Abstract
Testicular embryonal cell carcinoma is a germ cell tumor that can be poorly differentiated and quite aggressive. It is a fairly common carcinoma of the testis but is uncommonly found coexisting with the rare inflammatory process of granulomatous orchitis. This case study describes a male in his early 30s diagnosed with embryonal carcinoma of the right testis combined with granulomatous orchitis that was initially found with sonography. The main treatment options are an orchiectomy or chemotherapy. The patient in this case was treated with orchiectomy before the cancer could metastasize to distant organs.
Embryonal cell carcinoma is the second most common malignancy of the testis. 1 It often presents as part of a mixed germ cell tumor but can sometimes be found in pure form. 1 Most testicular cancers present as palpable, painful masses that are usually found by the patient or the primary care physician during checkups. Malignant germ cell tumors account for 95% of palpable testicular tumors. 2 Sonography is the first imaging modality ordered when imaging the testes to evaluate pain or a palpable mass. The most characteristic sonographic appearance of embryonal cell carcinoma is the presence of ill-defined, hypoechoic, heterogeneous lesions. Granulomatous orchitis is a rare inflammatory process with unknown etiology and is more typically seen in 40- to 70-year-olds.3,4 There have not been any cases found in the literature of embryonal carcinoma combined with granulomatous orchitis and, while it is unsure if there is a direct relationship between the two, they both exhibit similar characteristics.1,3
Case Report
A Caucasian male in his early 30s presented to the sonography department with a palpable, nonpainful lump on his right testis. He had no prior imaging or relevant medical history and had previously visited his primary care physician with concerns about a lump on his testis. His physician grew concerned after examining the lump and immediately ordered a sonographic evaluation of his testis.
A scrotal sonogram showed a complex, heterogeneous, unifocal lesion located on the right testis. The lesion measured 2.00 × 1.78 × 2.80 cm (Figures 1 and 2), was hypervascular, and demonstrated punctate calcifications throughout (Figures 3 and 4). Following protocol, the left testis was also imaged and the sonographic appearance was within normal limits (Figure 5). The sonographic appearance of the mass on the right testis was suggestive of malignancy, an impression strengthened by the comparison images of the left testis.
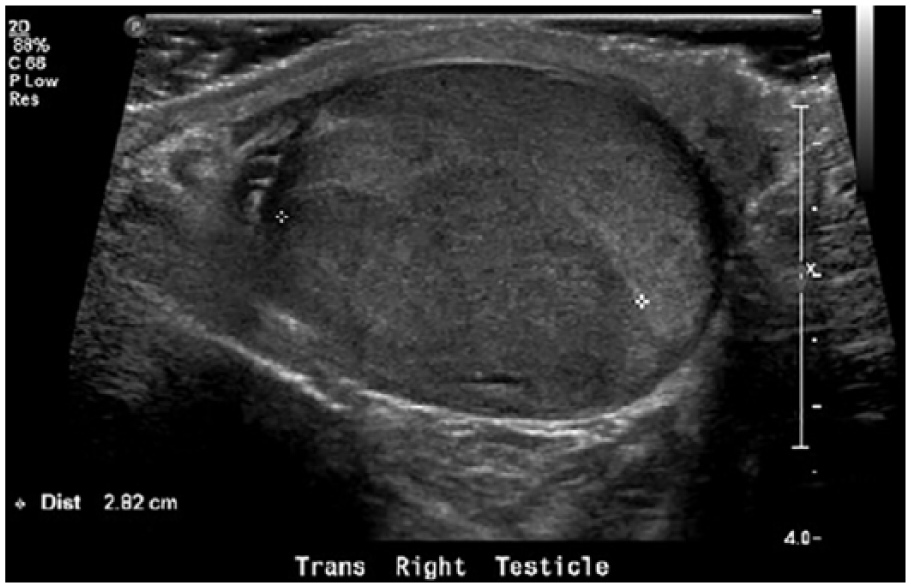
Transverse gray-scale sonogram of the right testis demonstrating a hypoechoic, irregular lesion measuring 2.82 cm in width.
