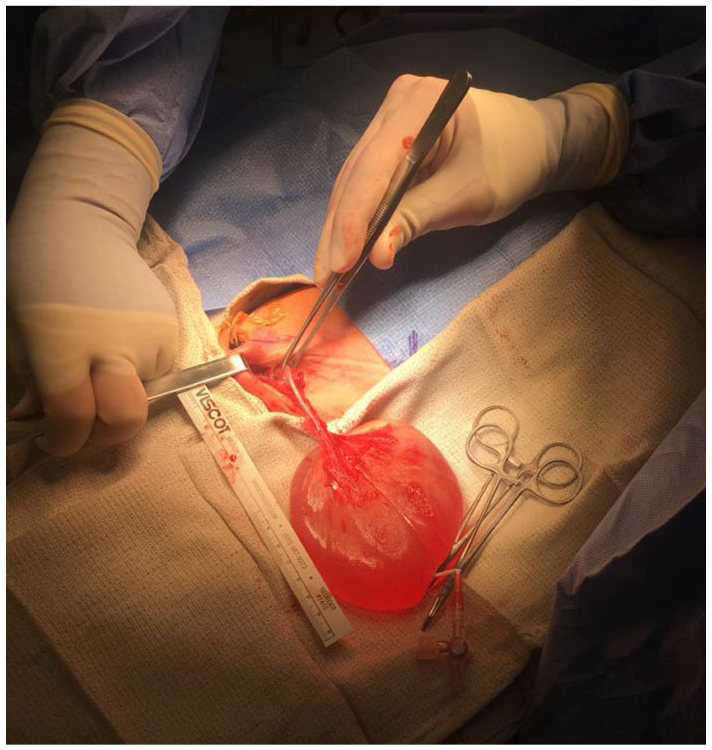
Intraoperative photograph taken during the exploratory laparotomy shows the exteriorized left ovarian cyst with no residual ovarian tissue noted. Pathology revealed a follicular cyst with negative fluid pathology.
Discussion
History and Incidence of Canal of Nuck Cyst
The canal of Nuck, which is the female equivalent of the processus vaginalis in the male, is a protrusion of the parietal peritoneum that travels along with the round ligament through the inguinal canal to its attachment on the labia majora. In the 17th century (1691), Dutch anatomist Anton Nuck published Adenographia curiosa et uteri foeminei anatome nova, a document where he described for the first time a cystic inguinolabial structure extending up from the labia majora in women. 1 In 1954, the condition was described in depth, naming it “cyst of the canal of Nuck,” giving credit to Anton Nuck. 2 A cyst of the canal of Nuck is a rare entity, with fewer than 500 cases described in the literature, most of which are case reports. 3 Due to the rarity of this condition and lack of awareness among health care professionals, the true incidence has not been established. In a retrospective study of 686 females undergoing inguinal operations, Akkoyun et al 4 reported 6 cases of a cyst of the canal of Nuck, resulting in an incidence of 0.76% in this population. Similarly, Huang et al 5 reported the incidence of a hydrocele of the canal of Nuck in the female child at 1% based on a series of 1800 infants and children who were evaluated for possible inguinal hernia due to an inguinoscrotal or inguinolabial mass. In this study, a total of 580 of the patients were female, and 32 of these presented with an asymptomatic palpable movable mass over the labium major. Ultimately, 6 of 580 cases of suspected inguinal hernia resulted in a diagnosis of a cyst of the canal of Nuck. 5 The true incidence of this condition may not be reflected, as many cases may not be reported or are treated conservatively and resolve on their own. Typically, the diagnosis is made on the operating table at the time of suspected inguinal hernia surgery. Of note, the term “female hydrocele” or cyst of the canal of Nuck is not mentioned in many comprehensive textbooks on surgery, urology, and gynecology. 6 The authors of this article also failed to find reference to this female condition in any sonography textbook on obstetrics, fetology, or prenatal diagnosis of congenital anomalies.
