Abstract
Advances in cardiac sonography have made it an essential tool for physicians in detection, surveillance, and treatment of patients with advanced heart failure (HF). Echocardiographic examinations are often pivotal in assisting physicians develop treatment plans. The Food and Drug Administration (FDA) approved the HeartMate II (HMII) ventricular assist device (VAD) for bridge to transplant (BTT) in 2008. Since then, there has been the addition of many devices and treatment options for these patients. Sonographers and vascular technologists can be expected to encounter increasing numbers of long-term VAD patients in facilities with limited experience with these devices. Cardiac sonographers are in a key position to assist physicians in the care of these patients such that data generated assist with the selection of advanced therapies or as part of a patient’s follow-up care. This article provides a review of these new technologies along with practical considerations for sonographers.
Keywords
Since the 1990s, advances in cardiac sonography and diastologic measurements have been regarded as essential tools to help physicians in detection, surveillance, and treatment of advancing heart failure (HF). In contemporary practice, echocardiographic techniques are directed at evaluating left ventricle (LV) systolic and diastolic function as well as spectral Doppler to characterize mitral inflow and pulmonary vein flow and tissue Doppler imaging (TDI) of the mitral annulus. 1 The amount of clinical information that cardiac sonographers must master has grown exponentially. As an example, The Echocardiographer’s Pocket reference (first ed., 1990) has increased from 185 pages to 444 pages (fourth ed., 2013). Additionally, it also includes sections for wall segments, TDI, coronary artery distribution, chamber quantification, myocardial strain, cardiac resynchronization therapy, real-time 3D echocardiography, and a peripheral vascular section.
At the time of this article, there are 158 hospital centers in the United States who are participating in left ventricular assist devices (LVAD) programs. Of those, 128 hospitals have been designated as sites for destination therapy (DT). 2 Between June 23, 2006, and December 31, 2014, 15 745 patients who received a US FDA-approved mechanical cardiac support (MCS) device were entered into the INTERMACS database. The rate of patient enrollment has continued at pace exceeding 2000 per year. 3 Mechanical cardiac support has been expanded to include bridge to transplant (BTT), bridge to decision (BTD), and bridge to recovery (BTR). With these changes, it becomes imperative that sonographers become more familiar with these types of patients and their varied devices.
The field of cardiovascular medicine continues to rapidly expand. As the field grows in expertise, so has the alternatives to standard open heart procedures, including new, minimally invasive cardiac surgeries. They include robotic mitral valve repair and minimally invasive aortic valve replacement. Cardiologists and cardiac surgeons are now collaborating in hybrid surgical/catheterization suites. Often patients require MCS with devices such as TandemHeart or an Impella pump. New procedures, techniques, and improvements in sonography have changed and expanded the responsibilities of the cardiac sonographer, especially those working in these types of treatment programs. Although complicating the diagnostic evaluation, MCS devices and cardiac transplant have extended the lives of many patients who would otherwise have died from advanced HF. As these patients experience improved health, their subsequent care is being followed in outpatient cardiology offices. As of August 2015, the ASE published guidelines for these devices. The ASE states that more than 30 000 patients have received LVADs. More and more of these patients are now receiving treatment in health facilities that have not had to manage ventricular assist devices (VADs) in the past. It appears that there will be many more in the future. The purpose of this symposium is to review VADs and some of the considerations for sonographers who are unfamiliar with these devices and transplant patients to consider.
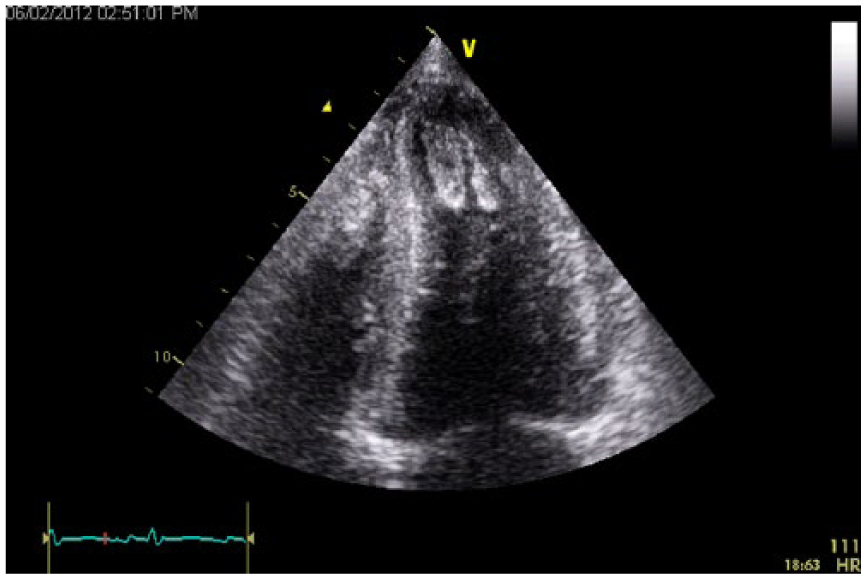
Transthoracic echocardiography apical four-chamber view transplant rejection with apical clot.
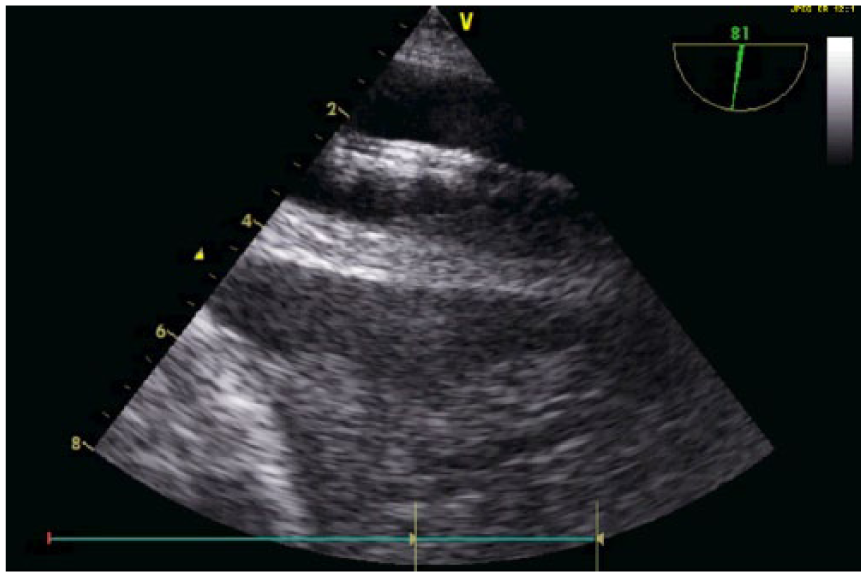
Transesophageal echocardiography of abdominal view of the descending aorta intra-aortic balloon pump. Aorta is from 1 cm to 4 cm marker. Balloon is at 2 cm marker.
