Abstract
Congenital diaphragmatic hernia (CDH) is an opening in the diaphragm in which the abdominal viscera protrudes into the chest as the result of an embryologic defect. CDH can be an isolated anomaly or occur simultaneously with a chromosomal abnormality or genetic syndrome. Prognosis for fetuses with CDH is widely variable and depends on numerous factors including premature birth, the presence of a chromosomal abnormality or genetic disorder, location of the herniation, lung volume, and cardiac involvement. This study presents details of a case of right-sided CDH with an intrathoracic kidney detected sonographically. Fetal echocardiography and magnetic resonance imaging (MRI) were used to further characterize the herniation. Both sonography and MRI aided in accurate diagnosis and postnatal treatment planning.
Keywords
A congenital diaphragmatic hernia (CDH) occurs when the pleuroperitoneal canal fails to close between 8 and 10 weeks of gestation. This sporadic defect results in the herniation of abdominal viscera into the thorax. 1 Displacement or shifting of the mediastinum or evidence of abdominal organs within the thorax are indications for the diagnosis of CDH. The etiology of the malformation is unknown, but it has been reported in association with maternal administration of medications such as thalidomide or antiepileptics. 2 The reported estimated rate of the incidence of CDH varies between 1 in 2000 and 1 in 5000 live births2–4 and has remained consistent throughout the past 2 decades. According to Graham and Devine, 5 the incidence numbers do not account for the CDH cases that end in stillbirth, intrauterine fetal demise, or neonatal death before being assessed at a facility where these data are reported. With the advancement of modern medicine, many cases are detected prenatally and diagnosed with the assistance of sonography and other noninvasive imaging modalities, such as magnetic resonance imaging (MRI).
There are three primary types of CDHs: Bochdalek, Morgagni, and hiatal. Defects occurring through the foramen of Bochdalek are located posteriorly and laterally and more commonly occur on the left side. Herniations located anteriorly and medially, through the foramen of Morgagni, are termed anterior Morgagni hernias. Hiatal hernias occur when herniation of the abdominal organs, most commonly the stomach, protrude through a physiological but overly lax esophageal hiatus into the thoracic cavity. 6
Case Study
The patient, a 33-year-old Caucasian woman, had a history of a singleton pregnancy that concluded with an uncomplicated delivery of a viable male at 40 weeks of gestation. With the second pregnancy, the patient was referred to the high-risk obstetric clinic for a complete sonographic evaluation at 33 weeks, 5 days after an initial inconclusive, abnormal sonogram with her primary obstetrician. The complete, targeted sonographic assessment of the fetus at the high-risk clinic focused specifically on the abdominal and thoracic cavities, as those were the areas of concern after the initial ultrasound. The gender of the fetus had previously been confirmed as female.
A complete sonogram was performed with a GE Voluson E10 scanning system, using a C1-5-D transducer. The fetus was in a cephalic presentation. Fetal movements were visualized, the fetal heart rate was 161 beats per minute (BPM), the placenta was identified on the posterior portion of the uterus and showed no evidence of previa, the umbilical cord was composed of three vessels, and the amniotic fluid was within normal limits with a maximum vertical pocket measurement of 6.3 cm. Fetal biometry confirmed correct gestational age. The estimated fetal weight was 2278 g.
Evaluation of maternal structures determined that the uterus appeared normal, and there was no presence of adnexal masses. Examination of fetal anatomy determined that all structures appeared normal, except for the palate, aortic arch, ductal arch, and right renal artery, which were not well visualized, and structural abnormalities were seen with the left kidney, right kidney, right lung, and the fetal heart. The left kidney was displaced superiorly near the stomach, which was on the left side (Figure 1). Within the thorax, the left lung appeared normal, but it was suspected that a mass measuring 5.4 × 3.2 cm occupied the right lung space (Figure 2). The etiology of this mass was unclear; it did not have the same echogenicity as lung tissue and did not appear to contain any vascular structures (Figure 3). It was suspected that this mass could represent a neoplasm related to the kidney, adrenal gland, or lung. The fetal right kidney, with vascularity, was visualized within the area of the right chest and appeared to be compressing the fetal heart (Figures 4 and 5).
