Abstract
Arteriovenous fistulas are known to be traumatic, congenital, or created for hemodialysis purposes in patients diagnosed with end-stage renal disease. In this case study of an immature Brescia-Cimino fistula, an outflow vein stenosis was assessed solely using sonography. This type of stenosis has been found to be the most common cause of fistula failure. This case study demonstrates how the use of sonography identified an immature arteriovenous fistula as well as assisted in further treatment planning and outcomes.
Introduction
Renal disease has become a prominent issue in today’s society and has a variety of predictable causes, such as hypertension, diabetes mellitus, and smoking. 1 As renal disease progresses to the designation of end-stage renal disease (ESRD), patients need dialysis to maintain the body’s filtration needs. One method of dialysis is through hemodialysis and the use of an arteriovenous fistula (AVF). In this particular case, a radiocephalic AVF was surgically created to provide easy access, achieve elevated flow velocities, and facilitate regular hemodialysis treatments. 2
In the process of assessing a patient for AVF surgery with sonography, a selected vessel should demonstrate patency, lack of calcifications, and lack of thrombus and be of an appropriate size. In the case of tributaries, ligation of a tributary vein associated with a potential AVF should be done to increase venous flow through the fistula. The vein of choice must measure 0.25 cm in diameter to be deemed viable for creation of a dialysis AVF. 3 To determine diameter of the vessel, the sonographer can use a tourniquet to dilate the veins for better visualization. Depth should also be adjusted to exclude deep veins because deeper veins may be undesirable compared with veins closer to the surface, which provide easier access. The vein being considered will fail to meet these criteria if any evidence of previous or current thrombus is present or any venous calcifications are noted. Venous tributaries should be marked or noted for the surgeon to ligate during the initial assessment with sonography.
Standard Doppler protocol should be used to study the artery for patency, lack of calcifications, or absence of stenosis and for recording appropriate measurements. The artery must be at least 0.20 cm in diameter. 2
Benefits of a Brescia-Cimino AVF include longer lasting hemodialysis access, lower likelihood of infection, or possible thrombosis. 2 Even though grafting and catheterization provide quicker hemodialysis access, they can be more susceptible to thrombosis and infection. The most common type of AVF is a Brescia-Cimino fistula, which is an anastomosis of the radial artery and cephalic vein near the wrist. 4 Once the fistula has been created, it must mature, which may take 2 to 3 months. 2 Follow-up serial postsurgical sonographic examinations should be completed to determine access maturity.
Flow volumes can vary across conduit types; in addition, there is a wide range of accepted flow rates that have been established, which can be confounding. For instance, some institutions use 800 mL/min as normal, 500 to 800 mL/min as borderline, and less than 500 mL/min as abnormal. These metrics are specific to an AVF. For a patent graft, 800 mL/min is considered an optimum flow rate. Commonly, 600 mL/min is the cutoff between normal and abnormal for Brescia-Cimino. 5 The example provided points to accepted criteria differences depending on the graft type. The most common complication of an AVF is stenosis at the anastomosis site or within the outflow vein. 5
Reproducibility and safety have made sonography the ideal imaging modality for investigating an AVF because it is noninvasive and does not require the use of iodinated contrast in patients with renal failure. It is used continuously to assess the preoperative maturation, postoperative arterial flow, and venous anatomy.