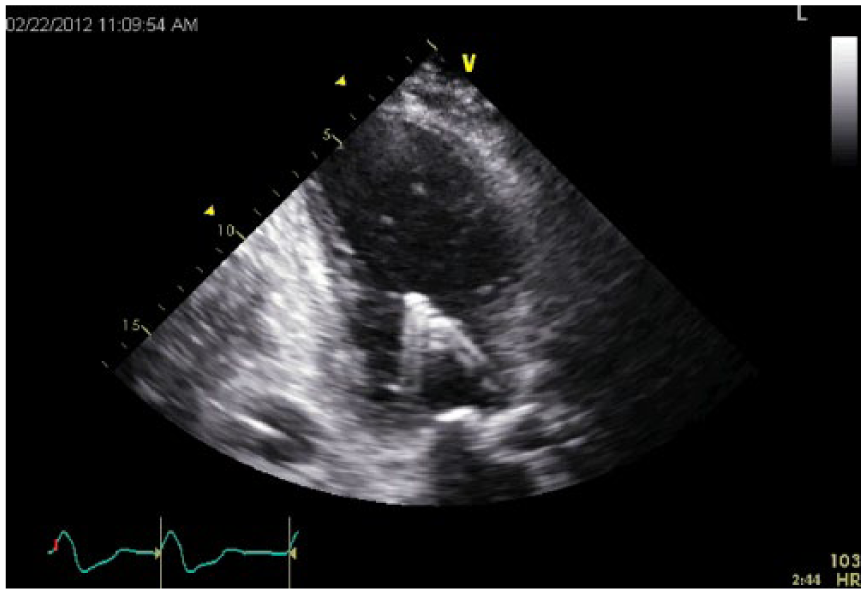
Transthoracic echocardiography apical long axis view. The Impella device cannula is inserted through the aortic valve into the left ventricle. There is artifact reflecting to the left of the catheter.
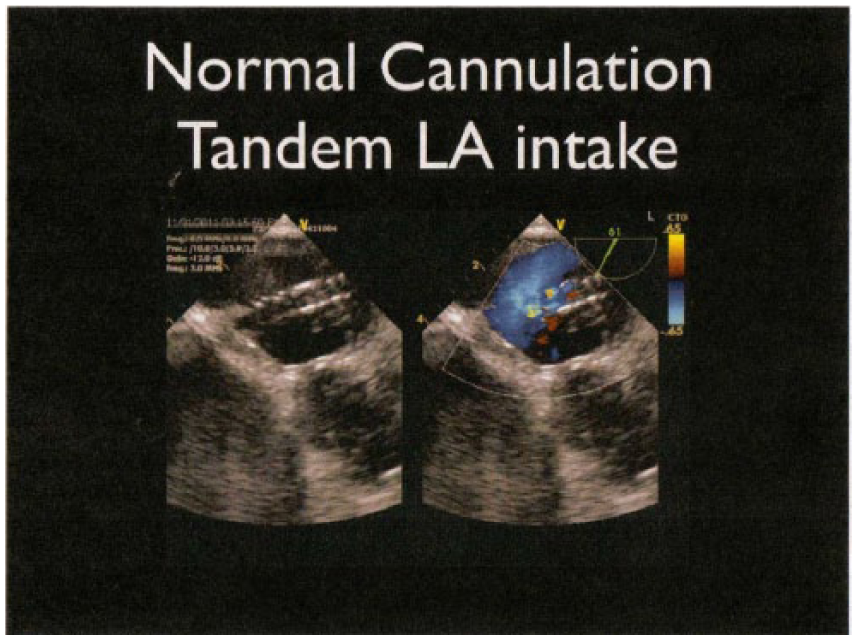
Transesophageal echocardiography of normal cannulation of the Tandem heart across the intra-atrial septum from right atrium to left toward the left atrial appendage.
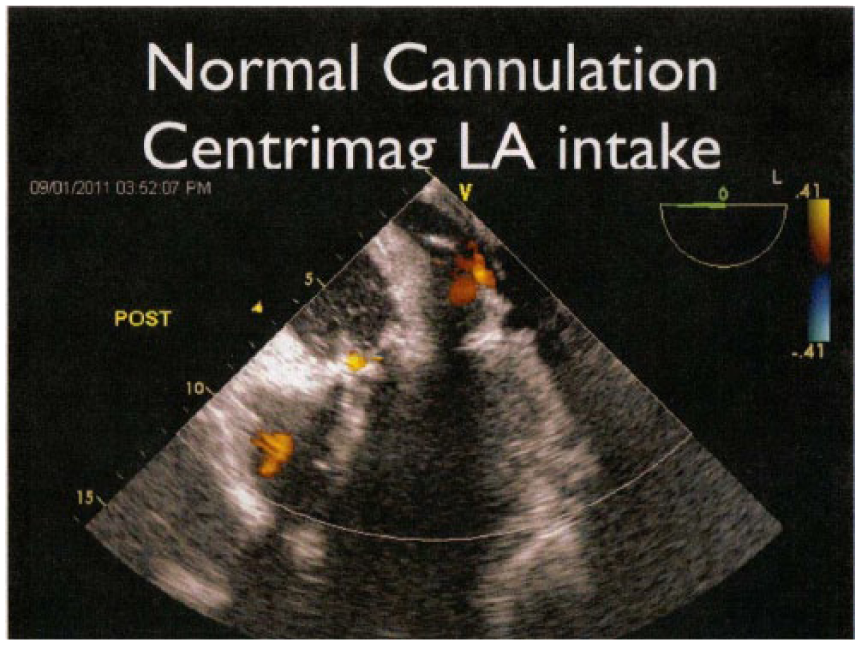
CentriMag left ventricular assist device cannulation of the left atrium by transesophageal echocardiography. The blood in taken into the heart here and leaves the heart through the apex of the left ventricle.
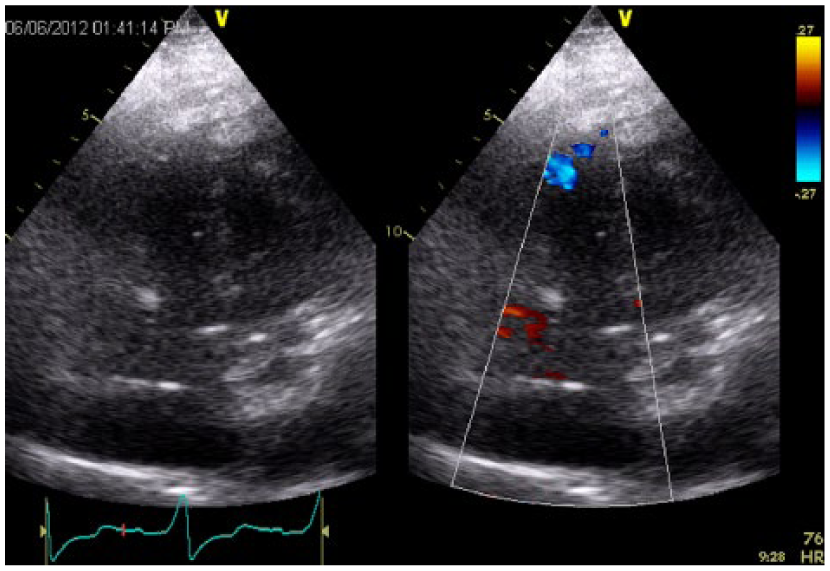
Right ventricular assist device apical cannula flow in transthoracic echocardiography in apical five-chamber view.
Veno-venous extracorporeal mechanical oxygenation. Arrow points to cannula tip in the right atrium on transesophageal echocardiography parasternal short axis aortic valve view (PSAX-AO).