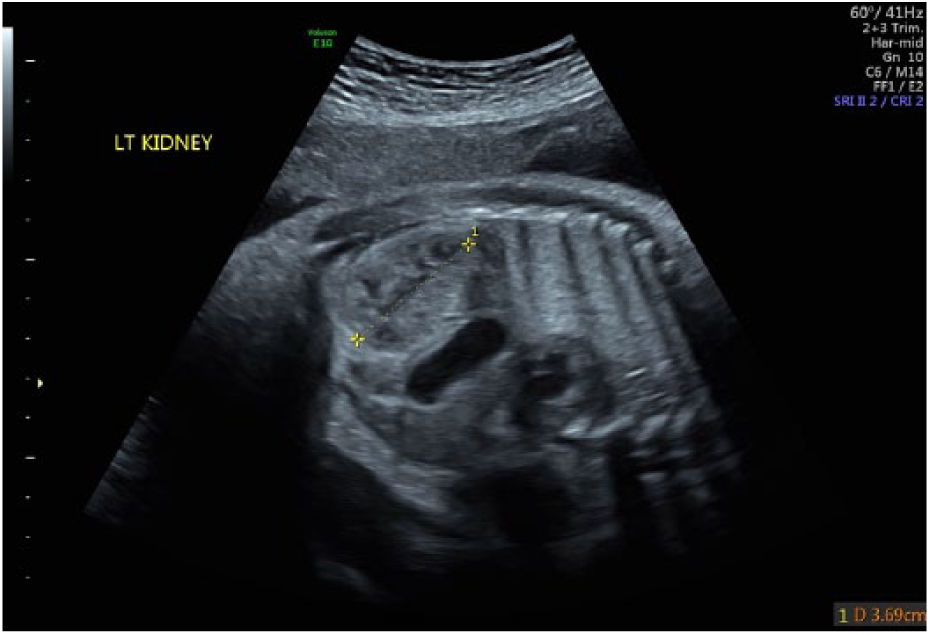
Sagittal view of the superiorly displaced left kidney, located near the stomach.
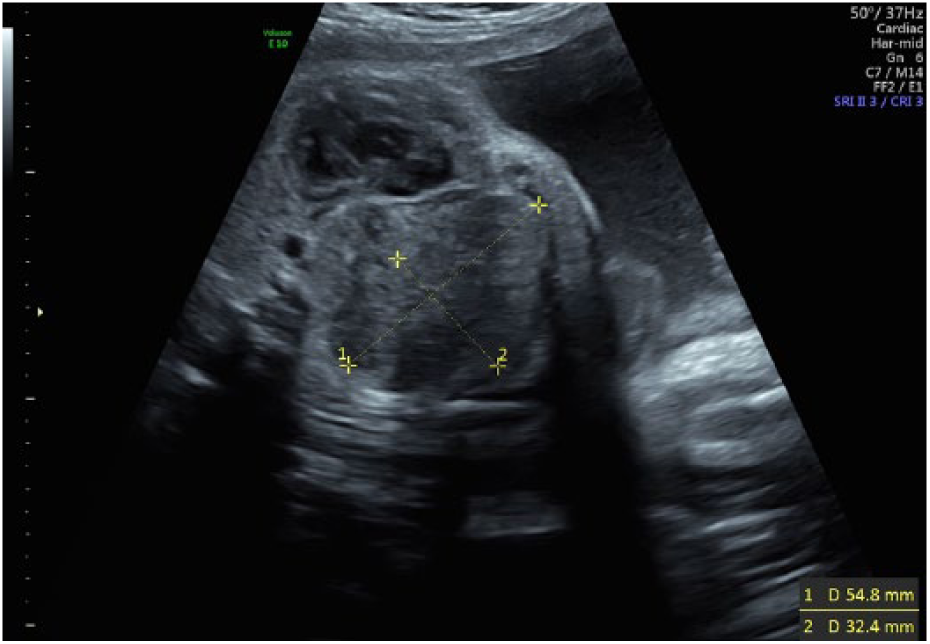
Transverse view of a suspected mass believed to occupy the right lung space.
