Abstract
Ultrasound imaging continues to break through scientific and engineering ceilings that have formerly restricted the type and quality of information available. Limited by the temporal, data acquisition, and processing constraints inherent in traditional beamforming technology, ultrasound systems did not have the capacity to acquire and process large amounts of raw acoustic data fast enough to move beyond standard imaging modalities. While traditional beamforming capabilities can provide high-quality and high-resolution images, sensitive Doppler modes, and other advanced imaging applications, there have been limitations to creating new applications that use the information contained within the received acoustic data set. This has all changed with the introduction of ultrasound imaging systems that acquire and process significantly more acoustic data quickly. Upgraded, state-of-the-art digital signal processing (DSP) capabilities have made new imaging possibilities, including enhanced B-mode tissue characterization. This modality helps to differentiate areas within a region of interest based on the unique acoustic characteristics of the tissues insonated.
Keywords
Traditional Limitations
The acoustic data set is the complete range of echo information received by the transducer after the transmit beam interacts with the human soft tissue and returns to the transducer face. There is an enormous amount of returning data that, in the past, could not be acquired or transferred to computer memory fast enough to take advantage of the totality of the information contained therein. Computer chips simply did not have the high-speed, high-capacity capabilities requisite to do the work.
Once the data were acquired, they were sent to a receiver. The long-established functions of a receiver (i.e., compensation, compression, demodulation, and rejection) resulted in only a fraction of the digital data outputting to the image creation device (scan converter) compared to what was actually received.1,2 This “integrate-and-dump” method, while still permitting high-quality sonographic images, obviated the possibility of more advanced software analysis of information stored in on-board computer memory. Memory capabilities were also limited by the capacity of the hardware storage components available in the past.
