Abstract
Abernethy malformation is a very rare abdominal vascular malformation. It can be defined as portal blood flow bypassing the liver. Without treatment, it can lead to high levels of ammonia, which can cause symptoms such as confusion and predispose the patient to develop lesions within the liver. To date, only a few reports in the literature have been seen using sonography to aid in the diagnosis of this pathology, the majority being discovered in the pediatric population. In this case study, a liver sonogram identified no evidence of a portal vein. This finding was confirmed by a contrast-enhanced computed tomogram of the abdomen. This is a case report in which sonography aided in the incidental diagnosis of Abernethy malformation in a 38-year-old man with Marfan syndrome.
Abernethy malformation, also referred to as congenital portosystemic shunt and congenital absence of portal vein (CAPV), is a very rare vascular malformation discovered by John Abernethy in 1793. 1 The main portal vein carries blood from the intestines and spleen, supplying up to half of the oxygen requirements of the hepatocytes of the liver. 2 Portosystemic shunting can cause inadequate perfusion to the hepatocytes of the liver, predisposing these patients to lesions within the liver. Portosystemic shunts that do not perfuse the liver are referred to as type 1 or extrahepatic, and shunts that perfuse the liver to some degree are defined as type 2 or intrahepatic. 3 These shunt configurations can be diagnosed prenatally or in the early pediatric stage with sonography. Treatment is essential and varies, depending on the location of the shunt and severity of the patient’s condition. Both types of shunts have been associated with various congenital abnormalities, including heart defects, gastrointestinal anomalies, and vascular anomalies, but never has it been related to any connective tissue disease such as Marfan syndrome. This case study discusses the presentation, treatment, and imaging components on a 38-year-old man with Marfan syndrome who had a congenital absence of a portal vein.
Case Study
A tall, slender, 38-year-old man presented to the emergency department with nausea, vomiting, and abnormal levels of ammonia measured at 134 µmol/L. The normal range for ammonia is between 11 and 51 µmol/L. The patient had a history of mitral valve prolapse, an internal carotid aneurysm, episodes of confusion leading to hepatic encephalopathy dating back to 2009, and Marfan syndrome. A right upper quadrant sonogram, with spectral and color Doppler, was ordered to rule out any liver disease as an explanation for the hyperammonia. There were no prior imaging studies of the abdomen available for comparison. Using a Philips iU-22 (Philips, Reedsville, Pennsylvania) with a 5-1 MHz curved array transducer, the liver measured 13.9 cm in length. The liver’s echogenicity, echotexture, and shape were all normal. Multiple subcentimeter cysts with posterior enhancement were seen throughout the liver. No evidence of masses or nodules was noted. With regard to the vasculature, the inferior vena cava (IVC) was dilated without dilation of the intrahepatic vessels (Figure 1). Gray-scale sonography failed to visualize the typically well-defined and echogenic wall borders of the portal veins in both the right and left lobes (Figures 2 and 3). Color Doppler did not demonstrate the portal vein within the porta hepatis region. The porta hepatis consisted of a single large hepatic artery and a common bile duct. In the transverse imaging plane, the portal triad revealed a dilated IVC, posterior to the common bile duct, and a prominent hepatic artery (Figure 4). When spectral Doppler was assessed at the porta hepatis, an antegrade arterial waveform was obtained with a resistive index (RI) tracing of 0.64 (Figure 5). This is considered a normal low RI as expected in the hepatic artery, perfusing the liver. In general, low-resistant hepatic arteries normally have an RI of 0.55 to 0.7. No portal venous waveform was captured on this patient. A cavernous transformation, secondary to a chronically occluded portal vein, was not seen. There was little collateral vessel formation and no signs of portal hypertension to support this differential. Portal vein obliteration from tumor invasion was also considered, but there was no evidence of mass effect from a tumor invading the adjacent liver parenchyma. The main portal vein was unable to be visualized with gray-scale sonography or Doppler. No vessel dilation was noted during the examination. The color Doppler scale, or pulse repetition frequency (PRF), was decreased in an attempt to detect slower flow. This adjustment caused aliasing from the hepatic artery, and yet no portal venous flow was documented. Spectral Doppler was placed in other areas within the porta hepatis, as well as the expected right and left portal venous areas, all of which demonstrated an arterial waveform. The splenic vein was not well visualized due to bowel gas. The spleen was not enlarged and a trace of ascites was seen throughout the abdomen.