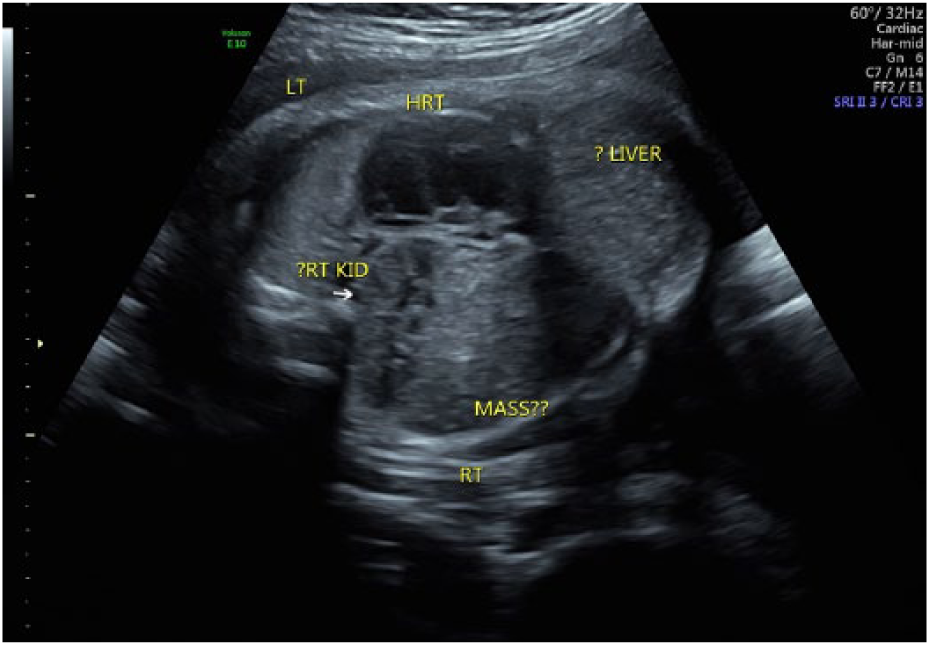
Coronal view of the fetal thoracic cavity demonstrating the presence of a mass.
Coronal view of the fetal thoracic cavity demonstrating intrathoracic kidney and compressed heart.
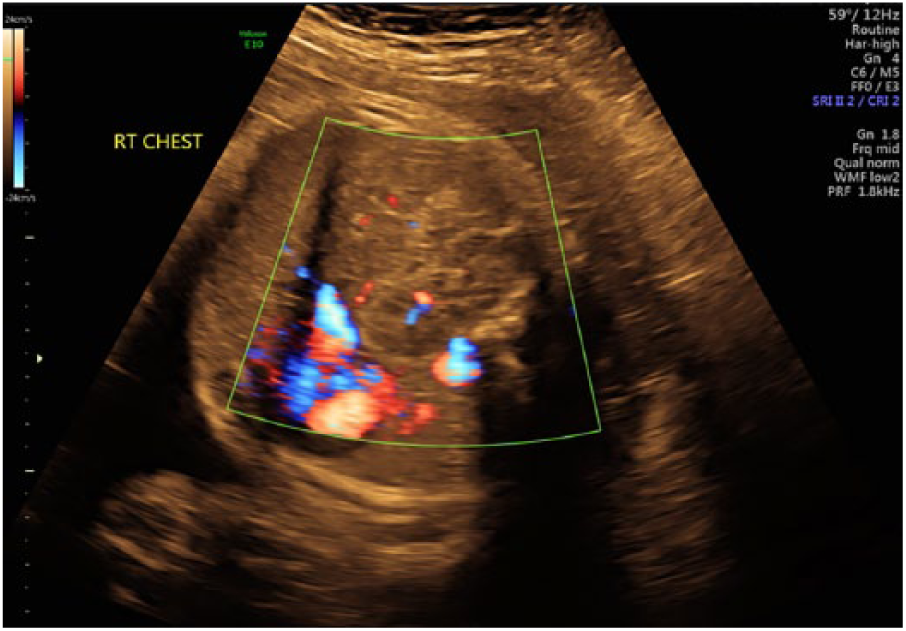
Coronal view of the fetal thoracic cavity with color Doppler demonstrating vascularity within the right kidney.