Embryology
The gubernaculum and the processus vaginalis are the two main fetal structures that contribute to the prenatal development of the inguinal canal. The superior aspect of the gubernaculum attaches to the lower pole of the fetal gonad, and the inferior aspect of the gubernaculum attaches to the skin of the fetal groin. 7 In the male fetus, the gubernacula are paired structures that assist in the descent of testis through the inguinal canal into the scrotum, a process that occurs at approximately 28 weeks’ gestation and is usually completed by 32 weeks’ gestation. 8 In the female fetus, the paired gubernacula connect the uterus with each inguinal canal and labia majora. They also connect the uterus with the ovaries because during fetal life, the midpoint of each gubernaculum attaches to the uterus and is designed to prevent ovarian descent into the ipsilateral inguinal canal. Postnatal remnants of the fetal female gubernacula include the paired ovarian ligaments, which extend medially from each ovary to the uterus, and the paired round ligaments, which extend from the uterus through each inguinal canal to the labia majora. 7 The processus vaginalis, a tubular fold of parietal peritoneum, penetrates into the inguinal region about the 13th week of fetal development, extends into the inguinal canal alongside the gubernaculum during the seventh month of fetal life, and usually is completely obliterated into a fibrous cord from eight months to one year of age in females. The obliteration proceeds gradually in a superior-to-inferior direction, and closure of the processus vaginalis on the left side precedes the closure on the right.2,9–11 This contrasts with male development, where the obliteration also occurs in a superior-to-inferior direction, but the most inferior portion remains patent and develops into the residual tunica vaginalis. The processus vaginalis is estimated to be longer in males than the canal of Nuck in females.2,12 In both males and females, failure of obliteration of the entire processus vaginalis provides open communication with the peritoneal cavity and allows fluid or pelvic contents to herniate through the inguinal canal to the labia majora or scrotum. This pathway may result in an indirect inguinal hernia, hydrocele, or both. The processus vaginalis may remain patent in approximately 80%–90% of full-term babies. 10 In a study by Toki et al, 13 22/40 neonates with a birth weight under 2500 g had a patent processus vaginalis, while 54% of premature neonates between 22 and 37 gestational weeks were noted to remain patent. The patency was noted to close spontaneously within the first year of life in 81% of low-birth-weight neonates and 91% of premature neonates. If no abnormality occurs, a patent processus vaginalis may continue the natural course to obliterate naturally or may remain patent without consequence.
Patent Processus Vaginalis: Males
Depending on the degree and level of processus vaginalis obliteration failure, different abnormal conditions can develop anywhere along the inguinal canal and into the scrotum. In the male, hydroceles can be divided into two categories: communicating and noncommunicating. When it comes to hydroceles situated in the spermatic cord, some authors divide them into three types: communicating, funicular, and encysted. The term “scrotal hydrocele” refers to the presence of a fluid collection inside the tunica vaginalis and is limited to inside the scrotum and surrounding the testes. 10 The prenatal diagnosis of a simple scrotal hydrocele is typically based on the visualization of anechoic fluid within the fetal scrotum. Hydroceles, a relatively common finding during ultrasound evaluation of the fetus, have been reported to occur in 15% of male fetuses in the third trimester. 8 The literature suggests the frequency of hydroceles in newborn male infants of 36-42 weeks’ gestation is as high as 58%. 14 This condition is well recognized in the literature, with the first prenatal case reports being published over 30 years ago.15–17 In a prospective study, Naji and colleagues 18 reported that 89% of patients with infantile hydrocele will have a spontaneous resolution owing to the ongoing changes in the patent processes vaginalis. In rare cases, if a fetus has developed meconium peritonitis due to leakage of meconium from a perforation in the bowel, a patent processes vaginalis may lead to migration of meconium into the scrotum, resulting in a meconium-filled hydrocele.19,20 Clinically, most hydroceles are considered physiologic and will resolve spontaneously during the first year of life due to ongoing changes in the processus vaginalis and natural resorption of the fluid. Operative intervention may be needed if there is an associated inguinal hernia or the hydrocele becomes symptomatic. 21
Indirect hernias occur when pelvic contents protrude through the deep inguinal ring, lateral to the inferior epigastric vessels, due to a patent inguinal canal from failure of closure of the embryonic processus vaginalis. 11 The overall incidence of inguinal hernias in infancy and childhood ranges between 0.8% and 4.4%, is highest in the first year of life, and may develop in up to 30% of premature infants.11,22 Indirect hernias may be reducible, incarcerated, or strangulated. Inguinoscrotal hernias usually occur in preterm neonates, and sonography can establish the diagnosis with demonstration of loops of bowel with peristalsis located within the scrotal sac. 10 An additional abnormality of the male that may be detected is cryptorchidism. This condition results when one or both testes fail to completely descend from the abdomen into the scrotum, in most cases retained in the inguinal canal or in the abdomen. 9
Patent Canal of Nuck: Females
In females, if the canal of Nuck remains patent within the inguinal canal, it creates a communication between the peritoneal cavity, the female inguinal canal, and the labia majora. 23 The two main categories of canal of Nuck abnormalities are indirect hernias and hydroceles. Inguinal hernia should be the first consideration in the differential diagnosis of an inguinolabial mass in females. This makes the diagnosis challenging because up to one third of canal of Nuck cyst cases are associated with an inguinal hernia.24,25 The patent canal of Nuck may allow for abdominal and pelvic contents such as bowel, omental fat, ovary, fallopian tube, and rarely the uterus and urinary bladder to herniate through the inguinal canal to the labia majora. It is reported that 15%–20% of hernias in female infants contain ovary.11,22 As with male inguinal hernias, potential reducibility, incarceration, and strangulation of these structures can occur.