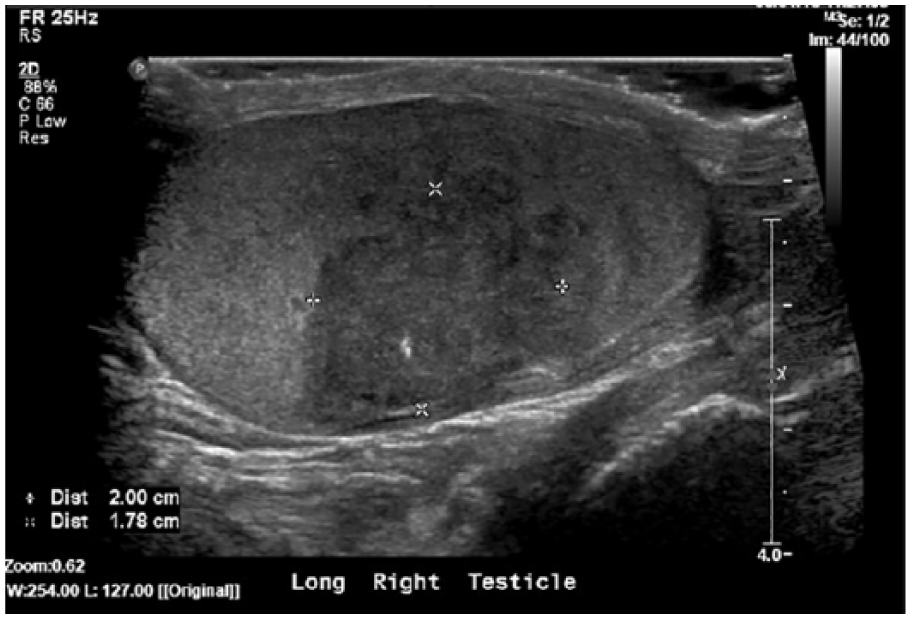
Longitudinal gray-scale sonogram of the right testis demonstrating a hypoechoic, irregular lesion with calcifications measuring 2.00 cm in length and 1.78 cm in height.
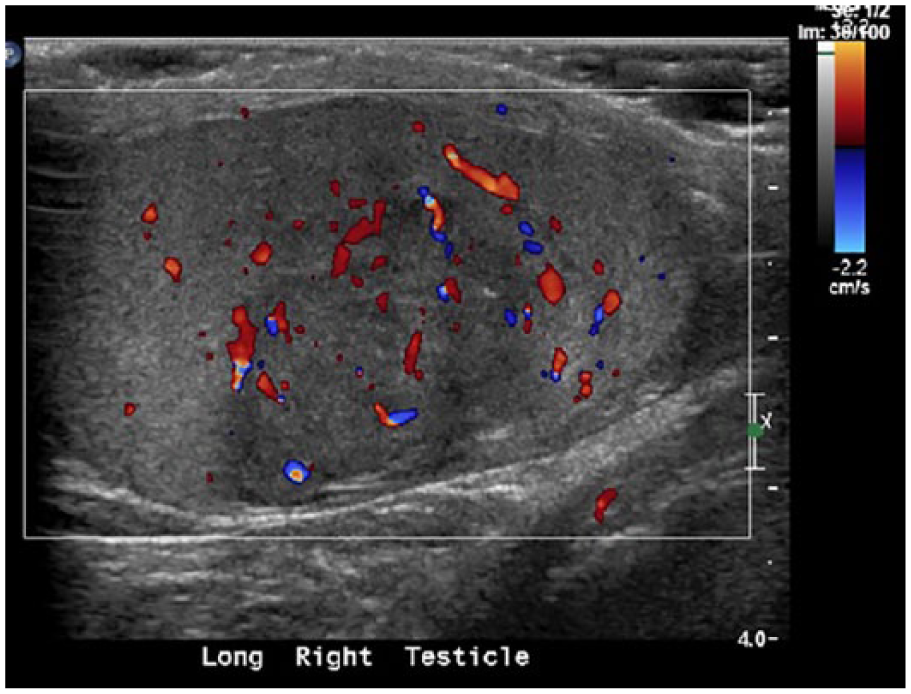
Longitudinal color Doppler sonogram of the right testis demonstrating hypervascular flow within the lesion.