The liver was seen predominantly midline, appeared to be displaced superiorly and to the left, and was situated partially in the fetal chest. Because of these abnormalities, the diaphragm was not adequately visualized, although the diaphragm was seen in the sagittal plane. It was determined that a right CDH (R-CDH) or a congenital absence of a portion of the diaphragm was possible.
The etiology of the mass in the right side of the chest and the abnormalities were difficult to ascertain with sonography. There was discussion with the patient by the maternal-fetal-medicine physician and genetics counselor about the potential for poor neonatal outcome. A fetal echocardiogram was performed the same day as the initial sonographic examination. This study verified normal conotruncal anatomy, normal left and right ventricular cavity size as well as systolic function, and sinus rhythm with 1:1 atrioventricular conduction. An echocardiogram shortly after birth to assess for pulmonary hypertension was recommended. It was determined that further information from a fetal MRI would be advantageous and was completed at another facility later the same day. Further follow-up and review of the MRI results, as well as a prenatal neonatology consult, was scheduled.
A follow-up evaluation was completed exactly 3 weeks (36 weeks, 5 days) after the initial assessment and included a limited sonogram and discussion of fetal MRI results. A transabdominal sonogram determined a normal amount of amniotic fluid, with a maximum vertical pocket of 7.5 cm, and confirmed the previously documented three-vessel cord and posterior placement of the placenta with no evidence of previa. The fetal heart rate was 143 BPM, and the fetus remained in the cephalic position. Fetal size remained consistent with the previously determined estimated due date. A biophysical profile was completed, assessing qualitative amniotic fluid volume, fetal breathing movements, gross body movements, and fetal tone with an overall score of 8/8.
The fetal MRI that was previously performed confirmed the finding of a large R-CDH containing liver, collapsed small bowel, and the right kidney (Figure 6). The superiorly placed left kidney was noted near the stomach (Figure 7). Observed fetal lung volumes were calculated to be 80%, based on MRI gestational age–based nomograms, and the predicted lung volume was 44%. The estimated liver herniation was approximately 25%, and no evidence of additional fetal anomalies was identified.
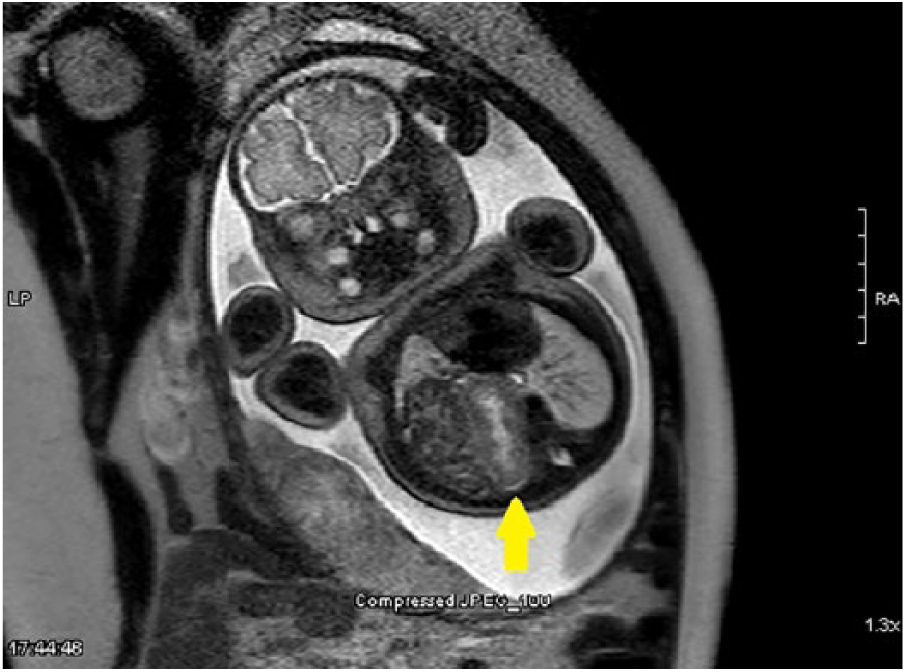
Fetal magnetic resonance imaging confirming the right kidney (yellow arrow) is located in the thoracic cavity, located next to the collapsed bowel.