Case Report
A 76-year-old male presented to the vascular lab for sonographic evaluation of his left Brescia-Cimino fistula 1 month after implementation per protocol. As stated, a 1-month postoperative follow-up sonographic examination is required to begin assessing maturity. His clinical presentation was not concerning, and he had no complaints. There was a palpable thrill at the site of the AVF. The AVF sonogram was performed using a linear array transducer with a central frequency of 9.0 MHz on a Phillips iU22 ultrasound system. The sonogram began by evaluating the radial artery proximal to the anastomosis, which had a peak systolic velocity (PSV) of 130 cm/s and an end-diastolic velocity (EDV) of 51.1 cm/s (Figure 1). The radial artery was measured in transverse, proximal to the anastomosis, with a diameter of 0.29 cm. The radial artery was seen distal to the anastomosis and was found to have a PSV of 162 cm/s and an EDV of 16.6 cm/s (Figure 2). Prior to the vascular surgery, the artery had high-resistance triphasic flow. If the AVF had matured properly, arterial flow would have demonstrated a low-resistance biphasic waveform, demonstrating maximum circulation throughout the AVF. 6
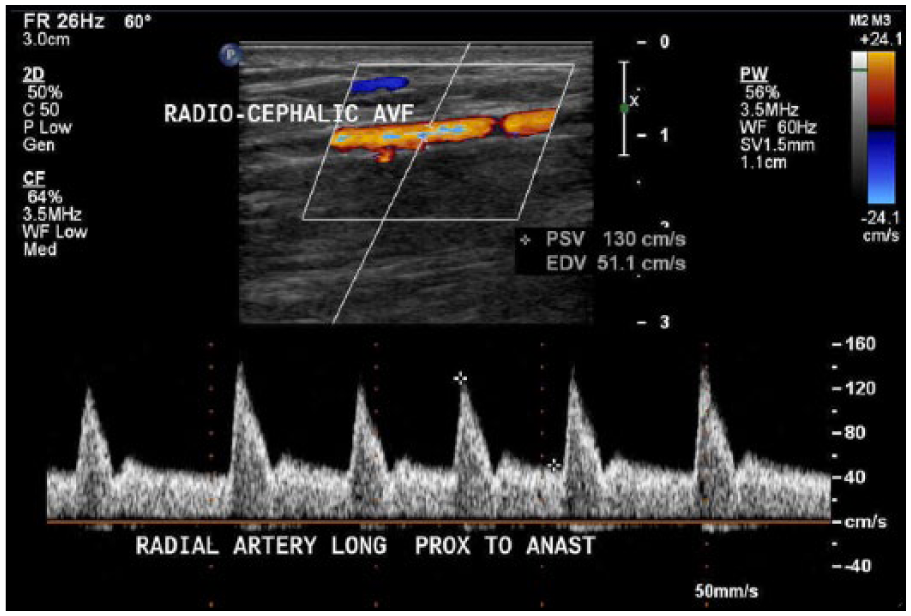
Pulsed Doppler demonstrates flow in radial artery proximal to arteriovenous fistula anastomosis.
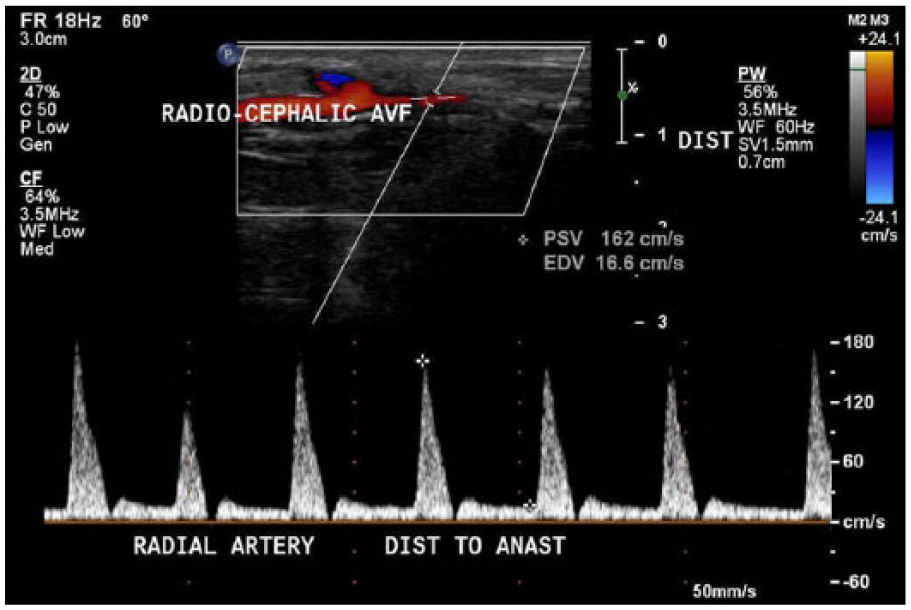
Pulsed Doppler demonstrates flow in radial artery distal to arteriovenous fistula anastomosis.