Historically, the term “hydrocele” has been almost exclusively associated with the male gender. There is little information available in the literature concerning the incidence of female hydrocele, also known as a cyst of the canal of Nuck. Three classifications of female hydroceles have been outlined. Type 1, the most common type, occurs when there is a cyst in the canal of Nuck that does not communicate with the peritoneal cavity and may be found anywhere along the course of the round ligament from the internal ring to the vulva. This type corresponds to the encysted hydrocele of the spermatic cord in the male. Type 2 develops as a fluid collection inside the patent canal of Nuck that communicates with the peritoneal cavity through the deep inguinal ring and corresponds to the communicating hydrocele in the male. Type 3, the least frequent type, presents as a bilocular appearance with two cystic volumes separated by a constriction of the deep inguinal ring, giving an hourglass appearance. The most superficial cyst is within the inguinal canal, and the deepest one retroperitoneally protrudes into the abdominal cavity, communicating with the peritoneal cavity through a small opening.9,26 It is reported that 80% of Nuck cyst cases appear predominantly on the right side. 2 Various mechanisms have been proposed for the accumulation of fluid in the canal of Nuck. These range from passive accumulation of physiologic free intraperitoneal fluid through a completely patent canal to an imbalance of fluid secretion and absorption functions of the secretory membrane lining of the epithelium. The hypersecretion or underabsorption imbalance may be due to inflammation, trauma, or impairment of lymphatic drainage, but in most cases it is idiopathic. 27
Cysts of the canal of Nuck can have a variety of sonographic appearances depending on the type of hydrocele encountered. Nuck cysts are usually small, rarely exceeding 3 cm in largest dimension, but may vary from between 23 and 55 mm.4,28 Jagdale et al 29 described a 4.3 × 2.6 cm well-defined, oval, anechoic cyst within the inguinal canal with a cranial-detected tail measuring 1.6 × 0.7 cm. The cyst showed thickened echogenic wall with a single septation. Khanna et al 28 described a 4.2 × 2.4 × 2.4 cm dumb bell–shape cyst with an internal septum. Other case reports describe a well-defined 5.5 cm hypoechoic, elongated septated mass that extended from the superficial inguinal canal to the labia majora; an anechoic tubular, sausage-shape cyst that appeared to extend along the round ligament; or a comma-shape cyst with a surface beak that was thought to represent the point of continuation with the peritoneal cavity.28–30 Yigit et al 25 described a cyst with tissue thickening deep to the inguinal canal resembling a tail or neck that was seen to open and distend with fluid upon application of graded pressure, thus delineating an entirely patent canal of Nuck. Stickel and Manner 6 described a cyst-in-cyst appearance. De Meulder et al 31 reported a cystic mass with a maximum dimension of 3.2 cm without communication with the peritoneum. Cysts are often described as sausage-shaped since they frequently conform to the elongated and narrow configuration of the canal itself. However, as more fluid accumulates, the cyst can become more rounded, and as in other locations throughout the body, a cyst of the canal of Nuck may be simple or complex with internal septations and debris. Simple cysts appear as an anechoic collection with a thin wall, posterior acoustic enhancement, and lack of internal vascular flow on color Doppler. A complex cyst will contain avascular septations and/or low-level internal echoes on sonography. This does not necessarily indicate superimposed infection; however, these same findings and a thickened wall can be seen in an infected cyst. 23 Rarely, a meconium hydrocele may develop if there is in utero perforation of the bowel and subsequent spillage of meconium into the peritoneal cavity. Free communication of the peritoneal space with the processus vaginalis during gestation permits formation of a meconium hydrocele.32,33