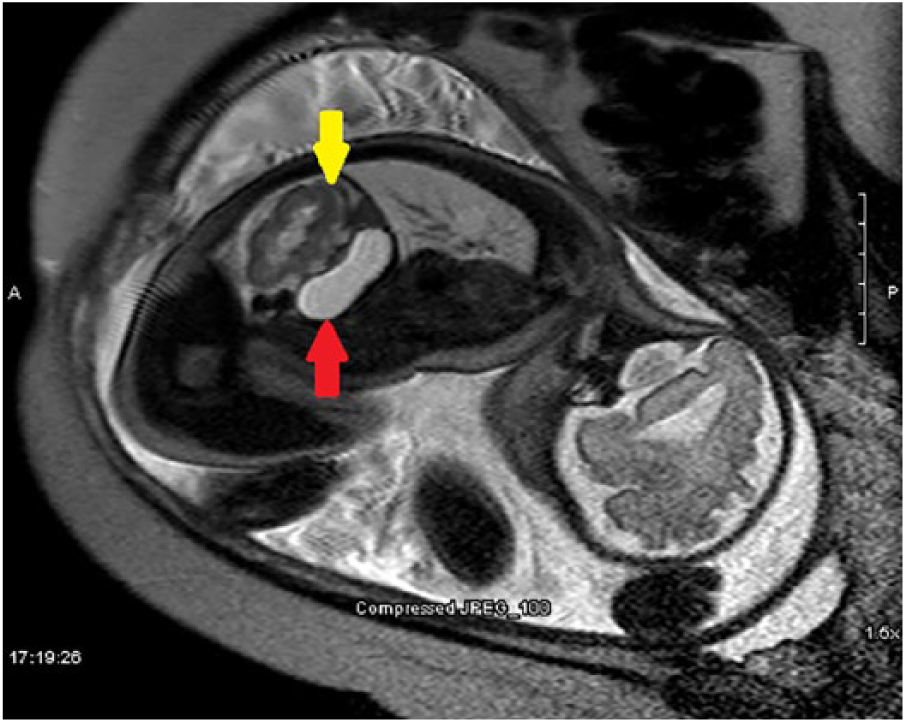
The left kidney (yellow arrow) is superiorly located next to the stomach (red arrow).
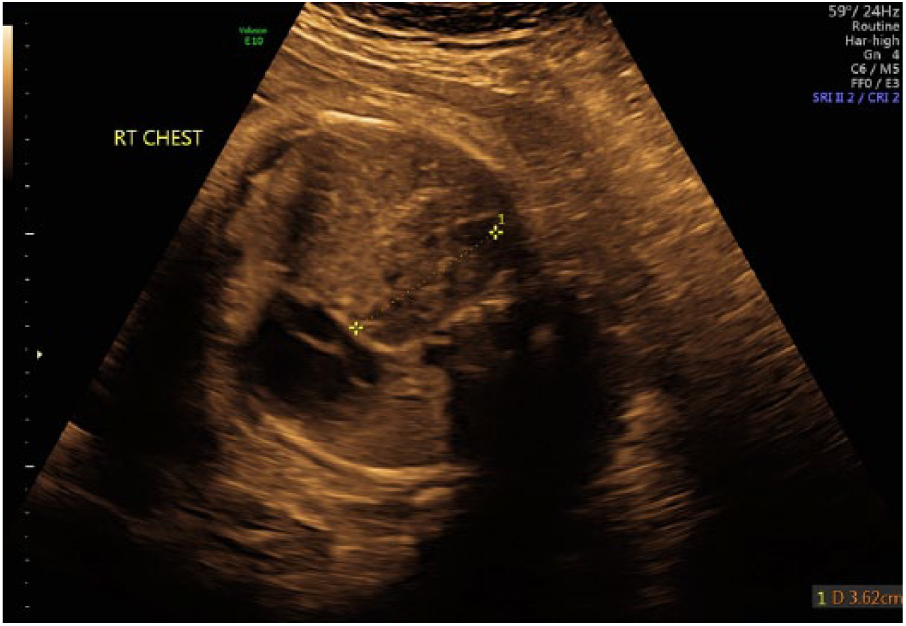
Review of the MRI results allowed the follow-up sonographic evaluation performed at 36 weeks, 5 days to be a more in-depth, targeted examination. With the information provided by the MRI, it was determined that there was no mass present in the right chest. An R-CDH was confirmed and visualized again, with fetal bowel, right kidney, and part of the liver in the chest. At its largest measurement, 4.85 × 3.43 cm of the liver resided in the right chest. color Doppler was used to demonstrate liver vasculature and to confirm its location, as shown in Figures 8 and 9. The right kidney was situated centrally within the chest to the left of the liver and right of the heart. The heart was well visualized in the four-chamber view and appeared symmetrical, although it was displaced to the left. The left and right lungs were compressed, with more severe effects on the right. Exact measurements of the lungs were not achievable with sonography as the surrounding echotextures could not be definitively differentiated. The left kidney, stomach, and bladder remained in the abdomen. The right and left kidneys were symmetrical in length, measuring 3.62 cm and 3.69 cm, respectively (Figures 1 and 4).