Next, the cephalic vein was identified distal to the anastomosis (Figure 3), with a PSV of 204 cm/s and an EDV of 99.9 cm/s (Figure 4). The time-averaged mean velocity (TAMV) is determined by obtaining 3 to 4 cardiac cycles with pulsed Doppler. 5 Usually, this is a calculation that the equipment software generates for the operator. The cephalic vein distal to the anastomosis demonstrated a TAMV of 197.2 mL/min (Figure 5). This finding demonstrates that the AVF was immature since it had a TAMV of less than 500 to 600 mL/min. 5 The diameter was also measured at 0.3 cm. The cephalic vein was then followed up the arm and evaluated every 2 cm. Two centimeters distal to the anastomosis, a tributary of the cephalic vein was identified (Figure 6). An incidental finding was noted of a tributary off of the outflow vein, which can cause failure of the fistula to mature properly due to decreasing flow velocities and the possible future development of a thrombosis. Upon evaluating the cephalic vein with pulsed Doppler at this site, a PSV of 636 cm/s and an EDV of 329 cm/s were recorded (Figures 7 and 8). Outflow vein velocities, such as these, are highly suggestive of a stenosis because the PSV at this site compared with the PSV 2 cm upstream was greater than a 2:1 ratio, meaning that there was greater than 50% stenosis. 5 The cephalic vein was followed 4 cm more distally (toward the heart) to the AVF anastomosis and was found to have a PSV of 34.2 cm/s and an EDV of 21.6 cm/s (Figure 9). These parameters can also be referenced to the ACR Appropriateness Guidelines provided for the evaluation of hemodialysis fistulas. 7
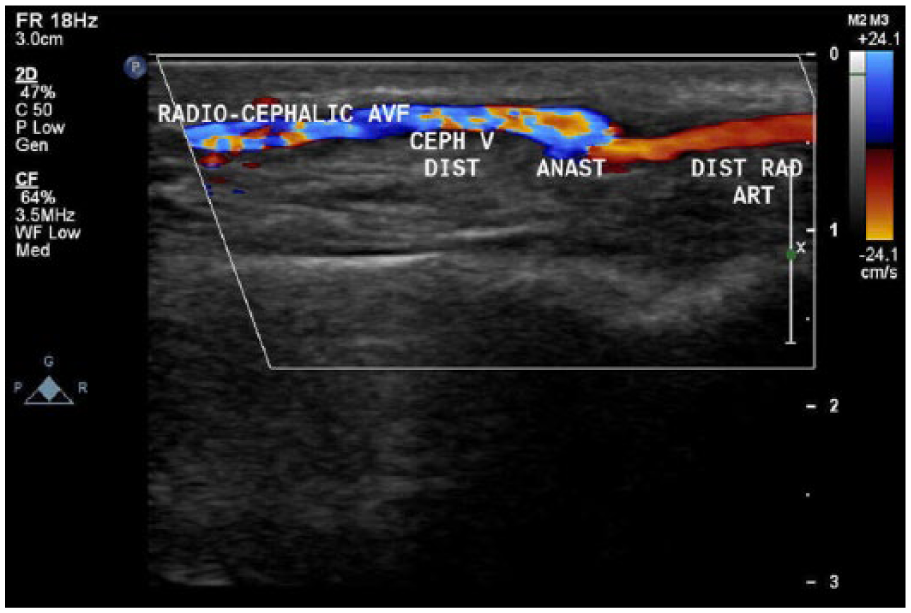
Sagittal view of distal cephalic vein departing the arteriovenous fistula anastomosis.