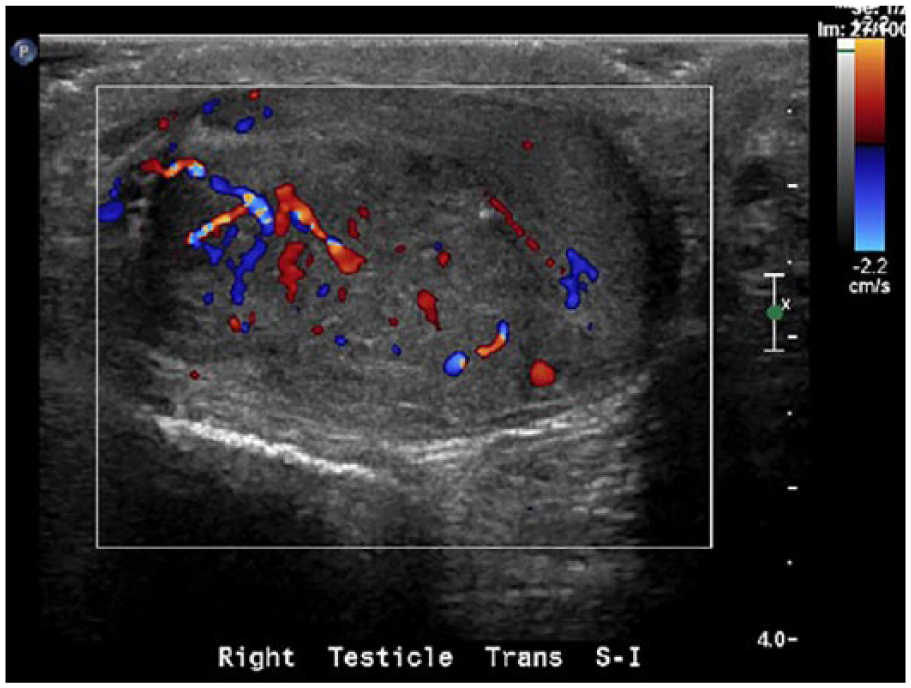
Transverse color Doppler sonogram of the right testis demonstrating hypervascular flow within the hypoechoic lesion.
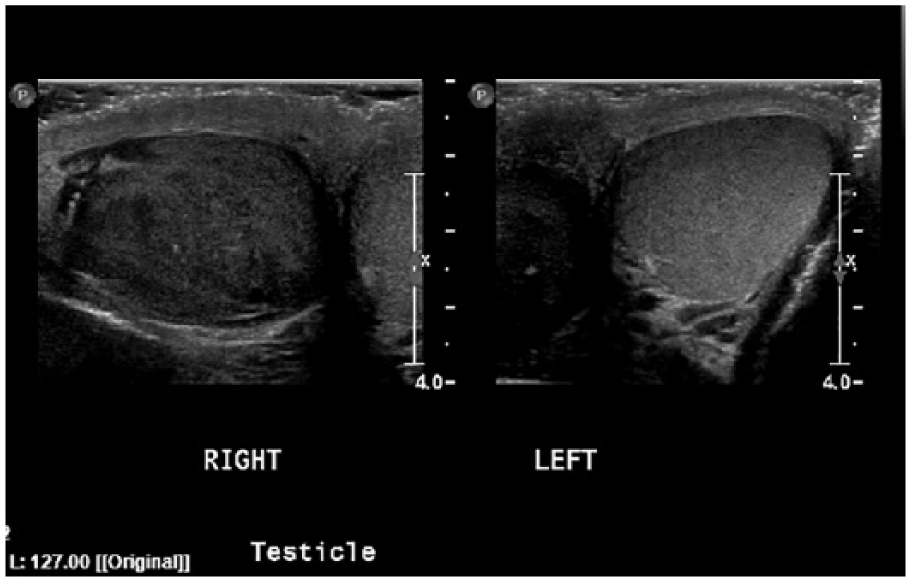
Transverse gray-scale sonogram comparing the size and echotexture of the testes.