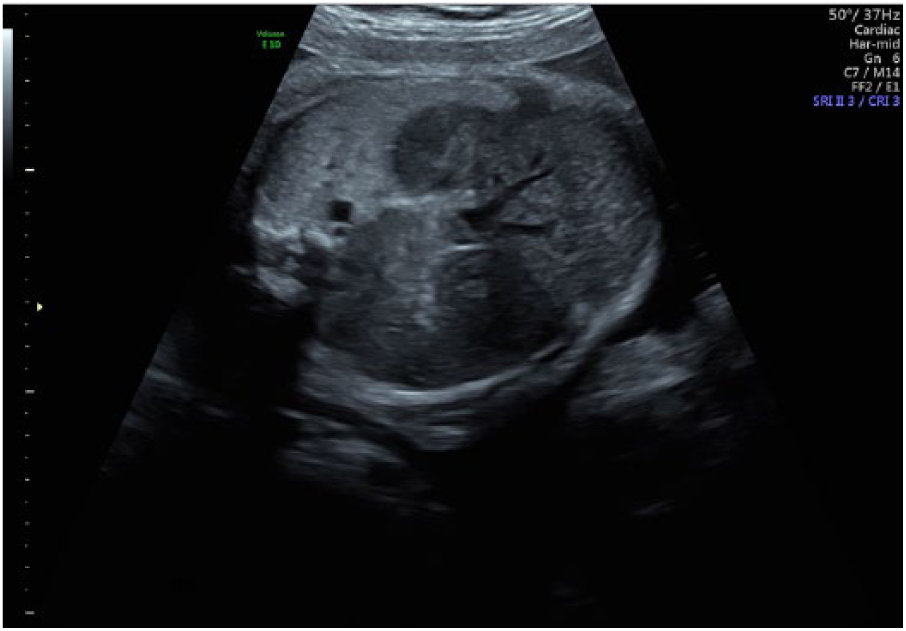
Coronal view of liver vascularity within the fetal thoracic cavity in gray-scale.
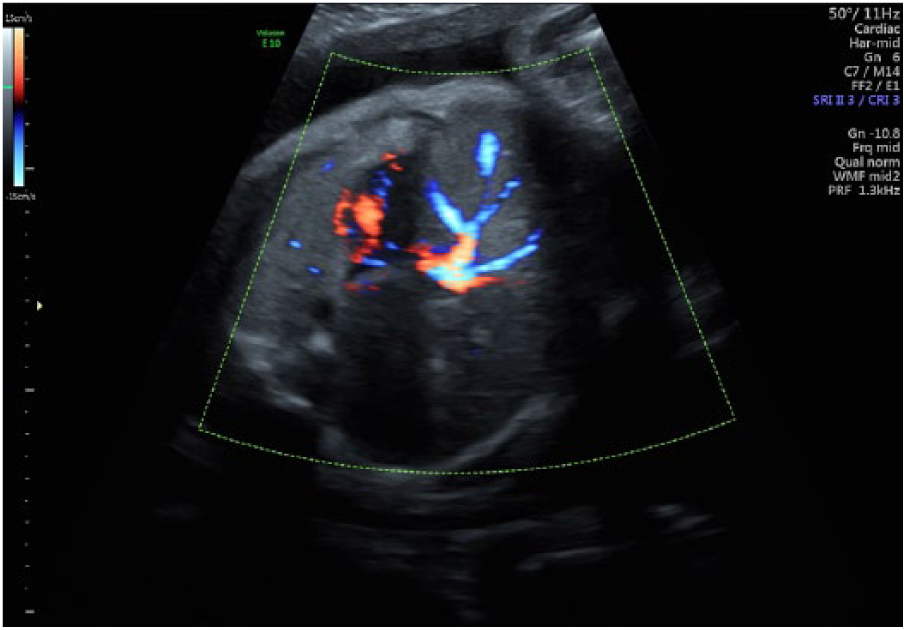
Coronal view of liver vascularity within the fetal thoracic cavity with color Doppler.