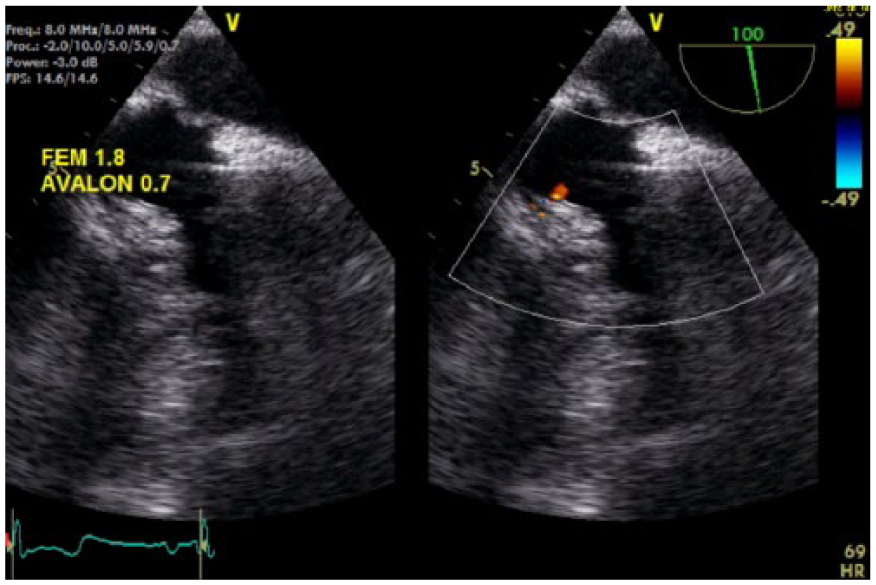
Transesophageal echocardiography of Avalon veno-venous extracorporeal mechanical oxygenation catheter. The outflow is directed at the tricuspid valve.
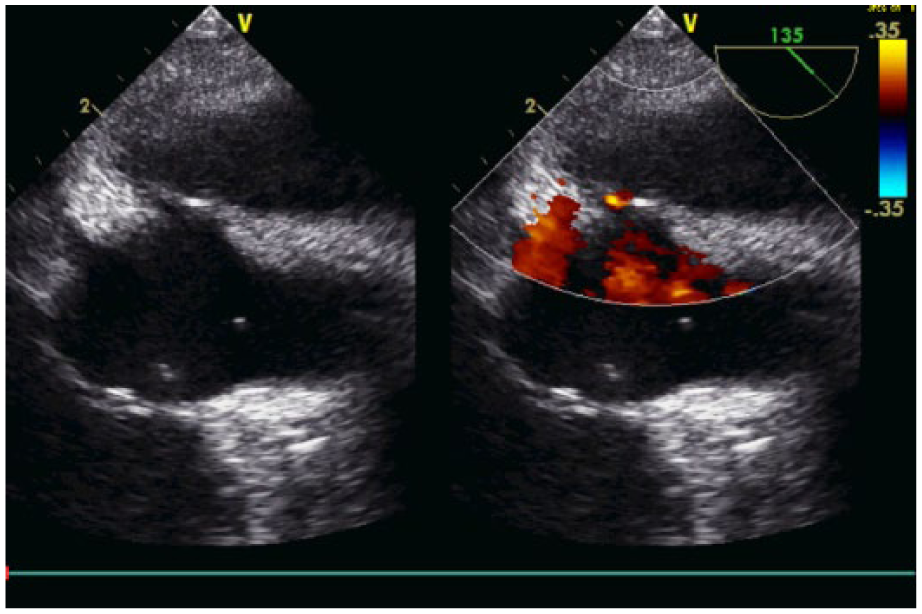
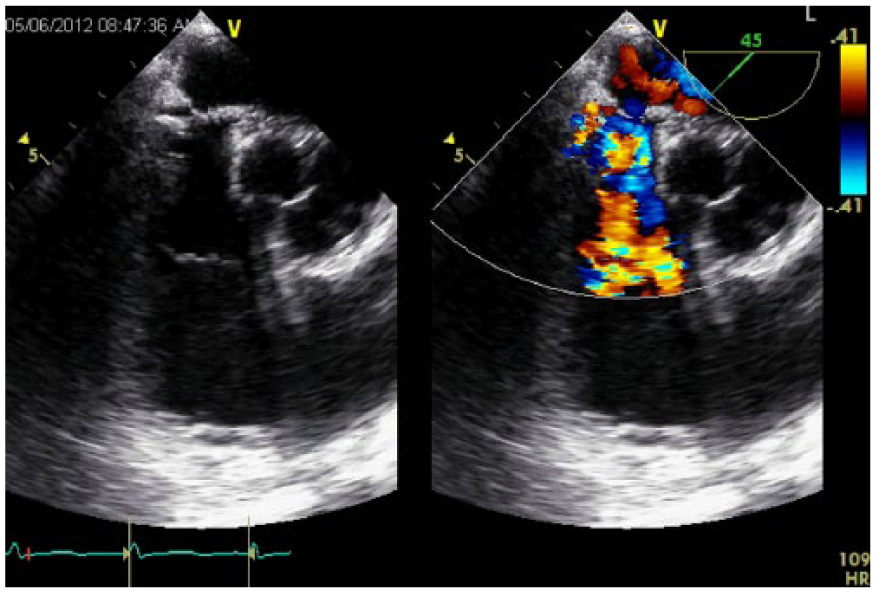
Transesophageal echocardiography bicaval view of veno-arterial extracorporeal mechanical oxygenation with patent foramen ovale flow.
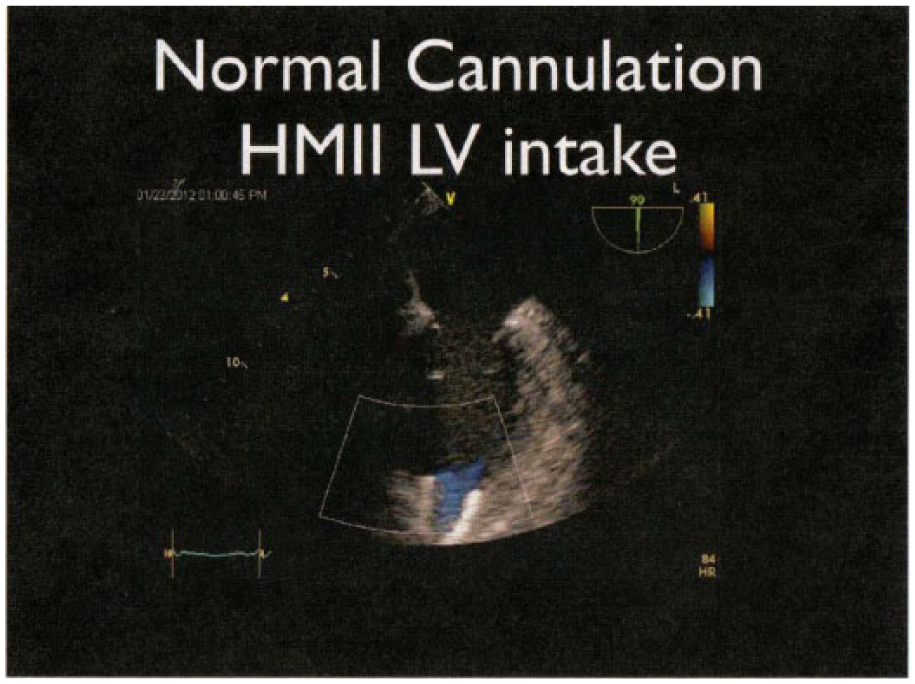
Transesophageal echocardiography of HeartMate II left ventricular assist device cannulation of the apex with it directed toward the mitral valve.
