Abstract
Bakri balloons are minimally invasive devices placed in the uterus and inflated with saline to apply extrinsic pressure to stop postpartum hemorrhage. Sonography is an ideal imaging modality to confirm position of a Bakri balloon because it is quick and precise in locating the device, does not use ionizing radiation, and is inexpensive. This case report describes the sonographic documentation and management of a malpositioned Bakri balloon.
Introduction
Maternal postpartum hemorrhage (PPH) is the leading cause of maternal mortality, with 80% of these deaths being from a delay in treating uterine atony, the suboptimal contraction of the myometrium following placental separation.1–3 The blood vessels supplying the placenta during pregnancy are severed when the placenta separates from the uterine wall and the bleeding resulting from these severed vessels usually stops when the uterus contracts, compressing the vessels. However, in uterine atony, the uterus does not contract enough and the bleeding continues.2,4 Currently, the Bakri balloon is one of two balloon products specifically designed and approved by the US Food and Drug Administration (FDA) for the control of PPH.5,6 An important consideration is that, to be effective, the balloon must be positioned correctly within the uterus to provide the desired hemostasis. When placed correctly in the uterus and filled with fluid (typically saline), the Bakri balloon has an 80% to 100% success rate in stopping postpartum hemorrhage.3–5,7,8 The balloon is able to control PPH by exerting significant pressure against the uterine wall, which slows or stops persistent capillary and venous bleeding. 9 Use of the Bakri balloon to stop PPH in cases of uterine atony avoids the need for more invasive interventions such as uterine artery ligation, uterine compression sutures, uterine artery embolization, or hysterectomy.
Case Report
A 25-year-old woman, gravida 3, para 2, presented with severe postpartum hemorrhage after an otherwise uneventful vaginal delivery. The patient was given 1000 mg of the synthetic prostaglandin E1 misoprostol to stop the bleeding. However, bleeding continued over the next 3 hours, and the decision was made to consider placement of a Bakri balloon. The patient was placed supine on the operating room table and her epidural catheter, which was still in place from her delivery, was used to provide an adequate dose for anesthesia. She was then placed in lithotomy position, the cervix was grasped with a ring forceps, and a large curette was passed through the cervix. The fundus was manually held in place with an upper hand while curettage was performed. Following curettage, the patient continued to bleed, although at a slower rate. At this point, the decision was made to place a Bakri balloon.
The balloon was placed through the dilated cervix as high in the uterus as possible and was inflated with saline. The initial attempt to inflate the balloon resulted in the balloon being pushed back down through the cervix, despite attempting to hold it manually. A second attempt was made and the balloon was retained with 360 mL of saline. It was then secured in place with vaginal packing. There was a small amount of bleeding through the Bakri balloon but no active hemorrhage. After Bakri balloon placement, the doctor palpated the patient’s uterus to be above the appropriate level, so sonography was ordered to check the position of the Bakri balloon. A transabdominal sonogram was performed using a Philips iU22 xMatrix (Philips Ultrasound, Bothel, Washington, USA) with a 5-MHz curved array transducer. Transabdominal images showed a postpartum uterus with an echogenic endometrium without endometrial fluid or sonographic evidence of retained products of conception (Figure 1). A malpositioned fluid-filled Bakri balloon was seen spanning the lower uterine segment and endocervical canal (Figure 2). At that time, it was noted that the patient’s bleeding had stopped, and the decision was made to remove the Bakri balloon instead of trying to reposition it.
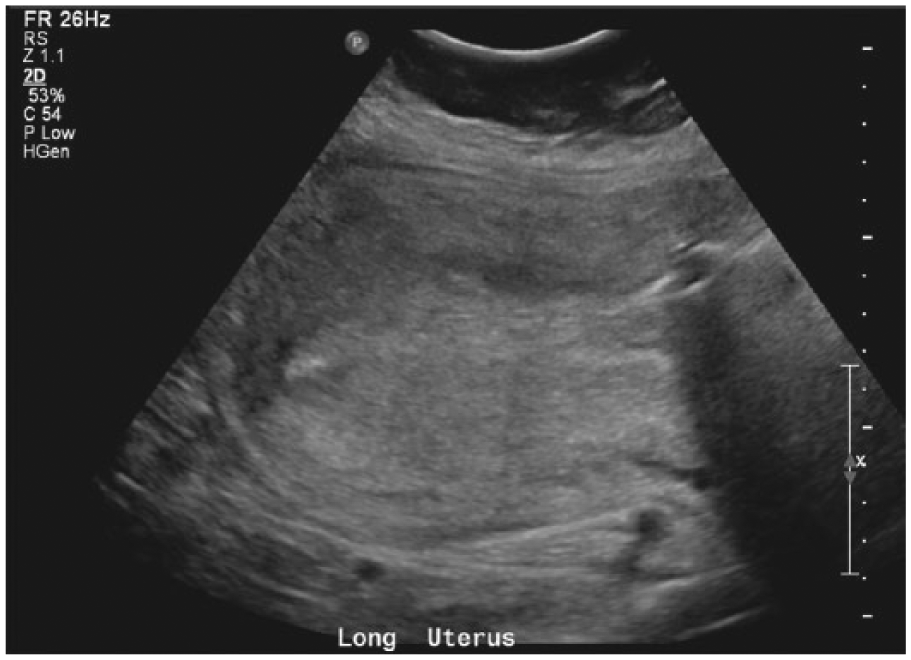
Longitudinal gray-scale image of the patient’s uterus postpartum, with an echogenic endometrium and no sonographic evidence of endometrial fluid or retained products of conception.