Based on the sonographic appearance and clinical presentation, a right radical orchiectomy was performed five days following the sonogram. During surgery, the right testis including the spermatic cord was removed. The mass did not invade the tunica albuginea. Tissue specimens were sent for pathologic evaluation, which determined the mass to be embryonal cell carcinoma combined with granulomatous orchitis. During surgery, immunohistochemistry was performed and showed a positive staining for pankeratin, CD30, and placental alkaline phosphate (PLAP) values. The tests for α-fetoprotein (AFP) and human gonadotropin (hCG) were both negative. Computed tomography (CT) performed after the surgery did not detect any evidence of metastatic involvement with distant organs or lymph nodes.
Discussion
Embryonal carcinoma is a germ cell tumor that usually presents as a mixed cell tumor and is highly aggressive if left untreated. 1 Of the patients who present with a pure form of embryonal carcinoma, 66% will exhibit metastasis at the time of diagnosis.1,5 However, when seen as a mixed neoplasm, it accounts for 40% of testicular cancers and typically presents in the second to third decades of life. 6 Testicular tumors of pure form include seminomas, spermatocystic seminoma, yolk sac tumor, embryonal cell tumor, and a teratoma. When in mixed form, they usually present as mixed embryonal cell carcinoma and teratoma, mixed teratoma and seminoma, and then others. 1
Granulomatous orchitis is a rare inflammatory process that, according to Roy et al., 7 is seen in the presence of granulomatous inflammation with admixed multinucleated giant cells. This process causes damage to the seminiferous tubules. With unknown specific etiology, granulomatous orchitis is thought to be caused by trauma or the cumulative effects of multiple urinary tract infections. Another hypothesized factor is the extravasation of sperm within the testis. It has also been seen as a reaction following chemotherapy in patients with Hodgkin and non-Hodgkin lymphoma. 8 It is seen more commonly in African Americans. 3 When granulomatous orchitis is present, the risk for testicular neoplasm increases. 3
The presence of a unilateral, often tender, palpable testicular mass is a characteristic symptom of embryonal cell carcinoma, as well as many other forms of testicular carcinoma. It is rarely bilateral but can be highly aggressive. 1 Granulomatous orchitis has not been found to be associated with embryonal cell carcinoma, but it will present with the same type of physical symptoms, including swelling and tenderness due to the inflammatory response. A “firm” area can be felt when the inflammatory process has spread. 7 One of the only clinical symptoms of granulomatous orchitis is male infertility, if the orchitis is bilateral. 8 The diagnosis of this process is difficult because clinical and sonographic aspects are mainly inconclusive for the inflammatory process.
While these symptoms are typically the first signs of malignancy, laboratory values are important as well. When embryonal carcinoma is suspected, the laboratory values might show an increase in AFP, hCG, pankeratin, and/or lactic dehydrogenase (LDH). 6 With this malignancy, it is also common for cytokeratin, PLAP, CD30, and OCT4 values to be elevated.9,10 Urine cultures and tumor markers are typically within normal ranges for granulomatous orchitis. 7
While sonography is used to diagnose testicular tumors, CT and magnetic resonance imaging (MRI) are typically used to determine the extent of metastatic involvement. CT is the most common study that is used when assessing the retroperitoneum for metastatic invasion. 11 After reviewing the sensitivity and accuracy of multiple imaging modalities used to assess testicular neoplasms, each were given a rating out of 10. Each modality was also assessed by the expense and examination time for patients. CT has a rating of 9 out of 10 when a CT abdomen and pelvis with intravenous (IV) contrast is used. 11 MRI is also commonly used, but there are limitations due to an overlap between malignant and benign lymph nodes. MRI has a rating of a 7 out of 10 when used with and without contrast. It also has a longer examination time and is more expensive for the patient. 11 MRI and CT both give a more global image and have better resolution of retroperitoneal lymph nodes than sonography. These are some of the reasons why CT was used in this particular case. However, practice guidelines suggest employing nonionizing imaging modalities when possible, particularly in the young group most commonly affected by testicular cancer.