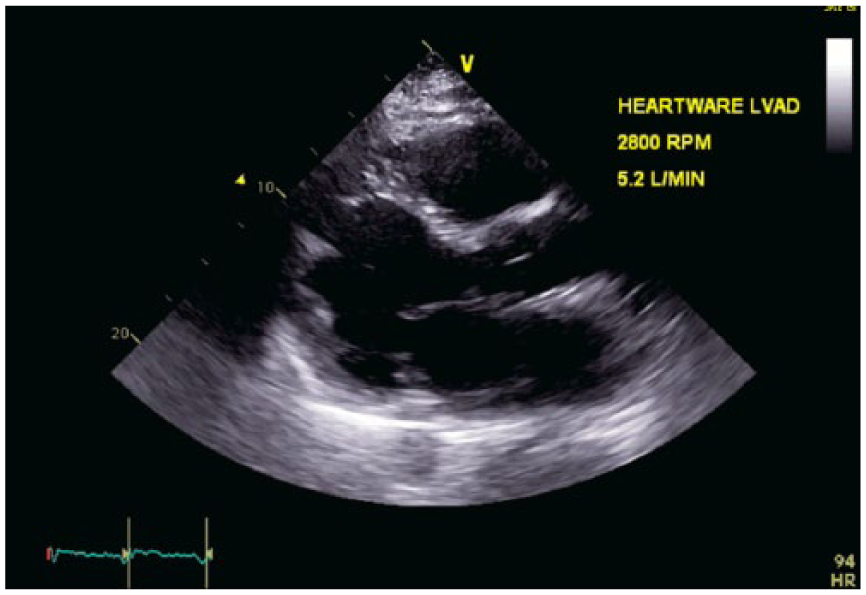
Transthoracic echocardiography parasternal long axis view. HeartWare left ventricular assist device. Outflow cannula in the left ventricular apex.
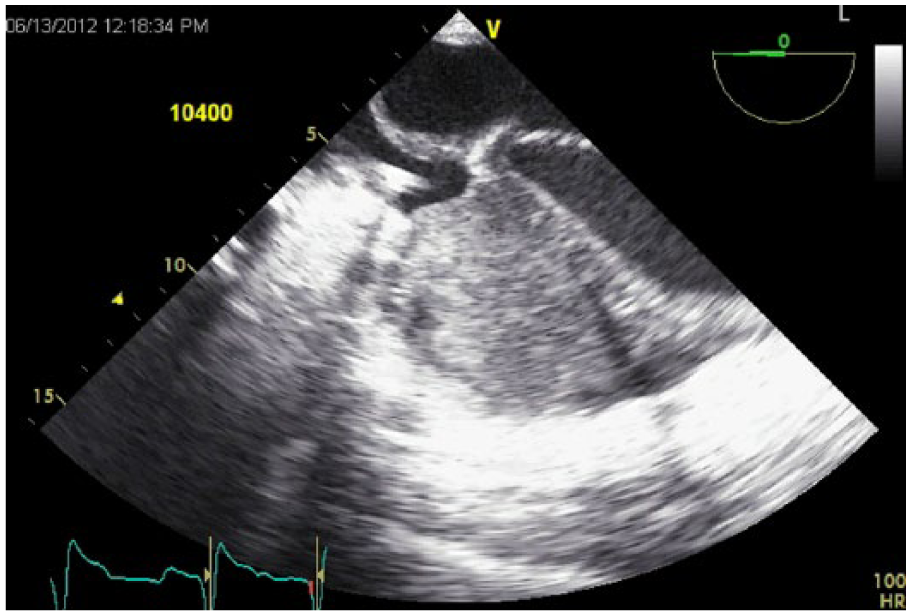
Right heart failure with large right ventricular clot. Transesophageal echocardiography four-chamber view.
Heart Failure
Some of the earliest descriptions of HF are recorded at the beginning of medicine. 4 Modern medicine has advanced greatly the care of these patients due to the mounting scientific evidence regarding the mechanics of HF. In the US alone, approximately 5 million people are afflicted by HF, with an annual incidence of 500 000 new diagnoses and 300 000 deaths. 4 Mortality rate is high after initial diagnosis, with one in five dying in less than a year. 5 Heart failure can be manifest across a vast range of patients and associated etiologies.
Heart Failure Pathophysiology
Patients can develop HF through several mechanisms. Myocardial stunning is a regional myocardial dysfunction after a brief episode of severe ischemia that recovers after a period of time. Myocardial hibernation is reversible myocardial dysfunction caused by chronic ischemia. Cardiomyopathy (CM) occurs when the heart muscle becomes diseased. There may be multiple causes, such as viral infections.
There are three types of CM that have been described: (1) In dilated cardiomyopathy (DCM), the heart cavity is enlarged and stretched. Many patients develop congestive heart failure (CHF) along with cardiac arrhythmias. (2) In hypertrophic cardiomyopathy (HCM), the muscle mass of the LV becomes very thick. In one form of the disease, the septum becomes thicker than the rest of the ventricle and obstructs blood flow from the LV. This is also known as hypertrophic obstructive cardiomyopathy, asymmetric septal hypertrophy (ASH), or idiopathic hypertrophic subaortic stenosis (IHSS). The mitral valve anterior leaflet or submitral apparatus can be drawn into the left ventricular outflow tract (LVOT) and can cause mitral regurgitation; HCM can cause sudden death. (3) Restrictive cardiomyopathy (RCM) is a type of CM in which the heart becomes stiff and filling can be inhibited. Each patient requires unique management by physicians. 6
Heart failure prevalence trends have changed over the past few decades. From 1994 to 2003, the incidence of HF declined from 32 per 1000 person-years in 1994 to 29 per 1000 person-years in 2003. 7 Heart failure rates continued to decline through 2014, but from 2012 to 2014, there has been a steady increase. 8 The incidence of HF in older patients seems to be declining; however, when elderly patients access the health care system, they come with the comorbidities associated with aging. Meanwhile, the incidence of HF appears to be increasing among younger patients. This means that younger patients enter the health care system earlier and are staying longer. Because of improved medical care for these patients, many are now living longer, and these points indicate an increasing number of advanced HF patients in the future.
The increase in patients with HF has also increased the types of HF medications available for treatment. Although HF medications provide some relief to these patients, it does not decrease the number of failing hearts. The increase in prevalence and the increased hospital lengths of stays for these patients require more options for treatment. Heart transplant is one surgical option, available to some qualified patients. There have been 118 788 total heart transplants reported since the inception of the technique. 9 However, the number of hearts available for transplant is insufficient for the demand. In addition, some patients who have managed to live longer have exceeded the age requirements for transplant eligibility. For these patients, MCS devices (ie, VADs) are an alternative.
One practice example is the cardiology team at Spectrum Health in Grand Rapids, Michigan. In 2009, they implanted their first VAD; currently, more than 258 long-term VADs have been implanted at that location. Additionally, more than 117 patients with devices are cared for in that community. The first Spectrum Health patient received a heart transplant in November 2010. To date, more than 74 heart transplants have been completed by this practice, with 56 of these patients being former VAD recipients.
Introduction
To assist students and practicing sonographers, the following sections provide a review of some clinical conditions and devices that sonographers can encounter in patients with suspected cardiomyopathies.
Cardiogenic Shock
Cardiogenic shock is a condition in which a suddenly weakened heart is unable to pump enough blood to meet bodily needs. 10 The results of cardiogenic shock can include poor organ perfusion, decreased mentation or sense of anxiety or doom, decreased kidney output and failure to respond to diuretics, development of metabolic acidosis, vasoconstricted extremities, hypotension, and a reduced pulse pressure (the difference between the maximum and minimum blood pressure during one beat) of less than 25%. 11 The condition is a medical emergency and is fatal if not treated immediately.