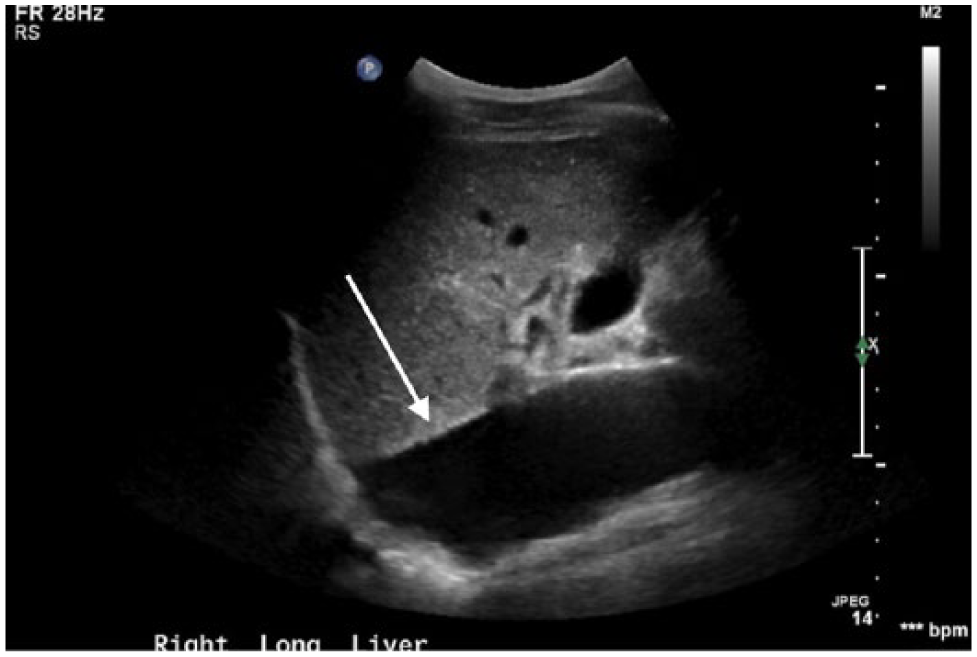
Dilated inferior vena cava without dilation of intrahepatic vessels.
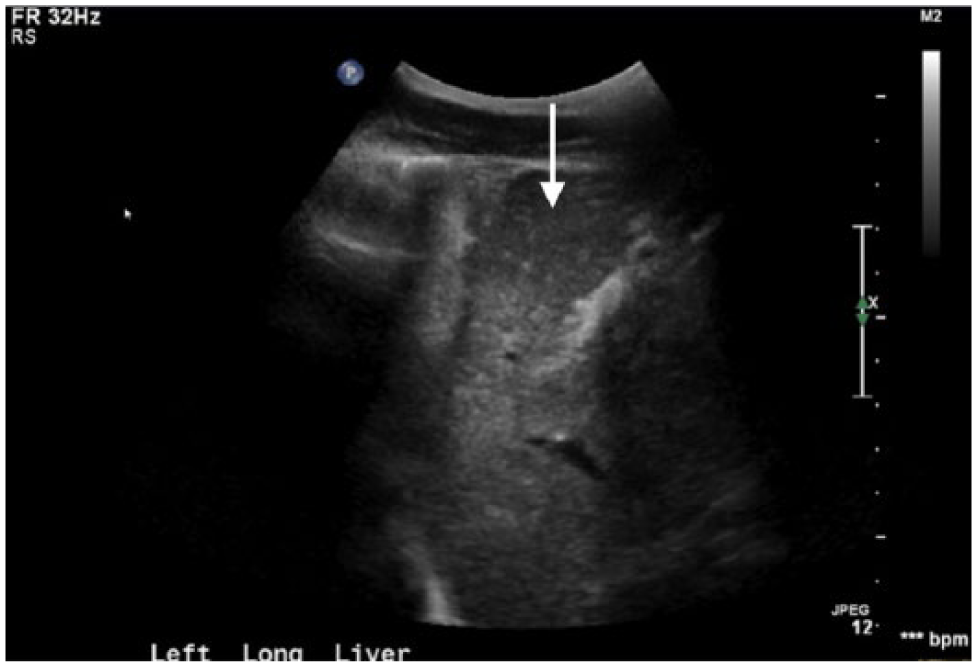
Absent echogenic borders of the left portal vein.