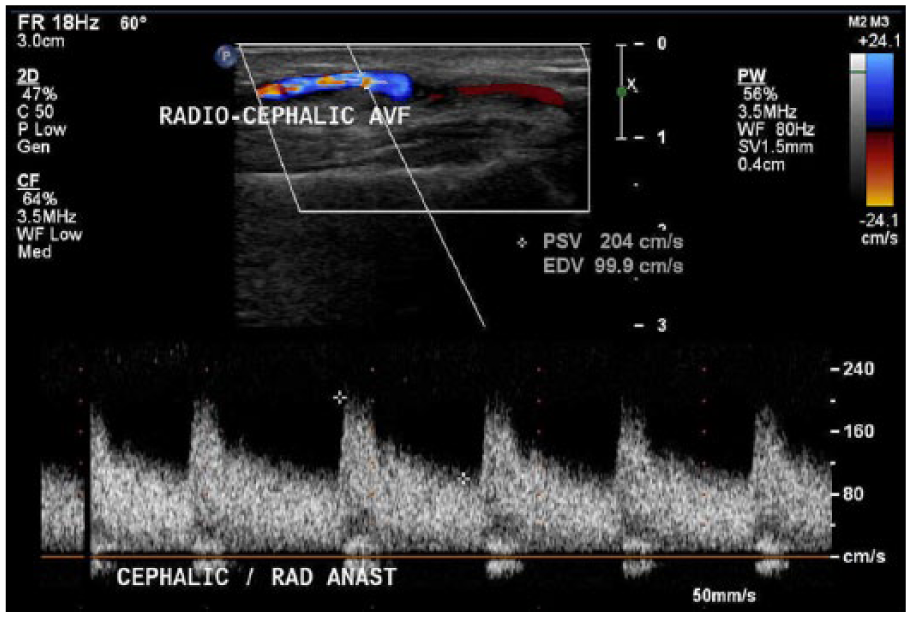
Pulsed Doppler demonstrates flow in the distal cephalic vein departing the arteriovenous fistula anastomosis.
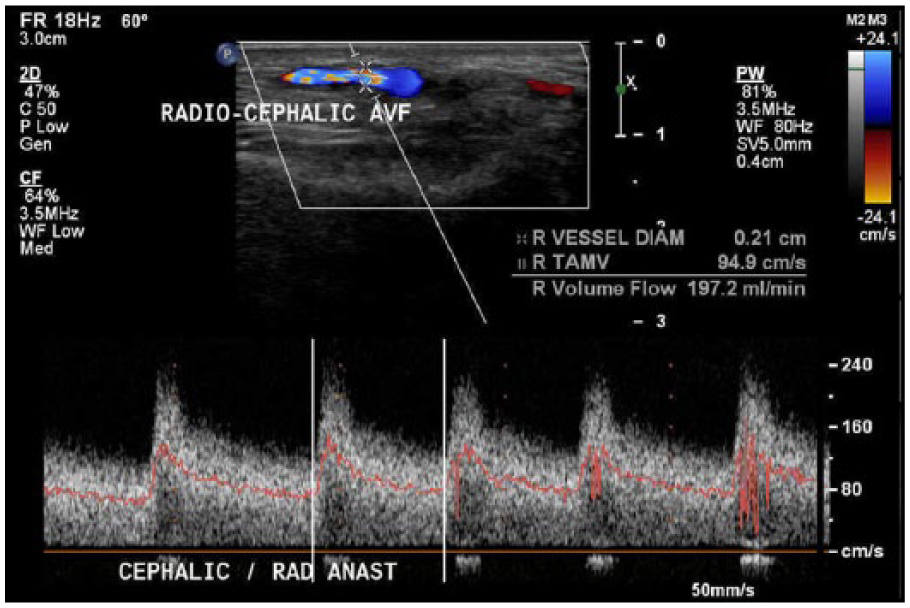
Time-averaged mean velocity of distal cephalic vein shows a rate of 197.2 mL/min.