Finally, the receiver would send the truncated data set to a scan converter that would build each image frame line-by-line. There are significant limitations to this method of image creation. The amount of time required to create each line was dependent on a number of practical clinical factors (i.e., imaging depth, number of focal zones employed, and the use of additional modalities such as color and/or pulsed Doppler). Increasing depth requires more “listen time” along each line of site, increasing the number of focal zones requires transmitting additional pulses along each line of site, and addition of Doppler modalities similarly requires more pulses per line, all of which increase frame creation time. Increasing individual frame time proportionally reduces frame rate. Therefore, the result was a noticeable and significant trade-off between frame rate, image quality, and the simultaneous use of additional modalities.3,4
Emerging Technology
A number of advances in digital signal processing and graphics display capabilities in the past decade have virtually eliminated these limitations. It begins with an alternative to the line-by-line transmit and receive cycles and the line-by-line creation of each frame. Using “virtual beamforming,” multiple transducer crystals can be fired simultaneously either in segments or across the entire array to create various-sized plane waves (Figure 1). The returning acoustic data are also received from these insonated zones and sent directly to computer memory, bypassing a receiver. The evolution of direct memory access capabilities enables large amounts of digital data to be stored and transferred directly to a device (i.e., the image former, without passing through the central processing unit). The use of traditional scan converters can also be eliminated, as each frame can be created pixel-by-pixel using software algorithms. As more of the complete acoustic data set is sent directly to channel domain memory (computer memory), there are more possibilities for DSP and the creation of advanced imaging modalities. Another advantage to these methods is the maintenance of a consistent and acceptable frame rate regardless of number of focal zones or modalities employed and a reduction in the amount of ultrasound energy required to generate an image. 5
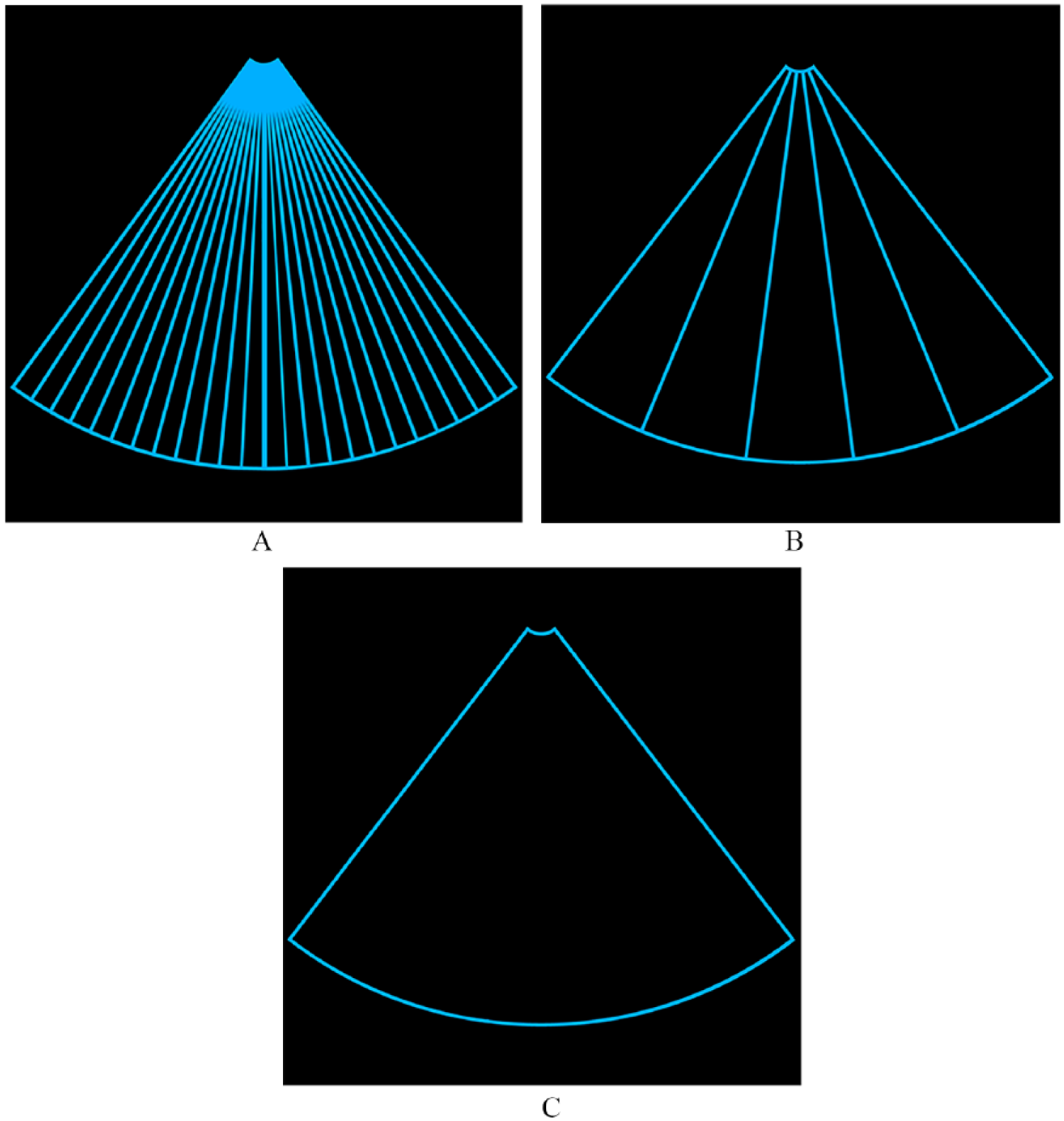
Schematic illustration of traditional beamforming with individual transmit and receive lines of sight, typically one line per transducer element (A). Virtual beamforming creating segmental transmit and receive zones by firing multiple transducer crystals simultaneously (B) or a full-array transmit and receive plane wave (C).