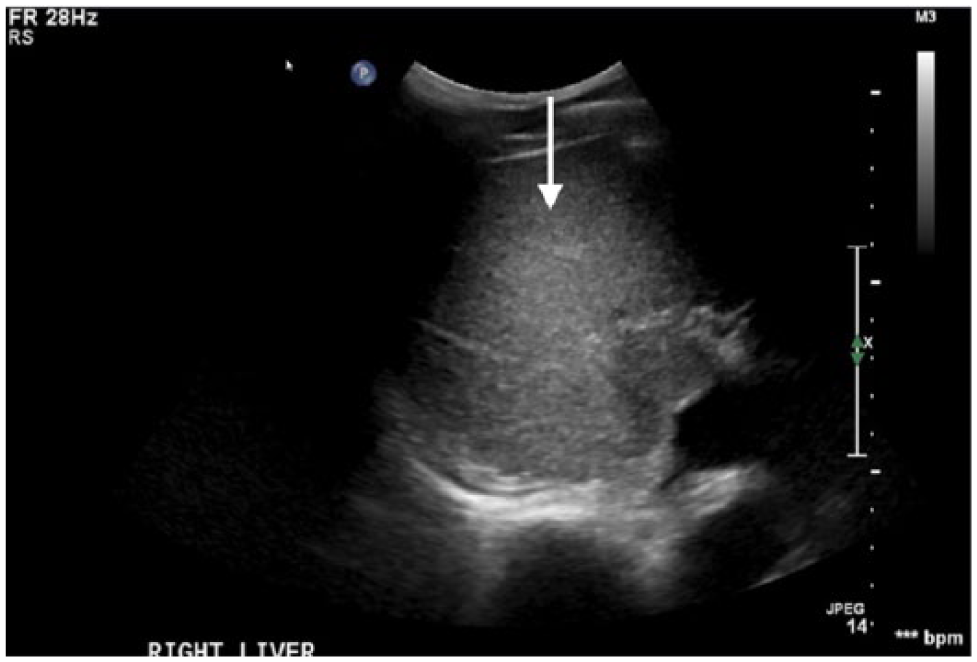
Absent echogenic borders of the right portal vein.
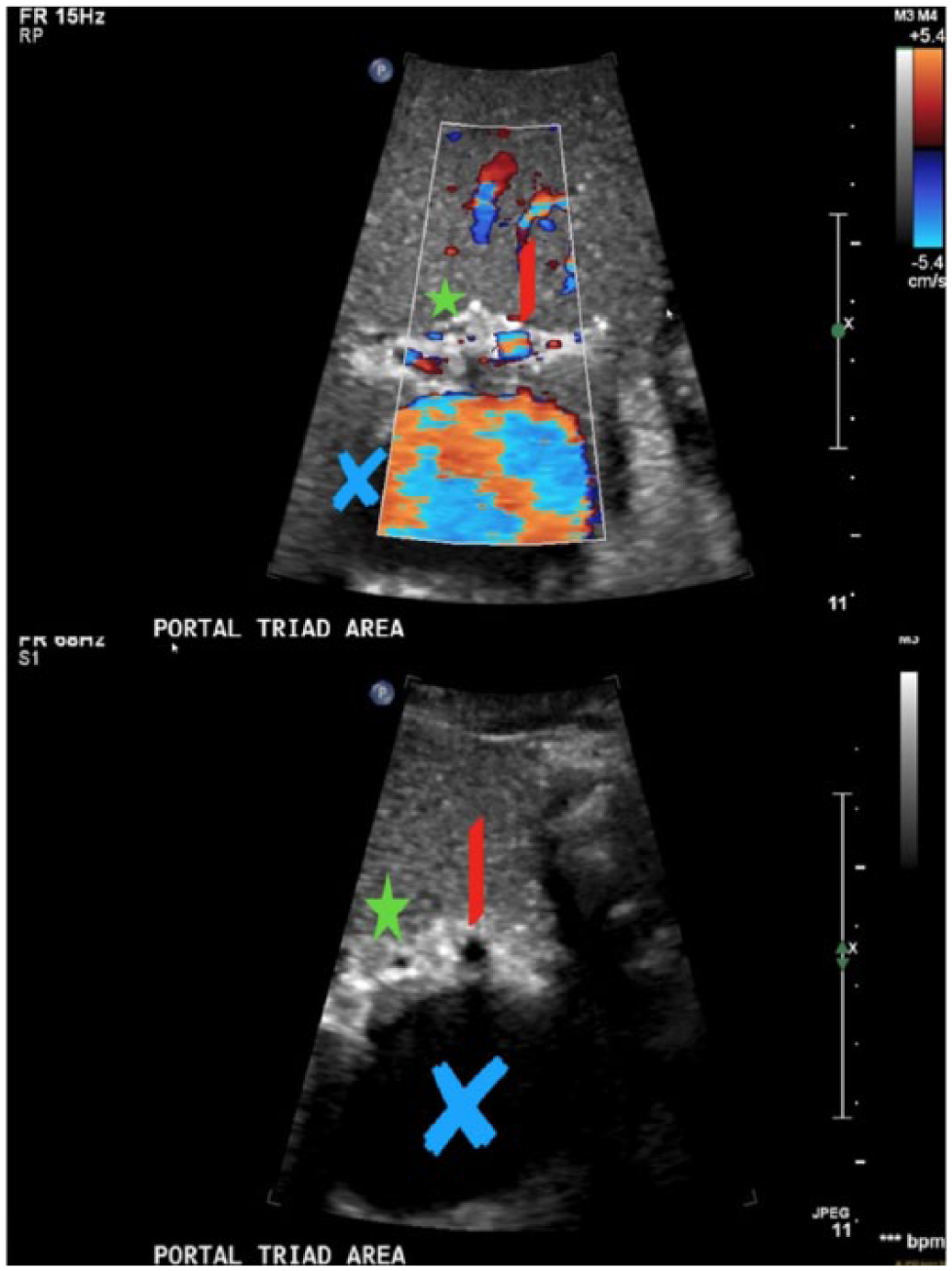
Portal triad with and without color showing a dilated inferior vena cava (blue X), presence of a large hepatic artery (red line), and common bile duct (green star). Absence of the main portal vein.
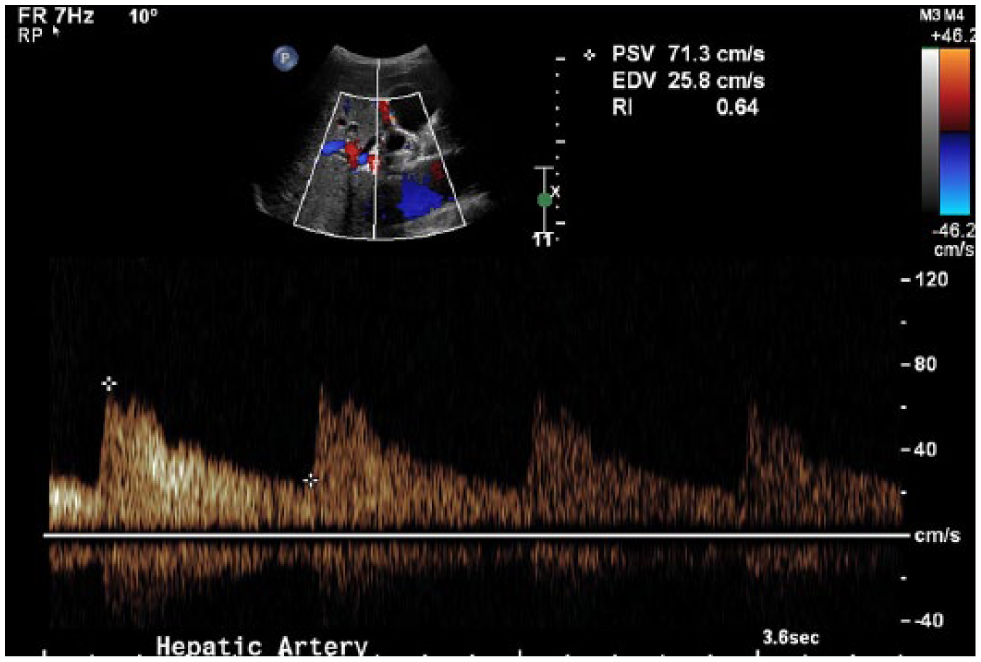
Antegrade hepatic arterial waveform obtained with a resistive index (RI) tracing of 0.64.
The interpreting radiologist reported that the main portal vein was absent. A large hepatic artery and a dilated IVC were suspicious of a shunt elsewhere within the abdomen. Further testing by computed tomography (CT) with contrast was recommended and obtained a day later (Figure 6). The results of the CT confirmed the documented findings from the sonogram. There was hypovascularity of the liver due to the absence of the portal vein and portal system, leaving only the arterial supply to perfuse the liver. The superior mesenteric vein and splenic vein merged to drain directly into the dilated left renal vein. The left renal vein drained into a massively dilated IVC. A visual illustration of the appearance of the abdominal vessels in this particular patient is provided in Figure 7.