The mean arterial pressure (MAP) is a very important measurement for HF patients, patients with cardiogenic shock, and patients receiving mechanical ventricular support. Mean arterial pressure is the average pressure during the cardiac cycle. The MAP formula is MAP = (2 × diastolic pressure) + systolic pressure / 3. The diastolic pressure is multiplied by 2 because it takes up two-thirds of the cardiac cycle. Mean arterial pressure can also be written as MAP = (CO × SVR) + CVP (CO, cardiac output; SVR, systemic vascular resistance; CVP, central venous pressure). A MAP of 60 mmHg is the minimum required to perfuse the organs. Mean arterial pressures below 60 mmHg are at risk of inducing ischemia. The normal range for MAP in non-VAD patients is 70 to 110 mmHg. A MAP of 65 to 75 mmHg is usually the goal in cardiogenic shock patients. In continuous flow VAD patients, a MAP of 60 to 90 mmHg is normal, with a MAP of 110 mm Hg or greater possibly causing organ damage. 11
Heart failure patients are at a greater risk of cardiogenic shock than the general population. When patients who have compromised hearts from previously diagnosed HF suffer new cardiac episodes, cardiogenic shock is a more common result. Intra-aortic balloon pumps (IABPs), percutaneous MCS devices, and VADs are short-term tools that cardiologists and surgeons can use when medical therapy alone no longer suffices. These devices can provide time to decide if more invasive interventions are required.
Tips for Sonographers
Patients suffering from cardiogenic shock are having a medical emergency. The most important factors a sonographer should note are LV and right ventricular (RV) function, wall motion abnormalities, and any signs of valve disease. Cardiac output should be measured and noted. Indications of CM or myocardial infarction (MI) should be noted as MIs are a major cause of cardiogenic shock. The sonographer should look for signs of ventricular rupture, aortic dissection, and outflow tract obstruction. If the patient has a VAD, the output and type of the device should be noted; this will give the physician the ability to distinguish the patient’s native contribution from what the device provides.
Heart Transplants
The first human heart transplant was done by Dr. Barnard in 1967, 12 and as a result, this greatly expanded the field of cardiovascular medicine. By 2011, a total of 50 000 heart transplants had been performed in the US. Approximately 50% of these patients survived 13 years, and approximately 16% survived more than 20 years. As of June 30, 2015, the International Society for Heart and Lung Transplant reported that a total of 118 788 heart transplants had been performed worldwide, with 113 472 of those being adults. Advances in immunosuppression have reduced the number of patients suffering severe episodes of transplant rejection. Surveillance for rejection is still a priority, however, since these patients are always at risk for organ rejection. A significant long-term complication is cardiac allograph vasculopathy (CAV). 9
Cardiac allograph vasculopathy is responsible for a significant proportion of deaths after transplant, and its contribution increases with time after transplant. Despite improvements, the prevalence of CAV remains high—20% at 3 years, 30% at 5 years, and 45% at 8 years after transplant. 9
Heart transplants are restricted by geographic distances due to the limited time an explanted heart can be outside of the body and still recover when implanted. New systems are being developed to create adequate perfusion of a heart for transplant once explanted from the donor. The organ care system OCS Heart, also known “heart in a box,” is technology that provides nutrients to the heart and increases explant time from 4 hours to 10 hours. This technology is available in Europe and Australia but is still in investigational phase in the US. 13
Tips for Sonographers
Heart transplants can have their anastomoses in the bi-caval or bi-atrial position. The position can affect the size of the atrium and affect the heart rate in the case of a stress test. Bi-atrial transplants may have double p waves on the EKG. When stress tests are required, these patients should have a chemical stress test, such as a dobutamine stress echo, due to the changes in the conduction system (the sino-atrial node is the origin of the conduction system and is altered by the excision of the heart). Although the bi-atrial technique of attachment was more common in the past, the bi-caval method is becoming more common. Bi-atrial patients are more likely to require a pacemaker more often. During the first 3 days after transplantation, a normal posttransplant echocardiogram should include an examination of the anastomosis sites for failure. There will be an anastomosis at the aorta, pulmonary artery, and attachment site of the heart. The patient’s ejection fraction should be calculated. All diastology measurements should be taken to examine the new heart for stiffness. Right heart measurements are required to inspect the right heart for dysfunction. These patients should always be evaluated for effusion and cardiac tamponade post biopsy. One example to consider is Spectrum Health’s cardiac team, which takes biopsies once per week for the first month, every other week for the second month, and monthly thereafter. In this practice, they have successfully reduced biopsies to one to two times per year. The frequency can vary postoperatively based on the patient’s rejection response. Cardiac allograph vasculopathy, rejection, and infection are the top complications for heart transplants. Echocardiography has been used in some institutions, instead of fluoroscopy, to guide biopsy procedures.
Day 3 posttransplant, ventricular function is the main focus of interrogation. Decreased ejection fraction can be an indication of rejection. New septal wall motion abnormality and decreased ejection fraction can be indicators of transplant vasculopathy. Diastology should always be done to inspect for increasing stiffness of the heart. If postoperative transthoracic imaging is too difficult, an imaging agent can be used. The cardiologist may choose to use transesophageal echocardiography (TEE). In recurrent visits for rejection surveillance biopsies, the patient should always be evaluated for tamponade post biopsy. 13
Lung Transplant
The lung transplant registry now contains from 51 440 lung transplants through June 2014. The disease process that is the most prevalent is chronic obstructive pulmonary disease. During 2013, more patients were recorded as having lung transplants than previous years. 9 As with heart transplants, the need for lung transplants are predicted to rise. Extracorporeal mechanical oxygenation (ECMO) is also being used by some physicians during the perioperative period for support to help oxygenate the organs. Extracorporeal mechanical oxygenation could be used in the future as a bridge to lung transplantation for patients who can no longer wait for a donor on medical management alone, as VADs have become an alternative for heart patients. The ex vivo lung perfusion (EVLP) monitoring is another investigational device. It is designed to give nutrients to the lungs and provide the surgeon with metrics of the donor lungs from the time it is explanted until implantation. This can assist the surgeon with viability status while the potential recipient is being prepared to receive the new lungs. 14
Tips for Sonographers
The lungs and heart work together as cardiovascular circuit, therefore issues with the heart can dramatically affect the condition of newly transplanted lungs. Similarly, lung transplant patients are likely to have comorbidities that affect their hearts as well. With a lung transplant, imaging is critical but can sometimes be very difficult to execute. Echocardiography is an important diagnostic tool to assess both heart and lung transplant patients. For example, it is important to establish if a patent foramen ovale (PFO) is present; PFOs can cause right heart dysfunction postoperatively. Right heart function is also an indicator of lung status and should be monitored.
Mechanical Cardiac Support
Physicians have been trying to find ways of supporting patients with advanced HF for many decades. The first experiments with VADs began in 1963. The National Institutes of Health established the Artificial Heart Program in 1964, culminating with the first LVAD that was successful for 10 days in 1966. 12 Mechanical cardiac support is being used as a BTT, BTD, DT, and BTR.
Ventricular Assist Devices
A VAD is a mechanical pump that assists the heart with CO. Pumps have different types of controllers that control, calculate, and monitor the amount of support produced by the pump. Ventricular assist devices have been pulsatile or continuous flow pumps; Spectrum Health uses continuous flow pumps. Ventricular assist devices can partially or completely replace cardiac function. Ventricular assist devices can be used to support the left or right ventricle.