Normally, the risk for malignancy with embryonal carcinoma is high (10%–40%), because it can be quite aggressive. 1 It is typical for testicular tumors to metastasize via the hematogenous or lymphatic route. Most will follow the lymphatic route and travel to regional lymph nodes on the same side where the primary lesion is found. 11 Vascular invasion with this type of malignancy is also common if not detected quickly enough. Embryonal carcinoma has an extremely high rate of lymphovascular invasion and extension into the paratesticular tissue when compared with other malignancies. 1 The most likely sites for metastasis include the lungs, liver, and retroperitoneum. 3 Granulomatous orchitis is an inflammatory process that can spread to surrounding structures, including the epididymis, tunica vascularis, tunica albuginea, and/or tunica vaginalis. 3 The inflammatory process for this case extended to the tunica vascularis, tunica albuginea, and possibly to the tunica vaginalis. There was no evidence of involvement of these structures by the embryonal cell carcinoma.
All types of cancerous processes have a tumor, nodes, and metastasis (TNM) classification that is followed. T describes the extent and the size of the tumor, N indicates lymph node involvement, and M indicates the presence and extent of metastasis. In this case, the embryonal cell carcinoma was classified as pT1b, which indicates that the primary tumor was confined to the right testis and did not extend into any blood vessels or surrounding/distant organs. The b explains that the tumor was found to be 3 cm or larger. 12 Regardless of size, if there was any vascular invasion, the lesion would immediately be classified as a pT2 tumor. 1 The lymph nodes were classified as pNX, meaning that the regional lymph nodes could not be evaluated. 12 However, results of a CT scan after surgery showed no primary lymph node involvement of the tumor.
Treatment of embryonal carcinoma and granulomatous orchitis is similar. It is difficult to diagnose pure granulomatous orchitis without an orchiectomy because its presentation is so similar to that of cancer and, statistically, solid testicular masses are more likely to be malignant than inflammatory. Therefore, the typical treatment is orchiectomy for any suspicious-appearing testicular masses. Past research has suggested embryonal cell carcinoma is radiation resistant, so orchiectomy and chemotherapy are the preferred treatment methods. 13 Typically, if the malignant tumor is unifocal and confined to the testis, orchiectomy is the only treatment that is needed. However, if there is metastasis to other organs or blood vessel involvement, chemotherapy is needed. 2 While orchiectomy may be considered by some as an extreme means of treating granulomatous orchitis, it does reduce the risk of recurrence. Also, the fact that granulomatous orchitis often presents in the presence of carcinoma helps justify that treatment option. Studies have shown that orchiectomy is justified because by the time it is found, the entire testicular tissue will be damaged by this inflammatory process. 7
Provided relapse of the tumor does not occur, most cases of embryonal carcinoma have a good prognosis after orchiectomy. Divrik et al. 14 conducted a study to examine the risk of relapse in stage 1 germ cell tumors. Of the 211 patients monitored longitudinally after an orchiectomy, 66 patients (31.3%) had relapse of the tumor. Recurrence of the tumor occurred within 2 to 32 months after the orchiectomy, with most occurring within the first year after diagnosis. They concluded that, in patients with no vascular involvement prior to the surgery, close surveillance and follow-up should occur but no chemotherapy would be needed. 14 Since tumor recurrence most often occurs within one year of surgery, close monitoring is recommended for these patients. Most recurrences of the tumor occur in the retroperitoneum, with a small amount occurring in the lungs. 15
In a study conducted by Kollmannsberger et al., 16 223 patients were examined following a diagnosis of nonseminomatous testicular cancer, with 49% having embryonal cell carcinoma. Following an orchiectomy, the patients were observed frequently in the first year. They were visited every four weeks in the first six months and then every six weeks for the first year. They were less frequently followed in the years after. Relapse occurred in 59 patients in a median time of just four months. In conclusion, 74% of patients required no other intervention other than a close follow-up following an orchiectomy. 16 When monitoring patients, it is important for physicians to understand the statistics of recurrence that occurs with embryonal cell carcinoma, so any tumor relapse can be detected early enough for treatment.