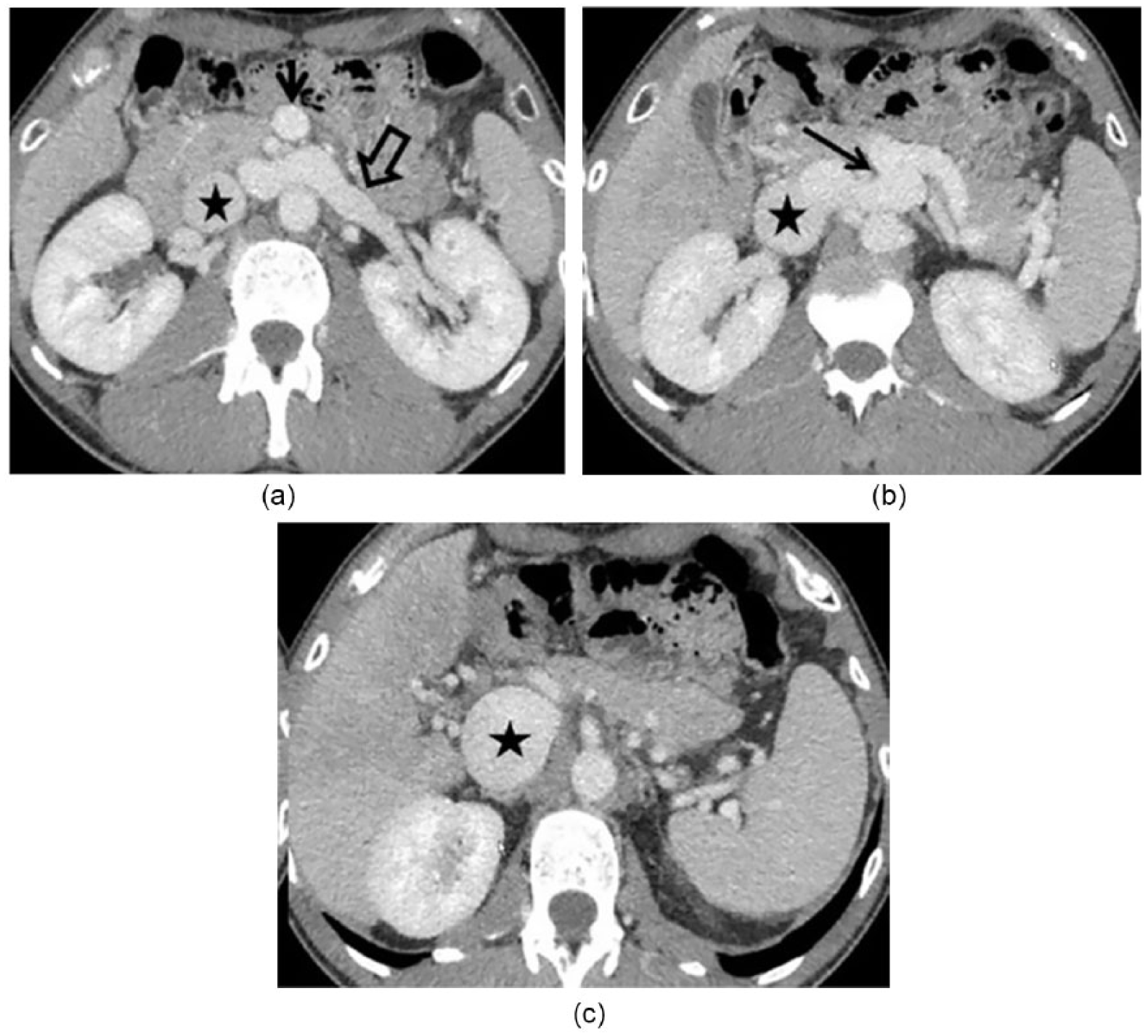
Axial computed tomography images in portal venous phase (A, B, and C) showing dilated left renal artery (block arrow) and inferior vena cava (star) with superior mesenteric artery (SMA; arrow in A) and splenic artery drainage into the left renal vein (arrow in B). The axial image at porta hepatic (C) showing hepatic arterial branches with absent portal vein and massively dilated inferior vena cava.