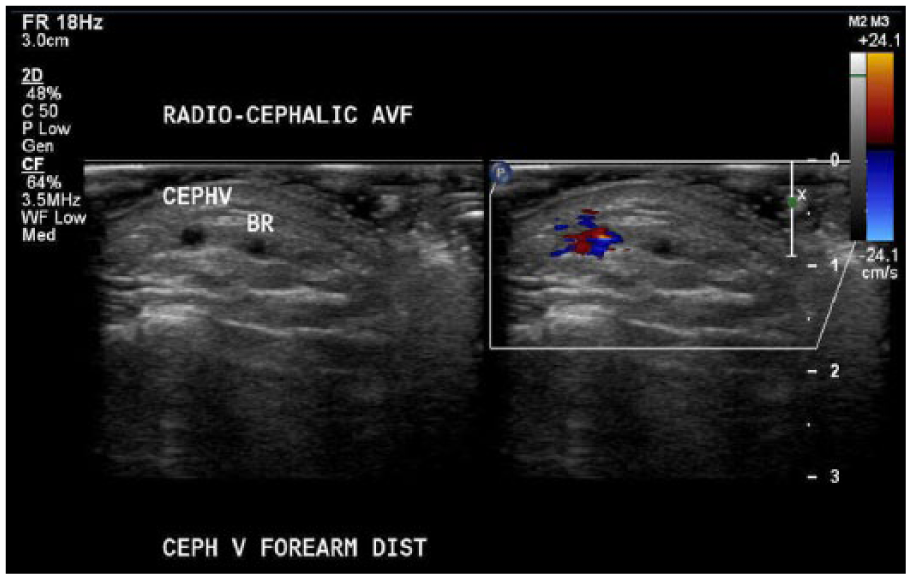
Transverse view of cephalic vein branch 2 cm above the arteriovenous fistula anastomosis.
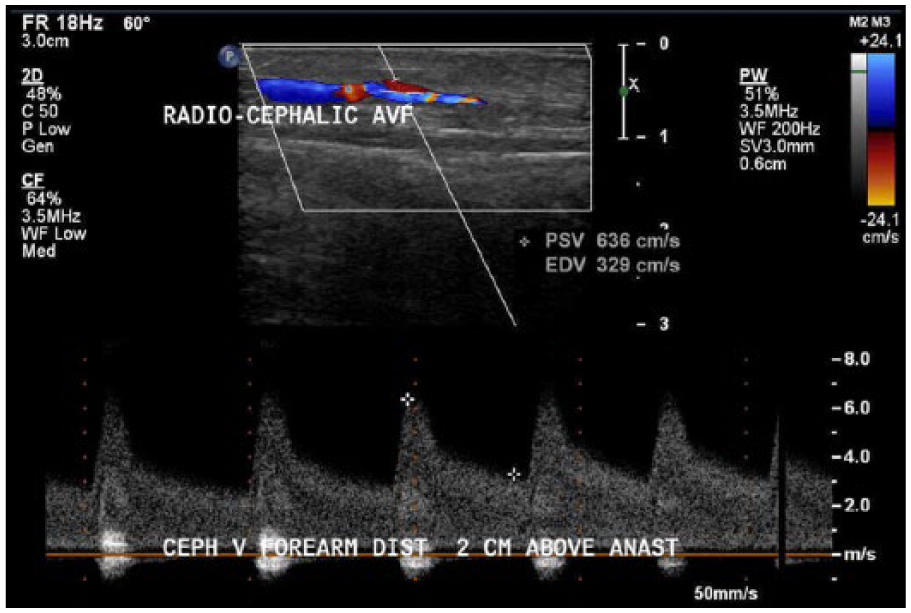
Pulsed Doppler demonstrates flow in distal cephalic vein 2 cm above arteriovenous fistula anastomosis.