On imaging studies, granulomatous orchitis and embryonal cell carcinoma often have a similar appearance. Sonography is currently one of the first imaging modalities considered when detecting and diagnosing testicular malignancies and inflammation. It has a 92% to 98% sensitivity and a 95% to 99.8% specificity when detecting testicular malignancies. 15 Sonography is also used to distinguish between intratesticular and extratesticular lesions. Intratesticular lesions are commonly found to be malignant, while extratesticular lesions are commonly found benign. 15 Typically, the sonographic characteristics of embryonal cell carcinoma are those considered suggestive of any malignancy, including ill-defined, irregular margins; hypoechoic, heterogeneous echo texture; and hypervascularity. Cystic components are found within 20% of embryonal cell carcinomas. 4 Granulomatous orchitis will be imaged as a hypoechoic lesion with cystic components and poorly defined intratesticular zones, very similar to embryonal carcinoma.3,7 This overlapping sonographic appearance of multiple pathologies highlights the importance of a thorough clinical history and laboratory values when evaluating sonographic results.
While gray-scale and Doppler sonography are the main imaging techniques used in the detection of testicular lesions, there are other common imaging modalities used. Sonoelastography is becoming more popular when imaging testicular lesions. This method allows for a more specific look at assessing these lesions to be benign or malignant. This sonographic technique uses the assessment of tissue elasticity that helps characterize certain types of cancers. 17 Calculation of tissue elasticity is accomplished by displaying colors for tissue stiffness. Typically, red demonstrates a soft lesion/tissue while blue demonstrates a hard lesion/tissue. A blue, or hard lesion, is more suggestive of malignancy. 17 The goal of sonoelastography in the assessment of testicular masses is to prevent orchiectomy when possible for lesions that appear malignant on sonography but are benign following removal. Following the results of a study conducted by Aigner et al., 17 sonoelastography successfully detected 34 hard lesions and 13 soft lesions. Of the 34, three were classified as hard lesions that ended up being a cyst, scar tissue, and an infarction. However, based on medical history examination and gray-scale sonography, surgery was not performed on any of these three patients. Of the 13 lesions that were soft, six were orchitis, four were infarction, and three were cysts. 17 The study concluded that sonoelastography successfully helps differentiate between tumorous and nontumorous lesions of the testicle. 17 If future research supports this conclusion, sonoelastography may become a routine method of evaluating testicular masses in order to avoid unnecessary surgeries. In the current case, the tumor was proven to be malignant, so it would be expected to have been classified as “hard” if sonoelastography had been available.
Conclusion
Embryonal cell carcinoma is one of the most common testicular malignancies. Testicular carcinomas most commonly occur in 20- to 30-year-old men. 1 Some common symptoms of embryonal cell carcinoma include swelling and a painful, palpable mass located on the testis. Granulomatous orchitis is an uncommon inflammatory process in this age range. In the rare cases that it does occur, it can accompany some form of malignancy. When chronic, this inflammation process presents similar symptoms and sonographic characteristics to embryonal cell carcinoma. A variety of imaging modalities can assess each of these processes, but sonography is the first imaging modality that will be ordered. This case demonstrates the importance of clinical history and laboratory values to help to narrow the differential diagnosis so that effective treatment can be pursued in a timely manner.
Footnotes
Acknowledgements
The authors thank Alli Egart, RDMS, RVT, Emily Luckenbill, RDMS, RVT, RDCS, Melissa Hirner, RDMS, RVT, Nicole Hoff, RDMS, RVT, and Kimberly Grandstaff, RDMS, RVT.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