Short-term Devices
These devices are recommended for use of up to 2 weeks for circulatory stabilization and/or myocardial recovery. 6 In severe cases, some have been used for up to 6 weeks. Short-term devices are intended for stabilization/recovery and can be subdivided two categories: pulsatile and centrifugal. Pulsatile devices require surgical implantation of cannulae through an open sternotomy and are most often are inserted on cardiopulmonary bypass and removed surgically. Due to the size and frequent complication rate of pulsatile devices, the continuous flow pump (CFP) for circulatory support has taken over the field. 15 Pulsatile pumps are still in use, although less frequently. Continuous flow pumps may be inserted percutaneously in the catheterization laboratory or surgically in the OR. Percutaneous VADs (pVADs), such as TandemHeart or Impella, devices are typically deployed in the cardiac catheterization laboratory. Surgically placed short-term mechanical circulatory devices may include Centrimag and others. Short-term use of MCS devices will aid in decreasing LV wall tension, decreasing myocardial oxygen consumption, and decreasing left atrial filling pressures while increasing CO. Indications for use of common short-term VAD or MCS devices include acute MI, post-cardiotomy pump failure, decompensated HF, post-cardiac transplantation allograft dysfunction, acute myocarditis, deterioration after catheterization lab procedures, prophylactic insertion for high-risk catheter-based intervention, cardiac arrest, and RV infarct. 6 Additives and an oxygenator may be placed at the same time as the pump, namely, ECMO. Oftentimes, cardiac flow of 5 L/min or better can be obtained.
Intra-Aortic Balloon Pump
Versions of the IABP have been an aid in cardiogenic shock both in and out of the OR since the late 1960s in the US. The IABP assists heart function through the inflation of the balloon during diastole, which pushes blood forward to the coronary arteries and increases myocardial blood flow. The balloon is deflated during systole, which creates a small amount of suction and reduces afterload on the heart. The balloon pump is usually inserted after MI to provide the patient with pump support while the heart heals. It can also be used before or after cardiac catheterization or open heart surgery to support pumping function.
The balloon pump can provide up to 2.5 L/min of CO. The balloon pump is a short-term solution. If the patient requires longer term support, the balloon pump can be removed and an Impella device or TandemHeart inserted.
Tips for Sonographers
The IABP is deployed in the cardiac catheterization laboratory under fluoroscopy or in the OR. The pump’s balloon should come to rest within 2 cm of the subclavian artery. The sonographer’s role comes after deployment; transthoracic echocardiography (TTE) or TEE can be used to check balloon position and follow cardiac function. The position and effects of the device will often be visible on ultrasound. If properly placed, the balloon should be visible in the suprasternal notch view and the subcostal view on echocardiogram. If the images are difficult to obtain, the turbulence of the Doppler signal can be helpful. In some patients, body habitus and positions may be difficult as the patient will be flat on their back. If possible, LV function, wall motion, ejection fraction, and CO are the most important things to acquire for this study to give physicians useful feedback on treatment progress. 15
Impella Device
The Impella catheter is an intravascular microaxial blood pump that supports a patient’s circulatory system. The Impella catheter is inserted percutaneously through the femoral artery, into the aorta, and across the aortic valve into the LV. The Impella catheter has a suction port that draws blood from inside the LV. There is a second port that expels the blood into the ascending aorta from behind the aortic valve. The Impella device is a pVAD that can be inserted by a surgeon in the OR or a cardiologist in the catheterization laboratory. The Impella is a short-term device. The following three versions of the Impella are used for LV support in the United States: Impella LP 2.5 (2.5 L/min), Impella LP 5.0 (5.0 L/min), and Impella LD (5.0 L/min). There is a European version of this device called the Impella cVAD, which was approved in Europe in April 2012 and is intended for use for up to 5 days.16,17
Tips for Sonographers
The Impella device is deployed under the guidance of TEE. The most important thing for a cardiac sonographer to know is that it is very important for the catheter to be midway into the LV. It should not be dislodged. The patient is often moved from the Impella deployment location to their room with the TEE probe still in place so that the location of the cannula can be verified to make sure the cannula has not been displayed during transit. Serial echocardiograms, which can be limited to a few images, can provide intermittent evaluation of cannula location.
Transthoracic imaging after deployment can be difficult as the patient cannot be turned to optimize imaging, but imaging is often possible. In the event that transthoracic imaging is suboptimal, TEE may be used. The revolutions per minute (RPMs) should be noted on the echocardiography console. The appearance of the ventricle at that particular speed provides the physician with guidance to make adjustments if necessary. If the cavity is too big, the revolutions may need to be increased. If the ventricle is too small, the revolutions may need to be decreased, or the patient may need to be hydrated. It is most important to see the catheter tip well deployed into the LV through the aortic valve. The inlet or tip of the cannula should be approximately 4 cm below the aortic valve, and the pump inlet should be well above the aortic valve.
The 2.5 L version of the device has a pigtail shape and can damage the mitral valve chords if deployed too far into the ventricle. Examine the catheter tip with 2D echocardiography as well as color Doppler. If possible, locate the inflow port in the LV and the return flow port in the ascending aorta with 2D echocardiography and color. The amount of aortic insufficiency (AI) should be noted. If there is too much AI, the patient may not have adequate CO as the blood is being delivered in a continuous loop inside the heart. The CO should be noted to help determine what the heart is doing on its own. For example, if the pump is delivering 2.5 L/min and the CO derived by the sonographer is 3 L/min, the patient’s heart is delivering approximately 0.5 L/min. The Impella 2.5 has a maximum speed of 51 000 rpm and delivers up to 2.5 L/min. 16
If the right heart is failing or right side pressures become elevated, the left heart can be compromised. The CVP should be checked on the vital signs monitor and noted at the time of echocardiogram. The Impella 5.0 is intended for circulatory support using an extracorporeal bypass control unit for recommended periods of up to 6 hours. It is also intended to be used to provide circulatory support during procedures not requiring cardiopulmonary bypass. 16 The Impella 5.0 can deliver 4.2 to 5.3 L/min at a speed of 33 000 rpm. 17 The Impella devices can be used for longer periods of time if necessary. It is important to note the rpm on the echo console.
TandemHeart
The TandemHeart is a short-term MCS device. The TandemHeart is a pVAD that requires atrial septal puncture. The TandemHeart cannula is inserted across the intra-atrial septum. It is an extracorporeal pump, meaning that circulatory support occurs outside of the body. It is a continuous flow pump. The TandemHeart can be deployed in the cardiac catheterization laboratory or the OR. The 2.5 L cannula can be deployed in the catheterization laboratory. The 5.0 L cannula must be deployed surgically. The TandemHeart is traditionally inserted in the catheterization laboratory. The inflow cannula is inserted through the femoral vein into the left atrium via a transseptal puncture. Blood flows from the left atrium to the pump. The outflow cannula is inserted into the femoral artery to return oxygenated blood. The device can deliver up to 5 L/min and is typically used to combat the effects of cardiogenic shock. 6
Tips for Sonographers
The TandemHeart is placed under TEE or intracardiac echocardiography (ICE) guidance to observe and record the transseptal puncture. The sonographer should look for tenting of the mid intra-atrial septum (IAS). The fossa ovalis should be located for the puncture site. The guidewire will be in the vicinity of the aortic root, and aortic root puncture is a potential complication.
After insertion of a guidewire and dilator into the femoral vein, the cannula of the TandemHeart device is advanced through the IAS. It is important that the cannula fenestrations are midway in the left atrium (well away from the intra-atrium septum). This is because the cannula draws oxygenated blood from the left atrium and deposits it into the pump. To facilitate visualization of the fenestrations, use color Doppler on the sonogram.