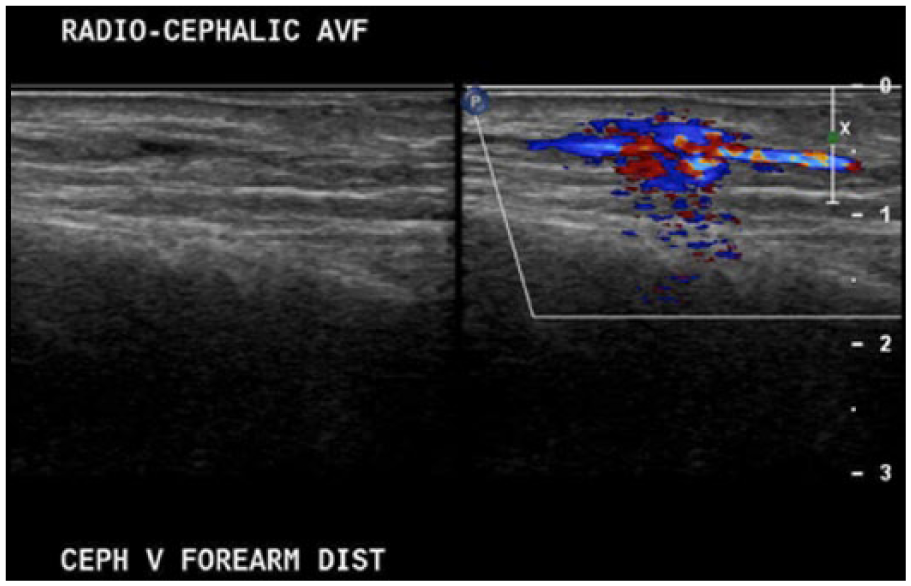
Sagittal view of cephalic vein stenosis 2 cm above arteriovenous fistula anastomosis.
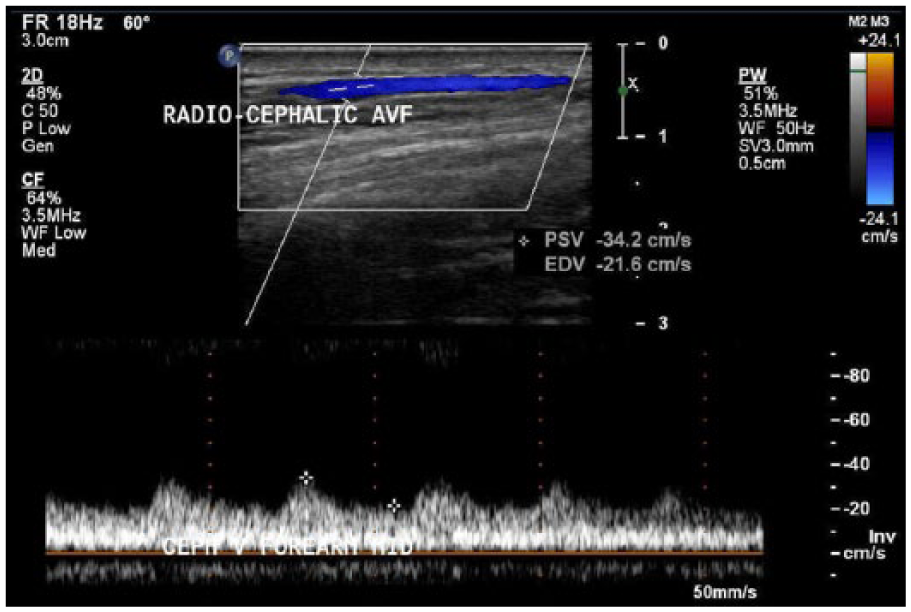
Pulsed Doppler demonstrates flow in mid-cephalic vein 4 cm above arteriovenous fistula anastomosis.
Sonography was initially used to map the vessels of the left upper extremity before the AVF surgery. This helped the surgeon determine vessel suitability for the AVF. Once the surgery had been performed, sonography was used to evaluate the fistula for any complications that had occurred. Since the patient had an outflow vein tributary and stenosis, the patient underwent repeated vascular surgery at the same facility to correct these issues. After this had been accomplished, sonography was again used to evaluate the AVF postoperatively.