Given the broad differential diagnosis and the frequency of cases that are in the pediatric group, grayscale and color Doppler sonography has emerged as the diagnostic imaging modality of choice for evaluation of superficial groin masses. Sonography provides a means for rapid acquisition of information, including size, shape, location, internal contents, and vascularity. It allows for maneuvers that provide additional dynamic evaluation of the mass and avoids ionizing radiation, injection of contrast materials, or sedation/anesthesia.25,28 On physical exam, a cyst of the canal of Nuck in the newborn usually presents as a nontender or mildly painful, mobile, irreducible mass lying superficially in the groin and medial to the pubic bone at the level of the superficial ring. Typically, the lesion will demonstrate positive fluctuation and transillumination. The existence of a cyst of the canal of Nuck is not associated with nausea or vomiting, and the cyst does not disappear or increase in volume with changes in position or with the Valsalva maneuver.2,28,34
The remaining differential diagnoses for an inguinolabial mass in the female fetus and newborn include gonadal and embryonic remnants such as Skene’s duct cysts (paraurethral cysts), hymenal cysts, Gartner’s duct cysts (mesonephric cysts), Bartholin’s duct cysts, and cysts of the canal of Nuck (hydrocele). However, Skene’s duct cysts, hymenal cysts, and Gartner’s duct cysts usually present as an interlabial mass. Bartholin duct cysts are the most common lesion arising in the vulva, usually in reproductive age, due to the accumulation of secretions. 35 In the prepubertal child, rare cases of vulvar hamartoma, rhabdomyoma, lipoma, fibroma, lymphangioma, granular cell tumor, neurofibromatosis, hemangioma, epidermal cyst, dermoid cyst, or malignant tumors such as embryonal rhabdomyosarcoma and endodermal sinus tumor have been reported.36,37 Additional considerations would be abundantly vascular lymphomas, hematomas, abscesses containing debris, femoral hernias, and hip joint ganglion cysts.23,28
Options for the treatment of a hydrocele of the canal of Nuck include observation with conservative management, aspiration, and surgical excision. 10 Due to the fact that the anticipated progressional obliteration of the processus vaginalis may not be complete until the first year of life, expectant management in this population may be reasonable unless the hydrocele becomes symptomatic. Surgical excision by hydrocelectomy is the definitive treatment of a canal of Nuck cyst. This involves with resection of the cyst, high ligation at the neck of the processus vaginalis, and closure of the fascial defect in the peritoneum. Although surgery prevents recurrence and has been reported as the treatment of choice, others feel this option should be reserved for symptomatic cases. Ultrasound-guided aspiration of fluid from the cyst is an option, but the potential for reaccumulation of the fluid is high and there is a risk of hemorrhage and/or infection. If infection is suspected and confirmed by ultrasound, the alternative management is drainage of pus and administration of antibiotics with surgery once the infection has resolved.38–42
Fetal Ovarian Cyst
The fetal and neonatal ovary is normally dormant, but follicular cyst development may occur. Small cysts are frequently found in neonatal ovaries and are accepted as normal, while cysts of > 2 cm are pathologic. 43 There is no consensus regarding the etiopathogenesis of ovarian cysts; however, the most widely accepted theory is that exposure to fetal pituitary gonadotropins, placental human chorionic gonadotropins, and maternal estrogens stimulates the fetal ovary and causes follicle production and maturation.44,45 Others report that maternal diabetes, toxemia of pregnancy, and Rhesus isoimmunization all increase the risk of fetal ovarian enlargement and cyst formation, and the cysts might therefore be expected to regress after delivery. Congenital hypothyroidism has also been linked to their formation.43,46 According to their review of cases of fetal ovarian cyst reported over a 25-year period, Bryant and Laufer 45 suggest the best estimate on the incidence of clinically significant ovarian cysts in live births is 1 in 2500. Similarly, Kirkinen and Jouppila 47 reported an incidence of 1 in 2625 based on a series of 21,000 pregnancies. Fetal ovarian cysts have been reported in 30% of neonatal autopsies and, following renal and gastrointestinal etiologies, are known to be the third most common cause of antenatally diagnosed intra-abdominal cysts. 