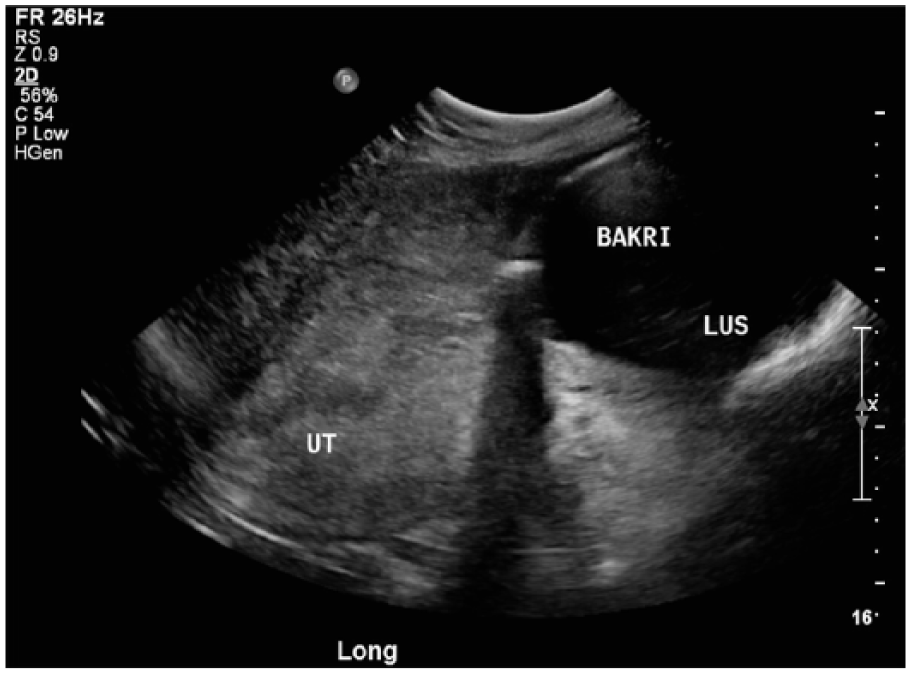
Longitudinal gray-scale image of the uterus following placement of the Bakri balloon, showing the malpositioned balloon to be in the lower uterine segment (LUS) rather than the appropriate mid-uterine (UT) position.