Discussion
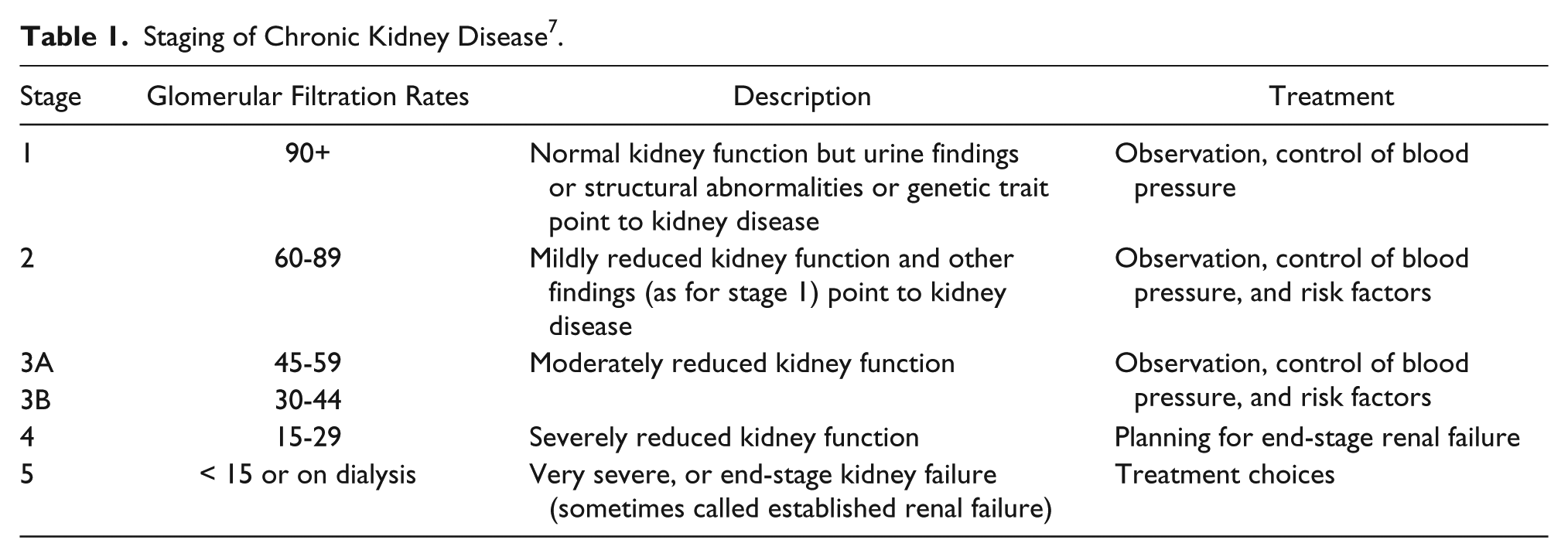
End-stage renal disease is most common in men 45 years of age and older and has been seen as a common affliction among African Americans.2,8 This disease begins as chronic kidney disease. Chronic kidney disease has been stratified into five stages, associated glomerular filtration rates, description of kidney function, and suggested treatment options for each stage (Table 1). 9 At the final stage, patients can become candidates for hemodialysis as a mechanism for maintaining proper blood filtration rates. As mentioned earlier, the preferred method for dialysis is an AVF, due to lower infection and thrombosis rates.
Staging of Chronic Kidney Disease 7 .