Enhanced B-mode Tissue Characterization
One of the advanced imaging modalities emerging from this new ultrasound imaging paradigm is enhanced B-mode tissue characterization (EBTC). Long the holy grail of sonographic diagnosis, tissue characterization offers the potential for delineating more specifically pathological versus normal tissue types. 6 Defining the characteristics of atherosclerotic lesions7,8 and breast lesions, 9 as well as classifying diffuse liver disease,10,11 has been investigated over the decades by employing a variety of research methods with different approaches and parameters of acoustic science. None have been particularly successful from a clinically practical way. The limitation has always been, at least in routine clinical practice, the loss of a significant portion of the returning acoustic data set due to the receiver “integrate-and-dump” design. There simply were not enough data available, nor were there advanced computational capabilities that made a more robust analysis and processing of the digital data possible.
EBTC capitalizes on the retention of approximately 90% of the returning acoustic data set and the high-speed, high-capacity DSP capabilities of virtual beam systems. This evolving application analyzes the acoustic characteristics of selected signals produced by various types of tissues within a user-defined region of interest (ROI) and segments them in different computer caches according to these tissue characteristics. The acoustic characteristics analyzed exceed the traditional frequency, phase, and amplitude parameters to include attenuation coefficient, sound speed, elasticity, and others. Various filtering and processing algorithms are then applied to each cache of signals to achieve optimal spatial and contrast resolution for each tissue type (Figure 2). The resulting image enhancement within the ROI is the result of tissue differences, not B-mode postprocessing (dynamic range, B-map, edge enhancement, etc.), although these methods may be factored into the analysis and display of each cache of signals. The results are striking and, while reliability and validation of results still need to be established, EBTC presents a first step in moving toward differentiating normal from pathological tissues using a standard ultrasound imaging system in day-to-day clinical practice.
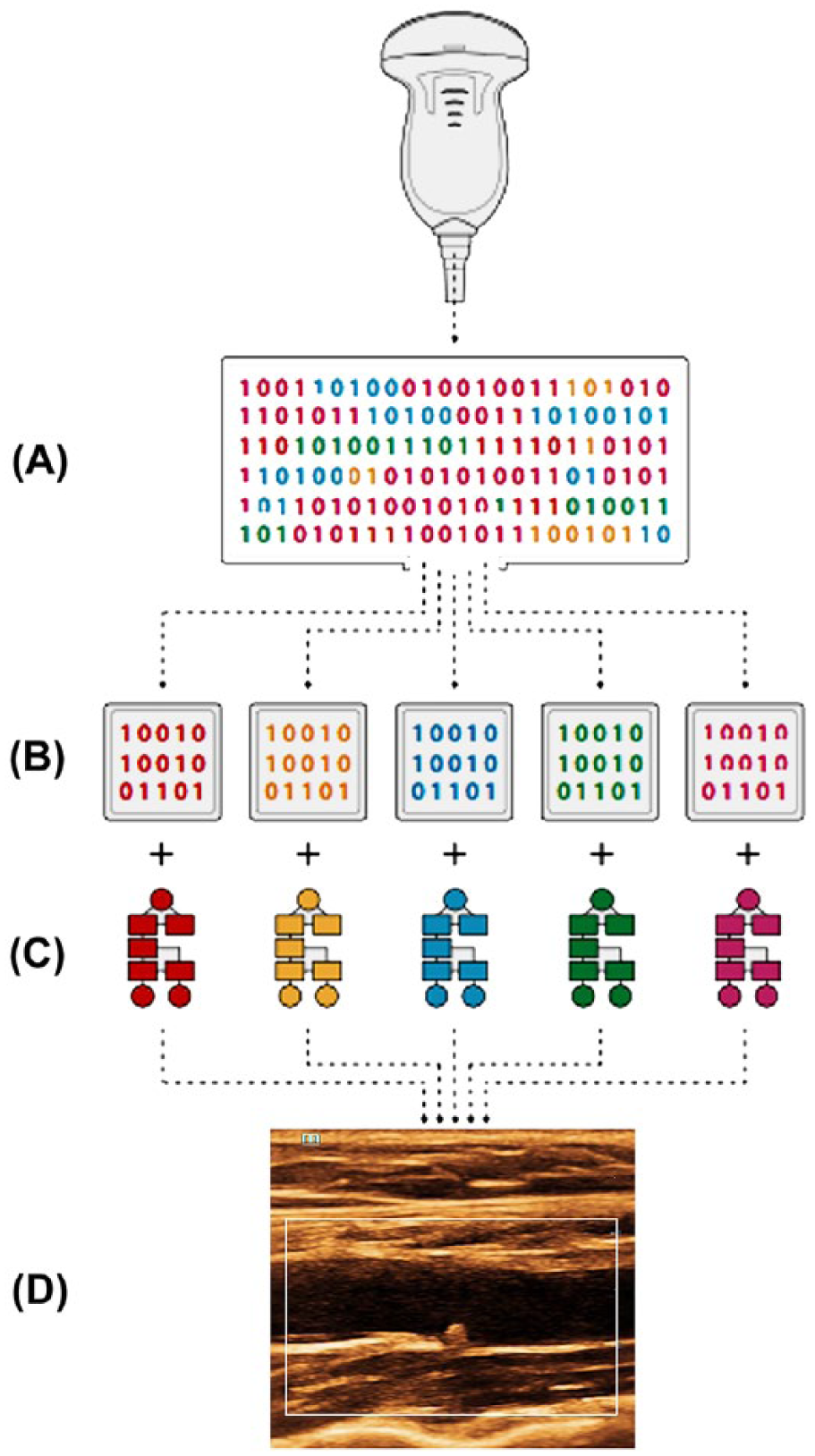
Schematic illustration of enhanced B-mode tissue characterization. The complete acoustic data set is received by the transducer (A). The data set is segmented based on acoustic characteristics of different tissue types and sent to separate computer caches (B). Unique filtering and processing algorithms are applied to each cache (C). The composite subframes are integrated to form a single frame with optimal spatial and contrast resolution based on differing tissue characteristics.
Clinical Examples
As B-mode tissue enhancement is an evolving technology and has only recently become available to the wide pantheon of sonography users, there is scant correlative imaging, surgical, or histological information currently available with identified lesions and abnormalities. More work needs completed before this new modality can move into the realm of “virtual histology,” but the potential is enormous. However, four clinical examples are included to demonstrate the imaging potential and capabilities of EBTC (Figures 3–6).
