Abstract
Background
There is a growing population of patients in the UK with advanced heart failure who are receiving a left ventricular assist device (LVAD) as a bridge to transplant. This is due to the plateauing number of heart transplantations and the increasing evidence of the effectiveness of these devices. It is, therefore, important that all clinicians working in an intensive care setting have an understanding of how LVADs work, whether as a district general physician referring a patient for consideration of implantation or a tertiary centre healthcare professional managing the complications.
Presentation
This case study describes the journey of a patient presenting with decompensated heart failure who failed to improve despite maximal medical intervention. The patient was not eligible for a heart transplant at the time, so an LVAD was inserted as a bridge to recovery of organ dysfunction and then eventual cardiac transplantation.
Discussion
This article will focus on providing an overview of the indications and anatomy of LVADs as well as the evidence behind their use so that intensive care professionals are aware of the potential of these devices. There will also be further discussion around complications of these devices and practical points to consider when managing a patient who has an LVAD in situ.
Background
The prognosis for advanced heart failure remains poor with a one-year mortality of 25–50% for patients with New York Heart Association Class IIIb-IV. 1 Many patients are not eligible for a heart transplant due to end organ dysfunction and, in suitable patients, donor availability is limited. Mechanical cardiac support is a rapidly evolving technology that can be lifesaving in these individuals. This case study considers a patient eligible for such a device and the evidence behind its use.
Example case
A 51-year-old woman presented to the emergency department with decompensated heart failure. She had a history of ischaemic cardiomyopathy. Her last echocardiogram showed a systolic pulmonary artery pressure (PAP) of 35 mmHg, an ejection fraction of 17% and a dilated right ventricle with preserved function. She had a CRT-D (cardiac resynchronisation therapy with defibrillator) and was on maximal medical therapy including bumetanide, ramipril, bisoprolol and spironolactone. Her angiogram showed triple vessel disease with no revascularisation options available. Other medical history included a high body mass index (BMI), insulin-dependent diabetes mellitus and she was an ex-smoker.
The patient was admitted to the intensive care unit due to type 1 respiratory failure and cardiogenic shock. She needed continuous positive airway pressure ventilation non-invasively with 50% oxygen to maintain her oxygen saturations at 94%. She was commenced on pharmacological treatment with furosemide, dobutamine and milrinone infusions to support her cardiac output. She did not improve and remained on inotropic support.
Management
She was referred to the cardiac multi-disciplinary (MDT) meeting where the consensus was that she would be a suitable candidate for a left ventricular assist device (LVAD) as a ‘bridge to decision’. Her high BMI and PAP were relative contraindications for a heart transplant at the time and the use of such a device would allow for these to be corrected.
She had a third-generation LVAD inserted into the apex of her left ventricle. Her cardiopulmonary bypass time was 75 min and she received no blood products intra-operatively. She returned to the cardiac intensive care unit where she remained intubated and ventilated. Her monitoring included central and arterial lines and a pulmonary artery catheter. She required multiple inotropes (milrinone and adrenaline), vasopressors (vasopressin and noradrenaline), nitric oxide as a pulmonary vasodilator to reduce right ventricular afterload and a furosemide infusion to aid diuresis. She received a five-day course of prophylactic antibiotics.
The patient had regular cardiac output (CO) studies performed, which initially showed a cardiac index (CI) of 2.5 L/min/m2, systemic vascular resistance index (SVRi) 1500 dynes/s/cm5, stroke volume (SV) 70 ml, CO 5.0 L/min and a PAP of 35/16 mmHg. By day 2, she was extubated and was weaned off the majority of pharmacological cardiovascular support except for a small dose of noradrenaline. She was started on warfarin.
Discussion
Overview
Ventricular assist devices are used as a lifesaving treatment option for patients with severe decompensated heart failure, with an ejection fraction of less than 25%, who have failed to improve with optimal medical therapy. Absolute contraindications for insertion include irreversible hepatic or renal failure and metastatic cancer. They can be surgically inserted into the left (LVAD) or the right (right ventricular assist device (RVAD)) chambers of the heart providing an alternative pathway for oxygenated blood flow into the aorta or pulmonary artery respectively (see Figure 1). The devices augment cardiac output and assist the failing ventricle.
2
They are preload dependent and afterload sensitive. The newer devices incorporate a fully magnetically levitated rotar that can generate a maximal continuous centrifugal blood flow of up to 10 litres/min.
3
Patients may, therefore, have a non-palpable pulse and it may be impossible to measure their blood pressure with a standard non-invasive cuff, requiring an invasive arterial line to be inserted with ultrasound guidance.