48 Fetal ovarian cysts were first described by Valenti et al 49 in 1975 and have apparently increased in incidence over the years, most likely due to the extensive use of sonography in prenatal diagnosis. A fetal ovarian cyst should be considered when a cystic abdominal mass is identified in a female fetus. The criteria that should be used to establish the diagnosis of an uncomplicated fetal ovarian cyst include the following: documentation of a female fetus, typically presents in the third trimester, location in the lower abdomen, round or ovoid in contour, anechoic in sonographic appearance, no peristalsis observed, and visualization of normal kidneys and urinary bladder. 50 A reported 95% are unilateral, while 5% are bilateral. Antenatal diagnosis is usually made in the third trimester with a range of 27–41 weeks with a median of 34 weeks. 51 One category of antepartum complications associated with fetal ovarian cysts results from compression effects and is usually associated with the relatively larger cysts. These larger ovarian cysts can cause gastrointestinal obstruction or perforation, renal compression/obstruction, upward displacement of the diaphragm causing compression of the lungs, cyst rupture, ascites, and torsion.43,52,53 An additional complication is “prune-belly syndrome,” a consequence of an overdistended abdomen. 51 Acute polyhydramnios has been associated with larger fetal ovarian cysts, with one review reporting the presence of polyhydramnios in 18% of cases. This relationship is also believed to be a result of the compression effects described above.51–55 The differential diagnosis for a cystic mass in any fetus includes renal cyst, hydronephrosis, ureterocele, urinoma, urachal cyst, dilated bowel, meconium cyst, enteric duplication cyst, lymphangioma, choledochal cyst, cystic neuroblastoma, abnormally distended urinary bladder, hydrocolpos, fetus in fetu, and ovarian cyst. 44
No consensus about the management of fetal ovarian cysts is currently available, and to date, no adequately powered randomized controlled trial has reported on the efficacy and safety of in utero cyst aspiration. 56 Management of a fetus affected by an ovarian cyst depends on the size of the cyst, the sonographic appearance, and the presence of complications. As experience has been gained, physicians have exercised increasing caution in applying diagnostic and therapeutic procedures, both pre- and postnatally, as many cases resolve spontaneously. 50 Bascietto et al 57 reviewed and analyzed the outcome of 954 fetuses with a prenatal diagnosis of ovarian cyst. The ovarian cyst regressed either in utero or postnatally in 53.8% of cases, and the likelihood of resolution was significantly lower in complex cysts and cysts measuring ≥ 40 mm. Change in the sonographic pattern of the cyst was associated with an increased risk of ovarian loss of 57.7% (surgical removal or autoamputation). The risk for ovarian torsion and need for postnatal surgery was significantly higher for cysts ≥ 40 mm. In cases undergoing aspiration of the cyst, the rate of recurrence was 37.9%; ovarian torsion and intracystic hemorrhage were diagnosed after birth in 10.8% and 12.8%, respectively; and 21.8% had surgery after birth regardless of circumstances. Tyraskis et al 58 performed a systematic review of the literature to compare outcomes of ultrasound-guided aspiration of fetal ovarian cysts with conservative management. In a review of 92 nonrandomized studies of 365 fetuses, the authors noted a total of 380 cysts. For those 324 cases of cysts that were observed, 56 cysts were aspirated. The overall spontaneous resolution rate of conservatively managed cysts was 46% yet decreased with increasing size. Risk of prenatal ovarian torsion in conservatively managed cases depended on the cyst size. The rate of prenatal torsion in simple cysts ≥ 40 mm was lower in the aspirated group than the conservatively managed cysts, and aspirated cysts had much lower rates of postnatal surgery when compared to conservatively managed cysts. In a retrospective multicenter study, the natural history of 37 unilateral fetal ovarian cysts over a 15-year period was reviewed. Spontaneous resolution occurred in 32% prior to birth; 18.9% of cases resulted in postnatal surgery, and of those, 10.8% were diagnosed with torted necrotic ovaries. Overall, 16.2% of cases resulted in ovarian loss. Rates of ovarian torsion increased incrementally with increasing cyst size, and prenatal resolution occurred less often with increasing size. Cysts ≤ 40 mm resolved spontaneously in 90% of cases as compared to only 44% of cysts > 40 