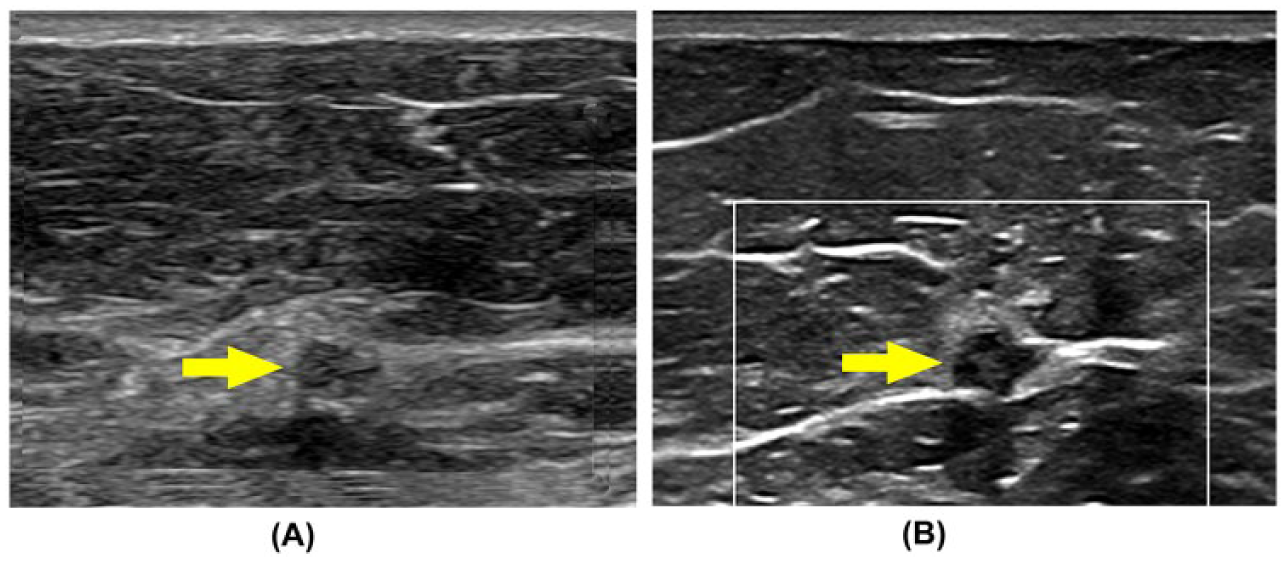
Patient with a nonpalpable breast lesion identified on routine mammography. (A) Indistinct hypoechoic, heterogeneous focal lesion seen deep to mammary tissue without enhanced B-mode tissue characterization (EBTC) (arrow). With EBTC enabled, the internal architecture and borders of the lesion are more clearly visualized, including the presence of a spiculation (beaking) along its lateral margin (B).
Patient referred to ultrasound with elevated liver function values. (A) Zoomed image without enhanced B-mode tissue characterization (EBTC) reveals a hypoechoic lesion in the posterior aspect of the right lobe (arrow). With EBTC engaged (B), there is improved resolution of internal architecture and borders of the same lesion (arrow) and the identification of a second lesion not immediately identified previously (arrowhead). Other B-mode image parameters were not adjusted between acquisition.
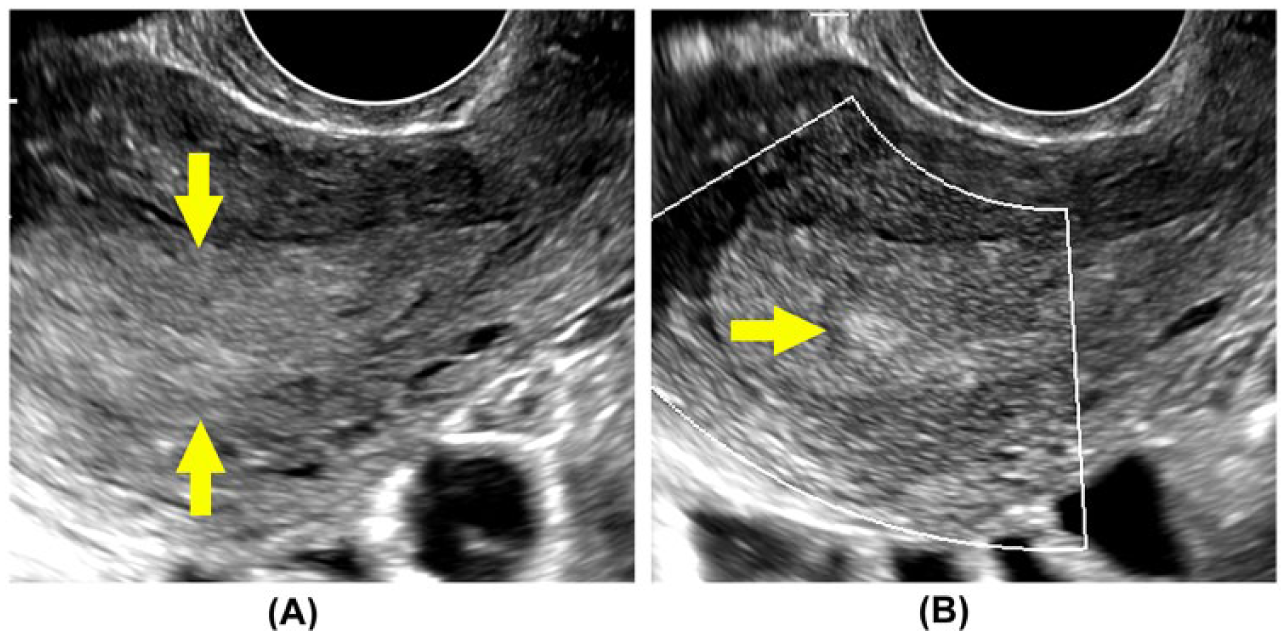
Patient with hysteroscopically diagnosed uterine polyp. (A) Endovaginal imaging through the uterus demonstrates a diffusely enlarged secretory endometrium without apparent focal lesion. With enhanced B-mode tissue characterization engaged (B), a discrete, hyperechoic lesion is identified in the midportion of the endometrium (arrow). Subsequent surgical excision and histological evaluation confirmed a benign endometrial polyp.