During implantation of the TandemHeart, the sonographer should watch for air embolism as the cannula is crossing to the left side of the heart. Once flow has begun, the sonographer should monitor the right heart for failure. If the right side of the heart already suffers from failure, it can decompensate further. In the event that this happens, the right heart will not be able to fill the left heart. If this situation continues, the cardiologist may choose to switch devices, to a veno-arterial (VA) ECMO or biventricular assist device (BIVAD). To evaluate the RV, the sonographer should evaluate the 2D image for RV size, tricuspid valve (TV) velocity time integral (VTI), tricuspid regurgitation (TR), and pulmonary regurgitation (PR). Increases of these parameters should be noted. The inferior vena cava (IVC) diameter and tricuspid annular plane systolic excursion (TAPSE) should be measured, and the left side should be assessed for LV decompression and function. The aortic valve should be assessed for opening, closure, and insufficiency. The patient may be taken from the catheterization lab to the intensive care unit (ICU), with the TEE probe left in to confirm proper insertion of the cannula. In the ICU, the patient can have serial echoes to verify the cannula position, assess both LV and RV function, and check for cardiac shunting. The CVP should be monitored. Elevation of the CVP may indicate that the patient may be going into RV failure. Calculation of QP/QS (pulmonary blood flow divided by systemic blood flow) can be used to evaluate for intracardiac shunts. A value of greater than 1.5 is significant. Limited serial echocardiograms may be ordered to check cannula position on an intermittent basis.
CentriMag
The CentriMag is a small centrifugal flow blood pump that can produce blood flow up to 10 L/min. The CentriMag is a short-term device that can be used on the left side, right side, or biventricular. The CentriMag is often placed in the OR through sternotomy, although catheters can be inserted percutaneously. The speed can be adjusted from 800 to 5500 rpm and can routinely provide flows of up to 9 L/min. For the left sided position, the outflow cannula enters the proximal portion of the ascending aorta. The inflow cannula enters the left atrium. It can also be placed in the LV. In the right sided position, the outflow cannula enters the main pulmonary artery, and the inflow cannula enters into the right atrium. 18
The rotation of the impeller occurs within a magnetic field, eliminating contact between components and resulting in frictionless movement with little heat generation or wear of moving parts. In addition, the contact-free frictionless impeller movement reduces the risk of hemolysis and thrombosis. 18 The CentriMag impeller can also be used for ECMO.
Tips for Sonographers
CentriMag implantation is guided by TEE in and outside the OR. Once the patient is admitted to the ICU, TTE or TEE can be used to examine the cannulation sites. For the left sided support position, examine the left atrial drain for inflow and the proximal aorta for outflow. The LV should be inspected for size and function in addition to swirling blood that gives the appearance of smoke and/or clots. The sonographer should check the aortic valve for opening and closure. Aortic insufficiency should be measured, and the proximal aorta should be inspected for clots. The intra-atrial septum should be inspected for PFO, which can cause the LVAD to suck venous blood across into the left atrium.
For the right sided support position, examine the right atrial drain and main pulmonary artery outflow. Inspect the pulmonary valve for pulmonary insufficiency. If the cannula is too close to the pulmonary valve, severe pulmonary insufficiency and recirculation of blood can result. If there is too much insufficiency, the pulmonary artery may have to be banded. The sonographer should inspect the intraventricular septum to confirm midline positioning. Both midline shift and PFO are problematic. The right sided support devices should be able to adequately fill the LV. In the presence of pulmonary hypertension, the LV may appear under filled.
Extracorporeal Membrane Oxygenation
Extracorporeal mechanical oxygenation is for full support of both circulation and oxygenation. The device requires surgical implantation and setup by a perfusionist. For the past three decades, ECMO has been used primarily to support neonatal patients and adults with respiratory distress syndrome. It has also been used to support patients with cardiogenic shock and severe hypoxemia. However, with the increase in incidence of HF, cardiogenic shock, and respiratory failure, ECMO is being employed more frequently and in more varied diseased states.
Veno-Venous ECMO
Veno-venous (VV) ECMO is a treatment option for acute lung injury and acute respiratory distress syndrome, when the heart function is capable of supporting adequate organ perfusion. A venous catheter removes blood from the vena cava and after oxygenation returns the blood to the right atrium, tricuspid valve, right ventricle, then pulmonary artery. This method can only be deployed in patients who have adequate LV function.
Avalon Catheter
The Avalon is a common VV catheter used for ECMO. Inserted into the internal jugular, the Avalon is able to match the body’s natural flow ratios by simultaneously removing blood from both the superior vena cava (SVC) and IVC and returning oxygenated blood to the right atrium. The Avalon catheter is VV, so the device is used to rest the lungs and not for treatment of cardiac issues. 19
Veno-Arterial ECMO
Veno-arterial (VA) ECMO is extracorporeal oxygenation in which the venous catheter can enter through the internal jugular vein or the inferior or SVC to drain the right heart into the oxygenator. The arterial catheter is typically situated in the ascending aorta, usually via insertion in the femoral artery or the subclavian artery. This type of ECMO is usually used for treatment of cardiogenic shock or HF.
Tips for Sonographers
Veno-venous ECMO is becoming more commonly used to support patients before, during, and after lung transplantation. It is a gentler way than a ventilator to oxygenate lungs that have been involved in a traumatic event and are at risk for damaging inflammation. Adult ECMO is usually placed under the guidance of TEE. For catheter placement with VV ECMO, the inflow and outflow portions of the catheter should be examined with color flow Doppler, and the flow across the tricuspid valve should be carefully evaluated. The TEE helps guide placement of the guidewire and the dilator as well as the individual ports of the Avalon catheter, which sit within the right atrium and the superior and inferior vanae cavae. Proper placement ensures simultaneous inflow and outflow. It is important for the sonographer to measure the right atrial size and examine placement of the outflow ports, which should face the right atrial septum. The sonographer should ensure that the drain portion is in the right atrium, pointing toward the tricuspid valve. 19 After deployment of the Avalon, subsequent echocardiograms can be by TEE or TTE. The catheter can be reexamined for placement.
For VA ECMO, the sonographer should assess cannula position in the right atrium. It is important to examine the patient’s head and neck. Swelling of the head and neck or “pumpkin head” can be a complication of SVC. In the presence of this sign, look for an obstruction in the SVC. Dr. Wilton, cardiothoracic critical care director at Spectrum Health, suggests that this may be indicated by a gap between the cannula and the SVC on echo. The ascending aorta may be difficult to visualize, but visualization of the cannula should be attempted. As with all MCS, it is important to check the LV and ensure that it has not become more dilated. The ECMO configuration may not always adequately drain the heart, potentially leading to LV distension and pulmonary hypertension. If the inflow cannula is in the ascending aorta, the aortic valve should be assessed for stenosis, insufficiency, and opening. If the patient has acute lung injury or acute respiratory distress syndrome, hypoxemia is a potential complication, in which deoxygenated blood is ejected into the aortic root, coronary arteries, and brain.
Long-term devices
Long-term devices are used primarily as bridges to cardiac transplantation or as destination therapies. Though placed in the hospital, these battery-powered devices are intended for use in a home setting.