At 39 weeks, 3 days of gestation, the patient was admitted for a scheduled cesarean delivery. A viable female newborn was delivered weighing 3583 g, with Apgar scores of 5 and 6 at the first and fifth minutes, respectively. Intubation was completed by 1 minute of life; initial heart rate was approximately 40 BPM and improved after intubation. Shortly after intubation, oxygen saturation improved to the high 90s. No audible heart murmurs were detected, and the infant was pink and well perfused with no dysmorphic features. The abdomen was soft and nondistended. The infant underwent nonemergent corrective surgery with a favorable outcome and was discharged from the hospital 20 days after delivery.
Discussion
Bochdalek hernias were first characterized by Vincent Alexander Bochdalek, an anatomist and professor from the Czech Republic, in 1848. 7 Right-sided Bochdalek lesions are rare (10%–15%) compared with left-sided (85%) lesions. An R-CDH carries a disproportionately high mortality and morbidity according to some authors, while others state a higher survival rate when compared to left CDH (L-CDH).4,8 Schaible et al 8 conducted research comparing survival rates of R-CDH versus L-CDH in a total of 106 patients, and the results revealed that patients with R-CDH had higher survival rates (94%) when compared with the L-CDH group (70%). Researchers have suggested that the higher survival rate in R-CDH patients could simply be attributed to location of anatomy. With L-CDH, there is a greater impact on the fetal heart and its atypical dextroposition, which can lead to hemodynamic instability and impaired cardiac function and development, while R-CDH has a less substantial impact regarding atypical dextropositon. 8 While the small number of R-CDH patients limits the study, it highlights the need for more research comparing the location of the defect and its relation to postnatal outcomes in a larger sample size across multiple facilities.
R-CDH is less accurately identified before birth with sonography alone. The fetal heart is not always drastically displaced with an R-CDH, and it can be challenging to identify what types of tissues reside in the thoracic cavity. One reliable sonographic indicator for identifying liver in the thorax is visualization of intrahepatic vessels crossing the hemidiaphragm. This can be demonstrated with the use of color Doppler in a sonographic examination, as in the case presented (Figures 8 and 9). Differentiation of the fetal lungs and liver is difficult sonographically because they appear homogeneous with moderate echogenicity, but color Doppler can help provide distinction.
Fetal MRI is able to supplement obstetric sonography to better discern unidentified masses or tissues when R-CDH is suspected. Fetal lung volumes and estimation of the percentage of liver protruding through the defect can also be assessed with fetal MRI to help predict postnatal outcomes, whereas sonography is unable to provide that detailed information. 9 In the case of suspected R-CDH diagnoses, fetal MRI can provide a definitive diagnosis and assist the care team with postnatal surgical planning. 9 The presented case demonstrated the effectiveness of MRI, as a supplement to sonography, in confirming the diagnosis of an R-CDH, estimating fetal lung volumes, and assessing the percentage of liver herniation as well as the location and differentiation of other organs that have migrated into the thoracic cavity.
Intrathoracic ectopic kidney accounts for 5% of all renal ectopias; its association with CDH is rare and has been reported to have an incidence of only 0.25%. 10 Further highlighting the rarity of an intrathoracic kidney, research conducted by Masturzo et al 11 assessed 15,919 autopsies of children, and only 1 case of intrathoracic kidney was found, with only a total of 22 ectopic kidney cases discovered. Embryologically, the kidney originates in the pelvis and ascends to interface with the adrenals, at which time their upward migration is halted. The failed cessation of this movement can result in the fetal kidney reaching the thorax before closure of the developing diaphragm. Sonographically, the echotexture of the intrathoracic kidney will be different than that of the fetal lung and liver. 11 Again, color Doppler can be a helpful tool when trying to discern an intrathoracic mass as being fetal kidney or another type of tissue. The structure may appear as an unidentified mass initially, but by using color Doppler, the sonographer can trace the origin of the renal artery from the aorta to the unidentified mass and reveal the mass to be an intrathoracic kidney. While challenging, the sonographer can use this technique to assist in providing a more definitive diagnosis of intrathoracic kidney with sonography.