Schematic representation of an LVAD providing a continuous flow of blood from the left ventricle to the aorta. LVAD, left ventricular assist device.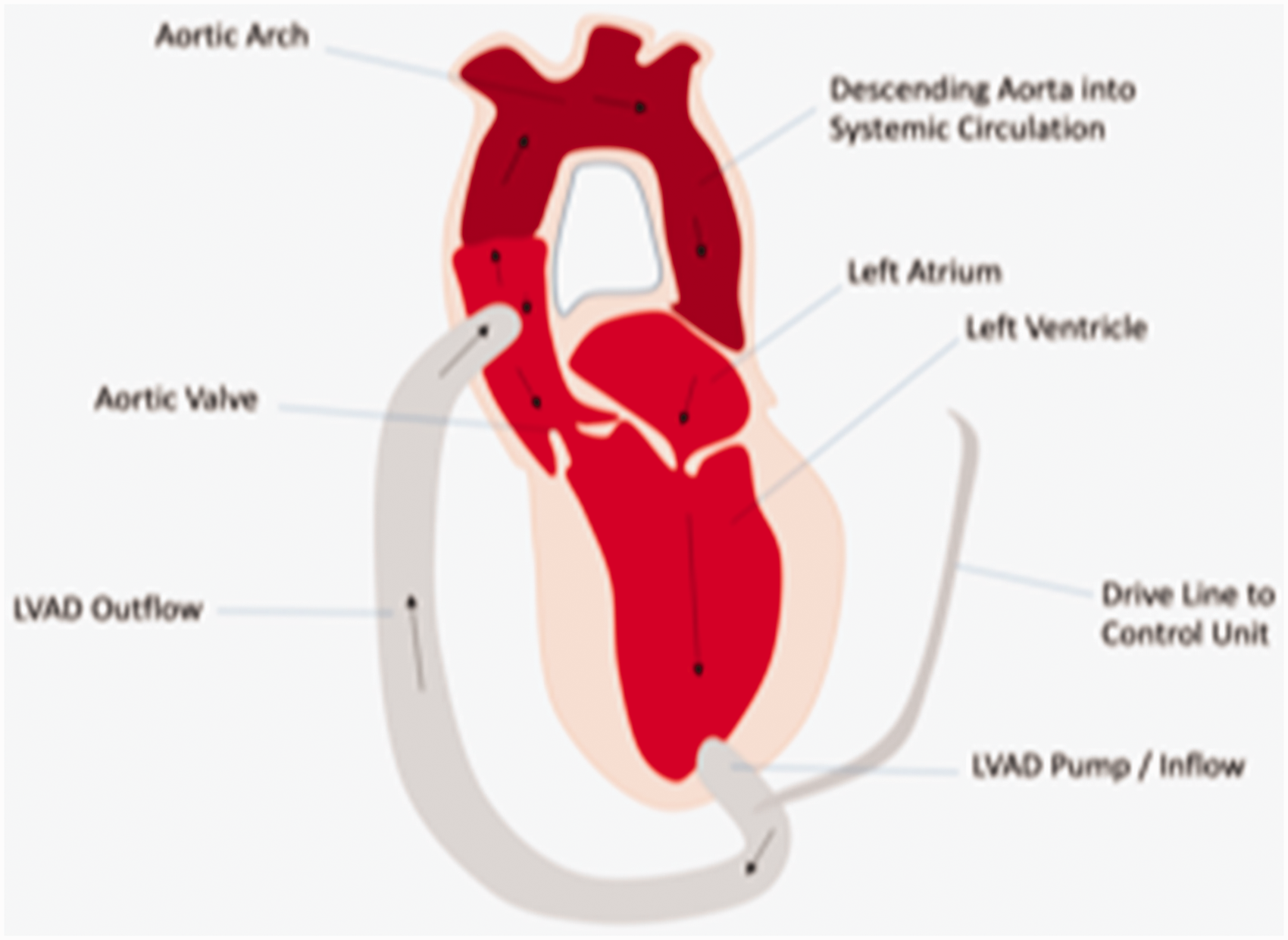
Implantation of a mechanical assist device is indicated as a ‘bridge to decision’ for cardiac transplantation by improving potentially reversible secondary organ failure, such as reducing pulmonary hypertension as described in the example case. 5 This can improve post-transplant survival. They are also indicated as a ‘bridge to recovery’ of native cardiac function with eventual explantation. 4 In the UK, NHS England does not commission mechanical assist devices as destination therapy for patients in whom a transplant is contra-indicated due to their high cost. However, it is important to know that patients receiving LVADs as permanent, life-long therapy may be seen in other countries. The majority of patients receive an LVAD with approximately 15% receiving both an LVAD and RVAD as biventricular support (BiVAD), also known as a total artificial heart. 6 These devices are fundamentally different from Veno-Arterial Extracorporeal Membrane Oxygenation (VA-ECMO) as an LVAD provides cardiac support only and requires adequate lung function and oxygenation. However, some groups of patients may be considered for peripheral VA-ECMO pre-LVAD insertion to improve organ dysfunction.