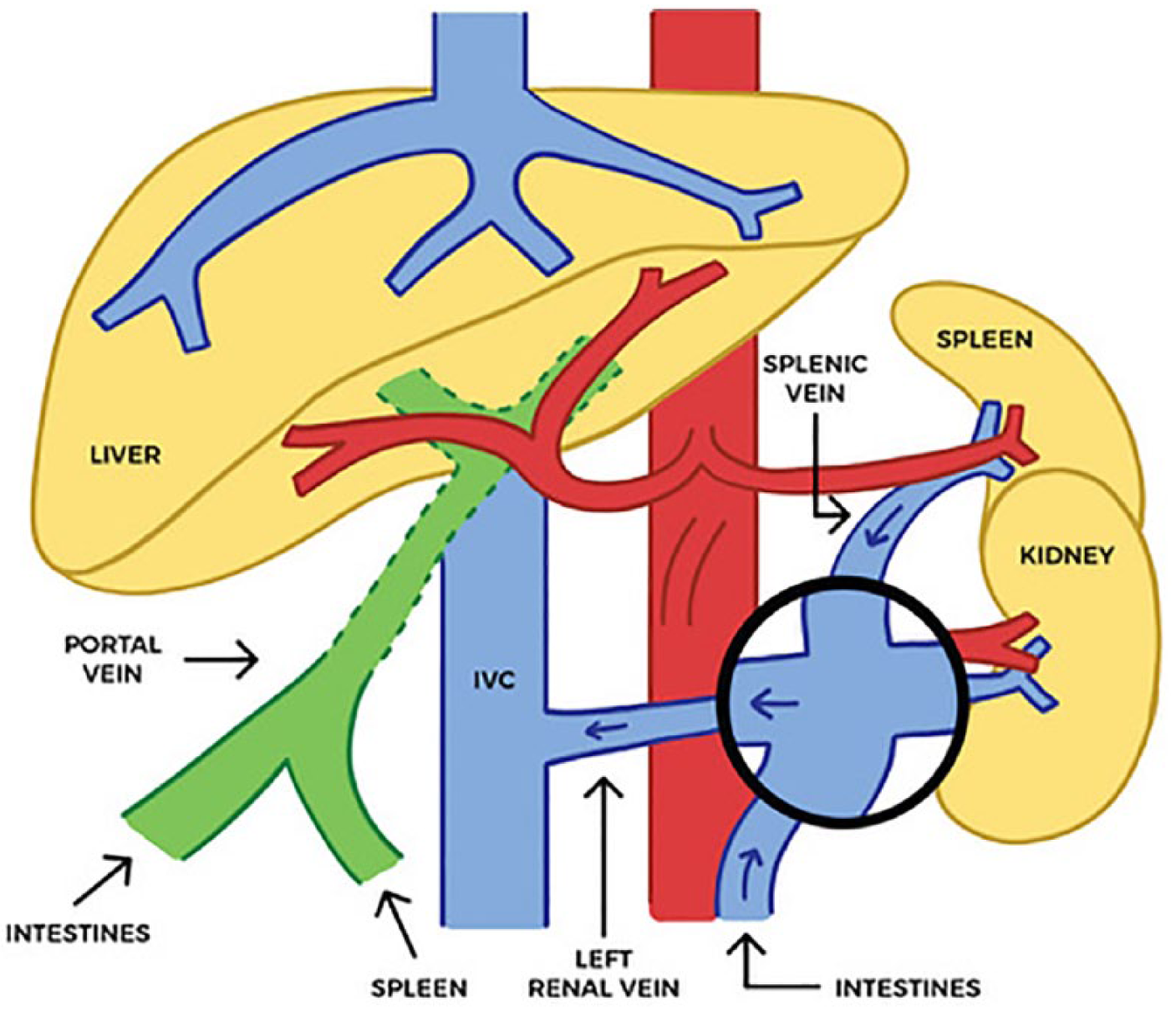
The superior mesenteric vein, coming from the intestines, and splenic vein merged (circle) and drained directly into the dilated left renal vein. The left renal vein drained into a massively dilated inferior vena cava (IVC).
This presentation of the vessels was confirmed and diagnosed as a congenital portal caval shunt type 1b, also known as Abernathy malformation. Upon completion of imaging, the patient was discharged from the hospital.
Eight months later, the patient returned for a transjugular core needle biopsy. Pathology revealed nonspecific portal, patchy lobular inflammation and inconspicuous portal vein branches, consistent with Abernethy malformation. The patient was diagnosed with Abernethy malformation type 1b, discharged, and treated with lactulose. Lactulose aids in the treatment of hepatic encephalopathy by controlling ammonia levels. The patient’s ammonia levels remained elevated one year later at 100 µmol/L.