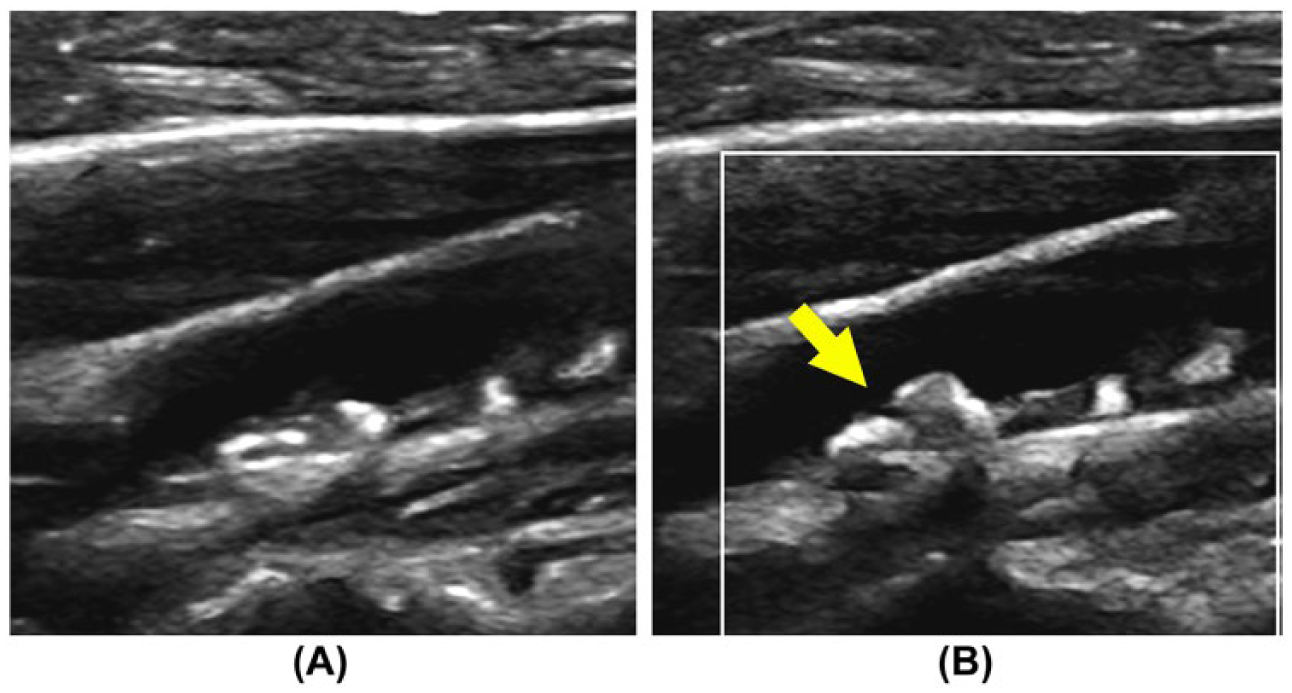
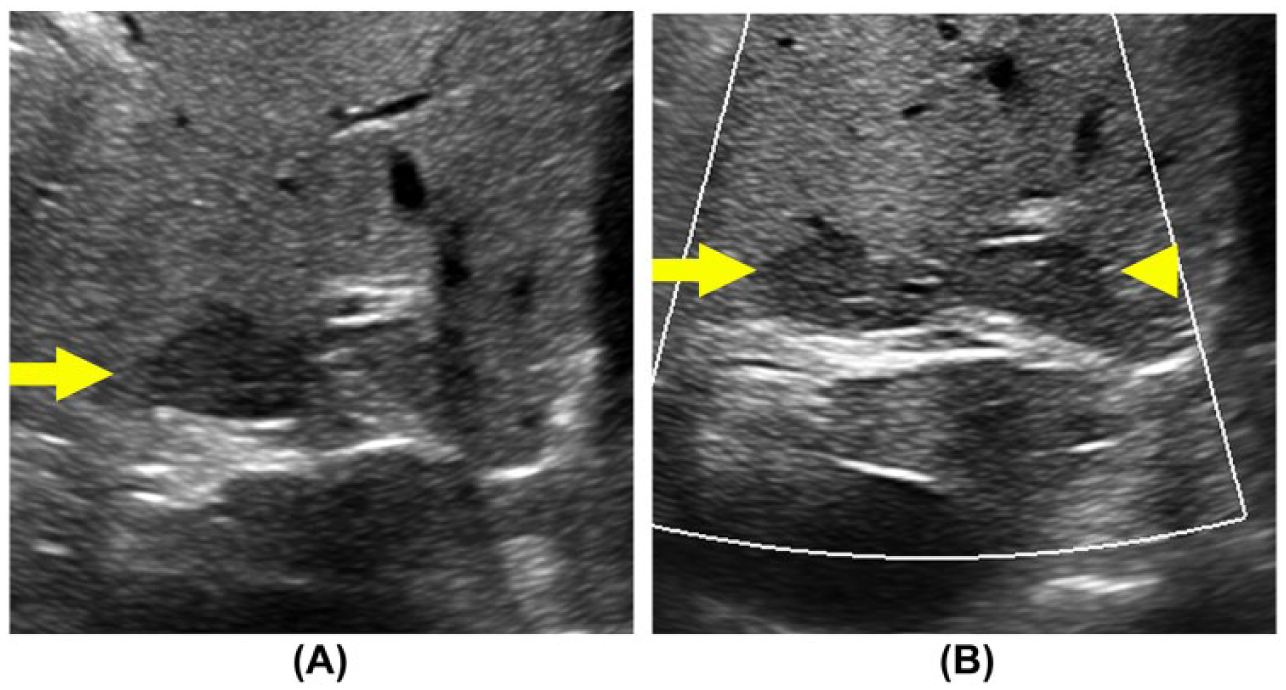
Improved visualization of arterial atherosclerotic lesions. Patient with a single episode of unilateral visual disturbance referred for carotid ultrasound. (A) Routine B-mode evaluation of the carotid bifurcation demonstrates heterogeneous atherosclerotic lesions in the proximal internal carotid artery. Pulsed Doppler waveforms were consistent with a 16% to 49% stenosis. With enhanced B-mode tissue characterization engaged (B), enhanced morphological characteristics of the lesion are demonstrated, including a possible surface disruption (arrow). Imaging, surgical, and histological confirmation is not available in this case.
Summary
While there has been significant evolution of traditional beamforming ultrasound systems over the past several decades, recent developments and advances in core imaging technologies, computational capabilities, digital signal transfer and processing, and graphics display capacity have created exciting new applications. EBTC, currently available on some commercially available imaging platforms, offers the potential for moving forward into a new realm of diagnostic possibilities, such as ultrasound tissue characterization. While large, prospective clinical studies need to be executed in a number of clinical specialties, the future of a new ultrasound capability may be at hand.
Footnotes
Acknowledgements
The author would like to extend his appreciation to Mr. Kendall Dea, senior corporate communications specialist, Mindray Innovation Center, for his contribution of the original illustrations contained herein.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