Evidence
The REMATCH trial in 2001 was the first study to reveal the longer term survival benefits and show that mechanical assist devices are an acceptable alternative to transplantation 7 ; 129 patients with end-stage heart failure ineligible for transplantation were randomly assigned to receive either an LVAD or optimal medical therapy. The rates of survival at one year were 52% vs. 25% respectively (p = 0.002) with a significantly improved quality of life in the LVAD group. This trial used the first-generation devices, which were pulsatile positive displacement pumps. They were problematic due to limited durability and the extensive surgical dissection required with high infection rates, malfunctioning and bleeding. This was evidenced in the REMATCH trial by the increased frequency of serious adverse events.
A further randomised controlled trial published in 2009 used the newer generation of devices. 8 These are centrifugal pumps with longer durability, a smaller size and simplified surgical implantation. Rapid changes in rotar speed generate an intrinsic artificial pump pulse that is designed to reduce pump stasis and thrombosis; 34 patients with advanced heart failure who were ineligible for transplantation received a continuous flow device and 66 received the pulsatile flow device. The newer devices showed a significantly improved probability of survival free from stroke and device failure at two years. Similar results have been shown in subsequent trials such as ENDURANCE 9 and MOMENTUM 3. 10
It is important to note that there are currently no randomised controlled trials comparing LVADs with best medical therapy in transplant eligible patients.
Observational studies of the continuous flow pumps reveal a 180-day survival of >90% 11 and improved survival while on the transplant waiting list. 12 The Interagency Registry for Mechanically Assisted Circulatory Support (INTERMACS) in 2014 contained data for >10,000 non-transplant eligible patients and showed survival rates of 80% at 1 year and 70% at 2 years. 13 This has led to LVAD implantation being recommended by the European Society of Cardiology, as a bridge to transplantation or as destination therapy, for acute and chronic heart failure. 5
Complications
Despite these technical advances, bleeding, thromboembolism, driveline infections, pump thrombosis, arrhythmias, strokes and device failure remain significant problems. 12 The continuous pressurisation of the aortic root can lead to aortic insufficiency with resultant loss of forward flow and left ventricular distension, which can be life threatening. 14 Hypotension in a patient with an LVAD can be related to the device itself such as malposition, thrombosis or suckdown of the pump. All patients presenting with a complication should be discussed with the nearest heart transplant centre.
Gastrointestinal bleeding is particularly problematic due to the concurrent use of a systemic anticoagulant and antiplatelet agent, such as the combination of warfarin and aspirin, the formation of arterio-venous malformations and acquired Von Willebrand factor deficiency. 15 Efforts are being made to decrease the amount of antithrombotic therapy given to patients with LVADs. The TRACE observational study of 100 patients taking aspirin only, warfarin only or no antithrombotic therapy showed that these regimes led to a low rate of device thrombosis. 16 However, despite the reduction in antithrombotic medications, 52% of patients still had significant bleeding. This highlights the multifactorial component to bleeding risk in these patients and the need to gain a better understanding of how LVADs affect platelet function, the coagulation cascade and thrombosis.
Another major cause of morbidity and mortality in patients with LVADs is the risk of early or late right heart failure. An increase in the right ventricular (RV) preload occurs due to the improved cardiac output and venous return, which is compounded by an inadequate RV reserve. 17 The post-operative incidence of RV failure ranges from 10 to 40%. Meticulous monitoring and recognition are essential to allow early initiation of treatment such as pulmonary vasodilators, inotropes, diuretics or insertion of a RVAD. In patients receiving positive pressure ventilation, improving ventilation perfusion mismatch, and minimising hypoxic pulmonary vasoconstriction will also assist RV performance.
Conclusion
Developments in the technology of cardiac mechanical assist devices have led to the increasing role of mechanical circulatory support in patients with advanced heart failure. In the UK, LVADs are indicated for patients as a bridge to transplant or recovery by improving reversible end organ dysfunction. They are life saving. Patients with cardiogenic shock should be managed in an intensive care environment with cardiac output monitoring. They should be referred early to a cardiac MDT for consideration of transplantation and LVAD implantation, before they develop multi-organ failure.
Nevertheless, adverse events such as life-threatening bleeding and RV failure limit their broader application. Newer generation pumps are being developed in an attempt to reduce some of the devastating complications.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