HeartMate II
HeartMate II (HM II) is a long-term CFP. The continuous flow of the HM II is driven by a small axial pump, which generates up to 10 L/min of blood flow; common pump speeds are at 8600 to 9800 rpm. 20 It is currently the most common pump for long-term support. Because the HM II is a long-term device, it can be used as a BTT, or it can be the DT for patients who are too old, too ill, or otherwise ineligible for cardiac transplant. More than 20 000 patients have been treated with long-term VADs, with the current implant rate more than double the rate of heart transplants. There are patients who have had this device as long as 10 years. Long-term VADs will become more of a mainstay for treatment of advanced HF patients. 20
Tips for sonographers
For preoperative echocardiography, it is important to inspect the patient for a PFO. If present, the PFO often is closed during implantation. 20 The condition of the aortic valve should also be inspected. The physicians will want to know if there is aortic stenosis and the severity if present. The competency of the aortic valve is important. Aortic insufficiency can cause a circulation loop from the heart to the VAD pump, and the aortic valve may need to be replaced or sewn shut. Such patients have low ejection fractions, so they should be inspected for thrombus in the heart. Mechanical prostheses should also be inspected for thrombus and position. Right heart assessment is important preoperatively because it can be an important factor in postoperative outcome.
In the immediate postoperative period implantation of the HM II, as with any post-cardiotomy surgery, sonography can be difficult. Transesophageal echocardiography can be used if necessary when transthoracic imaging is problematic. The RPMs and CO should be noted on the first echo image. Excessive speed can cause suction events in which the LV can become compressed or part of the apex can fold over the outflow cannula. This can cause ventricular fibrillation and must be remedied. If the speed is not high enough, the LV can become dilated. Ensure that the LV is properly decompressed. Left ventricle cavity obliteration may also be a sign that the heart is underfilled or the patient is dehydrated.
Patients with this device are in advanced end stage HF. The patient often will have mitral regurgitation preoperatively, and reduction in mitral regurgitation is a desired result that is often seen. No AI is desired, but small amounts are tolerated. Ideally, the aortic valve should open every few beats. Most do not open with every beat. Some will not open at all if the LV is in poor condition. If the aortic valve does not open, examine the aortic root for clot. All right heart measurements should be acquired. On TEE, the inflow cannula should be directed toward the mitral valve. The sonographer should pulse the inflow cannula in the apex, which oftens show phasic flow. There may be no antegrade flow by spectral Doppler in the aortic valve position. There should be phasic flow above the baseline.
While hospitalized in the ICU, these patients will have serial limited echocardiograms. Chamber dimension measurements and valve velocities should be taken if the walls and valves are visually discernible. In addition, the patient should be evaluated for pericardial and pleural effusions and for cardiac tamponade. Before each echocardiogram, examine the prior images to facilitate the grading of any recovery. Pump speed is adjusted with the aid of echocardiography to allow adequate filling of the LV cavity and avoid the development of suction. Adequate filling prevents suction events that can cause ventricular tachycardia. As always, evaluate RV function.
Heartware
The Heartware LVAD is a long-term centrifugal pump whose impeller is suspended within the housing without any mechanical contact by magnets and a hydrodynamic thrust bearing. 21 The Heartware device is smaller and may better suit patients with small torsos. The Heartware device delivers 4 L to 8 L of flow at 1800 to 4000 rpms. The rpms are usually set between 2000 and 29 000 rpms. 22
Heartmate III
Heartmate III (HM III) is a pulsatile pump that is currently in the clinical trial stage in the US. The first generation of pulsatile pumps was rarely used due to limitations with the patient’s body habitus, the noise of the pump, and limits of longevity of the pump. The rationale behind the HM III is that the propeller is magnetically levitated with intermittently artificial pulsatility. It is hoped that it would decrease the stress on the device to promote longer pump life and reduce complications for the patients. 23
Biventricular Support (BIVAD)
There are patients for which LVAD support alone is not sufficient to support life while waiting for a heart transplant. There are also patients who have an LVAD as their DT and require temporary right heart support post LVAD implant. Currently, BIVADs are only done in the hospital for short periods of time. Biventricular assist device patients are more ill and generally have worse outcomes than patients who have LVADs alone. 24
Right Heart Assessment
One of the most important aspects of the care of heart transplant and VAD patients is assessment of the function of the right heart. Stage D HF is left sided HF that produces signs and symptoms even with medication and therapy. This stage of HF has exhausted the limits of medical therapy and usually begins the conversation around surgical intervention between the physician and patient. 25 At this point, the right heart has now been exposed to chronically elevated left sided filling pressure. This creates high pulmonary vascular resistance, resulting in pulmonary hypertension and leading to right HF. 25 Chronic obstructive pulmonary disease (COPD) and history of pulmonary embolism can also complicate matters for a patient with left HF. Compromise of the right heart will limit function and negate any potential gains made from the LVAD on the left side. Thus, right heart assessment is a part of both preoperative transplant assessment and device evaluation. In transplant patients, such assessment provides an indication of pulmonary function and the degree to which it may complicate postoperative recovery. Poor postoperative right heart function increases mortality rates in these patients. The ASE’s “Guidelines for Echocardiographic Assessment of the Right Heart in Adults” is an excellent primer. 26 There are currently no approved long-term devices for the right ventricle. There are, however, approved temporary supports.
RV Impella
The RV Impella is a right ventricular assist device (RVAD) that received FDA approval in January 2015. The inlet cannula draws blood from its position in the IVC and delivers the blood to the outlet cannula in the pulmonary artery. This short-term device can be used for support post LVAD implant, MI, heart transplant, or open heart surgery. 25
Tips for Sonographers
With transplants and VAD patients, early postoperative imaging can be difficult to accomplish. Scar tissue, bandaging, ventilators, VAD equipment, and ECMO can crowd the surrounding area and make access to imaging areas difficult. When evaluating the RV, make note of RV size and function, making note of any septal shift to the left. Because of the potential for large volume shifts in the early postoperative period, echocardiography may be performed daily because these volume changes alone can alter RV and LV function. If the patient is decompensating, evaluate the patient for right HF. The VAD pump speed may need to be adjusted.
Outpatient Assessment
In the outpatient setting, echocardiograms can be limited or complete studies. It is important for sonographers to assess the LV for size, the amount of AI, and aortic valve opening or lack thereof. In the office setting, physicians may consider doing a RAMP study. A RAMP study consists of changing VAD speed to evaluate cardiac response. If the LV is dilated, VAD speed may be increased to reduce LV size. An increase in VAD speeds can also determine AI. If there is a significant amount of AI, the surgeon may consider closing the valve. If possible, the sonographer can measure RV size, RV FAC, TDI S’ RV strain, RIMP, and TAPSE to measurements of right heart function for record and comparison. 26
Conclusion
New developments in the treatment of advanced HF patients are proliferating and can be diagnostically challenging for sonographers. It is important to stay current on new technologies being provided to patients and their implications for sonography. Echocardiography is very useful for the assessment of postoperative complications because it allows visualization of the cardiac chambers and blood flow at LVAD inlets and outlets as well as evaluation of cardiac valve function.
The number of advanced HF patients has substantially increased in the past two decades. Heart transplant and MCS devices have helped advanced HF patients have a higher quality of life. Sonographers play a pivotal role in the treatment and ongoing assessment of these patients that have been afforded these new and evolving technologies. Continuing education in the utilization of these cardiac devices is paramount as continued innovation occurs.
Footnotes
Acknowledgements
The authors would like to thank my medical director, David Langholz, MD, FACC; Teresa Wainscott, PA-C; and Penny Wilton, MD, FANZCA, FRCA, FICM, for their explanations of the technology and which images are most important. Thank you also to Karen J. Vander Laan, PhD, MSN, RN, for helpful manuscript editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