Discussion
The Bakri balloon is a minimally invasive intrauterine tamponade device invented by Dr Younes Bakri. The obstetric balloon is a 24-French, 54-cm long, silicone catheter with a filling capacity of 500 mL.6,10 The device is FDA approved for the temporary control and reduction of PPH. Other balloon-based devices that have been used for PPH include BT-Cath balloon tamponade catheters (Utah Medical, Midvale, Utah, USA), Foley catheters, Rusch balloons, inflated condom catheters, and Sengstaken-Blakemore tubes. The Bakri balloon and the BT-Cath tamponade catheter are specifically designed for postpartum intrauterine tamponade and are the only devices approved by the FDA for this purpose. The Bakri balloon is the most commonly used.4,11
The Bakri balloon is a well-recognized, minimally invasive, rapid method for management of PPH, with few complications.12–16 According to the World Health Organization, around 100 000 maternal deaths occur every year from PPH, making it the leading cause of maternal mortality in the developed world. Eighty percent of these deaths are caused by delays in treating uterine atony.1,17 A correctly and quickly positioned balloon can prevent the need for more invasive interventions such as uterine artery ligation, uterine compression sutures, uterine artery embolization, or hysterectomy. Complications, although rare, can be serious. Reports in the literature include balloon migration and uterine rupture secondary to balloon overinflation.15,16 Contraindications for Bakri balloon placement are bleeding due to retained products of conception, allergies to the balloon material, and uterine rupture.2,4,6
Sonographically, the Bakri balloon is seen as an ovoid structure with a hyperechoic rim and anechoic center (Figure 3). Its size will vary, depending on how much saline is injected. When correctly positioned, the balloon should be seen in the center of the uterus, expanding the endometrial canal. If the patient’s condition allows the time, sonography should be considered to rule out retained products of conception before balloon placement. After the balloon is placed, sonography can be repeated to confirm correct position.13,18
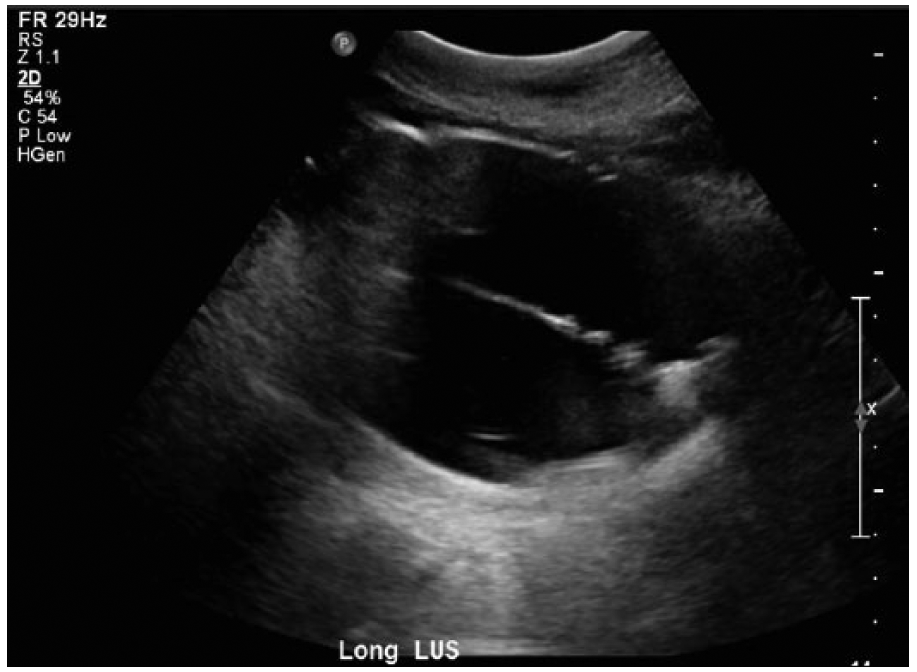
Gray-scale image of a saline-filled Bakri balloon showing the hyperechoic rim and anechoic center.
Other imaging modalities to verify Bakri balloon placement are not currently being used for several reasons. Computed tomography (CT) and magnetic resonance imaging (MRI) could visualize a balloon and its position relative to the uterus but are significantly more expensive, are not portable, and would take too much time with an unstable, hemorrhaging patient. Radiography could likely demonstrate the Bakri balloon but not the uterus. Computed tomography and radiography also use radiation, whereas sonography does not. Unlike CT, MRI, and radiography, sonography also is useful to check for evidence of retained products of conception before Bakri balloon placement.
Multiple case studies about the Bakri balloon make passing reference to sonography, but there are few cases reported of sonography used for documentation of Bakri balloon position.11,13,18 There was an anecdotal report of a similar case posted on a professional sonographic blog, 19 but there is not a case study focused specifically on the use of sonography and the Bakri balloon position. A case report by Cho et al 18 described a displaced balloon, but that report focused primarily on the mechanics of an intrauterine tamponade, not the Bakri balloon or specific usefulness of sonography. Although sonographic imaging in balloon placement is not standard practice, several case studies presented on this topic indicate that sonography may be useful.4,13,18
Conclusion
Postpartum hemorrhage is the leading cause of maternal mortality in the developed world. Uterine tamponade is a well-recognized therapy for severe PPH. The Bakri balloon is a minimally invasive, rapid method for the management of PPH with few complications. Sonographically, the Bakri balloon is seen as an ovoid structure with a hyperechoic rim and anechoic center. Sonographic documentation of Bakri balloon placement is rapid, precise, and in keeping with best clinical practice. A correctly and promptly positioned balloon can prevent the need for more invasive interventions such as uterine artery embolization, uterine compression sutures, uterine artery ligation, or hysterectomy.
Footnotes
Acknowledgements
The author would like to thank Dr Sid Green of Salem Radiology for his expertise and help and Kyle Katsinis of Chemeketa Community College for his editorial skills.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