As discussed above, there are several dialysis treatment options available for ESRD. Peritoneal dialysis requires the use of catheterization. However, a catheter is known to raise the risk of infection; therefore, this form of dialysis is chosen less often. The AVF graft is the second preferred choice for permanent access. An AVF graft is a piece of plastic tubing, generally made out of Teflon or fabric, that is attached on one end to an artery, and on the other end to a vein. 2 This method presents complications such as lower patency rates and higher infection rates. 10 There are several different AVF options that can be offered to patients. The most common is called a Brescia-Cimino fistula (Figure 10).4,11 This is a connection between the radial artery and the cephalic vein near the wrist. Other less common AVF connections are between the brachial artery and cephalic vein at the elbow and between the brachial artery and basilic vein at the elbow. 4
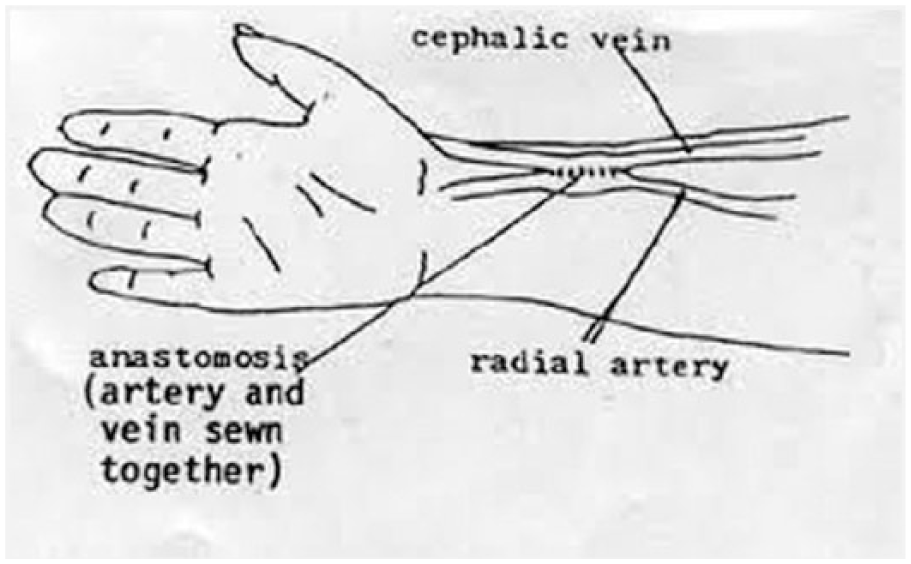
Brescia-Cimino fistula.
Whereas the most common connection sites are listed above, it is difficult to determine the percentage of each type of AVF in the dialysis population due to varying factors. However, in one particular university hospital, 245 patients with AVF outcomes were reported. In this descriptive study, the cephalic vein was used 59.2% of the time, the antecubital vein was used in 30.6% of patients, and the basilic vein was used in 0.4% of patients. The radial artery was used in 53.1% of patients, the brachial artery in 35.9%, and the ulnar artery in 1.2% of patients. After the AVF formation surgery, the patients were followed up at 6 months, 1 year, and 3 years postsurgery to evaluate AVF success rates. 12
The cephalic vein had 85% success at 6 months, 73% success at 1 year, and 59% success at 3 years. The antecubital vein had 81% success at 6 months, 67% success at 1 year, and 67% success at 3 years. The basilic vein data were indeterminable due to a dropout rate of 0.4%. The arterial aspect of the AVF was also examined, and the radial artery had an 80% success rate at 6 months, 72% success at 1 year, and 59% success at 3 years. The ulnar artery had a 66% success rate at 6 months and no success at 1 and 3 years. The brachial artery had an 86% success rate at 6 months, 79% success at 1 year, and 69% success at 3 years. Factors affecting these data could have been underlying disease and location of AVF. 12
In this outcomes study, patients become eligible for an AVF if the vessels demonstrated patency, had no calcifications or thrombosis, and were an appropriate size. The patient in the current case study had a Brescia-Cimino AVF for dialysis that was immature developmentally and stenosed in the outflow vein, one of the most common sites for this pathology. 5
Conclusion
In conclusion, vascular sonography was used to determine the patency and maturity level of a Brescia-Cimino fistula. Sonography was able to detect AVF immaturity based on TAMV independently. It was also able to characterize outflow vein stenosis in the cephalic vein based on waveform and velocities. Other imaging modalities could not have been used due to the patient’s ESRD, making the patient ineligible for iodinated contrast injection. These findings by sonography showed the extent of immaturity and stenosis. Although common, these pathologies must be documented to raise the rates of AVF success. Sonography remains the favored method of AVF analysis and documentation.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