Discussion
Abernathy malformation occurs when the umbilical and vitelline veins give rise to the portal system but do not correctly form during embryonic period. The portal vein develops embryologically between the 4th and 10th weeks.4,5 By the end of the fourth week, the umbilical and vitelline veins form a connection developing the intrahepatic and extrahepatic portal vein.5,6 Failure to form this critical connection leads to complete or partial absence of the portal system. As a result, mesenteric and splenic venous flow drains into the renal veins, hepatic veins, or directly into the IVC. The close relationship between the vitelline veins and IVC supports this explanation. 7
The umbilical portal venous system (UPVS) has been diagnosed prenatally since the early 1980s with sonography.8,9 In the normal portal system, the splenic vein and superior mesenteric vein join to form the portal confluence, giving rise to the main portal vein. The main portal vein then travels cephalic toward the liver, branching into right and left portal veins. The borders of the portal system are contained within connective tissue, providing the portal vein walls with a bright and echogenic lining. This echogenic wall lining is a reliable way for sonographers to distinguish between portal veins and hepatic veins in varied sonographic planes throughout the liver. 2 Between 70% and 80% of blood is brought to the liver by the portal vein and the remaining 20% from the hepatic artery. 2 The primary function of the portal system is to bring nutrient-rich blood to the liver for preprocessing. Once there, the liver regulates and detoxifies the blood before it enters back into systemic circulation.
Shunts that perfuse the liver to some degree are defined as type 2 or intrahepatic portosystemic shunts. 3 Congenital extrahepatic portosystemic shunts, referred to as type 1, have been reported as rare events. In 2014, a literature review of 101 cases of congenital absence of the portal vein reported 40 cases described as type 1. 10 Classified by Morgan and Superina, 11 type 1 is the absence of the intrahepatic portal veins and is often discovered in females. Type 1 can be subdivided into type 1a and type 1b. Type 1a is present when the superior mesenteric vein and splenic vein drain separately into the IVC. Type 1b is present when the superior mesenteric vein and splenic vein form a common trunk before draining into the IVC12,13 (Figure 8).
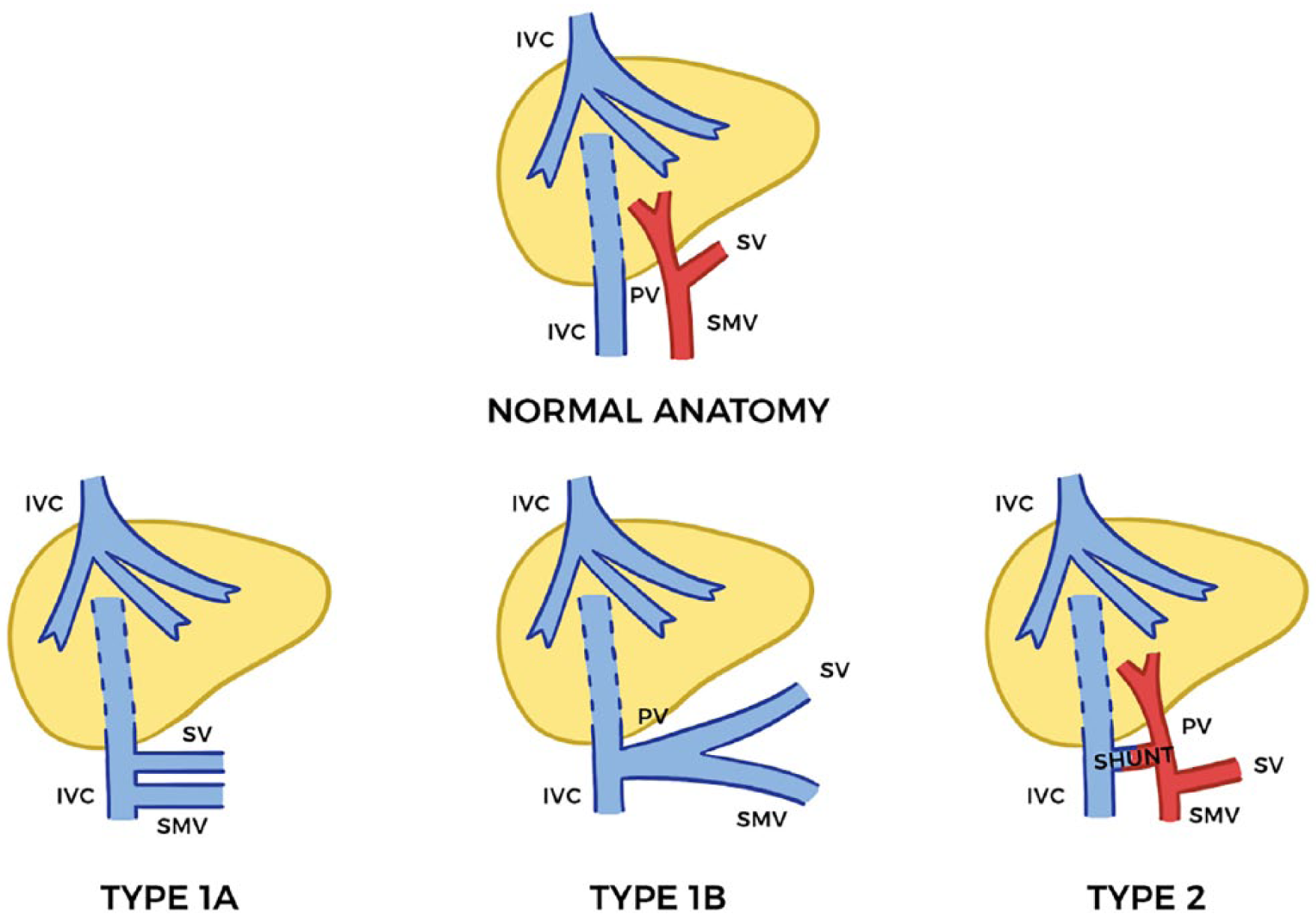
Abernethy malformation shunts: type 1a is present when the superior mesenteric vein (SMV) and splenic vein (SV) drain separately into the inferior vena cava (IVC). Type 1b is present when the SMV and SV form a common trunk before draining into the IVC. Type 2 perfuses the liver to some degree. PV, portal vein.