When combined with CDH, congenital heart defects worsen the prognosis. Proof of this degraded prognosis was confirmed with research conducted by Graziano through the Congenital Diaphragmatic Hernia Study Group (CDHSG) in which 2636 pediatric CDH patients from 82 centers were followed. The survival rate among CDH patients without any heart defect was 70%, whereas those patients with diagnosed heart defects had a significantly lower survival rate of 41.1%. 12 These data were validated by Menon et al., 13 on behalf of the CDHSG; the study found the survival rate of infants born with CDH and no heart defects to be 69%, and for those with a heart defect, the survival rate was 36%. This brings attention to the fact that although medical innovation and technology are constantly improving, little improvement has been made in the past 2 decades in terms of survival rates among those with and without heart defects and CDH. In all CDH cases, the use of fetal echocardiography is essential to assess fetal heart function and structure and to aid in the detection of pulmonary hypertension and concomitant pulmonary hypoplasia, and it plays a vital role in prognosis prediction.
Numerous structural anomalies are associated with CDH and can be divided into seven categories: cardiovascular, gastrointestinal, urogenital, musculoskeletal, respiratory, central nervous system/eye, and craniofacial. While most CDHs are isolated anomalies, chromosomal abnormalities can be associated. The most common include trisomy 12, 18, and 21, Turner syndrome, partial trisomy 5, partial trisomy 20, tetraploidy 21, and tetrasomy 12p. 5 Fetal syndromes such as Apert, Beckwith-Wiedemann, Coffin-Siris, and Pierre Robin can be associated with CDH, among many others. 5
With the assistance of routine obstetric sonographic examinations, approximately 50% of CDHs are diagnosed antenatally. 5 The skill of the sonographer, available technology, and quality of the study are the main factors contributing to missed diagnoses. The presence of abdominal organs within the thoracic cavity is the most dramatic sonographic finding. However, sonographers should be aware of less pronounced indications including polyhydramnios, cardiac axis shift, and mediastinal shift. Detection of an R-CDH is commonly dependent on the recognition of these subtle signs. There is no singular marker, identifiable with any imaging modality, that can accurately predict postnatal prognosis. Timely prenatal intervention allows for patient education and development of a postnatal surgical plan. The study presented demonstrates how early diagnosis is possible through the use of sonography, fetal echocardiography, and MRI.
The differential diagnosis of CDH is a highly involved undertaking that requires a broad spectrum of specialized health care professionals. Prenatal counseling should involve a multidisciplinary team involving maternal-fetal medicine, pediatric surgery, genetics, and neonatology. 5 The long-term outlook for CDH varies considerably and is based on associated anomalies, preterm delivery, hernia position, and fetal lung volume.
Conclusion
R-CDH is considered an uncommon finding that is challenging to detect and difficult to thoroughly characterize antenatally with just sonography. By using color Doppler to follow the renal arteries from their origin on the aorta to a mass in the chest, the sonographer can provide strong evidence to define the mass as a kidney and reduce the number of differential diagnoses. Color Doppler can also be employed by the sonographer to assess for liver herniation into the chest by careful evaluation of the hepatic veins and their location. In cases of suspected CDH, providing detailed information to clinicians as to what organs are protruding through the defect allows for better care management. Both sonography and MRI were employed in the presented case to examine and diagnose an R-CDH with an intrathoracic kidney. Accurate imaging across multiple modalities enabled continued care for the patient and the unborn fetus and provided valuable information for strategic postnatal surgical planning.
Footnotes
Acknowledgements
The authors thank the University of Arkansas for Medical Sciences High-Risk Obstetrical Clinic and Arkansas Children’s Hospital for providing the images for this case study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