mm. 59 In a retrospective cohort study, Nakamura et al 60 evaluated the outcome of 33 cases of prenatally diagnosed fetal ovarian cysts managed conservatively. Spontaneous regression occurred in 9% of in utero cases, while 42% of cysts regressed postnatally. Of the 14 cysts treated surgically, 4 cases had torsion confirmed. The incidence of torsion between complex cysts and simple cysts was not statistically different, and the incidence of torsion between patients with a cyst ≥ 40 and < 40 mm was similar. The ovaries were preserved in 85% of patients under conservative management. In a 13-year retrospective analysis of 64 fetuses who were suspected to have an ovarian cyst prenatally, spontaneous resolution of the cyst occurred in 53% of cases, 53% prenatally and 47% postnatally. Accompanying malformations are extremely rare, and in general, management and delivery are not affected by the prenatal diagnosis of an ovarian cyst. 50
Practice Parameters
The practice parameter for the standard diagnostic obstetric ultrasound examination (CPT code 76805) does not include the external genitalia as one of the recommended areas of assessment except in multiple gestations and when medically indicated. 61 The detailed second- and third-trimester comprehensive obstetric examination (CPT code 76811) includes all the components of the standard examination plus a list of potential elements based on the indication for the exam and the sonographic findings identified during the exam. The specific elements of a given detailed exam may be individualized based on these considerations. 62 While external genitalia remain listed as one of the elements that may be included, it may not be indicated in every detailed obstetric sonogram unless clinically relevant. However, fetal gender determination can be performed by sonography in pregnancy, as early as the late first trimester. Gender determination is usually prompted by patient request during the routine anatomy scan, if not sooner. Therefore, repeated evaluation of the fetal external genitalia is not usually pursued once the gender has been determined. Abnormalities of the external genitalia detected by sonography later in gestation are often an incidental finding noted during routine follow-up exams.
Conclusion
Delayed or defective obliteration of the processus vaginalis, or canal of Nuck in females, may result in a series of congenital anomalies in both genders, ranging from hydroceles to indirect inguinal hernias. Abnormalities of the external genitalia may be encountered in the prenatal period during the sonographic evaluation of the external genitalia during routine or detailed anatomy scans or, more commonly, in the third trimester during follow-up evaluations. In both males and females, there is a high association with a patent processus vaginalis and prematurity due to the progressional obliteration of this structure up to the first year of life. Thus, most uncomplicated hydrocele cases are expected to regress spontaneously without the need for surgical intervention. We report a case of a cyst of the canal of Nuck diagnosed in utero. To the authors’ knowledge, this is the first case report of a prenatally diagnosed cyst of the canal of Nuck in association with an ovarian cyst and acute polyhydramnios. A cyst of the canal of Nuck, or female hydrocele, is a rare entity with little information available in the literature. However, upon sonographic evaluation of a female fetus or newborn, if the source of inguinolabial swelling has been determined to be cystic, a cyst of the canal of Nuck is the most likely diagnosis. Similarly, fetal ovarian cysts can also occur in the female fetus, typically presenting in the third trimester, and research has shown that they are likely to undergo regression when they are simple cysts of less than 40 mm in size. With sonography being the imaging modality of choice for evaluation of these abnormalities, it is critical for the sonographer and other health care professionals to be aware of these conditions, understand the underlying anatomy and physiology, and recognize the spectrum of sonographic appearances in order to facilitate the timely and accurate diagnosis, management, and treatment of the fetus and newborn.
Footnotes
Acknowledgements
The authors would like to express their appreciation to Jeff Allen for the medical illustration associated with Figure 2.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