When the portal vein is absent, the development, function, and regenerative ability of the liver are affected. The liver plays a crucial role in the metabolic processes in the human body. Ammonia is a waste product from the intestines brought to the liver by the main portal vein. Within the liver, ammonia is broken down to urea and released by the kidneys. When the main portal vein is absent, toxic metabolites such as ammonia collected from the gastrointestinal tract have to bypass the liver directly and drain into the systemic circulation. This would lead to hepatic encephalopathy, since the liver is not breaking down ammonia into urea to be removed from the body. 3 Many cardiovascular manifestations as well as noncardiac abnormalities can be caused by the absent portal vein. Some of these malformations include compensatory changes in vessels like the left renal vein, as well as an enlarged and hypertrophic hepatic artery.3,14–17 Alternatively, patients presenting with a single hepatic artery are predisposed to the development of nodular dysplasia, hepatocarcinoma, or other benign and malignant hepatic tumors.3,18–20
Treatment
The type of shunt and its location will determine the proper treatment for the patient. In patients with type 2 malformations, coil embolization or surgical ligation of the shunt may be options.13,21 Usually in patients with type 1 shunts, occlusion of the shunt is not performed, as it is the only drainage route for the mesenteric venous blood, 22 and no surgical reconstruction of the main portal vein can be made of the native liver.23,24 In type 1, liver transplantation is necessary if the patient presents with liver disease or focal nodular hyperplasia. Many surgeons believe a liver transplant is necessary only when medical therapy cannot relieve complications, such as encephalopathy. 23 In the past, liver transplant was the only cure of these patients with type 1 shunts, 25 but other methods such as the use of lactulose have recently been used. Lactulose is an agent effectively used in cases of hyperammonia and hepatic encephalopathy. Lactulose induces remarkable growth of Lactobacillus to produce lactic acids that interfere with the urease-producing microorganisms.3,26,27 The prognosis of patients with CAPV depends on congenital heart disease, liver disease, and the site of portosystemic shunts. The outcome of patients with CAPV with no other abnormalities is different. 10 A long-term follow-up including laboratory tests and image screening is recommended for patients with CAPV. In this case study, a liver transplant was not an option due to the low model for end-stage liver disease (MELD) score; therefore, lactulose medication was used for detoxification. The patient’s ammonia levels continue to remain elevated 2 years later at 102 µmol/L.
Conclusion
Congenital absence of the portal vein is extremely rare and often is an incidental finding. Extrahepatic portosystemic shunts are usually identified early in pediatric populations but could go undetected until later in adulthood. Sonography can aid in the diagnosis of Abernethy malformation by documenting specific sonographic findings. If echogenic borders of the main portal vein are not visualized at the porta hepatis, right lobe, or left lobe, sonographers should document the presence of a potential thrombus or tumor invasion within the main portal. The proper documentation of the absence of the main portal vein gives rise to a potential shunt elsewhere. The location of the shunt must be determined, as this valuable information is crucial for proper treatment. Knowledge and experience of the sonographer are very important, as certain sonographic characteristics need further investigation for the accurate diagnosis of CAPV. A long-term follow-up with either magnetic resonance imaging or CT is recommended for patients with Abernethy malformation.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
